Triphenyltin Hydroxide: A Hard Look at History, Chemistry, and the Road Ahead
Historical Development: Lessons Learned, Warnings Issued
Chemical control in agriculture started picking up steam after the Second World War, when the promise of bumper crops gripped the world’s imagination. Triphenyltin hydroxide wouldn’t enter the story until organotin compounds caught attention for their powerful fungicidal properties. By the 1960s, triphenyltin-based treatments appeared in orchards and fields, especially on potatoes and peanuts. Industry and research teams pushed for these compounds because common fungal threats—blight, rust, mildew—threatened food security. Governments scrambled to register and regulate these new tools, but even then, scientists raised questions about their environmental and health impact. Today, the legacy of triphenyltin hydroxide rests on a complicated record: it kept food on tables, but environmental regulators found themselves playing catch-up with its byproducts showing up well beyond farm borders.
Product Overview: Unpacking the Compound
Triphenyltin hydroxide belongs to the organotin family, a group known for marrying organic molecules with the metallic punch of tin. Chemists gravitated toward triphenyltin because its phenyl rings—three carbon-based arms—give it staying power in wet, organism-rich soil. In fields faced with rot and mildew, persistence can seem like a blessing. These days, the compound still sees use where tough resistance and slow breakdown are wanted, though everywhere else, credible concerns about residues and long-term risks overshadow the benefits.
Physical & Chemical Properties: Understanding What We’re Working With
Anyone who’s ever stood in a chemical lab will know this: there’s rarely a mystery about organotins, and triphenyltin hydroxide proves it. It usually appears as a white to off-white powder, sparingly soluble in water but more approachable with organic solvents like acetone or alcohol. Heating doesn’t break it down easily, and its stubborn stability calls for careful handling. Topping off the list, that distinct tin smell lingers—something you don’t forget quickly. Formulators kept the blend stable to deliver field-long longevity, but it didn’t take researchers long to spot the flip side: once released, it sticks around in waterways and soils.
Technical Specifications & Labeling: Trust and Transparency
Over the years, technical data sheets grew from a few notes on purity and shelf life into a heavy stack of regulations. Labels in the past left much to the imagination, which fueled a rocky path for worker safety and environmental justice. Now, any shipment must declare not just active ingredient percentages but also significant impurities, recommended handling, and disposal steps. Hazard icons warn users of the stakes. While this development reflects a hard-earned respect for chemical risks, it also speaks to a larger shift: users demand to know what enters their environment, not just their fields.
Preparation Method: Chemical Craftsmanship
Synthesizing triphenyltin hydroxide involves a dance between organotin halides and strong bases, most often sodium hydroxide. Stirring and temperature control keep the reaction pointed in the right direction. Chemists balance input ratios and monitor every step since missteps leave behind unwanted metallic byproducts or incomplete conversions. The details echo my own time in university labs, sweating over glassware, never losing sight of yield and purity. In industrial settings, large reactors and airtight procedures scale up what students and researchers do a hundred times smaller—and always with the knowledge that any slip can introduce a new hazard into the batch.
Chemical Reactions & Modifications: Adaptation and Experimentation
Triphenyltin hydroxide doesn’t act alone. It reacts with acids to produce triphenyltin salts, while exposure to strong oxidizers may degrade its organic groups or split the tin off completely. In crop science, formulation scientists experimented by combining it with fillers, surfactants, or adhesives to shape how it clung to leaves or released into the soil. Research teams poked at the edges—looking for ways to blunt its toxicity or sneak in slow-release profiles—but always found themselves boxed in by its persistence. Anyone exploring the organotin class starts here, learning how each ring and bond changes real-world effects.
Synonyms & Product Names: Behind the Labels
Whether thumbing through old farming catalogs or browsing regulatory lists, you’ll find triphenyltin hydroxide wearing a host of names. Chemists call it TPT hydroxide. Farmers know it under badges like “TPTH” or trade names lost to time after bans or reformulations. Standard identifiers—like its CAS number—matter more now as international trade and treaty enforcement rely on clarity to track where chemicals flow. For those seeking clarity, looking up “triphenyltin hydroxide” alongside synonyms clears up confusion and helps avoid costly mistakes or regulatory lapses.
