Into the World of Tris(2-Methylaziridinyl)Phosphine Oxide: Past, Present, and a Cautious Future
Peering Back: Historical Development
People started paying attention to Tris(2-Methylaziridinyl)Phosphine Oxide when the race for new chemical agents reached its peak in the twentieth century. Anyone familiar with the intersection of chemistry and defense recognizes this molecule. It didn’t spring fully formed from the mind of a single scientist; its emergence grew out of years packed with experimentation, geopolitical tension, and trial-and-error synthesis. Researchers in the late 1950s and 1960s, riding the wave of new synthetic techniques, pulled together aziridine chemistry and phosphorus oxides. The result—what’s more commonly known in some circles as “Methyl-TEPA”—stood out for its peculiar ability to cross-link with biological molecules, something you just don’t see in most phosphorus compounds. History has shown, again and again, that once a synthetic route is found for a reactive compound, industries and research fields move fast, often leaving ethical questions to chase behind.
Product Overview: Understanding a Rarely-Discussed Compound
Tris(2-Methylaziridinyl)Phosphine Oxide rarely makes headlines outside of specialized circles. At a glance, it looks like a dense, white, crystalline powder. The structure holds three aziridine rings attached to a central phosphine oxide, making it jump out in terms of reactivity compared to typical organic molecules. Practically, most people never encounter it on purpose—it's not sold over the counter or found in consumer products. Its use shows up in labs running advanced organic syntheses and sometimes in defense research. The complexity of its structure, especially with those aziridine rings, means it packs more punch in reactivity than your garden-variety chemical.
Physical & Chemical Properties
What sets this compound apart is its volatility and unique reactivity profile. The molecule holds a certain stability thanks to the phosphine oxide backbone, but those methylaziridine groups open doors to dangerous and surprising reactions. In solid form, it melts at a relatively moderate temperature and dissolves in polar organic solvents such as dimethyl sulfoxide and acetone. This solubility boosts its usefulness in organic chemistry settings, especially where aggressive or targeted cross-linking comes into play. But these same features, useful as they are, demand serious respect for safety. Its reactivity with nucleophiles brings rapid transformation, meaning a slip in lab procedure can quickly turn risky. Even seasoned chemists know to treat every gram with the same care as explosives or strong acids.
Technical Specs & Product Labeling
Most chemical suppliers label this molecule with the CAS number 57-39-6 and often list its formula as C9H18N3OP. Labels highlight its high toxicity and warn about corrosive and mutagenic traits. Guides call for storage in tightly sealed containers, kept cool and away from moisture or incompatible reagents like strong acids or oxidizers. Regulations usually put limits on the handling, requiring full face shields, gloves, and fume hoods as a minimum standard. Nobody in the field talks about this compound without covering the technical specs—these numbers aren’t just admin requirements, they serve as strict warnings to those who know what substances like this can do.
Preparation Method: How It’s Made in the Real World
Synthesis of Tris(2-Methylaziridinyl)Phosphine Oxide doesn’t appeal to amateurs. Professionals rely on the controlled reaction of phosphorus oxychloride with 2-methylaziridine, typically in an anhydrous solvent. The entire process gives off hazardous byproducts, and slip-ups have led to severe injuries in poorly equipped labs. Each step—adding aziridine, maintaining low temperatures, handling toxic fumes—demands oversight and almost ritualistic attention. Long-time chemists warn that shortcuts end badly. Quality check follows at each turn, not just to confirm purity but to be sure no side-products linger, since a contaminated batch may turn an already toxic reagent into something even more unpredictable.
Chemical Reactions & Modifications
Tris(2-Methylaziridinyl)Phosphine Oxide reacts briskly with nucleophilic reagents, which makes it valuable for cross-linking DNA and proteins. This feature has kept it on the radar for both biochemical research and, less openly, for weaponization studies. The reactive aziridine rings undergo ring-opening reactions, leading to alkylation of target substrates. Chemists have modified the molecule to tweak its solubility or reactivity, sometimes by swapping in alternative aziridine derivatives or adjusting the central phosphorus atom. Every change brings a new angle—a fresh take for use in technical or research contexts. But each modification carries familiar risks, meaning new analogues of this molecule stay locked in tightly-controlled lab settings.
