Triphenylphosphine: An Essential Pillar of Modern Chemistry
Historical Roots and Development
Triphenylphosphine did not enter laboratories by accident. The chemical world has grown up around building blocks like this, and its history stretches back to the early 20th century. Chemists in those days were hungry for organometallic compounds that would open new chapters in synthesis, catalysis, and research. People discovered triphenylphosphine as part of a wave of experiments probing the physical limits between organics and inorganics. The aromatic, sturdy nature of those three phenyl groups bonded to a phosphorus atom gave researchers a versatile tool. By the 1950s and ’60s, labs across Europe and America leaned on the reliability of triphenylphosphine when shaping the future of applied and theoretical chemistry. Its name quickly became part of the essential vocabulary of a working chemist.
Product Overview and Practical Chemistry
Triphenylphosphine shows up as a powdery white solid, almost crystalline, sometimes with a faint, not quite unpleasant scent. It mixes well with organic solvents, like benzene or toluene, and resists water. The phosphorus at the heart of its structure bonds tightly with three phenyl groups, forming a stable molecule that stands up to moderate heat and everyday laboratory jostling. The appeal goes deeper than its form. Researchers prize it for the control it brings to synthetic reactions, whether acting as a ligand in transition metal complexes or driving reductions and functional group transformations.
Physical and Chemical Properties
Good chemistry begins with solid knowledge of what makes a substance unique. Triphenylphosphine melts at around 80 degrees Celsius, and its molecular weight clocks in at 262.29 g/mol. The compound dissolves well in chlorinated solvents, ether, and other organics—a detail anyone weighing out a reaction mixture can appreciate. Phosphorus gives it its edge: that lone pair on phosphorus makes the molecule an efficient nucleophile. These features mean that triphenylphosphine doesn’t just sit as a dusty bottle on a shelf. It goes out and gets involved in the chemical action, transferring atoms, changing bonds, and shaping new molecules.
Labeling and Handling on the Lab Bench
A bottle labeled “Triphenylphosphine, C18H15P” with hazard information and proper storage advice stands as a staple in academic and industrial environments. I recall many sessions in graduate labs where the telltale crystalline appearance of triphenylphosphine marked the starting point of a long afternoon of synthesis. Its packaging, hazard statements, and storage conditions don’t originate in bureaucratic whim—they reflect decades of practical lab experience. Store it away from humidity, in tightly sealed containers, and keep records of use for safety’s sake. These standards emerge from the reality that even mild organic powders have quirks and risks worth respecting.
How Chemists Prepare Triphenylphosphine
Right from the beginning, making triphenylphosphine wasn’t a chore for the faint of heart. The main route runs through the reaction of phosphorus trichloride with phenylmagnesium bromide. This Grignard reaction stands as a classical technique. Magnesium shavings, ether, controlled addition—chefs in the chemical kitchen have to mind their steps, but the process remains robust. Over time, methods improved, scaling for commercial manufacture needs and minimizing byproducts. Synthetic chemists keep tweaking the process, chasing greater purity and greener conditions. That search underlines a simple fact: nothing in chemistry stays static long.
Chemical Reactions and Transformations
Triphenylphosphine gained stardom thanks to how easily it slips into complex reactions. As a ligand, it pairs with metals, from palladium to platinum, forming complexes that drive cross-coupling reactions like Suzuki and Heck with astonishing reliability. The compound shows no hesitation when scavenging for allies, pairing with alkyl halides to form phosphonium salts—key intermediates in the Wittig reaction. Wittig chemistry opened new territory for making alkenes, letting generation after generation of chemists access molecules previously locked behind synthetic barriers. On my own bench work, using triphenylphosphine for azide reductions or as a gentle reducing agent has provided remarkable improvements in yield and selectivity. Each reaction has its quirks, but triphenylphosphine stands as a go-to reagent to untangle many synthetic puzzles.
Synonyms and Names in the Literature
Chemists know triphenylphosphine under a handful of aliases. Some catalogs and papers list it as TPP. Others reference it as triphenylphosphan, reflecting variations in international nomenclature. Walking through the older literature, “Ph3P” remains a familiar shorthand, a shortcut scribbled on countless blackboards and experimental notebooks. Clarity in naming matters because confusion over synonyms in chemical ordering or literature searches can cost time and sometimes much more in misdirected experimentation.
