Tris(1-Aziridinyl)Phosphine Oxide: Looking Beyond the Lab Bench
Historical Footsteps: A Compound Forged in Urgent Times
Back in the 1950s, as the world shifted from conventional warfare to the uncertain threats of the Cold War era, chemical research saw a wave of rapid innovation. Scientists didn’t work in isolation; their breakthroughs often came under military contracts. Tris(1-Aziridinyl)phosphine oxide, often referred to by its acronym TEPA or its code, found a niche not in the shelves of supermarkets, but quietly in military laboratories and specialized industrial spaces. Early studies placed it among a class of aziridine-based alkylating agents, with roots in the research for chemotherapy drugs and chemical weapon candidates. The world then looked for compounds that could damage DNA, sometimes to attack tumors, other times for more sinister uses.
Product in Focus: Little Known, Yet Highly Potent
On the surface, TEPA appears as a crystalline solid, usually white or off-white, depending on its purity and storage history. Its molecular backbone—a phosphorus atom snug between three aziridine rings and topped with an oxygen atom—gives it a reactivity that few chemicals match. Unlike household names in chemistry, such as sodium chloride or ethanol, TEPA keeps its reputation within the confines of research journals, patents, and the occasional government archive. It's rarely seen outside of research or specialized production sites, and for good reason. Its properties don’t lend themselves to casual use.
Tangible Characteristics: Weight That Matters
The molecule holds a formula of C6H12N3OP and tips the scale at a molar mass around 173 grams per mole. It melts at relatively low temperatures, enough that a warm room might soften it, and it stands up to air, though it doesn’t play nice with water or acids. TEPA’s three-membered aziridine rings put strain on the molecule, making it eager to react—a quality that’s both a blessing for synthetic chemists and a hazard for anyone who handles it carelessly. I’ve read about its use in organic synthesis, where those strained rings pop open and react with biological molecules, industrial plastics, or pharmaceuticals.
Labels Speak Volumes: Technical Specs Are More Than Numbers
Labeling for a chemical like TEPA means more than meeting regulatory demands. It often supports critical safety messaging. Containers—usually amber glass—warn of its reactivity, volatility, and toxic risk. This isn’t just for show: the compound can alkylate DNA, the sort of activity that turns a useful reagent into a dangerous poison. Certification calls for 98% or higher purity in analytical work, leaving impurities only in trace amounts, at least as far as reputable sources are concerned.
Building the Molecule: Preparation That Demands Respect
Every synthesis route for TEPA starts with a phosphine or phosphine oxide core and aziridine subunits. The classic method involves nucleophilic substitution where aziridine rings displace halide groups on a phosphorus precursor. Early synthetic protocols called for anhydrous and inert conditions, since aziridines and phosphorus-based intermediates can react with water, oxygen, or even glass if left unattended. Chemists who work with this compound often tell stories about preparation runs that needed all hands on deck, with monitoring not just for reaction yield but for unexpected venting of gas or exothermic surges.
Chemical Reactions: A Broader Canvas
The triple aziridine rings on TEPA make it a prime target for functionalization. It reacts eagerly with nucleophiles, acids, and even mild oxidants, sometimes leading to chain-extended polymers, sometimes to highly cross-linked network materials. In biological experiments, TEPA becomes an alkylating agent—transfering pieces of itself to DNA and proteins, disrupting their normal function. I’ve known colleagues who used it for cross-linking in biochemical studies, each time stressing about handling risks, ventilation, and cleanup.
The Language of Naming: Synonyms Tell Their Own Story
TEPA travels under many names. “Tris(1-aziridinyl)phosphine oxide” remains the most scientific, but among researchers, it shows up as TEPA, ‘Phosphamide,’ or its old military label, “aphosphamide.” Each name crops up in different corners of the scientific literature, and this patchwork of naming is a sign of its unusual origins. Sometimes it’s lumped with nitrogen mustards in cancer research, other times it’s filed under cross-linking agents in materials science.
