Trinitrofluorenone: A Writer’s Perspective on a Chemical Chameleon
Historical Development
Stepping back in time, trinitrofluorenone hit the chemistry scene around the mid-twentieth century, riding a wave of intensive research that prized molecules with complex aromatic structures and dramatic reactivity. Chemists digging into nitroaromatic compounds saw fresh opportunities in fluorenone derivatives. Compared to some showier explosives and dyes, trinitrofluorenone flew under the radar, yet its unique electronic properties turned heads in specialized research circles. Encounters with it in old German and Russian literature remind me how global this quest for new molecules grew, and also how quickly scientists began seeing potential beyond explosives—such as its role as a charge-transfer agent in all kinds of electronic investigations. There’s a lesson here. Even chemicals that start with military or industrial intentions sometimes find new life in laboratories and technologies that nobody expected back in those early years.
Product Overview
For scientists, trinitrofluorenone means a yellow crystalline powder used most often for its sensitive response to electrons and light. That distinct color never leaves your memory; it stains glassware and even seems to tint the air in handling. In the market, you see it show up in small glass ampoules from chemical suppliers, not in drums or sacks. It never joins the ranks of bulk chemicals you hear about at trade fairs; instead, you find it in the back corners of chemistry departments, lingering among samples used for niche experiments in photochemistry or electronics. People reach for trinitrofluorenone when looking for a compound that punches above its weight as an electron acceptor or an indicator in redox reactions.
Physical & Chemical Properties
Hold a sample in hand and you see an intense yellow powder with a faint, sharp odor. Its melting point runs high for this class of aromatic compounds, which hints at a stiff, conjugated structure. The poor solubility in water and greater affinity for organic solvents like benzene and acetone hint at its nonpolar, aromatic backbone. Stubbornness toward spontaneous decomposition sets it apart from some of its explosive cousins—still, you learn to treat it with respect. The molecule’s three nitro groups eat away electron density, making trinitrofluorenone strongly electrophilic, which opens doors in synthetic and analytical chemistry. In ultraviolet or visible light, its absorption spectrum stands out. For researchers tuning spectroscopy equipment, that strong absorbance isn’t a nuisance; it’s a feature that reveals subtle details about molecular orbitals and electron movement.
Technical Specifications & Labeling
Anyone handling trinitrofluorenone learns that purity makes or breaks an experiment. Small amounts of impurities—sometimes inevitable due to side reactions during synthesis or storage—play havoc with electronic spectra and reactivity. Professional suppliers list purity grades, normally above 98 percent for research work, and always include batch-specific data about melting points and spectroscopic analysis. Quality labels highlight the hazard class: toxic by inhalation, ingestion, and skin contact. Labels warn about the risk for environmental contamination. You often find it stored in amber glass, away from strong reducing agents and sources of ignition. From my own lab experience, the labeling standards on these bottles rarely look excessive. Every reminder pushes chemists to respect the consequences of mishandling a nitroaromatic compound with persistent toxicity.
Preparation Method
Synthesis usually starts with fluorenone or one of its close relatives, subjected to severe nitration. Fuming nitric acid and a touch of concentrated sulfuric acid, controlled at low temperatures, coax three nitro groups into the right positions on the fluorenone skeleton. Even with careful temperature management, yields sometimes drop due to competing side reactions. I’ve watched chemists improvise safer and milder nitration protocols, trying to balance efficiency with personal safety. Any accidental overheating releases choking fumes and risks dangerous runaway reactions. After nitration, extensive purification—often by recrystallization—pulls the product out from a stew of isomers and tars. From the student perspective, these purification steps teach hard lessons about patience and attention to detail, since careless work during isolation wastes an entire day (or week) of effort.
Chemical Reactions & Modifications
Once synthesized, trinitrofluorenone begs for further exploration. Its electron-deficient core invites reduction, making it an excellent tool in experiments where researchers want to probe electron transfer events. Reductive treatments, such as solutions of hydrazine or sodium dithionite, strip away some or all nitro groups or push the molecule toward semi-quinone states. Modification at the carbonyl group shifts reactivity again. Some organic chemists use it as a starting point for building more elaborate ring systems using condensation or coupling reactions. My own encounters involved experiments with nucleophilic aromatic substitution—those nitro groups aren’t just decorative; they change the entire reactivity game. Each alteration amplifies or tunes the properties that made the parent compound interesting, demonstrating the value of a well-chosen starting scaffold in synthetic work.
