Triphenyl Phosphite: A Closer Look at a Classic Organophosphorus Compound
Historical Development
Triphenyl phosphite entered the chemistry scene around the early 1900s, shaped by the expanding demand for organophosphorus compounds in both industrial and academic settings. The late 19th and early 20th centuries drew chemists like Michaelis and Arbuzov into exploring esters of phosphorous acid, discovering new reactions and uses almost accidentally as the needs of advanced materials and synthetic applications challenged the basics. The growing push for plasticizers, stabilizers, and flexible polymers during the boom years of plastics in the mid-20th century meant investment poured into understanding this class of chemicals, and Triphenyl phosphite turned out to play a key role.
Product Overview
Triphenyl phosphite, an organophosphorus compound with the formula P(OC6H5)3, shows up as a colorless, sometimes faintly yellow, oily liquid or solid. Chemists and manufacturers rely on it for its high purity and ease of integration into a slew of products. Classic applications include its role as a stabilizing agent in PVC, antioxidants in lubricants, and as a ligand in homogeneous catalysis. The material’s low volatility and resistance to hydrolysis have earned it a place on factory floors and in academic labs alike.
Physical & Chemical Properties
Physical form depends on the temperature: in a bottle at room temperature, Triphenyl phosphite comes as a viscous, colorless liquid, but solidifies if chilled. It melts around 22°C and boils at 360°C under atmospheric pressure. Although it looks innocent, a faint smell gives away its chemical nature, reminiscent of other phosphorus esters. Solubility extends to common organic solvents like benzene, toluene, and chloroform, yet water can trigger a slow but persistent breakdown. Chemically, it’s not terribly reactive at mild conditions, but it steps up in stronger acid or base environments, showing hydrolysis that releases phenol and phosphorous acid. Managing these traits makes routine handling safe and predictable, granted operators follow basic chemical safety rules.
Technical Specifications & Labeling
Reliable supply chains mean technical-grade Triphenyl phosphite arrives with clear documentation listing purity—often above 98%—and maximum allowable impurities, such as residual phenol or trace metals. Each drum or bottle comes tagged with batch numbers, manufacturing date, and recommended storage parameters. Regulatory markings, hazard pictograms for irritancy and aquatic toxicity, and standard GHS labels appear on good packaging. Storage conditions usually stress cool, dry, dark environments, because heat and light accelerate breaking down the compound, leading to off-odors and loss of function. Unopened containers generally last two years or more if stashed well.
Preparation Method
The manufacture of Triphenyl phosphite typically follows a direct route: phenol, itself cheap and plentiful, reacts with phosphorus trichloride under controlled temperature and inert atmosphere. Phosphorus trichloride is dripped slowly into a cooled flask charged with excess phenol and a basic scavenger like pyridine, which absorbs hydrogen chloride gas that forms as a byproduct. The product is then separated, washed to remove impurities, and distilled under reduced pressure to net a high-purity material. This straightforward synthesis echoes back decades and continues serving the needs of bulk chemical plants globally.
Chemical Reactions & Modifications
Triphenyl phosphite acts as more than just a static additive. In the lab, it finds use as a reactant and a ligand. Its ability to donate electron density to metal centers makes it valuable in organometallic chemistry, where it tunes the reactivity of rhodium, palladium, and nickel catalysts. The Michaelis-Arbuzov rearrangement features this very reagent: here, Triphenyl phosphite reacts with alkyl halides, transforming into organophosphonates, powering the synthesis of flame retardants and biologically active molecules. Oxidation is another trick up its sleeve, allowing conversion to triphenyl phosphate and phosphine oxides, which play their own function in plastics and lubricants.
Synonyms & Product Names
Across different catalogs and scientific literature, Triphenyl phosphite might carry several names: Triphenoxyphosphine, TPPi, or Phosphorus acid triphenyl ester. Distributors like Sigma-Aldrich or Tokyo Chemical Industry might vary the spelling slightly but mean the same chemical. Looking up its CAS number 101-02-0 provides a consistent anchor, as do trade names in European and Asian markets. It pays to scan technical sheets and regulatory filings using all known synonyms, avoiding surprises in inventory or compliance audits.