Safety Isn’t Optional: Hard Lessons from Risk
Handling TEPA means preparing for the worst. Lab protocols always command gloves, eye protection, fume hoods, and sometimes even full respirators. Personal stories from older chemists point to accidental exposures, with symptoms ranging from skin blisters to more severe, longer-term effects. Once, a smaller spill reached an unprotected hand and caused weeks of soreness and skin breakdown. Safety data sheets list TEPA as a proven mutagen and probable carcinogen, based on its DNA-alkylating activity. Waste disposal never happens through conventional channels; specialized chemical waste management usually takes charge, making sure nothing leaks downstream.
Out in the World: TEPA's Real Applications
TEPA’s main role lands in research, process chemistry, and carefully controlled medical studies. In the mid 20th century, it briefly figured as an anticancer agent, part of the same pantheon as nitrogen mustards and early alkylating drugs. Its strong cross-linking activity made it a candidate for probing DNA structure or for functionalizing laboratory polymers. Industrially, it sometimes appears in making specialty rubbers or toughening epoxy resins, where its reactive rings knit long chains together. That said, most major applications never went mainstream, stymied by toxicity and handling complexity.
A Field in Motion: Research and Development Ongoing
Even with its hazards, TEPA keeps a foot in the door of chemical research. Newer projects often look to soften its aggressive toxicity while keeping its powerful reactivity. Some research tracks focus on tethering the aziridine rings to less toxic scaffolds, aiming for targeted cross-linking in advanced polymers or biological systems. Others try to break it down safely after use, minimizing environmental risk and personnel exposure. Advances in laboratory monitoring help, but the compound’s intrinsic dangers mean every experiment needs thoughtful supervision and updated lab practices.
Toxicity: Hard Numbers and Hard Truths
TEPA doesn’t play fair with living tissues. Its mutagenic behavior stems from its trio of aziridine rings—each ready to snap open and latch onto DNA, causing breaks, crosslinks, and mutations. Animal studies from decades past set clear limits: small doses led to acute toxicity, while chronic exposure brought out carcinogenic effects. Cancer researchers watched promising cell-kill in tumors, only to see unacceptable side effects in normal cells. This intrinsic hazard closed the door on wide clinical adoption, leaving TEPA mostly in the hands of experts.
Mapping the Road Forward: Future Prospects in Sight
It’s easy to write off a compound like TEPA as a relic or chemical oddity. In truth, the future may hold new uses, as chemists chase safer alkylating agents or design controllable cross-linkers for smart materials. Tighter regulations and innovative engineering might help corral its dangers, making it a tool for very specialized settings. Advances in containment, waste treatment, and detection technologies could transform risks into manageable problems for research and industry alike. As always, success here depends not just on chemistry, but on thoughtful stewardship and an open dialogue about risk versus reward.
What is the chemical structure of Tris(1-Aziridinyl)Phosphine Oxide?
Breaking Down the Molecule
Tris(1-Aziridinyl)phosphine oxide grabs attention through its unique core: a phosphorus atom at the center, holding tight to three aziridine rings and a single oxygen. Chemists often draw this as OP(NC2H4)3. For those familiar with organic molecules, the three-membered aziridine ring adds a special strain and reactivity not common in larger, more relaxed ring structures.
What Sets the Structure Apart?
The phosphorus bonds with an oxygen through a double bond. Around it, aziridine rings attach directly to phosphorus with nitrogen atoms. Each ring forms a triangle with two carbons and one nitrogen. This geometry puts stress on the molecule at the atomic level—aziridine’s bond angles fall well below the usual, making the rings eager to react with other chemicals.
Throughout my time in academic labs, I’ve seen small rings like this go from stable in bottles to explosive during scale-up. The same tension makes it valuable for hard-to-pull-off reactions, especially in medicine and polymer chemistry. For example, these rings often make for strong cross-linkers or DNA-reactive probes in chemotherapy design.
Applications and Safety
Researchers prize tris(1-aziridinyl)phosphine oxide because its strained rings open up under the right conditions. This ability deepens its role in synthesizing cancer drugs. Chemotherapies sometimes use similar structures to link with DNA strands, disrupting tumor growth. Yet those same qualities demand respect—exposure can irritate skin, eyes, or airways and even cause mutations. All work happens behind fume hoods with gloves and constant vigilance.