Synonyms & Product Names
Trinitrofluorenone appears under a handful of names scattered across catalogues and research articles. Some call it 2,4,7-trinitrofluoren-9-one or 2,4,7-trinitro-9H-fluoren-9-one. Elsewhere, the shorthand TNF shows up, though confusion with other nitro-organic agents sometimes clouds literature searches. The core naming conventions always focus on the three nitro groups fixed to the fluorenone nucleus. Anyone searching databases soon realizes that slight spelling changes—like fluorenone or fluoren-9-one—surface in various international patents and research journals. These quirks of chemical naming might slow down newcomers, but they also encourage double-checking molecular structures before starting new research or ordering supplies.
Safety & Operational Standards
Nobody works with trinitrofluorenone without feeling the weight of its health risk. Inhalation hazards aren’t exaggerated. Even short exposure during weighing or transfer stings your nose and reminds you to reach for the fume hood. Labs handle it in small quantities, and protective gloves and goggles stand as standard-issue defenses. Disposal procedures treat spent trinitrofluorenone and its by-products as strictly controlled waste, since nitroaromatics persist in the environment and break down slowly. The rules might sound draconian, but a single careless spill or inhalation leaves lasting health effects. Years of lab experience taught me never to cut corners with this family of chemicals; medical literature details cases where chronic exposures led to blood and nervous system disorders. Regular safety training and clear emergency protocols back up each chemical order, making sure newcomers don’t underestimate the risks of outdated or leaky containers.
Application Area
Trinitrofluorenone doesn’t show up in manufacturing catalogs for paints or plastics. Instead, you see its influence in niche laboratory tests, mostly in the world of organic electronics. Engineers borrow it as an electron acceptor in studies exploring organic semiconductor behavior. Chemists use small doses as a colorimetric indicator in redox titrations or as a reference material in UV-visible spectroscopy. Beyond that, a handful of specialized applications—molecular sensors, certain charge-transfer complexes, and exploratory work on organic solar cells—draw on its high sensitivity to electrons and light. Most people never interact with trinitrofluorenone directly, but parts of the electronics R&D field quietly rely on its predictive properties. Knowing the difference that such a molecule can make in a controlled test system gives me a new appreciation for those rare compounds whose value lies in what they reveal, not in what they build.
Research & Development
In the current wave of organic materials research, trinitrofluorenone keeps popping up as a reference and sometimes as an active component. Researchers using it to probe electron transport investigate new organic light-emitting diodes and photovoltaic materials. For those designing electron-deficient moieties or looking for simple models of charge-separation processes, having access to high-quality trinitrofluorenone means clear, reproducible experiments. Investment in purer syntheses, improved characterization techniques, and digital databases connects researchers more efficiently with each other’s work. I’ve watched the evolution from hand-jotted melting point data to thorough digital archives, which raises the bar for reproducibility and accelerates discovery. These improvements sometimes seem invisible until a missed impurity or mislabeled spectrum ruins an entire batch of results. The dynamic field of molecular electronics needs standards, but also real curiosity to look at molecules as tools, not just objects.
Toxicity Research
Toxicity profiles matter. Trinitrofluorenone’s nitroaromatic skeleton means it gets absorbed by biological systems and sticks around longer than most would like. Animal studies, mostly in rodents, tell a consistent story: damage to the blood-forming organs and the nervous system shows up after repeated exposure. Some studies connect it to oxidative stress and disruptions in metabolic pathways. Waste handling requirements force academic and industrial labs to look for safer substitutes wherever possible and to track usage tightly. As a young chemist, I puzzled over why a compound with such promising electronic properties carried so many warning labels. Learning about environmental and occupational impact convinced me that curiosity and safety need to walk together. Even limited, well-documented exposure can teach us about better chemical stewardship and the need for innovative methods to reduce workplace and environmental risks.