Working With Triphenylphosphine: Safety as Habit
Nobody who works with chemicals should treat safety as an afterthought. Triphenylphosphine, though not the most hazardous molecule on the block, still deserves respect. The powder can irritate the skin and respiratory tract if mishandled, and it takes well-designed organization to keep containers, weighing boats, and spatulas clean. Inhalation, contact, or accidental ingestion can bring headaches, nose and throat discomfort, and the occasional allergic reaction. Safety glasses, gloves, well-ventilated hoods—these practices don’t just follow written standards, they allow chemists to push their research with confidence. The best labs I’ve worked in take this approach as gospel: treat each bottle as a challenge to do things right.
Applications: From Bench to Industry
The reach of triphenylphosphine covers a lot of ground. In pharmaceutical manufacturing, its ligands turn dull reactions into productive workhorses, enabling synthesis of critical intermediates on industrial scales. Agrochemical developers also rely on triphenylphosphine for efficient syntheses of bioactive molecules, lowering costs and streamlining development timelines. In specialty materials, triphenylphosphine-based catalysts helped bring new polymers and advanced electronics beyond the prototype stage. My own experience seals its reputation—as part of a small team optimizing routes to a novel aromatic compound, we found that triphenylphosphine outperformed higher-priced alternatives, made work-ups simpler, and shaved weeks off development cycles. Not every chemical delivers so much utility across such varied landscapes.
Pushing the Boundaries: Research and Technical Progress
Academic curiosity keeps triphenylphosphine at the frontlines. Advancements in transition-metal catalysis rely on its adaptability as a ligand. Researchers have explored modified analogues—sterically hindered, electronically tuned—to open new mechanistic doors that the original compound could not unlock. As pharmaceutical needs grow more sophisticated, chemists keep turning to triphenylphosphine in crafting smaller, more selective, and more active versions of itself. This ongoing dance between structure and function defines much of research in organophosphorus chemistry. Having worked alongside postdocs chasing better catalysts for cross-coupling, I’ve seen how a single tweak to a phenyl ring or the phosphorus center can shift a whole reaction paradigm. That creative process keeps pushing boundaries and rewriting textbooks.
Toxicity and Human Risk: Facing the Data
A seasoned chemist learns to separate rumor and reality. Triphenylphosphine has drawn criticism over the years due to its moderate toxicity. Data show that its hazard level remains manageable with standard precautions. Acute exposure produces mild skin and mucous irritation, with rare but reported allergic reactions after repeated exposures. Chronic toxicity studies are less conclusive, yet animal testing on related organophosphorus compounds makes careful handling the smart call. Waste management ensures that residues do not slip into the food chain or groundwater. Training young chemists on responsible use and giving them context about measured risks helps keep accidents in check without spreading fear or losing sight of efficiency.
Looking Forward: Prospects and Challenges
Triphenylphosphine’s story keeps evolving along with the needs and ambitions of the chemical industry. Interest in sustainable syntheses gives new urgency to finding greener preparation routes, aiming for less solvent use and fewer hazardous byproducts. Designing new ligands with improved selectivity, faster turnover, and increased stability remains a hot research area, and triphenylphosphine derivatives frequently headline those developments. As green chemistry grows into a guiding principle, the push continues to minimize the environmental impact of organophosphorus compounds, including effective recycling strategies. Triphenylphosphine won’t lose its relevance any time soon—it stands not just as yesterday’s hero, but a continuing force for innovation wherever molecular construction matters.
What is Triphenylphosphine used for?
Why Chemists Trust Triphenylphosphine
If you step into any college or industrial lab, odds are you’ll spot a bottle labeled Triphenylphosphine somewhere on the shelf. The name is a mouthful, but ask any organic chemist and they’ll tell you it’s as familiar as old jeans. It’s part of the reason many modern medicines and materials even exist.