Synonyms & Product Names
You’ll find this product under a handful of names. Besides Tris(2-Methylaziridinyl)Phosphine Oxide, the compound appears as Methyl-TEPA, abbreviated TMAPO, and less often as NSC 38721 in some scientific catalogs. Literature from decades past shows a variety of alternative spellings and nomenclature, reflecting changing naming conventions in chemical circles. Anyone researching older datasets or archived papers has to keep those synonyms in mind, or else risk missing important context about safety studies or reported accidents.
Safety & Operational Standards
Strict safety standards shape how anyone handles this molecule. Even seasoned chemists don anti-static clothing, full face protection, double-layered nitrile gloves, and rely on negative-pressure fume hoods. Spill kits and eye-wash stations aren’t decorations; they stand ready because laboratory accidents, once rare, have ended careers—or lives. Regulatory guidance pushes for closed system equipment and double verification of disposal methods. Any waste, even in trace amounts, heads straight to incineration in specialized facilities. I’ve seen firsthand how routine can breed complacency, and in compounds like Tris(2-Methylaziridinyl)Phosphine Oxide, even small mistakes snowball fast. Lab culture in reputable institutions refuses to treat this as “just another hazardous chemical.”
Where It Appears: Application Areas
Outside of the cloistered world of defense research, Tris(2-Methylaziridinyl)Phosphine Oxide rarely leaves secure laboratories. Its most significant application lies in cross-linking biomolecules, particularly in the study of DNA replication, cancer therapies, and enzyme modification. These uses aren’t casual experiments; the people working with the compound want to understand or control the most fundamental life processes. Governments once explored its potential in chemical munitions, drawn by the severe biological disruptiveness, but today most international agreements shut that door tight. Industrial-scale usage remains limited thanks to its safety profile and the costs tied to proper handling and disposal. Academic research still returns to it for its rare ability to produce specific modifications in nucleic acids or proteins—something that can’t easily be done with safer alternatives.
Research & Development: Pushing and Respecting Limits
Research involving Tris(2-Methylaziridinyl)Phosphine Oxide often runs up against ethical walls before it hits scientific ones. Early studies in oncology held promise, showing the molecule could disrupt rapidly dividing cells. This led to some clinical investigations, though the compound’s non-selective toxicity doomed large-scale therapeutic use. Decades of work have traced countless analogues, each chasing the grail of selective action with lower health risks. Many research teams with decades in chemical biology have left this compound behind, favoring targeted therapies that spare normal tissue. Yet, new analytical methods and bioengineering tools sometimes bring it back into focus as a benchmarking reagent or a way to probe just how far chemical modification of biopolymers can reach.
Toxicity Research: A Long List of Red Flags
Researchers quickly learned that Tris(2-Methylaziridinyl)Phosphine Oxide does not make for safe handling. Biological studies paint a clear picture: direct alkylation of nucleic acids leads to rapid cell death. Short exposures produce sharp toxicity, including mutagenic and carcinogenic effects. Regulatory agencies classify it among substances that can cause irreversible health effects, demanding controlled access and rigorous record-keeping. Real-world cases—such as accidental exposures in poorly managed settings—show everything from dermatitis to severe systemic failure. Toxicology investigations drove many early-stage therapies into the bin, overshadowed by the universal demand for worker and patient safety. It’s a tough truth in chemistry that the most powerful compounds make the loudest alarm bells ring.
Future Prospects: Where We Go Next
Most signs point toward a shrinking role for Tris(2-Methylaziridinyl)Phosphine Oxide outside niche biochemical research. Scrutiny from both regulators and the scientific community keeps tightening. Alternatives keep multiplying, offering lower toxicity with similar molecular effects. Still, there remains a stretch of terrain where its reactivity outpaces everything else. Specialized teams—especially in high-containment academic and government labs—continue picking through its reaction chemistry for answers about DNA behavior or protein engineering. Long-term, I expect the balance will tip further toward phasing it out except in scenarios where no better option exists. Increased emphasis on green chemistry and the steady rise of safer, more selective synthetic agents promise a day when people look back at compounds like this and wonder how anyone managed such risk for so long.
What is Tris(2-Methylaziridinyl)Phosphine Oxide used for?
The Realities Behind a High-Stakes Chemical
Tris(2-Methylaziridinyl)Phosphine Oxide, often shortened as TEPA or sometimes called “Thiotepa’s cousin,” brings up serious reactions from chemists and policy watchdogs alike. The compound caught my attention years ago during a late-night college discussion on chemical weapons control. Its formula ties into a complicated history and a future still uncertain, linking the worlds of science, ethics, and security.