Phosphorus-oxygen bonds give it water-resistance and chemical stability up to a point, but strong acids or heat can break the oxides and open the rings. Factory workers and researchers both need clear safety procedures, full training, and real emergency plans. Maybe the best solution for handling risk starts with education—hands-on demonstrations and visible reminders of what goes wrong with carelessness.
Quality, Sourcing, and Environmental Impact
Lab suppliers make this compound in small batches. They inspect purity with NMR spectroscopy and mass spectrometry. Trace impurities—halides, for example—can stop a whole synthesis or bring out unexpected side reactions. In academic research, the cost stacks up quickly, as only a few chemical suppliers ship it under strict regulations. Careful storage, secure documentation, and strict paperwork keep this reagent within responsible circles.
Environmental chemists continue to question what happens after use. Small molecules with reactive rings can linger and sometimes harm aquatic life. Improved waste collection, neutralization plans, and analysis of degradation products cut down risk at the source. Labs can push for greener solvents and reusable protective gear, shrinking the environmental footprint from the start.
Science and Society
Looking at tris(1-aziridinyl)phosphine oxide, the structure itself unlocks a range of chemical and biological capabilities. Medical science has benefited from this molecular tool, yet it brings challenges around safety and waste. Better education, responsible sourcing, and solid lab practices carve a path forward—keeping science moving without losing sight of the risks built into the very bonds of the molecule.
What are the main applications or uses of Tris(1-Aziridinyl)Phosphine Oxide?
What Sets Tris(1-Aziridinyl)Phosphine Oxide Apart
Tris(1-aziridinyl)phosphine oxide, better known as TEPA, doesn't show up in your daily news feed. Still, many scientists and regulatory agencies watch its use carefully. This compound earned its reputation as a powerful crosslinking agent, especially in materials and pharmaceutical science. Crosslinking means it connects different molecules, giving materials or drugs different strength or durability. It sounds technical, but the effects touch everyday life, sometimes in subtle but significant ways.
Heavyweight in the Fight Against Cancer
Most people outside chemistry circles first hear about TEPA from its link to chemotherapy. TEPA functions as an alkylating agent. In plain English, that means it attacks and disrupts DNA inside cancer cells, which slows or stops their growth. This use started back in the late 1950s, mostly for brain tumors and Hodgkin’s disease. Over decades, doctors and patients have wrestled with tricky side effects — nausea, immune system suppression, and more. Still, for certain cancers, it brings another chance when other drugs fail. The risks make clear why oversight and careful dosing matter so much.
Changing the Game in Industrial Polymers
Outside hospitals, TEPA pulls weight in high-performance plastics and resins. It pushes polymer chains closer together, building stronger materials. Think of airplane adhesives, electrical insulation, specialty coatings. Anytime a product faces heat, stress, or heavy wear, companies hunt for ways to toughen it up. TEPA steps in because those three aziridine groups make fast, irreversible bonds. One mistake, and the resulting plastic shows weakness or early breaking. That means only folks with training and protective gear work with it.
Why Safety Concerns Don’t Go Away
This chemical’s power works both ways. TEPA damages DNA — good for cancer cells, not for people handling it. Chronic exposure puts workers at risk for burns, breathing trouble, even long-term genetic damage. Many organizations, from OSHA in the US to the European Chemicals Agency, keep TEPA under sharp regulation. They require secure storage, fume hoods, and regular health monitoring. I’ve seen labs where a single spilled drop triggered a shelter-in-place for half a day. Mistakes happen, so prevention—through training and strong rules—saves lives.
Finding Alternatives and Next Steps
Every field leaning on TEPA also explores safer or greener options. Green chemistry has turned up alternatives, though for some jobs, TEPA still works best. A few research groups, like the EPA’s Green Chemistry Program, fund grant contests to reward novel chemicals with less hazard. Meanwhile, better protective clothing, improved glove materials, and digital exposure trackers give more control to people doing the hands-on work. If companies invest in regular training, honest reporting, and clear safety culture, accidents drop.
Tris(1-aziridinyl)phosphine oxide lives in the crossroads of medical innovation and industrial necessity. Its benefits demand respect; the hazards demand responsibility. By balancing both, we keep progress moving without forgetting the risks on the road ahead.
