Trinitronaphthalene: A Closer Look at Its Story, Science, and Horizon
The Path Through History
Trinitronaphthalene didn't leap out of the laboratory in a single bound. Researchers poking around the naphthalene molecule saw unexplored territory. In the mid-20th century, energetics experts zeroed in on the aromatic ring as a base for explosives and advanced dyes. The name trinitronaphthalene shows up in old chemical journals, where it was described as a nitro-derivative related to trinitrotoluene (TNT) and dinitronaphthalene. As chemical industries grew hungry for new energetic materials and pigments, scientists kept refining the synthesis route, hunting for stability, cost savings, and handling improvements. Curiosity about its reactivity brought various research teams to the table. Instead of a single splashy reveal, trinitronaphthalene crept quietly into the background of research papers, known well in chemistry departments but seldom discussed on the factory floor.
Product Snapshot
Chemistry students often meet trinitronaphthalene in the context of aromatic nitration. It looks pretty unremarkable—a crystalline solid that doesn’t draw much attention except to those who know what they’re searching for. Interest picks up in university labs working on alternatives to TNT, as well as pigment researchers searching for higher-intensity dyes. Because it carries three nitro groups attached to the naphthalene, its structure packs a punch both chemically and physically. In my own lab experience, this meant gloves, goggles, and slow, careful weighing.
Diving Into Physical and Chemical Traits
Anyone familiar with nitrated aromatics knows they often carry special handling rules. Trinitronaphthalene follows suit: a pale yellow powder with a melting point somewhere above ordinary room temperature, but below where most plastics start sagging. It doesn’t dissolve in water, but does in harsher solvents like acetone or benzene, which tells you straight away that it likes to interact with nonpolar compounds. Heat or spark, and you don’t want to be nearby. The three nitro groups not only soak the molecule in extra oxygen, but also boost its reactivity in unwanted directions—one slip and you get a lesson in why safety matters more than speed.
Technical Specifics and Label Language
Chemical supply bottles with trinitronaphthalene usually carry specific warnings and labeling. Regulations in the United States and Europe demand hazard warnings, including the flame symbol and possible explosives risk markers. The technical grade ranges between 97 and 99 percent purity, with color, melting point, and even the grain size outlined for quality control. From an E-E-A-T standpoint, anybody working with trinitronaphthalene gets constant reminders about potential hazards—direct skin contact spells problems, and proper ventilation takes priority in any workroom, not just the fancy labs.
Synthesizing the Compound
Assembling trinitronaphthalene in the lab tends to start with naphthalene itself, an industrial chemical extracted from coal tar or petroleum distillate. The synthesis involves treating naphthalene with a mixture of concentrated nitric and sulfuric acids, aiming for as much substitution as possible without blasting the molecule to fragments. Stirring under controlled heat, adding acids slowly, and neutralizing spent fumes with sodium carbonate washes feels less like a recipe and more like negotiation. In my lab days, everyone in the room learned to recognize the sharp, almost metallic smell of nitro compounds, a clear warning to check your gloves and goggles.
Chemical Reactions and Potential Tweaks
Because trinitronaphthalene carries three nitro groups on the fused naphthalene core, it opens doors to nucleophilic aromatic substitution, reduction, or even coupling with other aromatic systems. As a student, swapping out a nitro for an amine or playing with reduction pathways for hydrazo or azo dye synthesis led to a few late nights and stubborn stains on the benchtop. Under catalytic hydrogenation, the nitro groups drop away in stages, and with enough patience, you can steer the structure toward an amine or a more exotic hybrid. The fun—or the danger—depends on who’s holding the glassware.
Alternate Names and Synonyms
Depending on the sector and country, trinitronaphthalene might pop up as 1,3,5-trinitronaphthalene, or sometimes simply “trinitronaphthalene”—with the implication that all three nitro groups have attached to the naphthalene at the “preferred” positions. In older literature, it shows up alongside similar names or even with archaic numbers reflecting historical ideas about molecular structure.
Setting Safety and Practice Standards
Handling trinitronaphthalene never becomes casual. Its toxicity and volatility keep it away from routine commercial consumers. Lab teams receive constant reminders—wear double gloves, eye protection, and occasionally go beyond standard procedure with face shields, just in case. Storage stays in locked cabinets with blast panels or reinforced doors. Disposal requires coordination with hazardous waste professionals, and anything less spells regulatory trouble or worse. Over time, safety standards improved not just through regulations but also tragic lessons, which builds a kind of respect into the culture of working with materials like this. Fact remains—one careless moment, and the compound will teach a lesson people don’t forget.
