Uridine Triphosphate Trisodium Salt: A Commentary on Science, Industry, and Progress
Tracing the Roots of Uridine Triphosphate Trisodium Salt
Uridine triphosphate trisodium salt started as an emblem of curiosity. Early biochemists pulled together meager resources to understand nucleotides. Decades ago, most labs couldn’t imagine synthesizing complex triphosphates outside elite circles. Now, seeing it labeled in large glass jars on lab shelves signals how far we’ve come. Reading about the isolation of ribonucleotides in the early twentieth century, you bump into stories colored by near misses, laboratory accidents, and big scientific personalities. Discovery often grew out of an attempt to tease apart the building blocks of RNA and probe what keeps genetic information moving in living systems. Today, we deal with a much more accessible and consistent product, far removed from the fragile, hard-won samples of the past.
A Snapshot of the Modern Product
Uridine triphosphate trisodium salt looks and feels like a typical, fine crystalline powder but chemistry gives this simple appearance deeper meaning. The trisodium form keeps it stable and easy to dissolve—qualities that matter to more than bench scientists. Its solubility, taste, and color trace directly to the arrangement of sodium and phosphate ions. Bags labeled UTP hide that intricate dance, but for anyone who’s ever tried to weigh out a minuscule amount in a rush, consistency is a minor miracle. This isn’t a lab novelty any longer. Production lines, research facilities, and clinical labs all count on supplies that meet agreed purity and composition standards, but the leap from obscure biochemistry to life science staple happened fast.
Real-World Properties: More Than a Chemical Formula
I recall an early afternoon watching a technician weigh out uridine triphosphate trisodium salt with the kind of attention most people reserve for family heirlooms. That carefulness speaks to its properties. It keeps a moderate shelf life under sealed, dry conditions. Once open to air, moisture and even minor contaminants degrade both structure and function. One accidental spill—what looks like a wasted teaspoon—means lost money and lost hours. This hard-earned sense of importance ties back to what the salt delivers: a reliable, measurable substrate for enzymatic assays or an essential activator in various biological systems.
Bearing Labels: What They Actually Mean
Walking through storage rooms, you notice rows of containers with technical jargon, but behind those specs and acronyms lies regulatory, scientific, and ethical responsibility. Companies stamping purity percentages above 95% rely on proven chromatography and spectrometry, not guesswork. Label warnings about storage, contamination, and handling come from real cases—lab workers who picked up itchy hands or respiratory irritation after careless handling, or entire lots scrapped because of temperature excursions. This isn’t marketing or public relations fluff; filtering out impurities matters if you want meaningful, reproducible data. These standards serve more than lawyers—they protect end users trying to mimic conditions for their next experiment or product batch.
How It Comes to Be: The Making of UTP Trisodium Salt
Synthesizing uridine triphosphate trisodium salt takes more than enthusiasm. A mix of well-channeled chemical synthesis and biotechnological finesse allows access to the high-purity material that life science needs. The typical method involves phosphorylation of uridine monophosphate through specific enzymes or high-energy chemical reagents, followed by neutralization with sodium. Scale changes everything—small scale favors manual oversight, while commercial works require automated controls, routine quality checks, and waste management. Every new batch brings a subtle lesson. Researchers have tried optimizing steps to improve environmental impact, shorten reaction times, and extract higher purity with fewer inputs. As demand for precision and green chemistry grows, these processes attract more scrutiny and slow, steady improvement.
Chemical Tweaks and Expanding the Toolbox
Lab workers and scientists tinker compulsively. Given uridine triphosphate trisodium salt, minds go straight to modifications: labeling with isotopes for tracer studies, linking to fluorescent groups, or tweaking ring structures for drug design. These reactions stretch what’s possible, pushing UTP into fields ranging from metabolic tracking to genetic diagnostics. Not every modification works without trouble; sometimes side products gum up the works or altered salts don’t cooperate in critical reactions. Each success feeds new applications, each setback rewrites lab protocols and safety standards. What looks like routine nucleotide chemistry often disguises years of incremental advancement and broken glassware.
Different Faces: Names from Past and Present
Names tell a quiet story. In conversation, lab workers might say UTP, uridine 5'-triphosphate, or even just the “tri-sodium salt.” Patent databases list synonyms countless times, tracing the substance’s path across countries and decades. Small changes in naming can trip up newcomers, especially in regulatory filings or reviewing scientific literature. This jumble of synonyms reminds us how discoveries cross borders. Researchers from Tokyo to Berlin to Boston keep sharing and renaming the same stuff, nudged by culture, regulatory language, and evolving science. Knowing the polyglot language of nucleotides helps avoid confusion and speeds up collaboration, both in projects and problems.