Safety & Operational Standards: Real-World Protections
Decades of field studies, accidents, and lab reports have forced a blunt realization: even well-intended chemical tools can bite back. Today’s safety standards reflect slow but real progress. Handling triphenyltin hydroxide requires gloves, masks, and eye protection—no exceptions. Storage rules grew sharper, demanding cool, dry spaces away from food or animal feed. Training protocols go beyond surface warnings; users learn what to do if spills or exposures happen and who to call. Many countries now restrict its use or require certified applicators, a direct result of farmworker and environmental advocacy backed by medical research and tragic headlines.
Application Area: Once Everywhere, Now Under the Microscope
Triphenyltin hydroxide earned its keep in orchards and root crops, especially where fungal blights threatened wages and food. In humid climate zones, potatoes and peanuts faced black rot and blight, conditions hard to tackle without strong chemical lines of defense. For a generation, farmers counted on its broad-spectrum power to keep yields high. More recently, mounting pressures from consumers and regulators pushed the sector to rethink its reliance, with many switching to less persistent or biologically based options. Today, some countries still allow restricted use in specialty situations, but every deployment faces hard questions about downstream impact and residue management.
Research & Development: Always a Moving Target
Back in the 1970s and 80s, research meant chasing higher crop output and exploring new delivery methods. As environmental science matured, the research questions changed—what happens to triphenyltin residues in groundwater? How do they travel up the food chain? Laboratory teams mapped out soil persistence and studied how sunlight, microbes, and waterways broke the chemical down. I remember mentors painting a picture of progress: every answer raised new challenges, highlighting gaps where neither regulations nor industry could keep pace. Current research points at bioremediation—restoring soils and rivers through bacteria and fungal cleanup crews. Some labs now test innovative replacements, betting on safer, quicker-decaying compounds.
Toxicity Research: Hard Numbers and Human Stories
Toxicology didn’t wait long to sound alarms. Rodent studies confirmed that triphenyltin hydroxide can disrupt reproductive systems and immune responses. Wildlife sampling exposed problems for aquatic species—fish, frogs, and crustaceans suffer from even low doses carried far from treated fields. Human health findings followed, with agricultural workers reporting rashes, breathing trouble, and long-term effects traced to repeated exposure. Regulatory agencies set strict limits, but enforcement gaps remain a stubborn problem. After years in this field, I’ve seen firsthand how incomplete reporting and patchy monitoring schemes let risks slip through, especially in low-income or under-resourced rural areas.
Future Prospects: Toward Accountability and Innovation
Looking forward, no country stays unchanged by the lessons of organotins like triphenyltin hydroxide. Legal landscapes tighten every year as science uncovers new risks. Integrated pest management—combining biological, cultural, genetic, and chemical tools—offers hope of gradually leaving persistent chemicals behind. For triphenyltin hydroxide, the spotlight now shines on remediation: cleaning contaminated soil and water, supporting affected communities, and demanding restitution for damages long ignored. Industry research must set its sights on greener alternatives that drop quickly and leave no legacy. As climate conditions shift and global food production strains, the pressure mounts to explore safer, smarter ways to protect crops without paying an outsized price in health or habitat.
What is Triphenyltin Hydroxide used for?
The Roots of Triphenyltin Hydroxide Use
Walking through an apple orchard as a kid, I never thought about what it took to keep those trees healthy. Later, working on pest management teams with small farmers, I saw firsthand the trouble caused by fungi, especially the kind that eats away at crops many count on for daily income. Triphenyltin hydroxide stands out here as a chemical tool—some would say a necessary one—in the fight against fungal diseases in agriculture.
Why Growers Turn to Chemicals Like This
Some years, wet spring weather means scab outbreaks run wild in apple and potato fields, leading to rot, low yields, wasted work, and a world of frustration. Triphenyltin hydroxide works as a fungicide. Growers spray it to kill fungi like powdery mildew, late blight, and scab. The compound delivers a punch that older, weaker treatments struggle to match. Crops absorb it quickly, which stops fungi from spreading and lets fruit and leaves grow undisturbed.