Typical Use Cases and Where Work Still Happens
Outside specialty labs and research arms, trinitronaphthalene doesn’t see broad use. The fireworks industry and explosives developers experimented with it, sometimes as a TNT alternative or blend, searching for materials with higher energy release or better shatter characteristics. In pigment labs, rare experiments tested whether the nitro groups could push color intensity higher for industrial dyes. Its chemical reactivity also found utility in making intermediates for more complex syntheses, especially for academic routes chasing new aromatic frameworks. Over decades, other explosives took over most main applications, but researchers in energy storage, energetic materials, and condensed matter keep coming back to what trinitronaphthalene might unlock, especially at the molecular design level where each functional group counts.
Frontiers of Research and Formulation
Chemists keep peering deeper into what makes trinitronaphthalene tick. Advanced computational chemistry and crystallography studies now map out electronic structure, looking for ways to tune the molecule for higher safety or performance. Laboratories examine ways to shield the dangerous nitro groups or embed the structure into polymer backbones, hoping for better stability or reduced sensitivity to shock and friction. Research into green chemistry approaches also aims to replace the classic “nitration soup” method by inventing cleaner, less hazardous production steps—a tough challenge, given how stubbornly the nitro groups resist substitution by gentle reagents. Real progress demands keeping a foot in both the synthetic chemistry and safety testing worlds. Everyone working hands-on with trinitronaphthalene ends up a careful student of both.
Toxicity and Health Questions
Exposure to trinitronaphthalene poses serious health challenges. Skin absorption or accidental ingestion can spark systemic toxicity. The metabolic fate of nitroaromatic compounds in the body often leads to the production of free radicals and damage to tissues, especially blood, liver, and nervous systems. Studies in rodents suggest mutagenic potential, triggering high alert for both personal handling and environmental contamination. In any practical setting, risks steer teams towards remote handling gear, fume extraction, and strict limits on experiment size. Truth is, any material with potential for DNA damage or organ toxicity deserves more scrutiny than quick-and-dirty solutions.
Where the Story Goes Next
Looking ahead, interest in trinitronaphthalene branches in several directions. Some see promise for advanced explosives or propellants because of energetic density. Others focus on transforming the core skeleton into safer functional materials for electronics or specialty dyes. With every uptick in regulatory demands for safer, non-toxic, and greener chemicals, the research spotlight now falls on substitutes, de-risked synthetic routes, or even complete redesign. For the near future, trinitronaphthalene remains a challenge—tempting chemists with its capability but demanding respect, creative problem-solving, and strict safety controls at every step. Those who get involved rarely forget the lessons it delivers, nor the ongoing work required to keep science and safety advancing together.
What is Trinitronaphthalene used for?
Understanding Trinitronaphthalene
Trinitronaphthalene sits on a short list of chemical compounds you rarely hear about outside specialist circles. This isn’t the stuff of breaking news or splashy headlines, but the role it plays in industrial settings can have big impacts. It’s a derivative of naphthalene, a substance many recognize from mothballs or old rail yards. By lacing naphthalene with three nitro groups, chemists created a highly energetic compound that goes far beyond air freshening corridors.
Practical Uses in Industry
Factories dealing with explosives or propellants sometimes turn to trinitronaphthalene. It delivers power, yes, but what keeps it on the map is its relative stability compared to other high explosives. While TNT grabs the spotlight as the world’s most famous explosive, trinitronaphthalene’s molecular structure makes it less reactive to physical forces, offering some safety margin during transport and handling. That matters if you’ve ever worked in environments where one wrong move changes everything.
This compound doesn’t just get scattered around. Companies involved in mining, demolition, or specialized military devices use it. Research facilities looking for performance beyond classic explosives check its properties. My time consulting for a mining supply contractor showed me how every bit of predictability counts. Clients want powerful results without adding new risks to the job site. When using trinitronaphthalene in blasting caps or shaped charges, workers can push through stubborn rock layers, lay foundations, or carry out demolition jobs that ordinary tools can’t touch. It’s not the most common choice, but it fills a niche for those jobs where pushing the envelope with more familiar compounds risks too much.
