Trinitrobenzenesulfonic Acid: A Closer Look at a Powerful Chemical
Historical Development
Trinitrobenzenesulfonic acid didn't pop up overnight. Its story runs back to an era when scientists started exploring the world of aromatic nitro compounds in the late 1800s. The curiosity of that time pushed laboratories to tinker with benzene derivatives, searching for not just explosives, but also tools for organic synthesis and probing into the structure of biomolecules. Chemists learned early on that sulfonation of benzene could add versatility, while nitration created a new realm of chemistry altogether. Mixing these strategies, pioneers like Baeyer and his contemporaries laid the path for trinitrobenzenesulfonic acid, not only for academic reasons but also for industrial pursuits. Things ramped up in the twentieth century as folks realized the compound's usefulness in immunology and biochemistry, far beyond its roots in explosives and dyes.
Product Overview
You don’t find trinitrobenzenesulfonic acid lining the shelves of hardware stores or household supply shops. It mainly lives in research labs, where its strong reactivity and unique properties earn it a spot in many experiments. In my own experience working with protein labeling, I witnessed how a single drop of its concentrated form can kick off reactions that are tough to pull off with other chemicals. It’s got a reputation for being potent, a little hazardous, and extremely valuable for precise analytical work.
Physical & Chemical Properties
Trinitrobenzenesulfonic acid tends to show up as a yellow to orange powder, sometimes as dense crystals. Its intensity comes from the three nitro groups attached to the benzene, turning the molecule into a strong acid with notable oxidizing strength. This also makes it a sensitive compound, eager to take part in various chemical reactions and quick to stain surfaces. It dissolves well in water, giving a deeply colored solution. Prolonged exposure to air or light can cause decomposition, producing nasty fumes. I remember once forgetting a container without sealing it tightly—it was not a pleasant cleanup.
Technical Specifications & Labeling
There’s no getting around the need for clear and proper labeling with this compound. Mistakes can result in burns, inhalation risks, and legal problems. Reputable suppliers provide trinitrobenzenesulfonic acid with robust labeling—chemical name, concentration, hazard warnings, and date of manufacture. Specifications like purity percentages matter most to chemists who need predictability in outcomes. Safety data sheets demand attention to the combustion and explosion risks, with reminders about proper storage conditions. That’s something no seasoned technician ignores.
Preparation Method
Making trinitrobenzenesulfonic acid usually starts with sulfonated benzene, which then undergoes triple nitration. It’s not the sort of process that fits in a home lab. Concentrated nitric and sulfuric acids feature heavily, and this mix can get dangerously hot and volatile. Careful temperature control means more than just setting a dial; it calls for hours of vigilant monitoring. Even seasoned professionals keep blast shields and emergency showers close. The production scale hasn’t changed much in decades, displaying how powerful the old chemical engineering techniques remain.
Chemical Reactions & Modifications
Few reagents act with such brute force on biological molecules as trinitrobenzenesulfonic acid. Throw it into a solution of proteins or amines, and it launches into derivatization, tagging them with nitro groups that shift their properties and colors. Researchers rely on this property for quantifying certain types of biomolecules. Its electron-withdrawing capacity also makes it an interesting precursor for other explosives or high-density materials, though those applications raise serious safety questions and ethical debates. Reactive as it is, the compound doesn’t adapt well to casual tinkering—most modifications need careful, well-documented steps.
Synonyms & Product Names
Scientists like to use shorthand, yet trinitrobenzenesulfonic acid has a handful of aliases: TNBS, 2,4,6-trinitrobenzenesulfonic acid, and, less commonly, picryl sulfonic acid. For procurement and inventory, sticking to standardized names helps avoid confusion—no one wants the wrong compound showing up in the mailroom, especially not with this kind of risk profile. No matter the name, people in the know treat this molecule with real respect.
Safety & Operational Standards
Lab veterans never get casual with trinitrobenzenesulfonic acid. It burns skin, damages eyes, and releases toxic gases under the right conditions. Wearing goggles, gloves, and lab coats forms the baseline. Proper fume extraction and spill control gear sit within arm’s reach. Even disposal needs planning, since flushing it down the drain spells trouble for water treatment systems. Beyond basic lab protocols, local environmental and fire regulations dictate transport and storage. One slip-up can cause injuries or worse—a truth hammered home during every annual safety refresher.