Future Prospects
Looking ahead, interest in trinitrofluorenone hinges on its fundamental role in understanding molecular charge-transfer mechanisms and as a benchmark for nitroaromatic reactivity. As new organic semiconductors and sensing materials hit the market, researchers revisit old standards like this one to draw comparisons and extract baseline data. Advances in green chemistry will continue to pressure chemists to invent synthesis and disposal protocols that cut down on toxic by-products. The real future may not see trinitrofluorenone in mainstream consumer products; rather, it will serve as a foundational tool in emerging methods for diagnostics, molecular electronics, or analytical chemistry. The challenge lies in harnessing what makes it useful—stability under specific conditions and sensitivity to electrons—without repeating past mistakes in safety and waste management. As a specialist who has handled it, I see the promise and peril, and I hope the next generation builds on this legacy without the baggage of careless or underinformed use.
What is Trinitrofluorenone used for?
Looking Past The Long Name
You rarely see Trinitrofluorenone pop up in everyday conversation, but this curious-sounding chemical finds a home in some surprising corners of technology and science. Trinitrofluorenone, often abbreviated as TNF, looks a lot like something out of a chemistry textbook most people would rather forget. Still, its impact runs deeper than many realize.
Making Electronics Possible
At its core, TNF acts as an organic semiconductor. What does that mean in practical terms? Organic semiconductors help move tiny electrical charges through certain materials, which has real-world applications in electronics such as organic light-emitting diodes (OLEDs) and organic solar cells. TNF’s unique molecular structure lets electrons hop from one place to another, making it an ideal choice for early experiments with organic conductors. Back in the 1970s, one of the first organic electronic devices relied on TNF combined with tetrathiafulvalene, showing that even simple molecules could create new ways to move electricity. Without people tinkering with substances like TNF, there might not be the wave of advances in flexible displays, wearable tech, and clever lighting that fuel today’s new gadgets.
The Colorful World Of Dyes
People don’t just turn to Trinitrofluorenone for electricity. Chemists appreciate its deep yellow color, which brings the substance into play as a dye and stain. In the lab, TNF works as a color indicator, making reactions easier to track and analyze. This might sound like a minor role, but accurate detection of reactions in a sample can speed up drug discovery or help trace crucial changes in research. Nobody loves hunting for invisible changes under a microscope, and TNF gives a visible clue that something important has happened.
Testing And Detecting In The Real World
TNF steps up in the world of chemical tests, too. Forensic scientists and techs in quality control labs use it to spot the presence of specific substances, especially because it forms charge-transfer complexes with various organic molecules. In basic terms, mix TNF with certain compounds and you get a change you can spot. That gets used in tests for drugs and other chemicals, where reliability matters. Testing for purity and authenticity isn’t always glamorous work, but TNF’s role means fewer mistakes get through in finished products.
Risks And Awareness
Using TNF asks for caution. Like many nitro compounds, mishandling can lead to health hazards. Exposure irritates skin and eyes, and accidentally inhaling it creates bigger problems. Professionals always rely on proper protective gear and strict protocols with chemicals like this. As more consumers and companies put value on worker safety, the industry finds itself under more pressure to replace or control risky substances. Pushing for safer alternatives and more transparent safety practices pays off—not just for scientists, but for people whose lives touch these chemicals from a distance.
Looking Forward
Curiosity about substances like Trinitrofluorenone, and honest discussion about their risks, keeps progress grounded in reality. As science races forward, smart choices about which chemicals to use, and how to handle them, can foster safer research and better products. Real-world applications keep evolving, and so should the questions and solutions around chemicals like TNF.
Is Trinitrofluorenone hazardous to health?
What is Trinitrofluorenone?
Trinitrofluorenone, known in labs as TNF, works as a chemical compound mainly used as an oxidizing agent and electron acceptor. Chemists often use it to test for certain classes of chemicals. Its yellow crystalline appearance may look innocent, but there’s more beneath the surface.
How Harmful Can Contact Be?
Anyone who’s worked with TNF usually thinks about gloves, goggles, and lab coats long before picking up a vial. This chemical can irritate the skin and eyes. Even a small amount on your fingers may lead to redness or itching. Accidentally breathing in dust or vapors means you risk coughing, throat irritation, or even headaches.
OSHA and NIOSH don’t seem to have detailed workplace exposure limits for TNF. That spells concern, because lack of guidance often means research on health risks remains thin. Just because it’s not a household name doesn’t make it less harmful.
