Triphenyltin Acetate: A Closer Look at Its Path, Properties, and Perspectives
Historical Development
Triphenyltin acetate emerged in the mid-20th century, right at a time when chemists worldwide explored the power of organotin compounds for different practical uses, especially in agriculture. The search for effective fungicides and molluscicides led developers to molecules containing tin, as these seemed to promise more targeted control over crop-destroying organisms. Early successes in the lab and in the field stoked demand, and by the 1960s, triphenyltin products anchored themselves firmly in the toolkit of agricultural professionals. These compounds have been part of farming’s chemical arsenal through several decades of shifting practices and tougher regulatory scrutiny, making their footprint on both fields and environmental policy.
Product Overview
Triphenyltin acetate represents one of those organometallic substances that found a strong niche before others caught up. With an eye on molds, fungi, and pests, researchers gave this compound a long look due to the combination of three phenyl groups bonded with a tin core, completed by an acetate group. Its distinct structure set it apart, delivering efficient pest-killing ability without the volatility of some earlier inorganic fungicides. For years, this compound appeared in markets under many labels—agricultural suppliers offered it under names tied to the tin and acetate roots, but all signals pointed to the same fungicidal chemistry.
Physical & Chemical Properties
Triphenyltin acetate usually appears as a white or pale powder, often giving off a faint chemical scent. It doesn’t dissolve well in water, preferring organic solvents like chloroform or acetone. This low solubility in water mattered out in the field, since it stuck better to plant surfaces without washing off in light rain. Its melting point sits around 122 to 124 degrees Celsius, and the solid has shown real staying power under standard storage. One physical trait—its persistence—linked directly with its environmental reputation. Oxygen and sunlight eventually break it down, but the process is slow, so residue issues have made people take notice.
Technical Specifications & Labeling
In the past, labels on triphenyltin acetate packaging spelled out its chemical formula, purity grade (often over 95 percent for commercial uses), and advice on storage and usage, reflecting growing concern about worker safety and environmental impact. Technical bulletins from that era stress keeping it sealed, dry, and away from acids. Folks working with this compound always faced clear instructions: gloves and barriers needed for handling, tight protocols for spills, and plenty of safety messaging for professionals tasked with spraying or mixing solutions.
Preparation Method
The earliest lab methods to create triphenyltin acetate involved a reaction between triphenyltin chloride and a sodium or potassium salt of acetic acid. Chemists typically prepared the chloride compound first—by treating tin with phenylmagnesium bromide to whip up triphenyltin chloride—then swapped the chloride ion with an acetate. These steps drew from both classical organometallic synthesis and practical demands for reasonably pure product on a manufacturing scale. The organic chemistry behind it isn’t tricky, though controlling impurities and maximizing yield call for steady hands and well-maintained apparatus.
Chemical Reactions & Modifications
Triphenyltin acetate reacts with acids and bases, sometimes breaking down to triphenyltin hydroxide in moist, alkaline conditions. The phenyl-tin bond makes it less reactive than dialkyltin species that degrade faster in the environment. Chemists have tried to tweak its structure, hoping to find variations that retain anti-fungal power but lessen risks to non-target species. In lab settings, swapping the acetate group with other organic acids or attempting slight changes to the phenyl rings created related molecules with similar chemical properties, but regulation and toxicity questions always followed close behind.
Synonyms & Product Names
This compound carries several alternative names. Across literature and product sheets, it regularly appears as triphenyltin ethanoate, TPTA, and by less common systematic descriptors plugging the phenyl and acetate groups. Over the years, these names entered the regulatory lexicon, winding up in government lists of restricted or monitored pesticides, reflecting the broader debate about organotin products beyond this specific molecule.
Safety & Operational Standards
Long experience in chemical handling teaches respect for substances like triphenyltin acetate. It’s been classified by many regulatory agencies as a health and ecological hazard. Exposure carries real risks—skin irritation, eye damage, and long-term health concerns for anyone engaged in prolonged or careless use. Strict personal protective equipment has always stood as the line between safe practice and preventable accidents. Municipal guidelines often prohibit use near water, as aquatic life reacts strongly to even low concentrations. Out in the agricultural workplace, regular worker training, environmental monitoring, and tightly enforced application procedures went a long way in keeping mishaps at bay. Any company or farm crews relying on such a product keeps the binders full of Material Safety Data Sheets, audits, and compliance records close at hand.