Application Area
Trinitrobenzenesulfonic acid finds its way into immunology, especially as a reagent for labeling free amino groups in proteins. Assays built on this chemistry reveal how much glycine, lysine, or primary amine groups exist in a solution—a trick immunochemists have leaned on for half a century. Medical researchers use the compound for inducing colitis in animal models, a staple method for studying inflammatory bowel diseases. Outside the life sciences, it comes up occasionally in energetic material research, though concerns about safety and stability keep those uses somewhat restricted. Chemical manufacturers often partner with academic labs on refining protocols for protein modification, since the wild reactivity of trinitrobenzenesulfonic acid allows for elegant yet complex molecular designs.
Research & Development
R&D around trinitrobenzenesulfonic acid revolves around tightening up both its safety profile and its specificity. Chemists push to reduce accidental exposures and unwanted side reactions. In my time at a university lab, even small advances in containment and automation made everyone breathe easier. Researchers probe for new ways to harness the compound’s labeling ability, searching for methods that lower the need for such hazardous chemicals without sacrificing accuracy. Green chemistry movements have pushed teams to explore alternative reagents. Still, no perfect substitute matches the selectivity and intensity trinitrobenzenesulfonic acid brings to the table for certain assays. Funding for safer handling and decontamination methods continues to grow, following a trail of both safety incidents and regulatory pressure.
Toxicity Research
The hard truth about trinitrobenzenesulfonic acid is that it’s genuinely nasty stuff in untrained hands. Inhalation or contact can lead to digestive issues, respiratory problems, and, in the worst cases, severe organ damage. Colitis models involving this compound highlight how quickly it inflames tissue. Toxicologists keep a close eye not only on its acute effects but also on the lingering impact of low-level exposure. Institutions with large chemical inventories routinely audit where and how the substance is stored, mindful of the chronic risks for both humans and the environment. Calls for better antidotes and rapid neutralization methods reflect real-world risk—every misstep has prompted procedural reforms in research facilities.
Future Prospects
Folks working with trinitrobenzenesulfonic acid have their eyes set on several possible futures. Developing non-toxic, highly specific alternatives for protein labeling tops most wish lists in analytical chemistry. There’s a push for microfluidic devices and closed systems to keep exposures close to zero. With automation advancing quickly, tasks that once demanded personal risk can shift to sealed robots or reaction chambers. Environmental monitoring could also see upgrades, flagging leaks or spills before they spiral into bigger incidents. Startups and academic groups alike are hunting for green chemistry pathways that eliminate or minimize the use of intensely hazardous reagents across all industries. Progress takes time but history shows that the drive for safety and precision outlasts even the toughest chemical challenges.
What is Trinitrobenzenesulfonic Acid used for?
The Science Behind Trinitrobenzenesulfonic Acid
Walk into any well-equipped biochemistry lab, and someone’s probably reaching for trinitrobenzenesulfonic acid (usually dubbed TNBS). Its full name sounds intimidating, but it’s a go-to tool for scientists working with proteins and cells. TNBS isn’t something you’ll see on a shelf at the local pharmacy. Instead, it has built its reputation inside lab drawers, especially among researchers trying to measure and analyze complex molecules.
Protein Analysis and Beyond
TNBS shines when detecting amino groups in proteins or small peptides. Researchers rely on it for protein quantification—the acid tags onto those amino groups, causing a visible color change. That color tells scientists how much of the protein they have. The process is easy to grasp: mix your protein with TNBS, wait for the bright yellow hue, then compare the intensity with a standard. Labs use this every day for everything from food science to clinical research.
In everyday lab life, I’ve used TNBS to check the progress of chemical reactions that modify proteins. If you’re developing a new drug or just tweaking molecules for better function, you want to know where changes happen. TNBS gives a fast answer. The same reaction helps spot free amino groups in polymers or new biomaterials, driving progress in both medicine and material science.
Highlighting Immune Reactions
TNBS doesn’t just help with chemistry—its impact reaches into immunology as well. Scientists use it to induce and study inflammation in animal models, especially in the gut. By triggering inflammation in this controlled way, researchers can test new treatments for diseases like Crohn’s or ulcerative colitis. It’s tough work, but TNBS gives consistent results, letting teams compare therapies and understand the basics of immune system overdrive.