Pharmaceutical Building Blocks
I first encountered Triphenylphosphine in an undergraduate organic lab. The job? Help turn normal alcohols into much more interesting chemicals called alkenes. This wasn’t just an academic exercise. In drug development, the ability to make small, precise changes to molecules often separates a test compound from a future cancer therapy. Triphenylphosphine makes reactions like the Wittig reaction possible, turning boring carbonyl groups into double bonds in just a few steps. Factories making antibiotics, antivirals, or anti-inflammatory drugs rely on this chemistry because it offers control. If the end product has to hit a specific mark—purity, safety—Triphenylphosphine gets things there with fewer headaches.
Catalyst for Innovations
Outside the medicine cabinet, Triphenylphosphine helps manufacture plastics and advanced materials. It’s a staple in transition metal catalysis, where it binds to metals like palladium or rhodium to speed up reactions. These metal complexes help put together tough carbon frameworks with fewer steps and less waste, something that keeps both costs and environmental impact lower. Looking at the roster of products—polymers, electronics, specialty coatings—most take shape in part through steps that lean on Triphenylphosphine’s unique abilities.
Cleaner Reactions, Lower Waste
Old-school chemical manufacturing burned through resources and spat out nasty byproducts. Today, pressures for greener processes make chemists re-examine every bottle in the lab. Triphenylphosphine stands out because it packs a combination of strength and stability. By easing tough transformations, reactions require milder temperatures and fewer harsh chemicals. This drops energy bills and slicing waste output. My own work in process-development showed just how much difference this could make: switching out a clunky older reagent for Triphenylphosphine simplified isolating the target compound and reduced leftover contaminants.
Risks and Challenges
Triphenylphosphine isn’t perfect. It reacts with air—keep the cap tight—and the leftover byproduct, Triphenylphosphine oxide, builds up fast in labs and factories. You don’t want barrels of solid waste clogging up storage. Research teams now look for ways to recycle or reuse these byproducts in agriculture or other sectors. Some up-and-coming catalysts using phosphorus alternatives aim to bring the same benefits without the environmental baggage.
Facing the Future
Triphenylphosphine has enabled breakthroughs, but the scientific community recognizes room for improvement. Clear labeling, ventilation, training for workers, and investment in safer, greener alternatives all deserve attention. At its best, this chemical transforms how we cure disease and create modern conveniences. Its story proves that the right tools in skilled hands lead to simpler, smarter, and safer chemistry for everyone.
What is the chemical formula of Triphenylphosphine?
Understanding Triphenylphosphine’s Roots
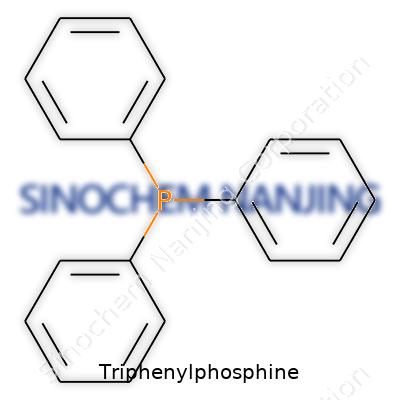
Triphenylphosphine is a staple for many chemists and lab workers. Its formula, C18H15P, pops up in texts and laboratories all over the world. Imagine a molecule built around a phosphorus atom, surrounded by three big benzene rings. That’s the backbone, and it’s changed how we approach a bunch of important chemical transformations. The way these phenyl groups stick to the phosphorus gives it its unique behavior. I’ve worked with this compound in reactions that needed something reliable and predictable, and it comes through every time — not with a bang, but with steadfast consistency.
A Real Player in Modern Chemistry
Triphenylphosphine isn’t flashy. It doesn’t bring drama. Yet, its role affects more chemistry than many would guess. It’s designed for organic synthesis, especially in processes like the Wittig reaction, which helps build organic molecules by stitching carbon atoms together. This reaction is essential for making building blocks in pharmaceuticals, agricultural chemicals, and even new plastics. In many research labs, students quickly learn that having a bottle of C18H15P handy expands the possibilities. It works hard behind the scenes—converting, reducing, and transforming molecules quietly while breakthroughs happen on the lab bench.
Why Accuracy in Chemical Formulas Matters
Mixing up numbers or elements in a chemical formula can derail more than just a lab experiment. With triphenylphosphine, getting the formula right—C18H15P—means avoiding wasted time, spoiled reagents, or worse, safety mistakes. Chemical formulas act as universal shorthand, making sure every scientist, from New Delhi to New York, speaks the same language. It’s not just about memorization, either. During my studies, I learned quickly that misunderstanding a formula could mean missing the point of a reaction or producing unwanted results.