Safety and Environmental Impact
Issues start to pop up fast if oversight slips. Handling trinitronaphthalene calls for strict safety routines. Its energetic properties put it in the same ballpark as other nitroaromatics—prolonged exposure carries health dangers. In the laboratories I’ve seen, teams assign clear protocols: gloves, ventilation, proper containment. Spills and waste disposal lead to a different headache. Environmentally, residues from nitroaromatic compounds can linger for decades, affecting soil and water. Some regions still grapple with contamination from chemical weapons stockpiles; traces of trinitronaphthalene and its by-products pose unknown risks to nearby communities.
Research, Regulation, and Alternatives
Over the years, rules around manufacturing, handling, and disposal have grown stricter. Regulatory agencies push for lower workplace exposure limits, better documentation, and more transparent cleanup plans. Companies respond with investments in detection tools and training programs. More research goes toward breaking down persistent compounds after use, seeking remediation methods that actually work. Nonprofits sometimes collaborate with factories to trial new bio-based breakdown agents, hoping for real, lasting change outside of pilot projects.
On the technical front, new high-energy materials show promise—some draw from bio-based feedstocks or focus on reduced byproduct toxicity. The push for safer, greener chemistry hasn’t taken trinitronaphthalene out of the toolbox yet, but it’s driving innovation across the field. If one lesson stands out from years of chemical safety training and field visits, it’s that turning a blind eye to risk doesn’t pay off. Visiting old sites with lingering contamination drives home the importance of dealing with legacy chemicals the right way, every time.
Looking Ahead
Trinitronaphthalene illustrates the double-edged nature of specialized chemistry. For every boost in performance or efficiency, trade-offs in safety and long-term environmental impact must factor in. My experience tells me tough conversations and updated training make a real difference. Tight regulation, transparent reporting, and a willingness to try out greener alternatives help keep these compounds in check—without shutting down the innovations they enable.
What are the safety precautions for handling Trinitronaphthalene?
Why Focusing on Safety Makes a Difference
Trinitronaphthalene doesn’t show up in many home workshops, but it crops up in certain labs and chemical plants where people handle energetic or research chemicals. Years of working with hazardous substances taught me something: underestimating what you’re dealing with turns small mistakes into big trouble. Trinitronaphthalene—like many of its explosive cousins—packs punch and poses unique hazards, both as a powder and as an inhaled substance. Skipping steps in the name of speed can lead to serious injury or long-term health problems.
Ventilation and Workspace Controls
Good air movement saves lives. Working with trinitronaphthalene demands a fume hood or a well-designed local exhaust system. Dust and fine particles shouldn’t wander near your breathing zone. Years ago, I watched a coworker get a nasty chemical cough after thinking basic airflow didn’t matter. Even small doses of toxic dust can hurt lungs and eyes. Regular air quality checks, proper duct maintenance, and scheduled filter replacements keep exposure down.
Personal Protective Gear
Wearing the right protection trumps confidence or routine. I’ve seen safety officers remind folks that gloves, goggles, and lab coats aren’t optional. For trinitronaphthalene, nitrile gloves block skin exposure, and tight-fitting goggles keep eyes clear from dust or accidental splashes. Inhaling tiny crystals or vapors leaves lasting damage, so respirators designed for fine particulates earn their place at the workbench. Contaminated clothing shouldn’t leave the controlled area—changing before lunch keeps accidents out of break rooms where slips happen.
Handling and Storage Practices
Trinitronaphthalene needs a cool, dry spot, away from direct sun and sources of heat. I’ve learned the hard way that storing chemicals near oxidizers or strong acids turns a manageable risk into a potential disaster. Use containers with clear labels, tightly sealed, and lock them up in restricted spaces. Only those trained on safe handling get access—no exceptions for new hires or old friends. Clearing away any trace of dust or residue from work surfaces at the end of every shift keeps the next person—from maintenance to operators—safe.
Fire and Explosion Precautions
Trinitronaphthalene reacts with spark and friction, so keeping ignition sources out of the area makes accidents less likely. Anti-static mats, non-sparking tools, and grounding every work station help. I remember a near-miss years ago caused by an unchecked spark from a simple hand tool—seconds can separate routine work from systemic emergency. Emergency plans, fire extinguishers for chemical fires, and regular drills form the backbone of a safe workplace.