Risks and Responsibility
Any tool this powerful carries risks. TNBS can cause burns or serious injury if mishandled. Long-term exposure, even to small amounts, may create health problems. In my own experience, the strong odor alone tells you to wear gloves and a mask. Lab safety guidelines treat TNBS with the caution it deserves: proper fume hoods, chemical-resistant gloves, and clear training keep researchers safe.
Moving Toward Safer Alternatives
More labs have begun searching for gentler substitutes. New reagents promise less toxicity and fewer environmental concerns, but the classic TNBS method keeps its following because it works so reliably. As labs share data and alternatives become affordable, the shift away from harsher chemicals grows. Every year brings new ways to tackle protein analysis with fewer health hazards.
Supporting Reliable Science
In research, precision and repeatability win out. TNBS offers a straightforward, sensitive way to detect and measure amino groups—a small detail that matters in everything from food testing to cancer research. My time in the lab has shown me the value of tools that deliver clear results. While the science world keeps pushing for safer, faster, and greener options, TNBS remains a testament to the power of solid chemistry.
What are the safety precautions when handling Trinitrobenzenesulfonic Acid?
Understanding What You’re Dealing With
Trinitrobenzenesulfonic acid isn’t something you roll around your garage with. The chemical packs a mean punch, both as a powerful irritant and as something that can do real harm with just a bit of carelessness. Skin might burn on contact. Eyes water and sting. Inhaled vapors take a toll on your lungs. The memory of that sharp odor lingers in your nose much longer than you’d like. I’ve worked with industrial chemicals, so I’ve seen what skipping a step costs—a burn here, a hospital trip there. Labs share horror stories about bottles knocked over, gloves springing leaks, or someone tossing a vial in the wrong waste bin. This acid isn’t shy about showing what it can do.
Personal Protection Isn’t Optional
No one wants red, angry skin or a trip to the ER. Lab coats help but a layer of nitrile gloves, splash goggles snug against your face, and sometimes a full-face shield give the best shot at keeping the acid away from you. I always pull on gloves that extend up the wrist and check each one for pinholes before starting. The acid can chew through latex, so stick with nitrile or neoprene gloves—no shortcuts. Many folks overlook shoe covers, but spills find shoes and socks fast. Closed shoes, long pants, and sometimes a plastic apron prevent drops from burning through your clothes and onto your skin. I’ve had an unexpected splash ruin my day once. It only took seconds.
Ventilation Keeps You Breathing
Decent ventilation turned a risky job into safer work. A plain workbench isn’t good enough. A certified fume hood, exhaust fans, and sometimes a portable air monitor knock down the risk of breathing in dangerous vapors. On a hot day, that smell becomes more pronounced, and coughing starts if the air gets heavy. No experiment or workplace rush is worth breathing in that fog.
Storage Rules That Can’t Wait
No matter how tough you think your container is, acids like this demand their own space. Never put it near organic combustibles, bases, or metals. I use glass containers with tight PTFE-lined caps. Every bottle gets labeled so nobody makes a mistake. The label should show the chemical’s full name in clear letters—no abbreviations or mysterious code words. Fireproof cabinets aren’t just for show. A locked one keeps both fire and curious coworkers at bay.
Cleaning up Spills
Here’s where I’ve seen the most trouble. Someone drops a flask, then grabs a random rag or throws sawdust on the puddle. Big mistake. Small spills call for a neat scoop of inert absorbent—vermiculite works—never paper towels, which may react or smolder. Neutralizing comes after containment. All waste, from gloves to soaked materials, lands in a sealed chemical bag and heads straight to hazardous waste disposal. The memory of chemical-soaked trash can catching fire sticks with me. You don’t forget that fast.
Emergency Readiness
An eyewash station and emergency shower should work without fuss. I always run a quick check before starting, whether people laugh or not. The first seconds after a splash or inhalation matter most. Bottles of clean water don’t take the place of a real eyewash station, no matter how clean the lab seems. Phones and emergency numbers need to be close. People waste critical time searching for help amid panic, but a sheet taped by the door speeds up response.