Safety & Operational Standards
Workers who deal with Triphenyl phosphite face typical liquid handling hazards, like slips or accidental contact, but its potential for skin and eye irritation means gloves and splash goggles stay necessary. Inhalation of vapors, while not acutely toxic, can lead to headaches and mild respiratory complaints. Spills, if left alone, can leave persistent slicks and odorous residues. Regulatory agencies, including OSHA in the United States, set workplace exposure limits and promote good ventilation in production spaces. Disposal should avoid drains and be routed toward waste incineration, since aquatic toxicity poses a risk to streams and rivers. Every year, companies review safety data sheets to stay in step with evolving standards and keep incident rates low.
Application Area
Industries banking on the resilience of plastics lean hard on Triphenyl phosphite for its stabilizing properties, slowing down degradation and yellowing of PVC. Lubricant formulators squeeze more value from base oils with the antioxidant edge this chemical provides, stretching maintenance intervals and minimizing sludge buildup. Synthetic chemists swear by it as a go-to ligand for transition-metal catalysts in fine chemical synthesis. Even the electronics world charts uses for it in flame retardant research. From wire coatings to automotive interiors, Triphenyl phosphite works as a quiet enabler behind the scenes, never in the spotlight but always missed when absent.
Research & Development
The last ten years saw growing academic investigation into greener versions, swapping phenol for biobased substitutes or reducing environmental load in synthesis routes. Catalysis research teams at universities deploy Triphenyl phosphite in new coordination complexes, aiming for better yields in pharmaceutical reactions or lower energy demands in industrial processes. Polymer chemists have tested blends to improve weather resistance and mechanical properties. There’s bustling interest in integrating phosphorus esters into high-performance polymers or as scavengers for trace metal contamination. Reports pile up on attempts to modify its aromatic rings, seeking properties like better solubility or lower toxicity.
Toxicity Research
Toxicity data from longstanding animal studies and aquatic assays show that Triphenyl phosphite ranks as moderately toxic to aquatic organisms and mild to moderate in mammalian tests at higher doses. Chronic exposure studies for factory staff hint at irritation and, with substantial chronic exposure, potential liver changes in rodents. Regulatory bodies classified it as an irritant, not a carcinogen, setting workplace limits and enforcing strict labeling. Emerging research in the environmental fate of phosphorus-based stabilizers keeps pressure on both producers and regulators to monitor water systems and improve capture and treatment technologies. Safer alternatives and process containment get attention from sustainability groups, seeking to lower the overall risk signature tied to organophosphorus chemicals.
Future Prospects
Looking forward, Triphenyl phosphite sits at a crossroads with rising sustainability expectations and stricter controls on specialty chemicals. Researchers keep pursuing derivatives that carry similar performance but break down more safely, hoping to side-step durability conflicts in recyclables and avoid aquatic toxicity. Polymer plants and electronics makers track the regulatory winds, ready to pivot to newer stabilizer blends or adapt legacy supply chains as needed. Advances in catalytic systems keep this molecule on lab benches and in patent filings, even as the market shifts toward bio-based and low-impact additives. Bridging performance and responsibility does not give easy answers, but lessons learned from decades of Triphenyl phosphite shaping industrial chemistry will keep guiding industry and policymakers.
What is Triphenyl Phosphite used for?
What Makes Triphenyl Phosphite Important?
Triphenyl phosphite sounds obscure unless you spend time around plastics or lubricants. In truth, this compound has worked its way into many parts of manufacturing. I remember helping a friend in a plastics recycling plant and hearing about additives that improve heat resistance. Only after a little research did I realize that one of the key players in this game is triphenyl phosphite.
Triphenyl phosphite acts as an antioxidant and a stabilizer. Certainly, plastics don’t hold up forever, but triphenyl phosphite helps them last longer. Manufacturers blend it into polyvinyl chloride (PVC) and other polymers, aiming to keep the final product from breaking down too quickly under sunlight or heat. If you’ve ever replaced faded garden hoses or brittle plastic chairs, you’ve witnessed why stabilizers matter.