Histories from U.S. and European agriculture hint at this story. Both continents saw large harvest improvements after tin-based fungicides took hold in the late 1900s. Crops lived longer, shelf lives extended, and food security got a little stronger in tough seasons. For plenty of family farms teetering on the edge, this mattered.
Risks Lurking in the Soil and Water
After years spent walking farmland and stream banks, I know many hesitate to trust strong farm chemicals, and they aren’t wrong to ask tough questions. Triphenyltin hydroxide breaks down slowly in soil and water, lingering for months. Scientific journals track traces found in rivers near treated fields, raising red flags about wildlife and water safety. Fish and water insects show damage from tin compounds, and accidental spills threaten sensitive ecosystems.
Farmers who handle this stuff see the risks up close. Repeated exposure can harm the nervous system, so gloves, masks, and strong storage rules are common-sense essentials. Improper application or storage opens doors to health claims and lawsuits that can wipe out small growers.
Trying Better Ways While Staying Honest About Tradeoffs
Big and small growers hold mixed opinions. Some in my local farming network push for organic solutions, encouraging more crop rotation, disease-resistant seeds, or biopesticides that serve the same purpose with fewer side effects. It’s tough, since natural treatments rarely pack the same one-stop punch—some years, they fail when heavy rain or drought tips the balance in favor of the fungus.
New research—often funded through public universities—aims to break this tradeoff by developing safer chemical alternatives, stronger fungi-resistant strains, and low-impact application methods. The European Union started restricting organotin use in the early 2000s, and U.S. pesticide regulators keep revisiting which chemicals count as "safe enough." Each review digs into more data, mostly driven by real-world use, accident reports, and long-term community health trends.
For now, triphenyltin hydroxide sits at the intersection between keeping food abundant and keeping soil and waterfit for future generations. Any real solution should admit both facts: growers feel pressured to protect crops, but downstream effects last longer than a single growing season. Smart policy and patient science have to catch up with farmers’ realities, not the other way around.
What are the safety precautions when handling Triphenyltin Hydroxide?
Knowing What You’re Working With
Triphenyltin Hydroxide gets its reputation from its use as a fungicide, especially in agriculture. It can help protect crops against mildew and fungus, but the chemical itself brings risks that just can’t be ignored. People exposed to it can experience eye irritation, skin reactions, and problems with breathing. After years of working in labs and running field trials, I’ve learned just how important it is to take chemicals like this seriously and plan every step of the job with personal safety in mind.
Wearing the Right Protective Gear
You won’t get far with safety if you skip proper gear. I never start a session with Triphenyltin Hydroxide without gloves made from materials like nitrile or neoprene. Ordinary latex can give out quickly around organotins. A lab coat, long pants, and closed-toe shoes are all required, too. Face protection becomes essential, especially when there’s a chance for splashes—think safety goggles and sometimes a face shield for good measure.
Ventilation Can’t Be an Afterthought
Chemicals like this one throw off fumes you really don’t want to inhale. I’ve always worked with these substances under a certified chemical fume hood, or if I’m in the field, at least outside and upwind. Inhaling organotin compounds has a way of sneaking up on folks. If there’s any doubt, a respirator with organic vapor cartridges gets added to my routine. Trust me, you’d rather look a bit over-prepared than come down with chemical pneumonitis.
Preventing Skin Contact
Skin absorbs some organotin compounds faster than you might expect. Even small spills can lead to problems if wiped up with bare hands or thin gloves. I never use my fingers—even gloved—for cleaning up. Absorbent pads, proper tweezers, and double-bagging waste mean less chance for a surprise rash or worse. A bottle of specialized chemical soap sits close so I can wash up at a moment’s notice. It’s a rule learned after seeing what a drop on the wrist can do by the next day.
Dealing with Spills and Waste
Every lab and facility using Triphenyltin Hydroxide keeps a spill kit at arm’s reach. Cat litter soaks up most small spills with ease, and everything heads straight to a hazardous waste container. Working alone never sits easy with a chemical this potent; pairs mean more hands for cleanup and someone to call for help if trouble hits. All liquid and solid waste from these jobs gets labeled specifically with contents and hazard warnings since these don’t just blend in with regular trash or recycling.