Training and Respect
Folks new to handling dangerous chemicals need solid, hands-on training—not just reading manuals or sitting through online modules. Seeing the right way to suit up, contain spills, and get rid of waste brings home the risks much clearer than slides ever could. Watching someone with gray hair and decades of scars show how to check gloves makes it stick. Mistakes cost. Respect for chemistry goes hand-in-hand with careful habits.
How should Trinitrobenzenesulfonic Acid be stored?
Challenges Faced in the Lab
Trinitrobenzenesulfonic acid shows up in research labs across the globe. Its reputation isn’t just built on its strong chemical properties—it also demands attention due to the risks it brings. Anyone who’s worked with strong acids or energetic compounds knows how a simple oversight leads to costly setbacks or, worse, injuries. Years spent moving between chemical storerooms and research spaces have taught me something about treating hazardous substances with respect.
This compound doesn’t forgive sloppy storage. Its sensitivity to moisture, heat, and potential incompatibles really doesn’t leave much room for shortcuts. An unventilated shelf or a loose cap makes a huge difference here. One time, I watched a senior colleague stop a junior tech from storing a sample in a cabinet shared with oxidizers. The warning wasn’t theoretical: Trinitrobenzenesulfonic acid can react with common chemicals, and houses can end up learning that risk the hard way if stacks of incompatible materials gather dust side by side.
Keeping Danger in Check
The first step always involves choosing the right container. Glass usually wins out because it doesn’t react, and acid-resistant plastic stands in for smaller samples. Labeled containers aren’t just for show—they alert newcomers and help veterans in a hurry. Seals need to hold tight. No one wants acid fumes leaking and settling dust on the next shelf.
Placement determines outcome. Low shelves, away from sunlight and heaters, serve best. Top shelves might seem convenient, but nobody wants to reach overhead for a spill-prone acid. I once saw the aftermath when a bottle fell from the top rack: the cleanup went on for hours, and questions about exposure didn’t fade quickly.
Humidity turns storage into a game of chance. That means a dedicated, dry cabinet, ideally equipped with a chemical-resistant liner or spill tray, earns its keep. Some labs use flammable and corrosive storage cabinets with proper ventilation—worth every penny in peace of mind. Temperature swings should stay out of the picture, so spaces with climate control work best. Trinitrobenzenesulfonic acid doesn’t handle sudden changes in humidity or heat. That’s one reason storing it next to a window or near radiators never makes sense.
The People Factor
Training shouldn’t get shortchanged. New lab members need live walkthroughs, not just PDF protocols. I remember a safety officer’s advice: “Trust the protocols, but double-check your neighbor’s storage, too.” Peer checks have caught more than a handful of near-mistakes over the years.
Everyone’s better off with policy that encourages honest reporting and regular inspection. A single missed inspection allowed a minor bottle leak to linger in one research group for weeks. When the smell grew sharp enough to catch attention, a system overhaul followed. Frequent, scheduled checks help catch corroding caps, faded labels, or unauthorized relocations before surprises set in.
Making Improvements Matter
Stronger use of inventory software matters. In my experience, digital logs alert us to shelf-life, record who moves a bottle, and flag needed inspections. In research settings, these tools keep chaos at bay and support insurance policies, too—an audit trail can mean the difference between getting claims processed or stuck.
Facilities can back up safety with spill kits, eyewash stations, and clear emergency procedures pinned on storage doors. No one expects a spill, but the right supplies and training keep small incidents from turning major.
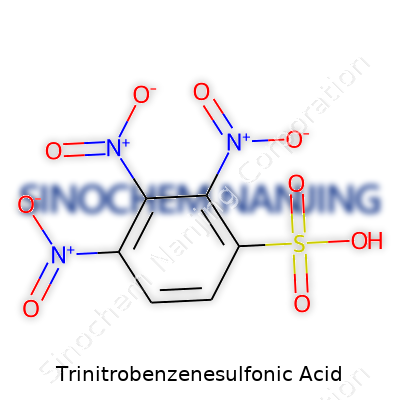
Focusing on these habits turns trinitrobenzenesulfonic acid from a terrifying prospect into a routine piece of laboratory work—and that confidence, built one good storage practice at a time, saves more than just glassware.What is the chemical structure of Trinitrobenzenesulfonic Acid?