Application Area
Triphenyltin acetate found its strongest foothold in large-scale agriculture. Farmers relied on it for decades as a go-to weapon against fungal blight or snail invasions, sometimes dressing seeds or spraying it directly on field crops. The struggle to balance food production with environmental protection complicates the story. On the one hand, this chemical played a role in reducing crop losses, but the environmental record shows strong signals of harm—tin residues accumulating in soil and water, and spiraling up the food chain. By the time integrated pest management and biopesticides became household terms among agronomists, restrictions and outright bans started to appear. Recent years haven’t witnessed widespread, legal use in countries with strict environmental and food safety regulations.
Research & Development
A steady stream of academic and industry research into organotin compounds—triphenyltin acetate included—helped sharpen the global picture of environmental persistence, bioaccumulation, and toxicity. Scientists continue to explore pathways to break down leftover residues and reclaim contaminated soils. Some teams dig into the chemistry, while others focus on big-picture monitoring of tin levels in agricultural regions. There’s no denying a compound like this pushed forward our scientific understanding, but it also underscored the importance of weighing short-term benefits against long-standing environmental debts. Less hazardous, biodegradable replacements sit at the top of the R&D priority list now.
Toxicity Research
Nothing shaped the narrative around triphenyltin acetate more than toxicity studies. Animal testing—along with widespread monitoring near heavy-use regions—helped document its impact: nervous system effects, reproductive impairment, and distorted development in fish and invertebrates. In humans, risks linked with occupational exposure sparked plenty of cautionary tales. International bodies from the World Health Organization to the European Food Safety Authority flagged organotin compounds as priorities for phase-out. The science is robust: even trace quantities stick around long enough to threaten aquatic communities and possibly affect food safety for people.
Future Prospects
Looking ahead, the story of triphenyltin acetate probably isn’t one of a comeback. More countries block or phase out its use each year, citing the dual demands of sustainable agriculture and environmental stewardship. This compound stands as a textbook case of how aggressive chemistry can outpace our ability to fully measure the fallout. Lessons learned from its rise and fall now guide the search for new solutions—safer chemicals, better pest control strategies, and a shift toward technologies that won’t leave future generations holding the bill for today’s quick fixes. Scientists still track residues, revisit legacy contamination sites, and occasionally experiment with remediation strategies. Where some see a relic, others take it as warning: solutions in farming and industry carry consequences, and the most successful ones will always build safety, transparency, and sustainability straight into the design.
What is Triphenyltin Acetate used for?
Old Pesticides, New Questions
Triphenyltin acetate found its purpose in the world of farming and industry. Growing up around fields, I saw older farmers mix chemicals with care, their eyes alert to any sign of blight or rot. Many fungicides crowded the shelves, but triphenyltin-based options stood out in the 1970s and 1980s. This compound worked well against fungi that liked to ruin crops, especially potatoes, sugar beets, and rice. It proved tough on fungal diseases such as late blight, a painful enemy for any potato grower. Watching seasoned neighbors spray their fields, you could tell triphenyltin acetate kept problems in check.
But There’s a Catch
Triphenyltin acetate doesn't stop at stomping out fungi. This chemical also cropped up as an anti-fouling agent in paints. Boat owners sometimes relied on triphenyltin to stop barnacles and algae from sticking to hulls. It kept watercraft cleaner, but the stuff didn't pick favorites—it harmed marine life along the way. The chemical built up and threatened oysters, snails, and all kinds of underwater creatures.