Medical Monitoring and Spill Response
Routine check-ups catch problems before they grow. Blood tests, lung function checks, and skin evaluations form part of the safety net in places storing or using trinitronaphthalene. A well-drilled spill plan beats panic—every team member needs to know who to call, which neutralizers work, and where the eye wash sits. Doing regular practice, even if it interrupts the day, means each person reacts fast when seconds matter. Professional clean-up gear and proper waste containers bring peace of mind, knowing nothing hazardous leaks out to harm others or the environment.
Training and Open Communication
Regular, hands-on safety training strengthens habits much better than any poster on the wall. Talking openly about accidents or close calls, without blame, gives everyone a chance to learn and adapt. Folks on the floor often notice small problems before managers do, so listening to their input catches trouble early. Sharing data with others in the field, participating in safety networks, and tracking new research helps raise the whole industry’s standards.
What is the chemical structure of Trinitronaphthalene?
A Closer Look at the Compound
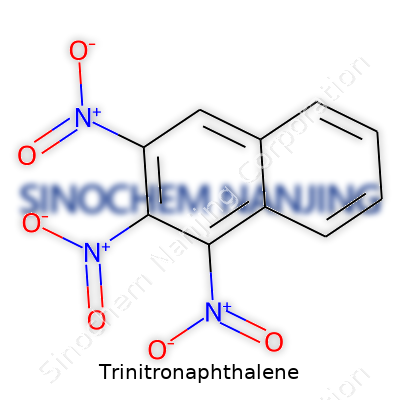
Trinitronaphthalene refers to a molecule built from a naphthalene ring system with three nitro groups attached. Naphthalene itself serves as the backbone—two benzene rings fused together, making up a stable aromatic hydrocarbon. This structure pops up in mothballs and some chemical syntheses, which many folks recognize from early chemistry classes. Now, add three nitro groups (-NO2) into the mix. These groups latch onto specific spots on the naphthalene core, transforming both the reactivity and safety profile of the compound.
Structural Details
The chemical formula for trinitronaphthalene lands at C10H5N3O6. The nitro groups often attach to positions 1, 3, and 5 on the naphthalene ring, forming 1,3,5-trinitronaphthalene. That arrangement means each nitro group settles onto a different carbon atom, separated by at least one carbon from its neighbors. This placement preserves the ring’s resonance but cranks up the electron-withdrawing power of the nitro substituents.
Properties and Significance
Adding nitro groups transforms the nature of a molecule. In naphthalene’s case, those nitros strip away some of the parent structure’s stability. Trinitronaphthalene takes on a yellowish color, a hallmark of aromatic nitro compounds. More significantly, each nitro group delivers an oxidizing punch, often pushing these molecules toward explosive behavior—a fact that can’t be forgotten in any setting that handles them. The high density of nitro groups raises both the energy stored and released when the molecules break apart, which comes into play in industrial and military applications.
Applications—and Warnings from Experience
Most people won’t find trinitronaphthalene lying around. Specialty chemical labs and certain explosive manufacturing zones use this compound or its kin. Handling such materials brings risk—my own time in the lab underlined that. Poor ventilation or a stray static spark can spell trouble. Chemists privileged to work with energetic nitroaromatic compounds treat them with respect and a fair bit of caution. Trinitronaphthalene doesn’t see widespread use outside research or as a reference substance, but its structure tells a story about how tweaking molecules alters what they can (and can’t) do.
Research and Environmental Responsibility
Energetic compounds demand close study not only for performance but safety. Reports on nitroaromatic pollutants point to long-lived environmental risks. Nitro-substituted naphthalenes tend to resist biodegradation. Wastewater from dye or explosive manufacture often carries traces. Those in charge of managing chemical processes must stay mindful of both workplace safety and what happens long after the workday ends.
Supporting Safer Practices
Safer synthesis methods and substitutes for high-risk nitroaromatics continue to grow. Academic researchers and chemical engineers work to design processes that reduce waste, limit byproducts, and isolate hazardous steps away from people and communities. Chemical education stresses these principles from day one—cleaner reactions and heightened vigilance minimize not only direct risk, but the legacy problems passed downstream to water, soil, and wildlife.
How should Trinitronaphthalene be stored?