What Makes Trinitrobenzenesulfonic Acid Tick?
Trinitrobenzenesulfonic acid—often shortened to TNBS—draws attention among chemists for good reason. It’s not just its tongue-twister name. In the lab, TNBS plays a key role in mapping out immune responses and checking protein structures. Curious minds might ask: what’s going on at the molecular level to give this compound its distinct punch?
Breaking Down the Molecule
Picture a benzene ring, the classic hexagon found in everything from gasoline to medicine. On this ring, imagine three separate nitro groups (–NO2) attached at positions 2, 4, and 6. That leaves positions 1 and another free. Take the first position and anchor a sulfonic acid group (–SO3H). The structure stacks up as 2,4,6-trinitrobenzenesulfonic acid.
So, we end up with a molecule that surrounds itself with electronegative groups—the nitros and the sulfonic acid. These heavy electron seekers drain internal electron density, which explains why TNBS acts so aggressively in chemical reactions. This matters, especially in the lab during protein and amine analysis, because it lets TNBS form stable, recognizable complexes.
Why the Structure Carries So Much Weight in Research
Most scientists know TNBS as a reagent for labeling amino groups or even for setting off experimental colitis in animal models. The underlying reason sits in its structure: multiple nitro groups packed onto a ring with a highly soluble sulfonic acid group. The nitro groups throw serious electron-withdrawing effect, ramping up the chemical reactivity. With the sulfonic acid group hanging off, TNBS dissolves well in water, giving researchers an easier time using it in biological settings.
This isn’t just an abstract chemistry exercise. When studying proteins or mapping immune responses, having a molecule that reacts fast and predictably gives people confidence in their results. That level of reliability isn’t a given in every chemical reagent.
Risks That Come With the Territory
Trinitrobenzenesulfonic acid isn’t some harmless kitchen ingredient. The same features that make it valuable in research give it a dark side. Nitro compounds like these tend toward toxicity. I’ve worked in labs where vigilance with TNBS made all the difference between a successful experiment and an emergency. Skin, eyes, and lungs—none of them fare well when exposed to this compound. Proper gloves, a fume hood, and some healthy respect for the dangers become non-negotiable. Safety data sheets on TNBS read like heavy warnings for a reason.
Seeking Better Practices and Solutions
Trustworthy lab safety protocols look boring at first glance. There’s really no shortcut here. Making sure containers of TNBS stay sealed, using minimal amounts, and always having spill kits nearby creates a safer workplace. Labs investing in containment—like gloveboxes or properly ventilated hoods—see far fewer mishaps. Teaching new researchers about real chemical risks, instead of downplaying the hazards, saves more trouble than most realize. Some teams experiment with less hazardous substitutes in their protocols, especially when moving to scale. It’s a slow road, but broadening the menu of reagents helps everyone in the long run.
TNBS in the Big Picture
The shape and makeup of trinitrobenzenesulfonic acid enable enormous progress in biology, immunology, and protein science. By truly understanding what’s on the molecular canvas, chemists and biologists sidestep guesswork and work more safely, driving their research forward while respecting the power bottled up inside those hexagons and nitro groups.
Is Trinitrobenzenesulfonic Acid hazardous to health or the environment?
Hazards in the Lab and Beyond
Trinitrobenzenesulfonic acid can bring real risks to the lab bench and outside the lab if handled without proper care. A bright yellow powder, this chemical draws attention not just for its appearance. Its structure contains both a sulfonic acid group and three aromatic nitro groups, combining aggressive acidity with a strong tendency for oxidation. This isn’t the sort of powder you leave lying around. In my own days spent hunched over lab benches, we marked containers with thick tape and red warnings, never trusting our memory alone.
One mistake, a small spill, or simple skin contact can lead to burns that stubbornly refuse to heal. The acid burns deeply, not just surface irritation, and the nitro groups only make things worse. Eyes, lungs, and skin—none of them fare well if exposed. I learned quickly that a fumbled procedure or a torn glove could turn a physical demonstration into a warning story for new students down the line.