How Industry Uses Triphenyl Phosphite
Plastic manufacturers lean on this compound because of its ability to snatch up damaging free radicals that form during processing. Excess heat or light can lead to chain reactions, which eventually crack or discolor plastic. Triphenyl phosphite helps break that chain. Adding a dose to PVC or polypropylene lifts durability, making these materials last longer out in the world.
Its role isn’t limited to plastics. Some brands blend it into lubricants to slow down oxidation in machine oils. In mechanical settings, this means less sludge in machinery and fewer breakdowns. A few major lubricant companies still advertise triphenyl phosphite on their ingredient lists for just this reason. For anyone in a workshop or a factory, protecting gearboxes and engines from corrosion or gumming up matters more than most would think.
Downsides and Safety: What’s the Real Story?
Triphenyl phosphite, like plenty of chemicals, brings risks alongside benefits. Working with it means paying attention to air quality and contact precautions. Breathing too much dust or fumes causes headaches and irritation, and folks who handle industrial stocks must use proper ventilation and gloves. In my time on that recycling line, a chemical safety briefing hammered home the point—masks and gloves saved us plenty of pain.
Some researchers have raised questions about its long-term effects on health and the environment. Triphenyl phosphite can break down in certain conditions, leading to byproducts that might cause concern. European regulations keep a close eye on this substance, and companies across the globe must check their safety data sheets every time they order it.
Looking for Better Ways Forward
The plastics and lubricant industries depend on stabilizers like triphenyl phosphite, but sustainability conversations are heating up. Scientists and engineers are exploring new additives based on natural sources or improved chemical designs with lower potential hazards. For now, triphenyl phosphite remains on many “approved for use” lists because nothing else on the market matches its balance of protection and cost.
Consumers looking for greener products should keep an eye on updated labels and push for more disclosure. Regulators continue to test these additives and re-examine exposure limits as new research rolls in. Change happens slowly, but each time a safer alternative emerges in pilot studies or technical trials, there’s a better chance for reducing hidden chemical exposures.
Everyday Impact
From a factory floor to household goods, triphenyl phosphite stays behind the scenes, keeping things stronger and lasting longer. As new studies surface and alternatives improve, everyone—consumers, regulators, and industry workers—has a role to play in balancing durability with health and safety.
What is the chemical formula and structure of Triphenyl Phosphite?
Looking at the Formula
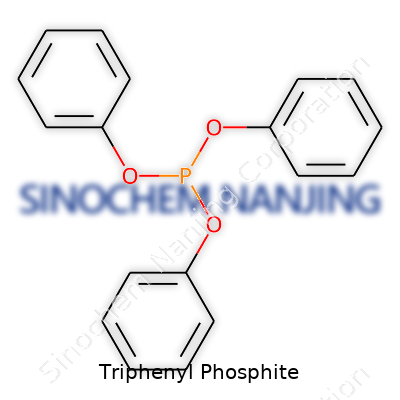
Triphenyl phosphite stands out in the world of organophosphorus compounds. Its chemical formula is C18H15O3P. Breaking that down, you have three phenyl rings, three oxygen atoms, and one phosphorous atom at the center. Imagine a central phosphorus forming stable bonds with three oxygen atoms, each of those oxygens linking up with a separate phenyl group.
Chemists usually draw the structure as a phosphorus atom with three single P–O bonds branching out. Each bond hooks to the oxygen of a phenoxy group (C6H5O–). It’s not simply a curiosity on a shelf; the arrangement influences everything it can do, from how it behaves as a stabilizer in plastics to its part in organic synthesis.
Why This Structure Matters
The structure means this molecule resists hydrolysis better than many phosphate-based cousins. As someone with experience making polymer blends, seeing triphenyl phosphite in an ingredient list always suggested a safeguard against reactive chemicals and heat. Its shape keeps the phosphorus protected, boosting the lifespan of the product.
No factory manager wants to shut down production because plastic has yellowed or broken down. Quality drops fast if the stabilizer can’t handle processing heat or humidity. Triphenyl phosphite performs reliably—even under tough conditions—because its molecular structure shields the sensitive phosphorus from attack.