Guardrails: Practical Safety and Handling
Veterans in life science treat uridine triphosphate trisodium salt with respect earned through years of trial and error. Despite its benign reputation, mishaps happen. Dust in open air triggers skin and respiratory sensitivity; unanticipated spills can spoil entire experiments. Safety goggles and gloves bring peace of mind, and even busy labs post handling rules near chemical storage. Waste disposal, air flow, and auditing of storage rooms are not red tape—they save budgets and, sometimes, skin. Regulatory bodies have stepped up scrutiny with increasing awareness of chronic exposure risks and environmental responsibilities. Within the ever-changing landscape of compliance updates, labs adapt old habits to meet today’s standards, swapping war stories and safety hacks in break rooms.
How UTP Steps Beyond the Lab
Uridine triphosphate trisodium salt features in more research areas than some scientists imagine. For years, I watched teams use it as a substrate in enzymatic activity tests, and in more recent times, pharmaceutical companies leverage its properties to design nucleotide drugs and explore neurological intervention. Its role in RNA synthesis, cell signaling, and molecular diagnostics bridges basic research and applied solutions like rapid genetic testing tools. Advances in molecular biology have turned this salt into a quietly essential ingredient in everything from food safety diagnostics to large-scale vaccine manufacturing. Each new research project pulls uridine triphosphate trisodium salt deeper into the machinery of healthcare, agriculture, and bioengineering, as needs shift toward faster, more reliable results.
The Evolving Field: What Research and Development Teaches
As research deepens, uridine triphosphate trisodium salt reveals fresh questions and uses. Labs investigate its influence on metabolic pathways, exploring therapeutic potential in neurological disorders and rare diseases. Biotech startups compete to make purer, more specialized versions for custom applications. Each published study generates as many questions as answers, with research circling back to practicalities like scalable synthesis and safety. Science thrives on this kind of constructive friction. Grants and collaborations sprout up, promising to unlock even greater value through automation, hybrid synthesis, or synthetic biology. The pace of innovation depends on how well technical teams can share findings and adapt—across universities, companies, and independent labs.
Sizing up Safety: Learning from Mistakes and Progress
Toxicity research doesn’t win awards, but it keeps everyone honest. Reports show that uridine triphosphate trisodium salt rarely causes harm when handled right, but long-term effects remain on the table for debate. Chronic exposure data lags behind its expanding use, prompting risk teams to update protocols and encourage transparency in reporting adverse incidents. I’ve watched risk assessments shift alongside changes in production scale and newer use cases. This caution makes sense in a world where new users emerge faster than regulators can draft new guidelines. Good habits, clear warnings, and ongoing education rank higher than wishful thinking or assuming old safety data will hold up forever.
Looking Forward With Eyes Open
The story of uridine triphosphate trisodium salt isn’t done. More researchers want customized forms. More sectors depend on fast, reliable supply chains. Environmental advocates ask for greener production. Scientists continue searching for new reactions, safer rinsing agents, and automated production controls. Regulators tighten standards as applications move closer to daily human health solutions. Each advance in synthesis, safety, or application opens new doors. Progress pushes up against practicality—scale, budget, and evolving standards. Teams who share failures and improvements collectively shape not just the future of uridine triphosphate trisodium salt, but the standards for how science and industry steward powerful tools, day in, day out.
What is Uridine Triphosphate Trisodium Salt used for?
The Brain’s Hidden Helper
Talking to neurologists and digging into biochemical research, I’ve seen how science circles back to small molecules with big impact—like uridine triphosphate trisodium salt. The name feels heavy until you look at what it does. This compound gears itself toward energy transfer and genetic information work inside our bodies. Uridine itself shows up naturally within all living cells, especially in our brain. Scientists in labs and clinics pay attention because of the way it feeds into the production of RNA and supports neural metabolism.
The Medical Side: Healing Nerves and More
Doctors prescribe uridine triphosphate trisodium salt mostly for treating nerve pain linked with diabetes and other neuropathies. Medications that combine uridine, folic acid, and vitamin B12 aim to repair nerves and reduce that persistent numbness or tingling. These ingredients bring together raw materials that the body uses to rebuild and maintain nerve tissue. I remember a friend battling diabetes-related pain, and after months on a supplement containing uridine, the burning in his feet eased up. It matches up with real studies—like randomized trials where patients noted clear improvements after steady use of uridine blends.