Routes to Exposure
People often underestimate how easily a hazardous chemical enters the body. Sure, you expect to keep your hands away. Yet, fine dust settles on counters, on sleeves, or even on a neighboring notebook. Dry particles float in the air after careless handling, and a quick swipe to the eyes spreads the chemical where it’s hardest to undo the damage. Personal protective equipment helps, but vigilance matters more. Inadequate ventilation, poor housekeeping, and skipped safety briefings lay the groundwork for harm in well-equipped and under-resourced labs alike.
Lingering Effects on Health
Immediate effects, such as chemical burns, tell only part of the story. Over time, exposure to nitroaromatic compounds like trinitrobenzenesulfonic acid has triggered allergic skin reactions and respiratory symptoms, with some workers reporting asthma-like responses. For those with repeated contact—researchers, production workers, even emergency responders—occupational health studies repeatedly link overexposure with chronic effects ranging from skin sensitization to more severe lung inflammation. Some nitroaromatics also raise concern for their links to methemoglobinemia, a blood disorder affecting oxygen delivery, though hard numbers remain sparse outside high-exposure settings.
Environmental Risks
This compound poses challenges that stretch beyond the walls of research facilities. Environmental release during improper disposal, accidents, or leaking containers leads to water contamination or soil pollution. Nitroaromatic substances tend to linger in nature, breaking down slowly if left unchecked. Fish, plants, and soil organisms face toxic effects at surprisingly low concentrations. There’s a well-documented record of similar compounds harming aquatic life and disrupting delicate food chains in rivers and lakes. Local advisories now often flag nitroaromatic contamination as a top concern in regions near chemical plants or legacy dump sites.
Better Management, Less Harm
Some real progress has come from tighter rules for chemical storage and better emergency response training. Every competent facility now keeps thorough records, trains staff in safe handling, and uses disposal contractors with a track record for safe destruction. Laboratories swap out the most hazardous chemicals for less dangerous forms whenever possible. Strong chemical fume hoods, eye wash stations, and gloves never substitute for a clear line of responsibility or accountability—both matter just as much for keeping people and the surrounding land safe. The lesson I learned: nothing cuts corners with a compound like this, and careful respect for the risks pays off for everyone involved.
| Names | |
| Preferred IUPAC name | 2,4,6-Trinitrobenzenesulfonic acid |
| Other names |
TNBS Picrylsulfonic acid 2,4,6-Trinitrobenzenesulfonic acid |
| Pronunciation | /traɪˌnaɪtroʊˈbɛnziːnˌsʌlˈfɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 88-39-1 |
| Beilstein Reference | 136654 |
| ChEBI | CHEBI:53059 |
| ChEMBL | CHEMBL1236077 |
| ChemSpider | 21419 |
| DrugBank | DB08326 |
| ECHA InfoCard | 100.036.375 |
| EC Number | '209-409-9' |
| Gmelin Reference | 8783 |
| KEGG | C01772 |
| MeSH | D014275 |
| PubChem CID | 657547 |
| RTECS number | XS9625000 |
| UNII | 5B8U4U31KN |
| UN number | UN2817 |
| CompTox Dashboard (EPA) | DTXSID0026937 |
| Properties | |
| Chemical formula | C6H3N3O9S |
| Molar mass | 289.14 g/mol |
| Appearance | Yellow powder |
| Odor | Odorless |
| Density | 1.682 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -0.50 |
| Vapor pressure | 0.0025 mmHg (25 °C) |
| Acidity (pKa) | -1.0 |
| Basicity (pKb) | -5.49 |
| Magnetic susceptibility (χ) | -64 × 10⁻⁶ cm³/mol |
| Viscosity | 1.33 cP (25°C) |
| Dipole moment | 3.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −46.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1446.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08, GHS09 |
| Pictograms | GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H290, H314, H317, H334, H335, H341, H373, H411 |
| Precautionary statements | P260, P264, P270, P271, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P311, P321, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | Health: 4, Flammability: 2, Instability: 2, Special: OX |
| Autoignition temperature | 150 °C |
| Lethal dose or concentration | Lethal dose or concentration (string): Rat oral LD50 640 mg/kg |
| LD50 (median dose) | 101 mg/kg (rat, oral) |
| NIOSH | WW4900000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Trinitrobenzenesulfonic acid sodium salt Picric acid Trinitrotoluene (TNT) Dinitrobenzenesulfonic acid Nitrobenzenesulfonic acid Trinitrobenzene Sulfanilic acid |