Health and Training Matter
I’ve seen teams skip the annual safety refreshers, but that’s how corners get cut. Anyone coming near Triphenyltin Hydroxide, whether in the lab or on the farm, needs hands-on practice with the equipment, not just a sheet of instructions. Emergency eyewash stations and showers stay checked and clear. Every person—not just the supervisor—knows what to do in a hurry.
Building a Responsible Approach
Handing down hard-earned habits to new staff or students brings a sense of pride. Approaching Triphenyltin Hydroxide like a curious amateur leads to trouble. Only respect and know-how get you home at the end of the day. Management’s job doesn’t stop at writing procedures. They show up, they ask questions, and they push for up-to-date chemical knowledge. That attitude trickles down, reinforcing a culture where chemicals never become casual, and safety feels like second nature—every shift, every bottle, every time.
What is the chemical formula and structure of Triphenyltin Hydroxide?
The Chemical Formula and Structure
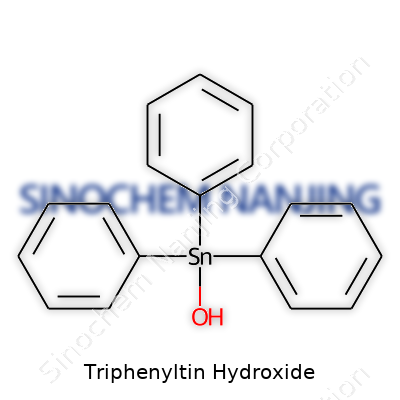
Triphenyltin hydroxide carries the chemical formula C18H16OSn. This compound features a tin atom at its core, with three phenyl groups (each consisting of a benzene ring attached to a carbon atom) and a single hydroxide (OH) group bonded to it. Looking at the structure, the tin atom connects directly to each of the three phenyl groups and the OH group. This gives triphenyltin hydroxide its typical "starburst" shape—a non-planar geometry that keeps the bulky phenyl groups from crowding each other in space. The substance owes its stability and reactivity largely to the arrangement of these four substituents around the tin center.
Why Triphenyltin Hydroxide Matters
Triphenyltin hydroxide hasn’t gained attention solely due to its intriguing chemical structure. Its primary claim to chemical fame comes from its action as a fungicide, especially in agriculture. Farmers handle a tough job keeping crops healthy; plant diseases threaten food production worldwide. This compound steps up by inhibiting the growth of fungi and protecting crops like potatoes and peanuts against stubborn fungal infections. While many folks may never hear about it, triphenyltin hydroxide has quietly played a part in stopping blights and boosting harvests.
Health and Environmental Considerations
While triphenyltin hydroxide carries practical value in the field, it also raises genuine concerns. Animal studies and research have shown it can be toxic if misused—exposure may cause harm to aquatic life and even humans, depending on dose and duration. Stories about organotin compounds like this one point to nervous system problems, immune dysfunction, and reproductive effects in some animal models. In my own time working in environmental monitoring, the persistence of tin-based pesticides in waterways became obvious. The residues can linger, slowly breaking down and impacting sensitive organisms. Researchers in Japan and Europe keep tracking residues in soil and water, tying some ecological disruptions to tin-based chemicals entering rivers and lakes.
Regulatory and Practical Solutions
The debate around triphenyltin hydroxide goes beyond chemistry labs. Regulatory bodies in the US, Europe, and other regions have put strict controls on use and disposal, especially since organotin compounds grew notorious for their environmental impact in the 1980s and 1990s. I remember field technicians testing streams miles downstream from potato farms, watching as countries tightened pesticide regulations and encouraged safer alternatives. Integrated pest management took off: farmers blended traditional crop rotation with minimal, targeted chemical use. Factoring in regular monitoring, soil health assessments, and the move toward new, less persistent fungicides, the hope is to curb the environmental footprint.
Looking Ahead
Triphenyltin hydroxide sits at a crossroads between helping feed more people and safeguarding environmental health. The science behind its structure seems elegant, almost academic, but the choices around its use ripple out onto fields, rivers, and dining tables. Ongoing research into greener alternatives and responsible practices will keep the conversation moving. Understanding what goes into these decisions isn’t just for chemists—farmers, consumers, and everyday people all depend on making food production safer and more sustainable, one compound at a time.