Why We Don’t See It Much Anymore
Over the years, environmental stories popped up, warning about the dangers of persistent chemicals in soil and water. Scientists tracked triphenyltin in rivers and lakes. Some of these stories came from Europe; others landed in the U.S., where people got uneasy about what these compounds did when they drifted from farm fields or boatyards. The risks hit hardest among folks living near water. Governments started tightening rules. Today, it’s rare to see legal use of this chemical as a fungicide or in marine coatings. The European Union banned many organotin compounds back in the early 2000s. The U.S. Environmental Protection Agency followed suit, responding to mounting evidence of toxicity in the environment. Many countries set strict limits or phased out registration for any agricultural uses.
Lessons from the Field and Water
I remember growers talking about shifting to other fungicides. Some options came with fewer health and environmental warnings, but pests and fungi don’t give up easy. They adapt. Even so, folks wanted less risk to their groundwater or seafood dinners. Science delivered better testing; more data rolled in; policymakers paid attention. In fact, long-term exposure to organotin compounds like triphenyltin acetate links to reproductive and developmental problems in animals. For humans, even low levels urge experts to say, “Let’s avoid this route.”
Searching for a Balance
We still deal with trade-offs. Farmers need ways to protect crops. Boaters want clean hulls. Instead of triphenyltin, copper-based coatings and biopesticides emerged. These don’t come free from risk, but they leave less mess behind. Rural communities and regulators now work to find safer pest-control practices. Newer coatings for ships draw inspiration from nature, making it tougher for barnacles to grab hold without relying on toxic chemicals. All this points to a world learning from earlier mistakes. The cost of getting rid of pollution proved steep—both in dollars and in what washed downstream.
Being Honest About Chemical Tools
Looking back, triphenyltin acetate filled a gap for its time. But the environmental and health costs were real. Factories and farms demanded results, and this compound delivered until science showed something better: not all solutions last. Tackling crop disease remains a tough job, but smarter choices make it easier to protect food and water. Building safer tools, listening to research, and sharing real evidence—these steps guide choices so communities and ecosystems don’t pay the price years later.
What are the safety precautions for handling Triphenyltin Acetate?
Understanding Why Caution Matters
Triphenyltin acetate has a way of raising eyebrows among professionals who handle chemicals, and for good reason. This compound carries toxic effects, especially for workers who long for a safe and healthy environment when clocking in every morning. It can irritate skin and eyes, harm the respiratory system, and—worst of all—slowly build up in the organs with repeated exposure. This kind of risk doesn’t call for panic, but it does call for respect and real-life preparation.
Real Risks, Real Experiences
A few years ago, a lab manager I knew landed in the hospital after just a few careless minutes handling this compound without gloves. He acted tough, skipping basic protection “just this once,” and paid the price with serious blisters on his hands. Hearing stories like that brings home that safety isn’t just a bunch of checklists. The consequences hit hard and fast.
To keep those risks at bay, the first step always starts with personal protective equipment. Nitrile gloves keep chemical splashes away from skin. Safety goggles mean an accidental splash won’t send you to the ER with an eye injury. Lab coats—ideally chemical-resistant options—add protection against spills, and a proper mask or respirator blocks dangerous fumes and dust from your lungs. The PPE gear might look a little over-the-top to outsiders, but inside the lab, it’s a basic necessity.
Ventilation and Containment
Nobody should open a bottle of triphenyltin acetate in a stuffy room. Fume hoods play a key role in keeping indoor air from turning toxic. Some labs also install dedicated exhaust systems just for handling heavy-duty compounds. If you ever breathe in a lungful of untreated chemical air, you remember the burning feeling for days. There’s no shortcut worth that kind of misery.
Handling and Storage Practices
From what I’ve seen, the way you store chemicals makes a real difference. Triphenyltin acetate belongs in a sealed, labeled container, stashed far from any food or drink. Store it in a cool, well-ventilated spot. Some colleagues use spill trays in case anything leaks, which helps prevent cleanup headaches and environmental contamination. Following a policy of “don’t eat, drink, or smoke near chemicals” seems basic to people with experience, but newcomers often overlook it until bad habits catch up with them.
Spill Response and Waste Disposal
Spills might sound rare, but even the most careful workers slip up. Standard practice calls for having spill kits—complete with absorbent materials and neutralizing agents—set out where accidents are likely. Trained staff respond fast, limiting the damage. Chemical waste from triphenyltin acetate can’t go down the drain or straight to dumpsters. Facilities collect this waste for licensed disposal to meet environmental regulations and keep water sources safe. Overlooking disposal steps means risking heavy fines and public hazards.