Applications in the Real World
TEPA’s story leans heavily on its power as a so-called cross-linking agent. In laboratories, researchers have used it to alter DNA or create new synthetic materials. The medicine world flirted with compounds of this nature for chemotherapy uses. Unlike everyday household chemicals, TEPA’s use stretches beyond cleaning or preservation. We see it touched most often in high-stakes research, not in mass-market products.
But that’s not why the name stirs up so much caution. TEPA belongs to a group of substances flagged by major international military watchdogs, including those monitoring the Chemical Weapons Convention. The compound played a part—at least on paper—in nerve agent development. Its ability to damage genetic material is what sparked interest from military labs.
The Shadows Over TEPA
Every time I see headlines about nerve gas stockpiles, my mind jumps to the hidden threat chemicals like TEPA pose. Manufacturing it requires skill and care that only specialized facilities can provide, but once produced, its danger blooms. Stories from my uncle, an industrial safety officer, highlight why tiny leaks or mishandling can lead to disaster.
In the wrong hands, this chemical is nothing short of terrifying. Accidental exposure risk sits high, given how it penetrates skin and disrupts biological systems. Protective suits and controlled ventilation are the norm in facilities handling it. Even a slight misstep leads to serious consequences—not just for the chemists, but for communities near production or storage sites.
Why Should We Care?
Beyond its specific place in chemical research and security policy, TEPA stands as a sharp reminder of the constant balancing act between innovation and responsibility. There’s no question about its value in scientific discovery, especially for pushing forward genetic engineering and cancer treatment studies. But every step forward comes with a shadow: regulatory loopholes, black market interest, and lapses in oversight.
For those of us not stockpiling chemical agents, the lesson comes down to vigilance and the weight of trust we place in our regulatory system. Governments do track and control substances like TEPA—at least in theory. Yet we’ve all seen cases, from Bhopal to more recent chemical accidents, where “in theory” failed real people.
Real Solutions, Real Responsibility
To move forward safely, we need transparency and enforced oversight from both public agencies and the private sector. Public records show some progress, like stricter licensing and inventory audits. My own experience with academic labs shows how peer review and whistleblower protections can help spot problems before they erupt. No solution fits every situation. We demand constant review, better detection technology, and above all, a culture that values safety over shortcuts.
The story of TEPA hits close to home for anyone who cares about what powers modern science—and what keeps us secure. Unchecked attention on breakthrough chemistry comes with a cost that society can’t ignore.
What are the safety precautions when handling Tris(2-Methylaziridinyl)Phosphine Oxide?
Respect the Hazards
Many folks who spend time in labs, like me, know that certain chemicals demand a kind of respect that comes only with stories told in break rooms and scars under sleeves. Tris(2-Methylaziridinyl)Phosphine Oxide is one of those compounds. This isn’t the sort of material anyone should keep on a shelf without clear labels or strict protocol. It has a dangerous bite, known for its toxicity and potential to mess with human DNA. Anyone who handles it—researchers, students, or seasoned pros—needs to pay close attention, or face consequences much worse than wasted time.
The Essentials: Gloves and More
Nobody forgets to grab gloves twice around this chemical. Standard latex won’t cut it. Nitrile or neoprene stand up better in this case, backed by eye protection that actually seals. Too many incidents come down to someone trusting safety glasses with open sides; a full face shield, rated for splash, pairs well with a proper laboratory coat. The route in isn’t always through the skin—this stuff vaporizes at a surprisingly low temperature, which means a good fume hood isn’t optional. Good labs keep emergency showers and eyewashes no more than ten seconds away, and nobody scoffs at a practice drill after seeing the nasty reactions in real cleanup scenarios.
Reactivity and Storage
Trying to store this chemical in the same spot as strong acids, bases, or oxidizers could turn a normal day hectic. A dry, cool, locked cabinet works best, away from sunlight and well-marked on every side. Anyone with a story about surprise reactions involving aziridines knows the value of double-checking storage plans and logging every transfer. Keeping inventory tight can prevent surprises during annual cleanouts—a nasty bottle at the back of a forgotten shelf almost guarantees trouble.
Spill Response: Not Just for Newbies
The first reaction to a spill often determines the outcome. Waiting to “see if it evaporates” doesn’t cut it. I saw a close call where someone assumed a small amount wasn’t a problem, leading to panic later. Labs I trust instruct staff to evacuate small spill zones, lock doors, call emergency teams, and never enter until the air readings clear. Absorbent materials soaked with Tris(2-Methylaziridinyl)Phosphine Oxide need labeling before disposal and immediate removal to a dedicated waste area—not left with regular trash.