Beyond Pain: Lab Research and Athletic Interest
Uridine triphosphate draws curiosity in Alzheimer’s research, stroke recovery, and even sports science. Data points out that uridine may help patch up synaptic connections in the brain. Research teams see hope for cognitive support, since healthy brains depend on steady RNA synthesis and membrane repair. Not every claim measures up yet, but early investigations show memory improvements in animals given uridine and choline together.
In sports clinics, uridine triphosphate trisodium salt sometimes finds its way into intravenous infusions—sometimes with the goal of speeding muscle recovery or supporting energy levels after long competitions. Here, the case isn’t clear-cut. The energy transfer from uridine might play a minor role, but the research still lags behind popular excitement.
Why It Matters
The story of uridine triphosphate trisodium salt isn’t just academic. Millions affected by nerve injuries, or memory loss due to age, call for more attention on how these basic compounds may change daily life. It’s an example where basic science translates into clinical use—where what’s discovered on the bench touches the bedside. When doctors talk about “nucleotide therapy,” they point to uridine as one of the chemical workhorses. Without enough, nerves don’t rebuild easily, and cognitive decline may pick up speed.
Finding Real Value and Safety
Bringing uridine triphosphate into treatment plans takes care. Physicians tend to favor combination therapies, since complex nerve damage rarely improves with one molecule alone. There’s always the question of dose; properly controlled studies use specific amounts and watch carefully for side effects. Too much phosphate can strain the kidneys or shift mineral balance, especially if someone’s taking other medications. So, close medical supervision means as much as the compound itself.
Looking Forward: Smarter, Safer Use
Healthcare researchers still sort out who benefits most and what combinations work best. Today, doctors and scientists call for longer studies—tracking both pain reduction and memory support over years, not just months. Patients and families deserve new options, but they also deserve answers rooted in careful, transparent science. Controlled trials, open reporting of results, and honest conversations between patients and prescribers set the foundation for using uridine triphosphate wisely. Real progress comes when basic chemical knowledge leads to practical, safe help in everyday life.
What is the recommended storage condition for Uridine Triphosphate Trisodium Salt?
Looking After an Expensive Reagent
Opening a container of Uridine Triphosphate Trisodium Salt feels like handling something precious, mostly because of its price and value in molecular biology and pharmaceutical labs. If you want reliable data and steady product quality, the way you store this salt matters, whether you run a busy research space or a quiet startup.
Why Storage Demands Precision
One summer, the team across the hall overlooked a single open freezer door overnight. By morning, their bin of reagents, including Uridine Triphosphate Trisodium Salt, was ruined. Salts absorb moisture and break down if they hang out in the wrong conditions.
This compound loves to pull water out of the air. If left in a humid room or even a carelessly closed bottle, it clumps and can lose its punch. Some powders degrade, lowering yields and creating questionable research. Each gram costs enough that replacement isn’t a casual expense for most labs.
Every researcher wants to keep contamination at bay. Moisture and fluctuating temperatures support molds and bacteria that nobody wants mixing with a high-purity product. I learned early on that even invisible contamination changes the way reactions unfold. It creeps into the data, especially in sensitive enzymatic assays.
Which Conditions Work Best?
Uridine Triphosphate Trisodium Salt stays freshest at -20°C, far from light and humidity. A strong, airtight container with a screw-cap lid cuts down exposure to air. The dark glass bottles often help, blocking UV and visible light, which can encourage unwanted chemical changes. Put the date on each new bottle and minimize how often it sits at room temperature. Small, single-use aliquots help avoid repetitive freezing and thawing.
I rely on dedicated, labeled freezers. Mixing reagents used for food or DNA work leads to costly mistakes and confusion. Sometimes the temptation pulls you in—just slide something into the crowded household freezer—but the frost fluctuations can wipe out irreplaceable stocks.
Don’t stack containers: crowding restricts airflow and can raise the temperature each time the door opens. Rotate old stock to the front. If possible, keep a low-grade hygrometer in the reagent closet and check it often. I spot-check all major reagents every month. If a vial starts to look cloudy or cakes up, it’s safer to discard it than risk an experiment.