Deeper Health Concerns
Some chemicals seem harmless until you read reports of chronic exposure. TNF falls under the category of nitro compounds. Similar relatives sometimes damage blood’s ability to carry oxygen, causing symptoms like fatigue or even cyanosis—the bluish tint skin takes on with low oxygen. Animal studies on other nitro compounds show potential for liver and kidney stress. With TNF, the long-term effects haven’t been studied well in people, so uncertainty looms. The lack of information doesn’t mean safety; it often means gaps in research funding and regulatory oversight.
Chemists who handle odd-smelling or brightly colored powders get good at listening to their instincts. If a compound has sharp odors and stands out in a crowded chemical cabinet, it never hurts to double-check the safety sheet. From my own years around research laboratories, I always noticed that people who cut corners ended up with burnt hands or worse. A moment’s carelessness with TNF could have consequences that last much longer than a single shift.
Is Regulation Keeping Up?
TNF doesn’t attract as much government attention as asbestos or benzene. Its industrial use stays small-scale, so there’s less pressure from workers’ unions and public health advocates. That means workers might not get the training or the equipment they would with better-known hazards.
Sometimes companies figure a less common chemical requires less safety oversight. In reality, those are the situations where problems slip through. Employers who really value health will make sure TNF gets the same respect as anything else with a long, complicated name.
Better Handling Means Fewer Risks
Proper glove material keeps TNF off skin. Good fume hood flow removes dust before it can get to your lungs. Regular eye protection covers accidental splashes. Places that train workers to respect all chemicals—including less regulated ones like TNF—see far fewer accidents.
Everyone from students to seasoned chemists gets told early to treat any new substance as potentially dangerous until proven otherwise. That’s not paranoia. For many compounds, including TNF, science hasn’t caught up to all the possible side effects. I’ve found that asking for more ventilation, stronger gloves, or extra training rarely slows down the work but always keeps people out of harm’s way.
How should Trinitrofluorenone be stored?
Understanding Why Storage Matters
Trinitrofluorenone might not turn up much in everyday conversation, but anyone who spends time in a chemistry lab knows its reputation. It’s a strong oxidizer, bright yellow, and sensitive to heat, friction, and shock. These details make the storage question more than academic. Real-world experience shows how easy it is for simple storage mistakes to invite risks you’d rather not face. Ignoring safe habits around such chemicals could lead to more than a mess—it can threaten everyone in the building.
Direct, No-Nonsense Storage Practices
For a compound like trinitrofluorenone, storage takes discipline. Rule number one: use an airtight glass container, protected from light. Light and moisture can trigger unwanted reactions, so a container made of amber glass and fitted with a tight seal does a lot of heavy lifting. Avoid any old bottle—choose one with chemical-resistant properties, and clearly label it with hazard warnings, not scribbled notes.
Every lab has its lazy days, but keeping trinitrofluorenone in a locked, dedicated chemical storage cabinet pays off every time. Place it away from acids, reducing agents, and flammable materials. Heat can sneak in through shared cabinets, so keeping oxidizers separate stops avoidable fires before they start. A high-quality, explosion-proof refrigerator becomes essential in settings where the heat outside can’t be trusted. I learned the hard way that crowded chemical fridges tend to hide damage or spills; regular checks keep small leaks from becoming disasters.
People on the Ground See the Hazards
Working with energetic chemicals, small oversights go a long way. In college, one missed lid led to a sticky yellow mess and a round of evacuations. That lesson sticks years later: drying trinitrofluorenone can ignite if storage falls short of best practice. Experience teaches to keep this material out of reach of students, restrict it to trained users, and double-check every element of the storage chain, from inventory to disposal.
Support From Science and Safety Guidance
Looking up the latest Material Safety Data Sheet (MSDS) pays off. According to the Centers for Disease Control and Prevention (CDC) and Occupational Safety and Health Administration (OSHA), trinitrofluorenone ranks as a chemical where stability matters as much as any other property. MSDS sources stress minimizing temperature swing and avoiding contact with metals or organic material that could spark a reaction.
Solutions For Safer Handling
Instituting a buddy system for tricky inventory makes sense. Don’t store trinitrofluorenone solo; have a second set of eyes inspect the setup. Digital logs and regular audits add an extra layer of safety. Outdated containers belong in a strict disposal pipeline, not the back of the shelf. Online resources, such as safety training modules, give both experienced chemists and newcomers practical reminders. Community colleges and universities investing time in regular drills help everyone know what to do if something leaks or breaks. Theft prevention also matters—locked cabinets and sign-out tracking keep tabs on every gram.