Medical Monitoring and Training
Places that put safety above speed schedule regular training seminars, including updated risk information from agencies like OSHA and the CDC. Personal exposure isn’t always obvious at first. Colleagues who work regularly with toxics submit to periodic medical exams and blood tests, comparing results over time. This proactive approach has caught early warning signs and saved careers, if not lives. Posters and SOPs only work if someone actually reads them; supervisors share real incidents to keep procedures fresh in everyone’s mind.
Building a Responsible Culture
Institutions that make safety a habit—rather than a checkmark—tend to avoid headlines and emergency room visits. I still remember an older technician who stopped a newcomer from reaching for a partially-labeled container, explaining calmly that one mistake could mean the end of a project or permanent harm. Clear communication, honest admission of unknowns, and a willingness to call out risks are worth more than any piece of personal protective equipment. In my view, building a trustworthy team culture means taking care of one another long before opening a bottle of something this tough.
What is the chemical structure and molecular formula of Tris(2-Methylaziridinyl)Phosphine Oxide?
Inside the Chemical Code
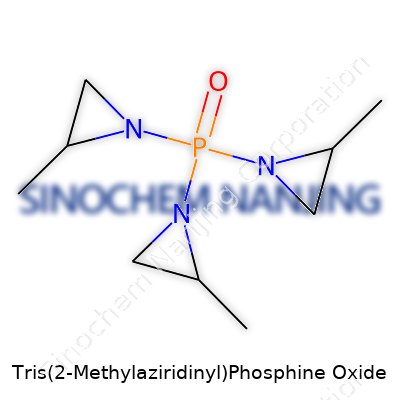
Tris(2-Methylaziridinyl)phosphine oxide has a name that feels like it belongs in a comic book, but it’s a real molecule with a punch in chemistry. The molecular formula, C9H21N3OP, captures its make-up in numbers and letters: nine carbons, twenty-one hydrogens, three nitrogens, one phosphorus, and one oxygen. Graphing its structure, each aziridine ring carries a methyl group on the second carbon. The phosphorus atom takes the center seat, double-bonded to an oxygen, with three arms, each clutching a 2-methylaziridine ring.
Why Structure Matters
In my lab days, structures translated to consequences. The three-membered aziridine rings pack significant strain, turning them into reactive hotspots. Phosphorus holding the rings together creates an interesting dynamic: the molecule doesn’t just float through reactions passively. In fact, those aziridine rings can swing open under the right conditions, engaging eagerly in cross-linking processes, which drew attention in the search for cancer drugs, chemical warfare agents, and polymer cross-linkers.
Holding this molecule up to the light, you notice the symmetry, but those rings are easily disrupted. This instability pushes chemists to respect its power and risk. The oxide on phosphorus pumps up polarity, boosting water solubility, and guides how the compound travels inside living systems or reacts in industrial uses. If you tasted the pressure of handling reactive organics in a chemistry lab, you’d know how quickly a structure like this can turn expectations sideways.
Industry and Health Risks
History writes this compound’s story on a rough page. Scientists found its knack for cross-linking DNA wasn’t limited to test tubes. The ability to mangle genetic material led to research on using it in anticancer drugs. That promise shadowed over fast. People making or handling Tris(2-methylaziridinyl)phosphine oxide paid a high price: those aziridine rings attack biological tissue, and studies found the compound mutagenic and carcinogenic. It walked into notorious company as an ingredient in chemical warfare agents.
Nothing hits home like safety data sheets filled with bold symbols and scarlet warnings. In real labs, strict controls, fume hoods on full throttle, and thick gloves become the minimum. Chemical companies walked away from this molecule for commercial applications. The safety risk eclipsed the rewards. Long-term, the only responsible way to treat these molecules is with zero shortcuts: protective gear, dedicated containment, and thorough training on spills and exposure.
Pushing Toward Safer Alternatives
Chemistry keeps moving forward, always under the shadow of what’s come before. Whenever a molecule like this crosses your path, the lesson is clear: every structural twist and turn holds consequences well beyond the lab bench. Smart science means learning from the data. New crosslinking agents now steer away from aziridine rings or tweak them to hold less danger. On the regulatory side, tighter approval standards block harmful agents from consumer or medical use. It comes down to understanding what the blueprint of a molecule tells us about its risks and then applying real discipline and creativity to dodge those hazards.