How should Triphenyltin Hydroxide be stored?
Looking at the Risks
Triphenyltin hydroxide is the sort of chemical that comes with a reputation. Its use in fungicides and paints means it pops up in a variety of places. Zoom in a little closer, it also brings a long list of safety notes. If you’ve spent any time working around industrial chemicals, you’ll know they can go sideways quickly if handled haphazardly. This one ranks pretty high on the list. Touching or inhaling dust can harm your skin, eyes, or respiratory tract. Let’s talk about what it takes to store it responsibly, drawing from years of work in labs where chemicals don’t always play nice.
No Shortcuts on Secure Containers
A big part of storage comes down to the basics. Tossing triphenyltin hydroxide into an old food-grade container or a leaky bucket just increases the odds of a problem. Sturdy, tightly sealed drums built from compatible materials—chemically resistant plastics or coated metals—protect the contents and everyone nearby. Labels stand out as one of the simplest safeguards. In my own experience, you won’t find anyone in a well-run chemical warehouse who lets unlabeled containers sit on a shelf. Making sure every container carries clear hazard warnings stops mix-ups before they start.
Keep It Cool, Keep It Dry
Like many chemicals, triphenyltin hydroxide reacts to its environment. High temperatures and humidity push things in the wrong direction. Facilities with climate control and regular monitoring of temperature and moisture help sidestep problems like clumping, leaking, or chemical breakdown. Once, I watched technicians lose an entire batch of a related compound because a single heat vent malfunctioned for a weekend. A minor oversight in climate can wipe out value and threaten safety, emphasizing the need for strict environmental controls in storage areas.
Distance from Incompatibles
Every seasoned warehouse manager learns fast that some chemicals just don’t get along. Triphenyltin hydroxide won’t mix safely with strong acids, oxidizers, or bases. For anyone in charge of inventory, segregation by compatibility matters. Dedicated shelving or even separate rooms for incompatible groups keeps stray spills from turning into emergencies.
Access: Only the Right Hands
In my first chemical handling job, training hammered home that not everyone should wander into a storage room. Restricted access policies keep only those with the right training near hazardous stocks. A sign-in sheet or an electronic badge scan limits exposure and keeps the accidental curious from getting in trouble.
Routine Monitoring and Housekeeping
Dust, residue, or even a strange new odor can be signs of trouble. Good storage is never just “set it and forget it.” Regular checks for leaks, corrosion, and container condition catch issues early. Any worn seal or unexpected mess signals a problem needing prompt cleanup. Years in lab environments have shown me how a strict cleaning schedule and regular inspection create a safer workplace.
Waste Isn’t an Afterthought
Old or contaminated chemicals can become dangerous if ignored. Following local hazardous waste rules, arranging for responsible disposal keeps things safe both on- and off-site.
What Makes a Difference
Accidents involving triphenyltin hydroxide rarely result from a single misstep. They crop up when corners get cut—when storage turns careless, or labels go missing, or the wrong person opens a drum. Consistent, practical steps pay off. Sturdy storage containers and climate control, clear labeling, careful access, diligent checks, and responsible waste removal keep people and facilities safe. These aren’t fancy solutions; they’re habits formed from lessons learned the hard way.
What are the health hazards associated with Triphenyltin Hydroxide exposure?
Understanding the Risk
Triphenyltin Hydroxide stands out in the world of agricultural fungicides. It works hard protecting crops from blights, but the reality is that the risks stack up quickly, especially for those who mix or apply it without solid precautions. Reading the data from the EPA and health agencies, anyone handling this chemical faces direct threats to their health. Just a small amount splashed or inhaled can turn a routine workday into a real emergency.
Breathe It In, Feel It Fast
Walking through a field just treated with Triphenyltin Hydroxide, the sense of something unnatural in the air feels hard to ignore. Inhaling dust or spray mist doesn’t just sting your nose—workers have reported headaches, throat pain, and skin redness. Several studies highlight that this isn’t a one-off fluke. Triphenyltin compounds irritate mucous membranes, trigger rashes, and set off asthma attacks. The chemical finds its way into the bloodstream quickly, and the liver and kidneys get hit with the clean-up job.