Staying Ahead Of The Risks
Preserving health and safety comes down to expecting the unexpected. Chemistry labs, research facilities, even museums housing old chemical samples, all benefit from treating trinitrofluorenone with sustained respect and habit-forming caution. Each step of careful storage doesn’t just follow a rule—it means another shift finishes without accidents, injuries, or property damage. Real safety is always personal. People who treat hazardous chemicals with gravity and care protect more than themselves—they set the standard for everyone around them.
What is the chemical structure of Trinitrofluorenone?
Why Structure Matters in Chemistry
Chemistry often feels intimidating, with its jargon and tangled diagrams. Still, every structure tells a story. Trinitrofluorenone (TNF) isn’t just another tongue twister. This chemical packs a punch in both its function and its atomic layout. TNF’s structure sits as a fluorenone core—a three-ring system fused together, something you would spot in an old organic chemistry textbook. Chemists add three nitro groups (NO2) to the mix, sticking them right onto that familiar fluorenone framework. Each nitro group lands in the 2, 4, and 7 positions around the core. That’s where the magic happens: these nitros pull electron density from the rings, making the whole molecule a much stronger electron acceptor than plain old fluorenone.
Drawing It Out
Think of the skeleton as a line of three benzene rings sharing borders, interrupted by a single ketone group (CO) at the central ring. The three nitro groups dangle on the periphery, looking almost like ornaments on a tree. The shape isn’t just for show. This arrangement lets TNF soak up electrons easily, a trait that underpins its tech uses and its hazards.
Why Should Anyone Care?
To some, a chemical structure feels distant. For anyone working in electronics, TNF means business. Its electron-sucking ability gives it a job as a charge transport agent. TNF often turns up in electrophotography—think of how copiers and printers make your images leap onto a page. TNF can be blended into organic semiconductors. These films enable some of the advances in organic light-emitting diodes (OLEDs) used in phone screens and high-end TVs. Without that particular arrangement of three nitro groups and one ketone, TNF would lose its edge. The molecule’s structure, dictated by the precise location of nitro groups, boosts its performance and reliability as an electron acceptor.
Handling Risks in the Real World
The fun stops if you ignore safety. Nitro groups, as any seasoned chemist knows, bring with them a potential for danger. Molecules packed with nitros can become shock-sensitive, prone to decomposition, or even explosive under the right conditions. Although TNF is more stable than some notorious relatives, handling always deserves serious care—proper storage, avoidance of excess heat, and solid waste protocols keep laboratories safe. Industrial settings leverage automated handling, not just for speed but for minimizing accidents. Over the years, mishaps have shown that underestimating such molecules can lead to consequences ranging from ruined equipment to much worse.
Solutions and Accountability
Researchers and manufacturers follow clear, strict standards. Regulatory agencies offer guidance rooted in past lessons, aiming to prevent health issues and fires. Using material safety data sheets (MSDS) and investing in proper personal protective equipment proves worthwhile every single time. Keeping up with current best practices, from containment procedures to regular audits, helps everyone—from academics to factory workers—avoid preventable mistakes.
Continuing Innovation
Efforts in green chemistry have started to look at substitutes for TNF in some contexts, aiming to reduce hazardous waste while maintaining the performance needed for hi-tech devices. Some labs are engineering derivatives that keep the electron-accepting kick but with tweaks that limit sensitivity and environmental persistence. Chemists, with trial and error, inch closer to safer, cleaner alternatives every year.
What safety precautions are required when handling Trinitrofluorenone?
Respecting the Risks in the Lab
Anyone who has spent time in a chemistry workspace knows that labeling and process consistency get drilled in early. Safety comes alive with compounds like trinitrofluorenone. This bright yellow powder doesn’t demand much—until it gets in your lungs, touches skin, or reaches an ignition source. Those of us who’ve spilled more than coffee in a lab learn the hard way: reliable habits beat shortcuts every time.
Understanding the Hazards
Trinitrofluorenone brings explosive and toxic risks. Breathing the dust can irritate your respiratory system. It stings and colors skin. Get careless with open flames, and the stuff can ignite or detonate—sometimes without warning. Memory holds the sound of glass shattering further down a bench. Stories like those from safety briefings or old lab hands hit different after you’ve worked with volatile organics yourself.