Looking for Solutions and Careful Use
Triphenylphosphine has some quirks. It doesn’t dissolve well in water, and if exposed to too much air, it starts to break down. Handling this compound calls for basic knowledge and respect—simple gloves, closed containers, and a clean bench. There’s also a push these days to cut down on waste and limit exposure. Chemists find greener solvents, use smaller amounts, and recycle whenever possible.
Education also plays a huge part. Accurate labels in classrooms, guidelines for storage, and up-to-date safety sheets set young chemists on the right path. More experienced hands pass down practical tips: don’t use it near strong acids, keep a lid on the container, and always wipe up residue. Passing down practical knowledge from one generation to the next gives everyone a better shot at handling chemicals safely and responsibly.
Impact Beyond the Lab
Triphenylphosphine’s reach extends far outside academic circles. Its formula appears in patents, manufacturing specs, and supply sheets for companies making life-saving drugs and everyday products. Precision and care for detail guide its movement through these global supply chains. By focusing on accuracy and teaching good practices, the scientific community keeps chemistry safe, effective, and ready to serve both present and future needs.
Is Triphenylphosphine hazardous or toxic?
Understanding the Chemical
Triphenylphosphine shows up often in chemistry labs and factories. It’s a trusty tool for chemists, especially in making pharmaceuticals, plastics, and a handful of other things most of us don’t notice in our daily routines. Its white, crystalline look makes it easy to recognize in a well-organized lab shelf. Still, beneath that clean appearance sit some very real risks.
What Makes It Hazardous?
Direct contact with triphenylphosphine can irritate skin and eyes. Breathing in its dust may upset the lungs and airway, bringing on coughing or shortness of breath – not a fun scenario for anyone working in chemistry. This isn’t just speculation. According to safety data, it has knocked people off their stride with headaches, dizziness, and nausea. Researchers at the NIH and several industrial safety agencies warn that the irritant factor is undeniable, especially if the workspace isn’t well-ventilated or basic precautions get skipped.
Its toxicity comes into play with bigger doses or prolonged exposure. If you push your luck and inhale a lot or spend days around it without gloves or proper gear, it can impact organs like the liver and kidneys. Animal studies, cited by occupational safety groups, show persistent exposure led to damage in both. Such examples underline why lab routines lean so heavily on gloves, masks, fume hoods, and solid cleanup habits.
Toxicity in Practice
Not all chemicals with bad side effects are automatically handled with the same caution by everyone in industry. I’ve worked alongside new chemists who shrugged off protective gear after a few months, thinking routine breeds safety. Every time I’ve witnessed that, the stories turned sour. Red eyes, sore skin, and a few near-misses with breathing trouble. Not every accident ends up as a statistic, but the stories travel fast through the lab.
Accidental spills have occurred. A friend once splashed a small amount on his arm and, assuming quick washing would be enough, kept working. The burning sensation didn’t go away for hours. So, yes, even ‘minor’ mishaps leave an impression.
Prevention Beats Damage Control
Staying safe isn’t about treating chemicals like monsters but recognizing their bite. Chemical manufacturers and regulatory agencies like OSHA call for strict labeling and solid ventilation setups. Most good labs follow a clear check-list: gloves, goggles, lab-specific coats, and regular refresher training. The threat drops a lot when these steps aren’t skipped.
Outside the lab, triphenylphosphine rarely appears. The odds of someone running into it at home or on the street are almost nil. Still, the risks it poses in industrial and research spaces hang over every container just waiting for a shortcut or slip-up.
Looking Ahead
The best fix lies in culture, not just safety data sheets. Training matters more than warnings pasted on bottles. Chemists and workers swapping stories about what’s gone wrong usually keep each other sharper than rulebooks do. I’ve learned more in casual conversations after an incident than any binder on the shelf ever offered.
Even as research into safer alternatives grows, triphenylphosphine remains a go-to in plenty of essential processes. Until a day comes that we retire it for something safer and just as effective, respect for its hazards and regular education are the best defense.
How should Triphenylphosphine be stored?