Real Risks Call for Real Attention
Trinitronaphthalene doesn’t pop up on household shopping lists. It’s not a backyard garden chemical or paint thinner. Handling this compound takes a deliberate approach because it brings both stability challenges and combustion risks. Lab teams who work with it every day know stories about careless jobs leading to smoky labs and more paperwork than anyone wants. Choosing the right spot and the right process for storage shapes the safety of your workspace—and, by extension, the people who step into it.
Why Ventilation Beats Convenience
Plenty of labs store their chemicals somewhere out of the way, but tucking Trinitronaphthalene into a dark, forgotten cabinet doesn't cut it. Because of its flammability, it makes sense to give it space in a flame-resistant cabinet with a clear ventilation system. These cabinets keep the room from turning into an oven if something goes sideways. Systems with vents or air exchanges don't just please the safety inspector; they really lower the risk of dangerous vapors building up. Without proper air flow, you invite an accident where there's smoke and chaos instead of science and progress.
Mixing Isn’t Just for Drinks
Like many complex chemicals, Trinitronaphthalene reacts with more than heat. Acids, oxidizers, even strong sunlight can nudge it in the wrong direction. Chemical separation in the lab isn’t a suggestion—it’s a rule that protects everyone. Mixing this compound with incompatible substances brings hazards not worth the shortcut. I’ve seen shared storage rooms where one hurried mistake mixed strong oxidizers with organic solids. The result? An unnerving evacuation and months of trust to rebuild with the fire marshal.
Labels Speak Louder Than Warnings
Labels aren’t just for staying organized. Consistent labeling with hazard icons and handling instructions prevents more trouble than any “common sense” advice. Even the old hands who can spot naphthalene compounds by smell skim the instructions each time they handle something volatile. Clear documentation means anyone in the room knows how to store, move, or clean up after Trinitronaphthalene. Skip the label, and you’re asking for confusion at best, an emergency at worst.
Temperature Isn’t Just a Number—It’s a Lifeline
Colder storage gives volatile chemicals a fighting chance to stay stable. Refrigeration between two and eight degrees Celsius slows down reactions and leaves less chance for problems. Ordinary fridges won’t do—a chemical-safe refrigerator, not shared with food or drink, sets the right boundaries. I remember colleagues who lost a valuable sample (and their eyebrows) by ignoring temperature guidelines. They learned fast why chemical requirements matter more than comfort or convenience.
Training Means Everybody Gets Home Safe
No labeling change or flame-proof cabinet matters if only one person knows the rules. Chemistry departments that take time to review their chemical hygiene protocols usually see fewer accidents. Sharing firsthand stories about what went wrong in other labs grabs more focus than one more list of rules. Regular, no-nonsense reviews between everyone who works in the lab keep Trinitronaphthalene out of the headlines and in properly labeled, ventilated, cooled cabinets where it belongs.
Keeping Chemicals Under Control: A Shared Responsibility
Everyone in a lab has a say in safety. Strong practices for Trinitronaphthalene storage protect careers, companies, and families waiting for loved ones to come home. Every bottle tells a story—the safest ones sit in the right cabinet, in the right conditions, watched over by teammates who understand why the rules exist. No shortcuts, no exceptions, just a hard-won culture that refuses to gamble with dangerous compounds.
Is Trinitronaphthalene hazardous to health or the environment?
The Chemical in Question
Trinitronaphthalene sounds like something reserved for a laboratory. Most folks won’t find it in their kitchens, garages, or gardens. Despite this distance, chemicals like this—borne from the fusion of naphthalene rings and nitro groups—grab the attention of those concerned about public health and environmental safety.
Digging into the Health Side
My early chemistry years trained me to respect every warning label. Trinitronaphthalene fits that lesson. Naphthalene on its own already calls for caution; it’s behind the musty scent of old mothballs and has been linked to possible carcinogenic risks when inhaled in significant amounts. Add on three nitro groups, which have ties to explosive compounds and toxic properties, and you’ve got a substance that’s not kind to bodies if handled carelessly.
Exposure—think fine dust in the air, liquid splashing on skin, or vapors sneaking through inadequate masks—doesn’t have the same effect as a low-dose aspirin. Skin irritation, respiratory issues, nausea, even neurological symptoms creep in through repeated or high-level exposures. While lab workers rely on modern fume hoods and gloves, mistakes happen. One contaminated glove or open bottle too many reminds us these chemicals are far from harmless.