Beyond industry, researchers lean on triphenyl phosphite for synthetic chemistry. In the lab, the molecule serves as a mild reducing agent. The robust structure lets it participate in reactions that shape everything from agricultural tools to medicine. That versatility flows straight from how phosphorus joins forces with phenoxy groups.
Supporting Facts and Solutions
Facts speak for themselves—triphenyl phosphite melts at about 22–30°C, boiling at around 360°C. That broad thermal tolerance lets manufacturers process plastic at typical production temperatures without losing any stabilizing power. High purity also protects against unwanted side reactions. Suppliers emphasize that careful handling matters, as oxidation can spoil the compound and drop performance.
Many modern wire and cable products wouldn’t survive without the reliable antioxidant properties triphenyl phosphite delivers. In PVC and other polymers, it prevents oxidation, blocks acid-catalyzed degradation, and guards the end product from turning brittle. Switching to triphenyl phosphite raises both the shelf life and the quality of the finished goods. For businesses, that means fewer complaints, less waste, and better confidence in their offerings.
Still, there’s always room to improve. Some reports flag concerns about potential skin or eye irritation. Good ventilation, gloves, and goggles remain must-have basics in a lab or plant using this substance. Improvements in packaging and delivery now help limit risks, keeping teams safe without extra hassle.
A Look Ahead
Advances in chemical engineering keep pushing the envelope. Researchers continue searching for even safer, more sustainable stabilizers. For now, triphenyl phosphite forms the backbone of countless applications because its chemical formula and structure give it staying power where others fall short. Understanding how that structure directs function pays off—from safer factories to higher-quality consumer products.
Is Triphenyl Phosphite hazardous or toxic?
Triphenyl Phosphite in Real-World Settings
Triphenyl phosphite steps into the scene mostly in plastics and rubber production. Chemists often look at this compound for its ability to protect materials from damage caused by heat and oxygen. Anyone who has worked around industrial additives gets familiar with the tang of these substances, and triphenyl phosphite proves no different. Anyone who pulls on gloves and goggles around this stuff already feels the tension between technological need and worker health.
Health Risks of Exposure
Direct contact sometimes causes eye and skin irritation. Inhaling dust or mist in a poorly ventilated workspace irritates the respiratory tract—burning in the nose or throat, coughing, sometimes even trouble catching your breath. Lab safety training hammers home basic facts: gloves, goggles, and proper air flow go a long way. Still, real-life lapses occur, especially in factories running full tilt.
Chronic exposure stories come up less often. There isn’t much evidence of cancer risk in humans linked to triphenyl phosphite, but odd cases of liver and kidney issues have floated in occupational medicine reports. Rodent studies hint at trouble after months of high exposures—liver changes, immune system oddities, weight shifts. Most workers never see these levels, but that doesn’t make complacency smart.
Environmental Concerns
Washing triphenyl phosphite down the drain rarely happens on purpose, but spills or leaks sometimes get into soil or water near industrial sites. Plants and aquatic creatures aren’t built to handle large doses. Even low levels upset the natural order, with potential for bioaccumulation. Unlike many durable chemicals, triphenyl phosphite breaks down a bit quicker once out in the wild, but the breakdown products aren’t always kinder to the environment. It pays to keep this substance out of waterways, not just for fish but for everyone downstream.
Ways to Protect Workers and Communities
Fixing hazards starts with basics most chemical plants already know: solid ventilation, spill containment setups, emergency eyewash and showers. Training every worker—especially new hires—matters more than any fancy notice on the wall. Companies investing in fume hoods, proper disposal routines, and regular air tests help their bottom line by keeping folks healthy.
Swapping less toxic additives where possible makes a big impact. The chemical industry isn’t shy about finding alternatives, and some offer similar antioxidant effects without the same baggage. Even small tweaks—tighter drum seals, better labeling, prompt cleanup—reduce chance of leaks or misuse.
Supporting Evidence and Ongoing Monitoring
Regulatory agencies like OSHA and NIOSH have set workplace guidelines for triphenyl phosphite. These call for exposure limits, air sampling, and medical surveillance in industries that use it. Studies from the National Toxicology Program back up the need for ongoing review of workplace standards, especially as manufacturing methods change or new uses emerge.