Why Storage Matters for Triphenylphosphine
Triphenylphosphine shows up in many labs thanks to its role in organic synthesis, most famously in Wittig reactions. This white crystalline powder seems harmless on the bench, but looks can fool. It reacts slowly with air and moisture, forming phosphine oxide — a byproduct that introduces headaches in purification and can ruin reactions.
Many chemists, myself included, learned the hard way about the price of sloppy storage. In my early days, I regularly found old containers with clumpy, off-white powder — a sure sign of moisture sneaking in. Disturbed reactions, odd yields, and extra time spent filtering out junk soon followed. Productive days get eaten up by preventable mistakes.
Solid Steps for Reliable Storage
Start with a strong, airtight container. Borosilicate glass works better than regular plastic for long-term storage, since some plastics let in tiny amounts of air over time. Child-resistant caps don’t hurt, but make sure the seal is tight. Screwcaps lined with PTFE or a solid rubber gasket help seal out moisture.
Find a dry, dark spot for the container. Triphenylphosphine doesn’t demand deep freeze, but low humidity makes all the difference. A desiccator charged with silica gel or another drying agent offers extra insurance. Exposing the chemical to air and light, even a little at a time, speeds up oxidation and spoils the batch. Light-proof, amber glass bottles cut down on UV damage too. I keep mine on a lower shelf, away from heat or direct sunlight, not up near the fume hood or above a radiator.
Reagents with oxygen scavengers can extend shelf life in difficult climates, though regular checks matter too. I mark purchase dates right on the container in permanent marker. An eye on color and texture flags trouble early. If the powder clumps or turns yellowish, problems have started. It makes sense to label containers with hazards and storage tips, especially in busy community labs where personnel change often.
Seeing the Bigger Picture: Safety, Cost, and Performance
Poor storage leads to more than wasted chemicals. Triphenylphosphine dust irritates skin and eyes, so spills from cracked, brittle or open containers pose a hazard. People handle dozens of bottles every day — the margin for error shrinks as the workplace gets busier. Upset reactions mean more failed syntheses and wasted time, driving up costs. Students and researchers learn caution by handling sensitive reagents right, which reinforces a culture of respect for chemicals throughout the lab.
Good storage pays off. One clean, dry batch can support months of research without interruption. It saves money, cuts down on waste, and produces reliable results. Talk to the more seasoned staff in your lab if new processes are confusing. Even a few small tweaks — a better cap, a drier shelf, a clearer label — can keep triphenylphosphine fresh and safe, making life smoother for everyone down the line.
What Keeps Labs Accountable
Institutions carry responsibility for lab safety, but culture comes from people. It only takes a single careless moment to spoil weeks of work or risk a nasty exposure. Reliable labeling and clear policies help, but every chemist develops habits through practice and shared experience.
Investing in proper storage gear costs less than buying replacement reagents or combing through failed experiments. Simple vigilance and proactive labeling save frustration and, sometimes, prevent true emergencies. Protecting chemicals like triphenylphosphine at the source keeps research flowing and people safe — a lesson every lab can take to heart.
What are common applications of Triphenylphosphine in organic synthesis?
The Appeal of Triphenylphosphine in the Chemistry Lab
Anyone who's spent real time in an organic chemistry lab has probably opened a bottle of triphenylphosphine. Its white, crystalline appearance and faintly musty smell are almost familiar fragrances in advanced courses or synthetic research. Unlike pure textbook chemicals, this one gives practical value that goes beyond the classroom. It’s a staple because it delivers on tough, real-life problems synthetic chemists run into all the time.
Making Reactions Work—The Wittig Reaction
Take the Wittig reaction as an example. This application produces alkenes by stitching together aldehydes or ketones with phosphonium ylides, made from triphenylphosphine. In my grad years, I learned quickly how this route can build carbon–carbon double bonds with more control than simple elimination reactions. Pharmaceutical chemists rely on this for making the building blocks found in antibacterial and antiviral medicines. The reason: triphenylphosphine offers unique control over stereochemistry, a key factor for active drug molecules.