Pitfalls and How to Deal With Them
Labs get busy, people cut corners, and mistakes slip in. Sliding a container onto a hot countertop or leaving it near a window usually causes trouble down the line. Even opening these salts in a humid room can introduce problems. I make it a lab rule to open new containers in the cold room or at least in a dry, air-conditioned space. Printed guides right on the reagent shelf remind users about quick handling: “Keep cold, keep dry, seal fast.” This simple sign keeps most mistakes from happening.
For anyone running low-volume operations or remote fieldwork, dried silica packs inside a secondary bag buy some extra time, but routine cold storage still wins out. Once, at a pop-up clinical lab, keeping the salt in a rented office fridge led to mystery degradation. It only cleared up after staff switched to a bench-top, lab-certified freezer with humidity control.
Reliable Storage, Reliable Results
Taking storage seriously saves time, money, and scientific headaches. Good storage habits protect your reagents, keep research honest, and help build trust in lab results. Careful conditions count, and they start with knowing your product and treating it with respect—every single day.
Is Uridine Triphosphate Trisodium Salt soluble in water?
The Story Behind the Science
In any lab, you spot bottles with long technical names like uridine triphosphate trisodium salt and can’t help but wonder if the story behind those labels matters. Dig a little deeper, and you discover scientists are not just filling shelves with fancy words—they look for substances that blend seamlessly with water. Water solubility makes or breaks so many experiments, especially in biochemistry and medical research, where prepping buffers and solutions demands predictable behavior from every ingredient.
A Simple Answer with Real Consequences
Uridine triphosphate trisodium salt may sound complicated, but it’s straightforward with water. This salt, a form of uridine nucleotides, dissolves easily in water at room temperature. Even a rookie grad student swirling a test tube can see the powder disappear without shaking for hours or warming past comfort. Real-world proof comes from labs using this salt in ATP regeneration, enzyme assays, or RNA synthesis. If it clumped at the bottom, none of these protocols would run smoothly. Reliable water solubility means no extra tricks to coax it into solution.
Why Solubility Matters to More Than Scientists
For anyone working on treatments for rare diseases, or those exploring mRNA technology, water solubility isn’t a nerdy detail. It streamlines dosing, keeps injected solutions safe for the bloodstream, and helps companies create uniform product batches. Solubility keeps waste low and results more predictable. One mistake with a poorly dissolving ingredient doubles costs and leaves projects stranded—resource mismanagement hurts both labs and patients.
Troubleshooting: When Things Go Wrong
Even with decent water solubility, problems sneak in. Old containers grow clumpy, or maybe lab water teems with minerals that interfere. New researchers sometimes dump in large amounts at once, overwhelming the water. In my experience, using fresh, room-temperature deionized water and adding small increments of the salt prevents issues. Stirring steadily, not furiously, works better for getting everything mixed in. Store containers tightly closed and away from humidity to keep powders from absorbing water before actual use.
Quality Counts
Not all uridine triphosphate trisodium salts are equal. Some suppliers offer products with extra stabilizers or higher purity—less risk of strange residues at the bottom of your tube. Many universities and pharma labs vet vendors by running purity tests before purchase, which pays off in the long run.
Demand for Reliable Research Materials
The world needs honest talk about ingredients like uridine triphosphate trisodium salt. Scientific research doesn’t happen in a vacuum. Supply chain disruptions, poor-quality batches, and mislabelled products slow progress. Regular checks and transparent documentation from suppliers give researchers confidence. Labs sharing their favorite techniques for dissolving, storing, and testing these chemicals raise the bar for everyone.
Seeking Solutions
Better storage, more open communication, and tighter controls on material quality serve everyone who relies on these biochemicals. Standard documentation and easy-to-read safety data help both newcomers and seasoned researchers. Connecting researchers with suppliers producing reliable and pure uridine triphosphate trisodium salt keeps projects on track and discoveries moving forward.
What is the purity specification of Uridine Triphosphate Trisodium Salt?
Why Purity Levels Matter in the Lab
Researchers know the drill. You run a reaction, you expect a result. It’s the idea that keeps students up late and scientists coming back for more experiments. But when a product like uridine triphosphate trisodium salt (often called UTP-Na3) lands in the lab with questionable purity, headaches follow. Purity isn’t a trivial checkbox—it’s a signal of whether data can be trusted or if resources will disappear chasing down contaminants. In fields like molecular biology and pharmaceutical development, inconsistent purity means lost research hours and unreliable results.