For people living near chemical plants, environmental watchdogs and community groups can play a role in keeping companies accountable. Shared environmental monitoring and transparent public reporting keep pressure on operators to stick to safe practices.
So, triphenyl phosphite carries risk, but clear-headed management and open information help prevent unnecessary harm. Tools exist; it’s using them every day that counts.
How should Triphenyl Phosphite be stored and handled?
Understanding Triphenyl Phosphite’s Hazards
Triphenyl Phosphite pops up in a lot of products across different industries, from stabilizers in plastics to flame retardants. Despite this wide use, not many actually talk about the real risks hiding behind the chemical’s mild-looking appearance. Exposure can irritate the skin, eyes, and respiratory system. Inhaling its fumes for even short bursts can set off headaches or dizziness. I learned pretty quickly that improvising with safety isn’t worth it—especially after seeing a poorly ventilated storage room cause training drills to turn serious simply because someone didn’t take the warning labels seriously.
How Professionals Store It
You can see a big gap between labs and warehouses that treat chemicals properly and those that just look for empty shelf space. Triphenyl Phosphite breaks down faster in moisture or heat. Humid environments trigger hydrolysis, which can then release toxic phosphorus-containing acids. Piling drums right next to windows or heat sources isn’t just lazy—it accelerates breakdown, increases pressure in sealed drums, and even creates a fire hazard. In my years working near industrial storage sites, the place with the lowest incident records always kept triphenyl phosphite in a dry, cool, shaded corner with active climate control. Double-sealed containers stood well above the ground, not hidden under old tarps or stacked with incompatible chemicals.
Why Proper Labeling and Segregation Matter
Bottles without clear tags or older drums missing hazard warnings set me on edge. A simple mix-up between Triphenyl Phosphite and strong acids or oxidizers can start a chemical reaction before anyone realizes what happened. The most responsible teams block off a dedicated spot in their chemical storage, always separated from strong bases and oxidizers. Even the color-coded labels and visible inventory logs send a message: “We take this stuff seriously.”
I remember one fire department drill that found half the containers with faded labels—nobody knew exactly what was inside, and in an actual emergency, that means real danger. Clear, updated signage helps responders and workers stay safer, not just following regulations.
Handling with Real Care
Pouring from a heavy bottle or opening a drum creates more risk than most new workers think. Splash-proof goggles, chemical-resistant gloves, and proper respirators aren’t extravagant—they’re basic. My early years on the job saw someone skip eye protection “just for a second.” He spent days in the hospital with eye damage after vapors leaked during a refill. You don’t get warnings before things go wrong.
Exhaust hoods and fans running while transferring triphenyl phosphite make a big difference. Simple habits like not working alone with it, and keeping spill kits handy, helped prevent bigger messes. I’ve seen well-meaning but rushed workers mop up spills with old rags, only to spread contamination or release more fumes.
Reducing Risks Through Training and Routine
Companies run safer operations if their teams actually know the risks. Regular training on chemical compatibility, spill response, and emergency contacts separates smooth handovers from chaotic accidents. Running drills and making safety equipment available at arm’s reach isn’t overdoing it; it sets up a workplace that doesn’t gamble with health.
These steps aren’t extras—they’re common sense for anyone who respects their own health and that of coworkers. Good habits, clear procedures, and a healthy respect for chemicals like Triphenyl Phosphite keep people out of harm’s way and businesses out of court.
What are the main applications and industries using Triphenyl Phosphite?
Plastic Manufacturing: The Backbone of Modern Convenience
Plastics show up in every corner of life, from grocery bags to smartphone cases. The production behind these items relies on materials that can handle heat and stress. Triphenyl phosphite comes in handy here. It serves as a key antioxidant and stabilizer in PVC and other polymers. Manufacturers trust it to protect plastics from breaking down or losing strength during processing. Without this, car dashboards might crack under sunlight, and electrical cables could fail fast.