Long-Term Trouble: Much More Than a Rash
Chronic exposure packs an even heavier punch. Researchers link Triphenyltin Hydroxide contact with nerve problems and weakened immune function. The chemical disrupts enzymes that keep the nervous system steady. Persistent, low-level contact on the job can mean memory problems, muscle weakness, even tremors—issues that never get better with just a day’s rest. Rat and dog studies, and long-term farmworker surveillance, point out that this isn’t just lab talk. People really do get sick over months, not just days.
Threats Beyond the Field
Fields don’t keep secrets. Once sprayed, runoff during a good rain can send Triphenyltin Hydroxide into streams, ponds, and groundwater. At low doses, fish and water bugs start acting strangely. Eggs and larval forms show stunted growth or weird malformations. Because these contaminants build up up the food chain, local families who fish or use well water end up with health questions of their own. In the European Union, these spills contributed to tighter rules—including strict bans.
Finding a Safer Way Forward
For anyone working with Triphenyltin Hydroxide, information isn’t enough—equipment matters. Tight-sealed goggles, nitrile gloves, and well-fitted respirators don’t just check boxes, they truly cut risk. Farmers can switch to less hazardous fungicides as agronomy research improves. Local governments step up by tracking water quality and warning families of contamination risks. In my own experience visiting family-run farms, just seeing the difference between those who invest in personal protective gear and those who don’t is a wake-up call. Folks who receive regular safety training, know the symptoms, and see their own exposure readings work more confidently and face fewer health problems.
Keep Talking and Asking Questions
No chemical, including Triphenyltin Hydroxide, solves problems without drawbacks. Workers, families, and policy makers should push for more long-term safety studies and share findings with each other. Keeping eyes wide open to these hazards makes it possible to protect both the people putting food on our tables and the land delivering those harvests.
| Names | |
| Preferred IUPAC name | triphenylstannanol |
| Other names |
TPTH Fentin hydroxide Triphenyltin(IV) hydroxide |
| Pronunciation | /traɪˈfɛnɪl.tɪn haɪˈdrɒksaɪd/ |
| Identifiers | |
| CAS Number | 'TPT-OH': '薉-76-4' |
| Beilstein Reference | 1463773 |
| ChEBI | CHEBI:34802 |
| ChEMBL | CHEMBL508128 |
| ChemSpider | 20806 |
| DrugBank | DB13521 |
| ECHA InfoCard | 100.034.099 |
| EC Number | 215-183-2 |
| Gmelin Reference | 87048 |
| KEGG | C18594 |
| MeSH | D014278 |
| PubChem CID | 66273 |
| RTECS number | WK4900000 |
| UNII | 2U70C9Y2V5 |
| UN number | UN2786 |
| Properties | |
| Chemical formula | (C6H5)3SnOH |
| Molar mass | 385.15 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.32 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.9 |
| Vapor pressure | 1.55 x 10^-7 mmHg (25°C) |
| Acidity (pKa) | 11.3 |
| Basicity (pKb) | 4.75 |
| Magnetic susceptibility (χ) | -36.0e-6 cm³/mol |
| Refractive index (nD) | 1.674 |
| Dipole moment | 2.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 384.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -171.2 kJ/mol |
| Pharmacology | |
| ATC code | QO649 |
| Hazards | |
| Main hazards | Toxic if swallowed. Fatal if inhaled. Causes damage to organs through prolonged or repeated exposure. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P310, P302+P352, P304+P340, P308+P311, P330, P361, P391, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | Flash point: 214°C |
| Autoignition temperature | 180°C |
| Lethal dose or concentration | LD50 oral rat 132 mg/kg |
| LD50 (median dose) | LD50 (median dose): 130 mg/kg (oral, rat) |
| NIOSH | TY2450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Triphenyltin Hydroxide: "0.1 mg/m³ (as Sn) |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Triphenyltin chloride Triphenyltin acetate Tributyltin oxide Triphenyltin trifluoroacetate |