Defining a Real-World Specification
Lab teams usually look for a purity level of 95% or higher in UTP-Na3. The best suppliers deliver upwards of 98% assayed by HPLC, with only trace amounts of water, sodium chloride, and other nucleotides left behind. People buying this stuff for enzyme assays, mRNA synthesis, or signal transduction work rely on these tight margins. If the purity dips, competing nucleotides can throw off results. Even a small spike in chloride or sulfate levels can mess with the outcome.
Experienced suppliers post certificates of analysis for every lot, not just a single product sheet on a website. They show heavy metal levels, report bacterial endotoxin counts, and specify residual solvents well below legal limits. Labs appreciate this transparency—trust builds on it. Rigorous companies pull lots from shelves that fail testing, no excuses.
What Quality Control Actually Looks Like
Quality speaks through data, not platitudes. UTP-Na3 should present as a white or off-white powder, soluble and stable at ambient conditions. HPLC analysis should show a single peak corresponding to the compound, with UV absorbance matching theoretical expectations at 262 nm. Sodium content stays within a narrow range, and a pH check in aqueous solution falls near neutral. People who’ve run low-quality samples through sensitive assays can tell heartbreaking stories of signal loss or unexplained background. Reliable purity keeps those stories rare.
Impurity profiles matter, too. Modern labs can pick up impurities below 0.1%. Endotoxin testing stands especially important in RNA-related work, given how sensitive many of those downstream applications are. Often, labs specify less than 0.1 EU/mg. Those who’ve seen results spoiled by contamination never want to repeat that experience.
What Happens Without Enough Purity?
Research isn’t forgiving with shortcuts. Impure UTP-Na3 brings risks no one wants—batch-to-batch result swings, misinterpreted kinetic data, and even safety problems for people handling unpredictable chemicals. In drug development, strict regulators demand clear proof of purity before any clinical work or patient samples.
Seeing suppliers cut corners with shoddy paperwork or vague purity ranges sets off alarm bells. Sourcing from a company with third-party verification and open, thorough documentation changes everything. It saves money in the long run because experiments produce actionable data, not question marks or do-overs.
How to Guard Against Low-Grade Product
Solid purchasing relies on more than brand names. Check for batch testing, ask for specific impurity lists, and push for supplier responses to technical questions. A good lab doesn’t take “trust us” as gospel—they get the test data. When user communities swap notes on chemical quality, word gets around about who meets specifications and who cuts corners.
Labs that keep copies of certificates, record expiration dates, and stay alert for product recalls protect their credibility—and their results. In my own time organizing materials for biochemistry labs, I learned fast that cutting costs by skimping on purity almost never pays off. You pay double later, sometimes in lost weeks or bad science.
How should Uridine Triphosphate Trisodium Salt be handled and disposed of safely?
The Reality of Laboratory Chemicals
Uridine Triphosphate Trisodium Salt crops up in more research labs than you might imagine—especially in life science settings looking at cellular metabolism, mRNA synthesis, or enzyme reactions. Like most specialty chemicals, it comes with a set of safety expectations. Too many times, people rush to open new bottles or handle powders without reviewing the particulars. My old lab had a whole shelf devoted to nucleotides, and even experienced grad students sometimes skipped past the safety data just to get to the fun part. More than once, this casual approach led to headaches—figuratively, thankfully, and literally, if the air got dusty.
Personal Protection: No Substitute for Basics
Diving into a tube of uridine triphosphate without proper gear invites trouble. Lab coats, gloves—preferably nitrile or latex—and safety goggles matter. This compound doesn’t smell or sting like acid, yet dry powders can irritate skin or eyes. The dust lingers in the air if you’re hasty, and nobody wants to sneeze in their mask afterward. Ventilation stands as your silent ally; chemical fume hoods do more than keep up appearances for audits—they pull floating powder and vapor away before there’s a problem. I learned this lesson early on after watching a buddy spill nucleotide powder on the bench; nobody felt sick, but cleaning it all up with gloves and towels took twice as long as it should have.
Careful Storage Keeps Labs Safer
Leaving uridine triphosphate near heat, sunlight, or open containers creates risk where none belongs. This salt prefers cold, dry, and dark places, usually in clearly labeled containers tight enough to keep out moisture and dust. Any time a chemical sits where it can get knocked over, the odds of an accident go up. I once saw a jar left too close to a hot plate in a summer course; a careless hand almost melted the label off. Keeping chemical stocks on dedicated shelves, separated by hazard category, stops small spills from turning into cleanup drills.