What safety precautions should be taken when handling Tris(1-Aziridinyl)Phosphine Oxide?
Understanding the Risks
Tris(1-Aziridinyl)Phosphine Oxide doesn’t show up in every workplace but packs a heavy punch in terms of risk. Coming from a research background in synthetic chemistry, I’ve seen how carelessness with reactive chemicals can end in accidents. This compound carries strong alkylating power and becomes hazardous by inhalation, skin contact, and ingestion. Cancer-causing, corrosive, and toxic, it doesn't need much exposure to do harm.
Wearing More than the Basics
Lab coats and gloves work for common chemicals. For this one, the gear has to be serious. Chemical-resistant gloves, goggles, and a face shield form the basic armor. I always go for double-gloving—nitrile under neoprene—because one splash reaching the skin could mean severe burns or worse. Clothes should cover as much skin as possible, and a lab coat alone often falls short. Chemical-resistant aprons add another layer of defense, especially if there’s any risk of splashing.
Containment: Not an Option—A Must
Any time I’ve seen this compound at work, it’s always handled in a properly functioning chemical fume hood. No exceptions. Tris(1-Aziridinyl)Phosphine Oxide gives off fumes you do not want near your lungs. Fume hoods help keep vapors and dust away from your breathing space and community spaces. Not only does this protect the person working with the compound, but it also protects anyone nearby. Filter maintenance shouldn’t be skipped, since a failed exhaust system turns personal protection into wishful thinking.
Storage That Takes No Chances
Leaving dangerous materials out in the open is just asking for trouble. This chemical goes in airtight, labeled containers in specialized storage cabinets—those designed for corrosives or toxins. I always check for solid seals and store away from heat, sparks, and incompatible chemicals. Segregation makes sense—store it far from acids, oxidizers, or anything that could trigger a reaction.
Clean-Up Plans Aren’t Enough—Practice Counts
Accidents happen, even with the best intentions. I remember drills in grad school where mock spills forced us to grab spill kits, don the right gear, and contain the hazard as fast as possible. Absorbent pads, neutralizing agents suitable for the compound, and heavy-duty bags should all be on hand. Proper disposal doesn’t get solved with a standard trash can—this chemical demands hazardous waste handling procedures. Nobody likes piles of paperwork, but waste manifests and chain-of-custody logs keep communities safe and accountable.
Training Builds Good Habits
Formal training helps, but everyday practice matters more. Every time I train someone new, repetition and reinforcement drive home what’s at stake. If even one person gets complacent—skips a step or cuts a corner—the results can be serious and permanent. Encouraging a no-blame environment where people feel free to raise safety concerns has always led to better outcomes in the labs I’ve worked in.
Solutions from Experience
Routine safety audits catch bad habits before they turn into incidents. Easy-to-find safety documentation—wall charts, MSDS fill-ins, digital apps—help remind busy teams what to do before and after an emergency. If budgets allow, investing in safer alternatives or engineering controls, like glove boxes or automatic dispensers, reduce the need for direct handling altogether. Small teams, regular check-ins, and open dialogue around safety standards go a long way toward preventing mishaps.
Bottom Line: Safety is Non-Negotiable
Anyone who works with something as dangerous as Tris(1-Aziridinyl)Phosphine Oxide owes it to others to slow down, suit up, and follow every protocol. Speaking from years handling hazardous chemicals, each step taken to prevent harm supports health, community trust, and professional credibility. Skipping the hard work around safety isn’t worth the risk.
How should Tris(1-Aziridinyl)Phosphine Oxide be stored?
Why Storage Demands Extra Attention
Nothing throws off a day in the lab quite like an unexpected chemical reaction. Over the years, I've come to respect just how easily a small mistake can turn into an emergency. Tris(1-Aziridinyl)phosphine oxide carries a reputation for being more hazardous than most. Handling it requires both knowledge and care, but safe storage stands out as the biggest deal-breaker between routine work and a bad accident.