Training: The Unsung Hero
I’ve watched summer interns walk through the door with no clue what triphenyltin acetate can do, picking up bottles bare-handed. After real training sessions that show the risks up close, their habits change for the better. Regular safety training, clear signage, and open discussions about close calls create a culture where caution becomes automatic—not an afterthought.
A Shared Responsibility
Taking shortcuts with toxic chemicals like triphenyltin acetate always seems tempting when workloads pile up, but nothing slows down progress like an accident. Keeping everyone aware, giving new staff practical hands-on training, investing in clear safety procedures, and checking up on storage and ventilation all add up. That takes commitment from every worker and manager, not just “the safety person.” Trust can only grow in a lab when everyone knows the risks and tackles them together.
Is Triphenyltin Acetate hazardous to the environment?
A Closer Look at Triphenyltin Acetate
Triphenyltin acetate shows up in the world of pesticides and fungicides, especially in agriculture. Its main job includes controlling unwanted fungal growth on crops and protecting materials like wood from rot. Anyone with a background in chemical safety or farm work has seen debates about its risks. The name sounds technical, but what matters sits in how it behaves once released into the ecosystem. If you consider chemicals that stick around in soil and water, triphenyltin compounds land among the more persistent ones.
Staying Power in the Environment
Once introduced, triphenyltin acetate doesn’t break down quickly. In water, it binds with sediments, hanging around long enough to affect aquatic life. Some studies have tracked it staying active for weeks, even months, depending on sunlight and temperature. That means it builds up, and fish or shellfish can pick it up over time. I’ve seen environmental surveys showing sharp drops in aquatic population numbers where these compounds were regular players. The U.S. Environmental Protection Agency lists triphenyltin as a hazardous pollutant, not for bureaucracy’s sake, but for its track record in harming ecosystems.
What Actually Happens to Wildlife?
Wildlife can’t filter out toxins the way water treatment plants filter tap water. Triphenyltin targets a basic piece of biology: the nervous system. Research out of Japan and Europe links triphenyltin to stunted growth, abnormal development, and even death among aquatic creatures. Bottom feeders, like snails and some small fish, take the first hit since they live closest to contaminated sediments. Over time, the chemical crawls up the food chain, eventually landing on dinner plates for predators and people alike. Health risks to humans include immune system disruption and hormone changes, based on studies in animals. That’s enough for regulators in several countries to call for tighter controls or outright bans.
How Farmers and Industries Use It
On the farm, triphenyltin acetate’s appeal comes from its power to protect high-value crops. In the timber industry, it keeps pests away from wood. Short-term, these seem like wins. In practice, communities near treated fields or industries using these chemicals have sounded alarms about health and water safety. Growing up in a farming community, stories of “off-color” wells or sick livestock often came with whispers about persistent chemicals.
Finding Better Paths Forward
Alternatives exist, but switching costs money. Safer fungicides and integrated pest management can reduce chemical fallout. Research from universities in California and the Netherlands both show that combining better crop rotation with biological controls drops fungus levels without the long-term consequences tied to organotin compounds. On a larger scale, stricter rules and better monitoring of runoff help spot problems early. I’ve watched NGOs organize testing days, turning science into community action. These grassroots projects bring facts to local decision-makers, speeding up responses when contamination threatens food or water.
Making Choices with Future Generations in Mind
Triphenyltin acetate shows just how much chemistry and ecology depend on each other. Decisions made for today’s short-term gain echo through the fields, rivers, and diets of tomorrow. Solutions begin by accepting what the evidence makes clear: some chemicals bring more trouble than any immediate benefit. Local knowledge and proven science, paired with honest conversations, can build safer, more resilient communities.
How should Triphenyltin Acetate be stored?