How should Tris(2-Methylaziridinyl)Phosphine Oxide be stored?
It’s Not Just Another Lab Chemical
Tris(2-Methylaziridinyl)Phosphine Oxide often pops up in research discussions about specialty chemicals. I learned early on in my lab days that treating every substance like a bag of sugar is a recipe for disaster. This molecule fits right into that hard-earned lesson. It’s known for high reactivity and toxicity, so casual storage routines just don’t cut it.
Why Extra Caution Is Worth It
No one in the lab wants a mess on their hands. Spills or leaks with this compound create some of the nastiest cleanup jobs, not to mention health risks. Its aziridine groups look harmless on paper but can set off chemical reactions with water, acids, and open air. Wrap your head around the fact that it causes mutagenic and carcinogenic effects, and the stakes get real. Labs aiming for a serious safety record never treat this compound lightly.
Basic Storage Practices Mean Nothing Without Consistency
Keep the container sealed shut unless you’re weighing or transferring. Even the best ventilation systems won’t make up for leaky lids or casual placement near a window. Strong, airtight glass bottles with Teflon-lined caps make a world of difference. Metal and basic plastic containers just don’t stand the test—they break down or corrode after contact, releasing fumes nobody wants to inhale. Every experienced chemist I know prefers clearly labeled, high-grade packaging to avoid the all-too-common ‘who left this open?’ horror show.
Temperature and Light Are the Real Enemies
Room temperature tends to speed up breakdown and reaction risk, so refrigeration matters. I once watched an overworked tech stash a bottle on a random shelf, only to catch high humidity in the air after a storm. Minutes later, the bottle showed signs of swelling. This chemical lasts longer and stays less reactive in the 2–8°C range, and direct sunlight is always a fast track to decomposition. Darkness isn’t negotiable; even short bursts of UV light can spark problems.
Controlling Access Matters for Peace of Mind
No one likes keys, but with these reagents, locked chemical cabinets have saved countless headaches. My own lab saw fewer accidents after upgrading storage to specialty cabinets with proper warning signs and restricted entry. Safety officers often perform random checks, and honestly, every lab worth its salt encourages this. Audits catch mistakes before they snowball. Overlooking this means anyone could grab the wrong bottle for the wrong experiment, putting health on the line.
Don’t Forget About Spill Response Plans
It shocked me to learn that some places only address procedures after an accident. Real responsibility means keeping spill kits close and updated. Activated carbon, acid scavengers, and full-face respirators should sit within arm’s reach. Drills aren’t a boring formality: in an emergency, just having the kit won’t help if no one knows how to use it properly.
The Bottom Line for a Safe Lab
Most of the horror stories I’ve heard started with tiny lapses. Small steps—better containment, cold storage, clear labeling—mean less risk. In real life, skipping one of these is all it takes for things to go wrong. For anyone responsible for this chemical, respecting its dangers makes the difference between a safe workday and trouble nobody wants to deal with.
Is Tris(2-Methylaziridinyl)Phosphine Oxide hazardous or toxic?
Getting Curious About Chemical Safety
Anyone working around specialty chemicals learns pretty fast that each one has its own personality. Put a handful of chemists in a room and sprinkle in a conversation about Tris(2-Methylaziridinyl)Phosphine Oxide—often called TEPA or THA—and you’ll see some eyebrows go up. The name alone sounds like a warning label, and for good reason. Here’s what actually matters when talking about the hazards and toxic risks related to it.
The Toxic Face of Tris(2-Methylaziridinyl)Phosphine Oxide
I’ve spent time in research labs where even the rumor of a mutagenic chemical put the whole team on alert. Tris(2-Methylaziridinyl)Phosphine Oxide has a reputation among folks who know carcinogens and mutagens aren’t just technical labels—they’re flags telling us to slow down and take extra care. Animal testing in the 1970s and 1980s revealed this compound can cause mutations in genetic material. That’s not just a detail for lab rats. Mutagenic chemicals often tie into stories of cancer, birth defects, and chronic illness for humans.
One big reason scientists pay such attention comes down to how the aziridine group reacts. Aziridines punch above their weight—causing changes to DNA, sometimes with tiny exposures. Regulatory agencies use that data to stay alert and keep workers protected. The U.S. National Toxicology Program and the International Agency for Research on Cancer include chemicals with strong mutagenic evidence in their lists of substances to handle with respect and robust safety controls.