Understanding Its Behavior
This compound’s aziridine rings give it an edge in chemical reactivity. Those same rings also raise the stakes—contact with air, light, heat, or moisture increases the risk of dangerous chemical changes. I’ve seen labels torn or soaked off bottles before, leaving everyone guessing whether a mystery container might contain something nasty. In this case, guessing isn’t an option. The right approach starts before the chemical even arrives, making sure the storage space won’t cause unnecessary risks.
Storage Conditions That Keep Everyone Safe
Cool, Dry, and Dark: The TrifectaFrom my experience, it’s easy to assume any shelf will do if a bottle looks well-sealed. That mistake leads to disaster. Tris(1-aziridinyl)phosphine oxide keeps best at temperatures below 8°C—refrigerated storage helps keep it stable. Humidity creates trouble fast, so desiccators or boxes with silica gel help keep the air dry. Storing chemicals in see-through glass jars might look nice, but with this one, a dark or opaque bottle prevents breakdown from light.
Physical Barriers and LabelingLast year, a friend mixed up containers of different aziridine compounds. Lab tape faded and a bottle ended up out of place. Luckily, sharp eyes caught the error before anything spilled. Clear, printed labels and color-coded stickers saved the day. Every single bottle gets labeled with the full name, hazard symbols, and a date received. Segregated compartments or locked cabinets keep reactive chemicals far apart from each other and from anything they might react with, especially acids, bases, and oxidizers.
Addressing the Real Hazards
Getting too comfortable in the lab makes people forget that one slip can mean burns or toxic exposure. Tris(1-aziridinyl)phosphine oxide reacts fiercely with moisture and releases toxic fumes. Spill containment setups are not just a formality—they’re a must. Absorbent mats and chemical-resistant trays catch the small leaks that could turn into big problems. I’ve seen old freezers stuffed so full you couldn’t find anything quickly, and that’s a nightmare if you’re responding to a spill. An organized space with spill kits in reach might sound like overkill, but when I’ve needed it, I’ve been grateful every single time.
Regular Checks and ResponsibilityKeeping chemicals out of sight and mind doesn’t mean pretending they aren’t there. Monthly inspections catch corroded lids, deteriorating containers, or forgotten stock. Any bottle showing signs of trouble deserves immediate containment and disposal. Training isn’t a box-ticking exercise; everyone who works with dangerous compounds ought to see real examples of how storage failures happen and what the costs are. Stories stick better than warnings written on a wall chart.
Better Storage Means Fewer Regrets
Safe chemical storage doesn’t boil down to following a checklist—real trust builds from habits, reminders, and respect for the risks. Tris(1-aziridinyl)phosphine oxide, like many specialized reagents, rewards that approach with safe, reliable performance instead of avoidable emergencies. The chemical’s reputation for danger comes from real accidents, not superstition. Careful storage methods and honest communication are the only answers that have made sense in my experience.
What are the typical purity levels and specifications of Tris(1-Aziridinyl)Phosphine Oxide available commercially?
Diving Into Purity Levels
Ask anyone working in specialty chemicals, and they’ll tell you purity matters far more than most want to admit. Tris(1-Aziridinyl)Phosphine Oxide, often tagged as TEPA or TAPO in labs, rarely gets sold at absolute perfection. In the catalog from most chemical companies, purity runs between 95% and 99%. Sometimes, you spot a technical grade for process work—a purity at the lower end makes economic sense in tough syntheses or big-batch jobs where trace contaminants won’t throw off your yields.
Still, if a researcher wants consistent results or is moving toward a regulatory milestone, anything shy of 98% throws a wrench in things. Most syringe pumps or glove boxes react poorly to leftovers from manufacturing, like unreacted aziridine or residual solvents. Impurities can torch an entire run, especially in medicinal chemistry or fine electronics, where one molecule off turns a project to waste.
What the Data Sheet Tells You
Labels for TEPA usually list moisture (below 0.5% for most top-shelf suppliers), pale yellow to almost clear appearance, and maybe a melting point (68–70°C). Those numbers aren’t decoration—phosphine oxides pick up water from the air. I’ve had bottles go gummy after a few days on the benchtop. Handling them without a drybox or a proper desiccator isn’t just a risk; you’re rolling dice with every reaction.