Protective Equipment that Actually Gets Used
Lab coats and chemical goggles sound basic, but life runs smoother when you never skip them. Nitrile gloves keep yellow off your hands and chemicals from entering your skin. Dust masks or, better yet, fitted respirators go from optional to mandatory when weighing or handling solids. I remember a project mate who put comfort over protection; it only took one batch of nitro compounds before he chose equipment over fashion. Failure to protect eyes risks lasting injury—most underestimated this until a single splash.
Fresh Air and Fume Hoods
A glorified kitchen fan does not cut it. Fume hoods with steady airflow catch vapors and dust before you do. A well-maintained hood trimmed risk more than any warning sign. That moment you smell something bitter through your mask and realize the hood isn’t running reminds you to check airflow every session. Never work with hazardous organics in the open.
Controlling Static, Sparks, and Ignition
Static charges built up from synthetic clothing have triggered more than one surprise. ESD-safe smocks—at first glance, overkill—just make sense for trinitro products. Cell phones belong outside the lab; uncontrolled sparks from electronics or even metal tools can light up a bad day. Every chemist I know keeps necessary items grounded, never shuffling feet more than needed.
Waste and Storage: More than Just Rules
Trinitrofluorenone needs cool, dry, and locked-up storage. Containers lock tightly. Labels matter—mistaken identity leads to stories best left untold. Don’t let waste stockpile or get confused with safe material. Mark containers clearly and dispose through official hazardous waste channels. A single shortcut with disposal set up legal, social, and health disasters that affected whole teams.
Training, Checklists, and the Culture of Speaking Up
Real safety builds from more than a binder full of protocols. Training sticks when people believe it applies to real lives. Seasoned workers lead by example, triple-checking steps and reminding others to slow down. After seeing senior chemists ask questions and flag risks, confidence builds for everyone to do the same. Fresh eyes sometimes spot overlooked dangers, so open comments never get dismissed.
Constant Vigilance and Real Accountability
Lab safety culture proves itself when nobody gets complacent. Documentation, strict PPE, and clear communication win each time. The compounds never forgive laziness or bravado. Everyone earns the right to go home in one piece by respecting chemicals like trinitrofluorenone and pushing for improvements where gaps appear.
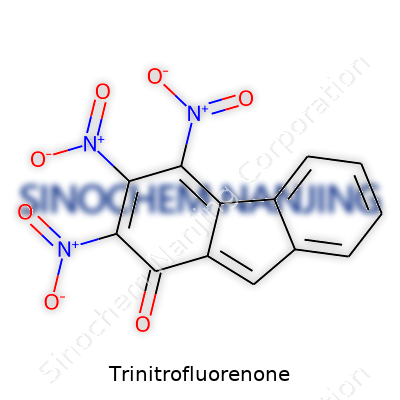
| Names | |
| Preferred IUPAC name | 9,10-Dioxo-2,4,5-trinitro-9,10-dihydroanthracene |
| Pronunciation | /traɪˌnaɪtroʊfluːəˈnoʊn/ |
| Identifiers | |
| CAS Number | 1510-30-3 |
| Beilstein Reference | 1428248 |
| ChEBI | CHEBI:52725 |
| ChEMBL | CHEMBL353225 |
| ChemSpider | 28528 |
| DrugBank | DB04174 |
| ECHA InfoCard | 100.009.204 |
| EC Number | 208-329-2 |
| Gmelin Reference | 73692 |
| KEGG | C14122 |
| MeSH | D014269 |
| PubChem CID | 66636 |
| RTECS number | WN2625000 |
| UNII | 3091K3R2N2 |
| UN number | 1325 |
| CompTox Dashboard (EPA) | DTXSID6049156 |
| Properties | |
| Chemical formula | C13H3N3O7 |
| Molar mass | 347.185 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.67 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.60 |
| Vapor pressure | 1 mmHg (196 °C) |
| Acidity (pKa) | -0.21 |
| Basicity (pKb) | 11.86 |
| Magnetic susceptibility (χ) | -74.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.684 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.71 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -372.2 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P210, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 113°C |
| Autoignition temperature | 285 °C |
| Lethal dose or concentration | LD50 (oral, rat): 320 mg/kg |
| LD50 (median dose) | 1130 mg/kg (rat, oral) |
| NIOSH | TT2975000 |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | Unknown |