Understanding What’s at Stake
Triphenyltin acetate belongs to a class of organotin compounds known for both their effectiveness in industrial use and their reputation for toxicity. Exposure can lead to serious health risks, including respiratory irritation and organ damage. I’ve come across the consequences of improper chemical storage more than once in lab settings—spills, ruined floors, and worst of all, health scares. Overlooking storage rules isn’t just careless, it invites trouble into homes, storage rooms, and workplaces.
Picking the Right Container
A chemical with this toxicity level can react with metal and light, so always turn to tightly sealed glass or high-density polyethylene (HDPE) containers. Never touch unlabelled bottles; misplaced faith in memory leads to confusion and possible accidents. Labels should survive splashes and remain legible for the long haul. My old habit of scribbling with a marker vanished after a single solvent leak washed out my notes and left me guessing what was inside a jar.
Location Makes the Difference
A quiet, cool, and dry place makes the safest home for this chemical—far from sunlight, moisture, and heat sources. Moist air feeds chemical degradation, while direct sunlight may trigger hazardous reactions. I’ve seen colleagues pack harsh reagents together, creating a cocktail of risks. Avoid stacking or crowding containers, since one topple can break a bottle and start a chain reaction. Separate storage of flammable, corrosive, and toxic materials keeps things under control, as safety manuals recommend.
Good Practice—Not Just Good Habits
Ventilated chemical cabinets outfitted with locking mechanisms limit unauthorized access. Kids, pets, and even uninformed adults end up in areas they shouldn’t if cabinets offer no resistance. In my experience, nothing replaces a quality lock and a clear ‘Toxic—Authorized Access Only’ sign. Storing incompatible substances together—like oxidizers with organotin compounds—has caused dangerous fire accidents at several facilities, most avoidable with a little forethought.
Why Personal Protection Matters
Storing hazardous chemicals is no small task. Gloves and goggles protect against splashes while moving or inspecting the containers. Always check for damaged seals, rust, or cracks. Once, a leaking bottle wasted hours of clean-up and left an acrid odor that wouldn’t disappear. It’s tempting to rush, but slow, deliberate handling saves time and nerves later on.
Spill and Emergency Readiness
Spill kits and absorbent materials should remain nearby, not tucked in some forgotten cabinet. Quick access to these tools means a minor mishap doesn’t turn into an emergency. Regular checks for expired chemicals, corroded containers, or damaged labels keep everyone safe and save money by reducing spoilage or cleanup costs. No substitute exists for up-to-date emergency plans posted on storage room doors. Periodic drills keep those plans fresh in everyone’s mind.
Bottom Line
Storing triphenyltin acetate isn’t just a matter of compliance; it’s common sense built on respect for hazardous substances. If rules seem strict, it’s because experience has proven what happens when corners get cut. Every clear label, every reinforced shelf, every checked lock keeps both chemicals and people safe. Sometimes solutions are as simple as slowing down, labeling thoughtfully, and choosing the right spot.
What is the chemical structure of Triphenyltin Acetate?
Understanding the Backbone: A Closer Look at Triphenyltin Acetate
Triphenyltin acetate doesn’t often make headlines, but anyone with a background in chemistry or environmental science has crossed paths with it. Its chemical formula, (C6H5)3SnOCOCH3, packs a lot of story into a small space. At its core, the molecule combines three phenyl rings linked to a tin atom—hence the “triphenyltin” part. The acetate group hangs off this tin anchor, forming the compound’s functional package.
Picture three benzene rings jutting out from a central tin atom like spokes on a wheel. Acetate comes in as a small but crucial member, featuring a carbonyl group paired with a lone methyl branch. This structure isn’t just a drawing on a chemistry exam—it defines how the molecule interacts with the world, especially with living systems and the environment. The phenyl rings provide bulkiness and lend lipophilic character, nudging the molecule to dissolve more in organic materials than in water. The central tin gives the whole structure weight and reactivity, while the acetate tweaks its activity.
Why Should We Care What It Looks Like?