Reductive Tools—The Staudinger Reaction
Some stubborn molecules, like azides, refuse to cooperate without a little extra help. Triphenylphosphine pairs up with azides to generate amines through the Staudinger reaction. That secondary product, an iminophosphorane, comes out cleanly and leaves behind fewer side products than more aggressive reductants. This feature pops up a lot in peptide synthesis, where chemoselectivity demands attention. Biochemists dealing with sensitive amino acids find triphenylphosphine far less destructive to other groups on the molecule.
Activation and Deoxygenation—Mitsunobu and Beyond
Another staple in my experience, especially in labs driven by complex natural product chemistry, is the Mitsunobu reaction. Triphenylphosphine teams up with diethyl azodicarboxylate (DEAD) to convert alcohols into esters, ethers, or other derivatives—often with complete inversion of configuration. No other reagent system achieves this level of versatility so reliably for secondary alcohols. These transformations show up in total syntheses of antibiotics and steroids, where the difference between success and failure hinges on one oxygen atom’s position.
Deoxygenation—Appel and Related Reactions
Oxidations and reductions can frustrate even careful chemists. For deoxygenating alcohols to halides, the Appel reaction introduces a combination of triphenylphosphine and a halogen source (usually CCl4 or BrCCl3). In my own work with challenging alcohols, this method consistently produced clean, predictable yields—something not every traditional halogenation delivers. The controlled reaction conditions keep functional groups safe from overreaction, an advantage in growing complex molecules where every functional group counts.
Cleaner Workups, Sustainability Questions
Every experienced chemist knows the struggle with triphenylphosphine oxide, a white byproduct notorious for clogging up purifications. Last year, I tried newer extraction techniques with biphasic solvents that separate the oxide, but the waste still piles up. In industry, managing byproducts and solvent load directly affects project cost and environmental risk. Green chemistry researchers continue to push for recyclable phosphines or catalytic systems to cut down on phosphorus waste. Successful developments here will transform triphenylphosphine from a necessary evil into a more sustainable staple of synthesis.
The Practical Chemistry Perspective
Ask any organic chemist about triphenylphosphine, and the feedback comes quickly—it's reliable, affordable, and proven. Its broad application covers pharmaceuticals, agrichemicals, and materials science, all industries where tailored synthesis can unlock new properties and higher value. Standard protocols with triphenylphosphine show up in published research from labs worldwide, underscoring its international status as a workhorse reagent. Finding a better, greener approach remains an ongoing project, but for now, triphenylphosphine keeps delivering on its promises.
| Names | |
| Preferred IUPAC name | Triphenylphosphan |
| Other names |
Triphenylphosphorus TPP Triphenyl-phosphane Phosphorane, triphenyl- Triphenylphosphan Phenylphosphine, triphenyl- Triphenylfosfine |
| Pronunciation | /traɪˌfiːnɪlˈfɒsfiːn/ |
| Identifiers | |
| CAS Number | 603-35-0 |
| Beilstein Reference | 1208733 |
| ChEBI | CHEBI:44718 |
| ChEMBL | CHEMBL1387 |
| ChemSpider | 546280 |
| DrugBank | DB03837 |
| ECHA InfoCard | 100.042.292 |
| EC Number | 207-989-7 |
| Gmelin Reference | 82880 |
| KEGG | C00515 |
| MeSH | D014277 |
| PubChem CID | 6629 |
| RTECS number | TC3850000 |
| UNII | 9C6Q8ASK43 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C18H15P |
| Molar mass | 262.29 g/mol |
| Appearance | White crystalline powder |
| Odor | pleasant |
| Density | 1.188 g/cm³ |
| Solubility in water | Insoluble |
| log P | 5.7 |
| Vapor pressure | 0.01 mmHg (25 °C) |
| Acidity (pKa) | 7.6 |
| Basicity (pKb) | 15.0 |
| Magnetic susceptibility (χ) | -34.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.668 |
| Viscosity | 4.74 mPa·s (160 °C) |
| Dipole moment | 3.02 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 348.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -58.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1597 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P270, P271, P272, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0-0 |
| Flash point | 130 °C |
| Autoignition temperature | 300 °C |
| Lethal dose or concentration | LD50 oral rat 1,600 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 1,600 mg/kg |
| NIOSH | TT4200000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | Not established |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Phosphine Triethylphosphine Trimethylphosphine Triphenylphosphite Triphenylphosphine oxide |