Most suppliers sell TEPA in solid form, with particle size rarely specified unless you’re buying for polymerization work. Instead, the key markers look like this: GC assay for purity, NMR peaks to verify all three aziridine rings are attached, and sometimes LC-MS for those who want a spectral print-out. Companies with an eye on Western markets run extra heavy metals checks—for areas like pharma or biotech, even single-digit ppm of lead, arsenic, or cadmium can send safety teams into overdrive.
Practical Quality Concerns in the Lab
Working with TEPA, clean product starts to matter when you’re linking it with sensitive substrates. I’ve seen cases where lower-purity batches produce side-products that eat hours of troubleshooting. If the process requirements get strict, specs like “99% minimum purity, ≤0.05% water, and identifiable lot traceability” pop up in purchase orders. Labs put a premium on documented stability data, too; a few grams of spoiled product costs more than the sticker price of a high-purity option.
Try reaching for a bottle that lacks documentation, and problems multiply. Regulatory audits demand certificates of analysis. Without proof, that lot becomes a liability. I’ve personally seen researchers lose six months of work in fine chemical development, all because one spec drifted beneath a compliance cut-off. For those shipping TEPA internationally, controls on hazardous substances require SDS and REACH compliance, so buying off-brand is risky business.
Raising the Bar for Quality
Suppliers take calls from demanding clients every day who want guarantees—not just a number on a label, but full batch records, impurity profiles, and long-term stability under shipping conditions. If you push for 99.5% and higher purity, the cost jumps, but sometimes that’s the best route. For research and process-oriented businesses, it pays to build a relationship with a trustworthy supplier who doesn’t shy away from detailed batch disclosure. Quality control starts with knowing what’s on the inside, and making the call to pay for it upfront saves headaches down the line.
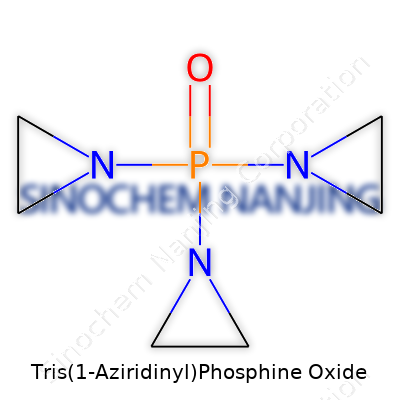
| Names | |
| Preferred IUPAC name | Tris(aziran-1-yl)phosphine oxide |
| Other names |
TEPA ThioTEPA Tepa Tris(1-aziridinyl)phosphine oxide |
| Pronunciation | /ˈtraɪs wʌn əˈzɪrɪdɪnɪl ˈfɒsfiːn ˈɒksaɪd/ |
| Identifiers | |
| CAS Number | 545-55-1 |
| Beilstein Reference | 136140 |
| ChEBI | CHEBI:39068 |
| ChEMBL | CHEMBL1230262 |
| ChemSpider | 152971 |
| DrugBank | DB01193 |
| ECHA InfoCard | 100.033.619 |
| EC Number | 208-843-6 |
| Gmelin Reference | 85798 |
| KEGG | C19308 |
| MeSH | D014285 |
| PubChem CID | 65703 |
| RTECS number | TZ1925000 |
| UNII | 56L5G1D71F |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID6020159 |
| Properties | |
| Chemical formula | C6H12N3OP |
| Molar mass | 207.16 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.44 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -0.5 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 13.2 |
| Basicity (pKb) | 3.37 |
| Magnetic susceptibility (χ) | -53 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5100 |
| Viscosity | Viscosity: 29.4 cP (20°C) |
| Dipole moment | 5.59 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 253.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1316 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | L01AX04 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled |
| Precautionary statements | P261, P280, P301+P310, P304+P340, P308+P311, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 107°C (225°F) |
| Lethal dose or concentration | LD50 oral (rat): 2 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 1 mg/kg |
| NIOSH | TX1750000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Tris(1-Aziridinyl)Phosphine Oxide: Not established |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 3 mg/m³ |
| Related compounds | |
| Related compounds |
Aziridine Phosphine oxide Hexamethylphosphoramide Phosphoramide Tris(2-chloroethyl)amine |