People have used organic tin compounds like triphenyltin acetate to combat pests and fungus, particularly in agriculture and as a marine antifoulant. Its structure isn’t just a chemical curiosity—the arrangement directly shapes its toxicological profile. The bulky phenyl rings shield the tin, making the compound more stable and less likely to break down quickly in the environment. Stability means triphenyltin acetate can linger for a while, building up in soil or water. Studies from agencies like the US EPA and academic chemical journals report the persistence and bioaccumulative tendencies of organotin compounds, flagging them as environmental red flags.
The three phenyl rings, tied so tightly to tin, allow it to slip into fatty tissues in wildlife and humans. This isn’t just trivia; it means exposure can result in higher-than-expected bioaccumulation, a point the World Health Organization has raised in toxicity reviews. As a chemist, seeing phenyl-heavy tin compounds often means dialing up concern for chronic exposure. And environmental techs worry too—structure predicts fate, and fate determines where the poison goes.
Facing the Risks: Finding Smarter Paths
Handling toxic chemicals demands both respect for their power and a hunger for safer solutions. Structure explains much of their behavior, so understanding it helps direct efforts to replace old, risky compounds with molecules tailored for lower risk and easier degradation. Reworking the structure—removing phenyl rings or swapping out tin—could open doors to alternatives that break down more readily in the environment. Throughout the past two decades, research teams have pushed for greener approaches, focusing on making molecules less harmful by design. Regulations tied to scientific reviews and industry safety calls have limited triphenyltin acetate’s reach, based directly on what its structure reveals.
Solid science brings clear answers about health and environmental risks. That’s where experience and built-up evidence matter. Scrutinizing the chemical backbone of triphenyltin acetate does more than solve a textbook riddle—it shapes policy and guides the search for more responsible chemistry that doesn’t leave a toxic legacy trailing behind.
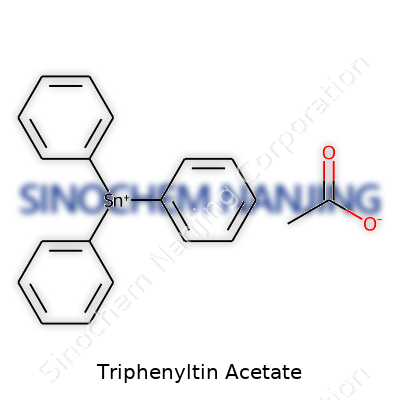
| Names | |
| Preferred IUPAC name | Acetyloxytriphenylstannane |
| Other names |
Fentin acetate Acetic acid triphenyltin ester Acutin Brestan TPTA Fenacet |
| Pronunciation | /traɪˈfɛnɪl.tɪn ˈæsɪteɪt/ |
| Identifiers | |
| CAS Number | 639-58-7 |
| Beilstein Reference | 1914126 |
| ChEBI | CHEBI:34720 |
| ChEMBL | CHEMBL4206882 |
| ChemSpider | 22006 |
| DrugBank | DB13521 |
| ECHA InfoCard | 100.011.345 |
| EC Number | 208-927-1 |
| Gmelin Reference | 324128 |
| KEGG | C19597 |
| MeSH | D014278 |
| PubChem CID | 66261 |
| RTECS number | TP8750000 |
| UNII | 8GB0M0DS8N |
| UN number | UN2588 |
| Properties | |
| Chemical formula | (C6H5)3SnO2CCH3 |
| Molar mass | 591.11 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.32 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 3.9 |
| Vapor pressure | 2.1 × 10⁻⁷ mmHg (25°C) |
| Acidity (pKa) | 6.1 |
| Basicity (pKb) | 4.11 |
| Magnetic susceptibility (χ) | -74.0e-6 cm³/mol |
| Refractive index (nD) | 1.627 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.92 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -578.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1628 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | S01AE05 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P310, P330, P362+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 2-2-2 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 280 °C |
| Lethal dose or concentration | LD50 oral (rat): 160 mg/kg |
| LD50 (median dose) | LD50 (median dose): 160 mg/kg (oral, rat) |
| NIOSH | WK2625000 |
| PEL (Permissible) | PEL: 0.1 mg/m3 |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 25 mg/m3 |
| Related compounds | |
| Related compounds |
Triphenyltin chloride Triphenyltin hydride Triphenyltin hydroxide Tributyltin acetate |