The automotive, construction, and electrical industries all bank on the reliability of stabilized plastics. Factories across Asia, Europe, and North America keep triphenyl phosphite on hand to improve product lifespan. Seeing fewer yellowed cables and more durable window seals partly comes down to this compound.
Lubricants and Greases: Keeping Machines Moving Smoothly
Industrial machines, cars, and even bicycles need lubricants that don’t gum up over time. Triphenyl phosphite acts as an antiwear additive, especially in applications that see harsh conditions. It keeps oil formulas more stable, fighting off metal corrosion in engines or heavy-duty machinery. The steel industry, for example, has to keep its giant rollers moving without grinding themselves down. Lubricants that include this compound help cut down on costly repairs and downtime.
Over my years working alongside engineers, I’ve seen what cheap lubricants can cause: seized engines, ruined bearings, wasted hours on repairs. Adding a stabilizer like triphenyl phosphite doesn’t grab headlines, but it preserves equipment worth thousands or even millions of dollars.
Flame Retardants: Building Safer Spaces
Nobody wants to worry about house wires catching fire or office furniture fueling a blaze. Phosphate-containing flame retardants step in here, and manufacturers often choose triphenyl phosphite for its effectiveness. Mixed into plastics and coatings, it slows down how quickly things burn. I recall safety engineers showing data from fire resistance tests—materials with this compound often met stricter standards and helped save lives in real accidents.
Agrochemicals: Supporting Modern Farming
In agriculture, manufacturers rely on chemical intermediates to make pesticides and herbicides more effective. Triphenyl phosphite finds a spot here, helping in the production process by controlling reactions and boosting purity. The outcome is more reliable crop protection, which means fewer insects eating the food on supermarket shelves. Farmers, especially in large-scale operations, look for any edge to safeguard their harvest. It’s not dramatic, but this compound helps them feed more people with less waste.
Potential Solutions to Environmental Concerns
With all these applications, there’s a real need to consider health and environmental impacts. Factories and labs working with triphenyl phosphite have to follow strict safety protocols, but there’s always room to do better. Investing in green chemistry and looking into biodegradable alternatives could reduce risk. Companies can team up with researchers to develop safer replacements without sacrificing performance. It’s a collaboration worth pursuing, because every improvement in industrial safety benefits workers, end-users, and the planet.
| Names | |
| Preferred IUPAC name | Triphenyl phosphite |
| Other names |
Triphenyl phosphite Phosphorous acid triphenyl ester TPPi Trifenil phosphite Triphenoxyphosphine oxide Phosphonic acid, triphenyl ester |
| Pronunciation | /traɪˈfɛ.nɪl ˈfɒs.faɪt/ |
| Identifiers | |
| CAS Number | 101-02-0 |
| Beilstein Reference | 1208733 |
| ChEBI | CHEBI:35293 |
| ChEMBL | CHEMBL138967 |
| ChemSpider | 17847 |
| DrugBank | DB14335 |
| ECHA InfoCard | 200-928-9 |
| EC Number | 262-149-1 |
| Gmelin Reference | 777 |
| KEGG | C01741 |
| MeSH | D014265 |
| PubChem CID | 7286 |
| RTECS number | TF3325000 |
| UNII | E8M8HG63BQ |
| UN number | UN2325 |
| Properties | |
| Chemical formula | C18H15O3P |
| Molar mass | 310.29 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Odorless |
| Density | 1.183 g/cm³ |
| Solubility in water | Insoluble |
| log P | 4.8 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | 13.1 |
| Basicity (pKb) | 13.4 |
| Magnetic susceptibility (χ) | -98.0e-6 cm³/mol |
| Refractive index (nD) | 1.585 |
| Viscosity | 36.8 mPa·s (at 25 °C) |
| Dipole moment | 2.40 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 270.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -174.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6216 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H411 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0-0 |
| Flash point | Flash point: 220°C |
| Autoignition temperature | 450°C |
| Lethal dose or concentration | LD50 oral rat 3,600 mg/kg |
| LD50 (median dose) | 1,600 mg/kg (rat, oral) |
| NIOSH | TT3990000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Trimethyl phosphite Triethyl phosphite Tris(2,4-di-tert-butylphenyl) phosphite |