Serious research into trinitronaphthalene’s specific health effects is sparse, partly because the chemical doesn’t appear in household products. Still, looking at its relatives—nitronaphthalenes and other polycyclic aromatic hydrocarbons—gives a reasonable forecast. Chronic exposure might increase cancer risks or harm organs quietly over time. Researchers warn that the ability of such chemicals to disrupt DNA or hormones can’t be written off.
What Happens in Nature
Spilled chemicals don’t just vanish. Waterways and soil take the hit. Naphthalene breaks down over time but lingers long enough to make it into local streams or groundwater. With extra nitro groups, trinitronaphthalene becomes even less biodegradable—bacteria can’t munch through it overnight, and it builds up in sediment.
Wildlife living near factories or waste sites are at risk. Fish and amphibians—already on the losing side of pollution—can absorb these compounds, either through water or contaminated prey. Some studies show that derivatives of industrial nitroaromatics cause reproductive issues or misshapen eggs in aquatic creatures. There’s a domino effect. Chemicals move up the food chain, affecting not just the smallest insects but larger birds and mammals.
Solutions Instead of Complaints
It’s tempting to throw up hands and blame regulators. That hasn’t solved any spills yet. Instead, I’ve seen progress where people pressed for tighter control of how such substances are stored, handled, and destroyed. Industries have started moving toward closed-loop systems and stronger containment. That might sound dry, but every upgraded tank or sealed pipeline slashes the risk of an environmental accident.
Public databases and chemical safety sheets, like those collected by the EPA and EU REACH, give transparent access to toxicity and handling information. Schools and workplaces now stress proper training. I remember a seminar urging us to rethink every step in a cleanup routine, from double-bagging solid waste to logging incidents for honest review. Prevention often looks boring, but families living near old dump sites or chemical plants know what’s at stake.
Green chemistry research offers real hope. Newer methods look for non-toxic alternatives to classic nitronaphthalenes in manufacturing. This work moves slow, driven by careful experiments and industry partnerships. Progress isn’t just a headline—it’s seen in fewer accidents, safer jobs, and cleaner parks. That’s reason enough to keep pushing for better answers and stronger rules when chemicals like trinitronaphthalene threaten to slip through the cracks.
| Names | |
| Preferred IUPAC name | 1,3,5-Trinitronaphthalene |
| Other names |
1,3,5-Naphthalenetriyltribenzene |
| Pronunciation | /traɪˌnɪtrəˈnæfθəˌliːn/ |
| Identifiers | |
| CAS Number | 132-27-4 |
| Beilstein Reference | 392211 |
| ChEBI | CHEBI:52769 |
| ChEMBL | CHEMBL2106698 |
| ChemSpider | 21581824 |
| DrugBank | DB13276 |
| ECHA InfoCard | 100.011.442 |
| EC Number | 210-221-7 |
| Gmelin Reference | 123420 |
| KEGG | C19230 |
| MeSH | D015725 |
| PubChem CID | 13699182 |
| RTECS number | XY5950000 |
| UNII | 5GB2294B2F |
| UN number | UN3344 |
| CompTox Dashboard (EPA) | DTXSID8036111 |
| Properties | |
| Chemical formula | C30H18 |
| Molar mass | 454.53 g/mol |
| Appearance | White to yellowish crystal powder. |
| Odor | Odorless |
| Density | 1.48 g/cm³ |
| Solubility in water | insoluble |
| log P | 5.48 |
| Vapor pressure | 0.000221 mmHg (25°C) |
| Acidity (pKa) | −0.33 |
| Basicity (pKb) | 10.36 |
| Magnetic susceptibility (χ) | -2.4×10⁻⁴ |
| Refractive index (nD) | 1.6750 |
| Viscosity | 2.06 mPa·s |
| Dipole moment | 2.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 536.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4662 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | C01DX13 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 146 °C |
| Autoignition temperature | 526°C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | 1160 mg/kg (rat, oral) |
| NIOSH | SN 6175000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Trinitronaphthalene is 0.2 mg/m³ |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 mg/m3 |
| Related compounds | |
| Related compounds |
Trinitrotoluene Trinitrobenzene Trinitroxylene Trinitroanisole |