Spill Response Relies on Preparation
Spills of uridine triphosphate demand quick thinking, not panic. Anyone nearby needs to know the plan—close off the area, put on fresh gloves and eye protection, and use damp towels or spill kits to limit powder spread. Sweeping up chemicals only pushes dust into the air; wet methods keep the particles from drifting. Everything picked up—towels, gloves, bits of filter—goes into a dedicated waste container. If something bigger happens or there's risk to breathing or water sources, alerting environmental safety teams right away beats the alternative of silently hoping for the best.
No Shortcuts with Chemical Waste
Proper disposal keeps hazardous waste out of drains, bins, or regular trash. Pouring leftover uridine triphosphate solution down the sink seems quick, but it invites environmental headaches and legal fines. Most research institutions have strict protocols: labeled chemical waste bottles, filled and logged, and routine pickup for disposal. Sodium salts from these nucleotides don’t break down fast, so water treatment systems can’t always catch everything. Following local regulations protects staff and neighbors—and keeps the lab running without unscheduled inspections.
Accountability Through Training
Science moves quickly, but safety should never fall behind. Regular training and open conversations about chemical hazards foster a culture where new and seasoned researchers support each other. In my experience, questions about handling or disposing of uridine triphosphate—no matter how basic—signal a team that takes stewardship seriously. Respect grows from shared experience, not just printed rules. Peer reminders, posted instructions, and walk-throughs together help cement best practices much better than email bulletins or paperwork.
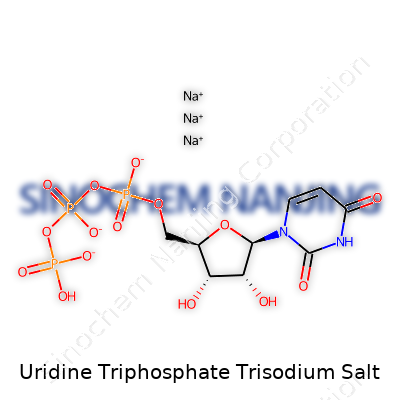
| Names | |
| Preferred IUPAC name | trisodium [(2R,3S,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl triphosphate |
| Other names |
Uridine 5′-triphosphate trisodium salt UTP trisodium salt Uridine triphosphate sodium salt Uridine 5′-triphosphate sodium salt |
| Pronunciation | /ˈjʊər.ɪˌdiːn traɪˈfɒs.feɪt traɪˈsoʊ.di.əm sɔːlt/ |
| Identifiers | |
| CAS Number | 19817-92-6 |
| Beilstein Reference | 82263 |
| ChEBI | CHEBI:61430 |
| ChEMBL | CHEMBL1235561 |
| ChemSpider | 21606619 |
| DrugBank | DB02631 |
| ECHA InfoCard | 22-211-960-379-48 |
| EC Number | 205-162-5 |
| Gmelin Reference | 82118 |
| KEGG | C00075 |
| MeSH | D000073666 |
| PubChem CID | 65109 |
| RTECS number | YQ7875000 |
| UNII | 8N287T0399 |
| UN number | Not regulated |
| CompTox Dashboard (EPA) | urn:uuid:75cfd132-686f-47e3-9381-04e6ac849ce3 |
| Properties | |
| Chemical formula | C9H11N2Na3O15P3 |
| Molar mass | 788.17 g/mol |
| Appearance | White to off-white powder |
| Odor | Odorless |
| Density | 1.45 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -4.3 |
| Acidity (pKa) | 2.8 (first phosphate group) |
| Basicity (pKb) | 21.34 |
| Viscosity | Viscous liquid |
| Dipole moment | 6.8 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 365.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | M01AX28 |
| Hazards | |
| Main hazards | Suspected of causing genetic defects. |
| GHS labelling | GHS labelling: "Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008. |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Not a hazardous substance or mixture according to the Globally Harmonized System (GHS) |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | Health: 1, Flammability: 0, Instability: 0, Special: - |
| LD50 (median dose) | LD50 (Median dose): >2000 mg/kg (oral, rat) |
| NIOSH | Not listed. |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Uridine Triphosphate Trisodium Salt: Not established |
| REL (Recommended) | 0.03 mg/m3 |
| Related compounds | |
| Related compounds |
Uridine Uridine monophosphate (UMP) Uridine diphosphate (UDP) Adenosine triphosphate (ATP) Cytidine triphosphate (CTP) Guanosine triphosphate (GTP) |