Where Exposure Risks Rise
Most people never run into Tris(2-Methylaziridinyl)Phosphine Oxide off the street. This chemical mainly shows up in controlled work settings, where it gets used to make other products or as a cross-linking agent for polymers. Danger climbs in labs where safety goggles fog up and gloves become questionable after hours of experiments. In real life, I’ve seen new lab members miss a droplet on their gloves and wonder, hours later, about a skin itch.
Direct skin contact or breathing in dust from TEPA gives it an open door to cause trouble. Respiratory health takes a hit, skin can burn or develop sores, and in some reported accidents, workers suffered from headaches, nausea, and fatigue. Chronic exposure delivers the higher risks—accumulation in the body opens the page for long-term conditions tied to toxicity.
Choosing Straightforward Safety Steps
The most effective way to minimize trouble with this substance always comes back to what we can control: personal protective equipment, solid engineering controls, and clear training. In labs where I’ve worked, spill kits sat within reach, and staff ran drills like students prepping for a fire alarm. Proper fume hoods, air filtration, and locked storage became part of daily routines.
It pays to read the fine print on safety data sheets. Anyone handling Tris(2-Methylaziridinyl)Phosphine Oxide should use impermeable gloves, face protection, and disposable lab coats. Keeping hazardous chemicals sealed, labeling them honestly, and never cutting corners on hygiene measures work better than hoping luck stays on your side.
Managers have a duty to set the example. Fixing poor ventilation or slow chemical disposal isn’t just about compliance—it means looking after real people. Co-workers talk, and what gets shared between shifts can help someone avoid a bad exposure.
Informed Choices Matter
Trust and transparency smooth the path forward. Having a workplace culture where folks respect the risks leads to safer labs and fewer problems. Being serious about training and documentation, cutting out shortcuts with hazardous chemicals, and focusing on proper disposal habits protects everyone.
| Names | |
| Preferred IUPAC name | Tris(1-methylaziridin-2-yl)phosphine oxide |
| Other names |
Thiotepa TSPA Tris(1-aziridinyl)phosphine oxide Tris(2-methylaziridinyl)phosphine oxide Tris(2-methyl-1-aziridinyl)phosphine oxide |
| Pronunciation | /ˈtrɪs tuː ˌmɛθɪl.əˈzɪrɪdɪnɪl ˈfɒsfɪn ˈɒksaɪd/ |
| Identifiers | |
| CAS Number | 5455-55-1 |
| Beilstein Reference | 1737704 |
| ChEBI | CHEBI:38919 |
| ChEMBL | CHEMBL2103836 |
| ChemSpider | 14214 |
| DrugBank | DB04951 |
| ECHA InfoCard | 13d1b0b3-7a5a-43f8-a2ea-dc225d0e074a |
| EC Number | 216-172-5 |
| Gmelin Reference | 122021 |
| KEGG | C14366 |
| MeSH | D014277 |
| PubChem CID | 66707 |
| RTECS number | TN6140000 |
| UNII | UF86VWI0N6 |
| UN number | 2810 |
| CompTox Dashboard (EPA) | DTXSID5036794 |
| Properties | |
| Chemical formula | C9H21N3OP |
| Molar mass | 221.23 g/mol |
| Appearance | White crystalline solid |
| Odor | Amine-like |
| Density | 1.14 g/cm3 |
| Solubility in water | Soluble |
| log P | 0.22 |
| Vapor pressure | Vapor pressure: <0.01 mm Hg (20 °C) |
| Acidity (pKa) | 13.2 |
| Basicity (pKb) | 6.73 |
| Magnetic susceptibility (χ) | -74×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.497 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.77 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 338.6 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4748 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | L01AC02 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes severe skin burns and eye damage, may cause an allergic skin reaction, harmful if inhaled. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P280, P304+P340, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 4-3-2-W |
| Autoignition temperature | 280 °C |
| Lethal dose or concentration | LD50 oral (rat): 2 mg/kg |
| LD50 (median dose) | 1 mg/kg (intravenous, mouse) |
| NIOSH | TT4110000 |
| PEL (Permissible) | PEL: 0.01 mg/m³ |
| REL (Recommended) | REL (Recommended): Ca (NIOSH Potential Occupational Carcinogen) |
| IDLH (Immediate danger) | 1 mg/m3 |
| Related compounds | |
| Related compounds |
Hexamethylphosphoramide Tris(2-chloroethyl)phosphate Tris(2-chloroethyl)phosphine oxide |

