Trinitroanisole: Scratching the Surface of History, Chemistry, and Future Applications
Historical Development
Trinitroanisole doesn’t show up in folk tales or popular media, but it’s been a quiet player in the world of energetic materials since the mid-20th century. It showed up in research after World War II, a period when countries threw themselves into innovation for new explosives, propellants, and specialty chemicals. Trinitroanisole made the cut due to its bench-stable nature and decent detonation characteristics compared to more sensitive nitroaromatics. Although overshadowed by TNT and RDX in field use, chemists kept trinitroanisole on their drawing boards because it gave them something different to work with: a blend of safety, power, and modifiable structure. It never stole the limelight but stuck around for good reason. Any growth in knowledge around energetic materials makes the whole field less hazardous and a lot more interesting.
Product Overview
Trinitroanisole sounds fancy, but at its core, it’s a nitroaromatic compound where three nitro groups and one methoxy group attach to a benzene ring. It looks like golden-yellow crystals, packs some heat, and gives manufacturers and researchers a chance to sidestep some of the issues associated with legacy materials like pure trinitrotoluene. Trinitroanisole brings a tempting mix of moderate sensitivity and reliable performance, plus it lends itself to various chemical tweaks thanks to that methoxy group. Nobody grabs trinitroanisole for backyard fireworks or construction blasting, but in a lab or pilot-scale operation, it pulls its weight where precise control and analytical ease count.
Physical & Chemical Properties
The first thing any chemist notices is the powder's yellow hue and crystalline shape. It melts above normal room temperature, doesn’t readily absorb moisture, and keeps its head down unless provoked. Those three nitro groups promise serious oxidizing energy, while the methoxy group lowers the shock sensitivity compared to other nitroaromatics. It dissolves in common organic solvents, which makes purification jobs less of a headache. Chemically, you get a stable core structure but enough sites on the molecule for interesting modifications—perfect if you want to develop derivatives or study push-pull electronic effects. Anyone handling it needs solid lab discipline because, like most nitroaromatics, sloppy handling can invite trouble.
Technical Specifications & Labeling
Trinitroanisole stands apart because of its straightforward formula and defined melting point, which makes quality control honest and verification reliable. Packaging information often mentions crystal size, moisture content, and purity levels. In practice, high-purity samples get flagged for research, while less-refined batches might see use in preliminary or scale-up trials. Labels on trinitroanisole warn about flammability, toxicity, and shock risk—traits no one should gloss over. My own experience taught me that, despite relative stability compared to nastier compounds, a little respect goes a long way toward keeping everyone safe.
Preparation Method
Historically, trinitroanisole synthesis starts with anisole—an off-the-shelf chemical—which then goes through nitration reactions. That old-school sulfuric and nitric acid mix gets the job done as long as the temperature stays under the boiling point of destruction, so to speak. Control the reaction, and you get respectable yields; rush or overheat it, and you end up with tars and trouble. Waiting for crystal formation and then washing and drying in a controlled environment helps keep the final compound pure. In modern labs, automation and closed systems handle these steps, cutting down on accidents, but the principles haven’t changed since the earliest patents.
Chemical Reactions & Modifications
This is where trinitroanisole earns its reputation in research. That methoxy group attaches like a handle for chemists, letting them swap or tack on new functional groups. Substitution reactions, reductive steps, and nucleophilic attacks work well on this molecule, opening doors for tailored energetic materials or tagging with traceable elements. In some studies, the methoxy group is replaced with bulkier or electron-withdrawing groups to tweak stability and detonation velocities. The benzene ring stays mostly intact unless harsh conditions step into the picture. Real-world reactions aren’t textbook perfect—impurities pop up, byproducts fight for attention, and separating everything involves column after column, but that’s the price of innovation.
Synonyms & Product Names
Trinitroanisole doesn’t parade under many trade names, but chemists love their aliases. You’ll find it listed as 1-methoxy-2,4,6-trinitrobenzene on reagent bottles. Its synonyms include trinitroanisole, TNA, and variants that label the positions of nitro and methoxy groups. Unambiguous naming helps avoid mix-ups, which can turn dangerous in the wrong hands. Markets don’t see much in the way of marketing for this chemical; use is mostly tucked away in technical catalogs and academic publications.
Safety & Operational Standards
Anyone comfortable working with trinitroanisole should carry respect for nitroaromatics in general. Even though it won’t detonate from a stray cat brushing alongside the bench, careless heat, static, or friction could still set off a nasty surprise. Personal protective gear, grounded equipment, and step-by-step protocols matter. Furnace drying, open flames, or sharp tools up the risk profile quickly. I remember a research group that moved too fast during a recrystallization; instead of a yield increase, they wound up conducting a fire drill. Regulatory standards, including those from OSHA and EU REACH, require tight access controls, specialized storage, and record-keeping for every gram. Waste disposal follows strict paths—no shortcuts, no sink-dumping—since environmental release can lead to fines and contamination headaches.
Application Area
Trinitroanisole doesn’t spread into agriculture or food industries. Its stronghold is research, pyrotechnics, military applications, and energetic formulation studies. It helps fill a gap between common explosives and experimental materials—sometimes used as a test case for sensitivity studies, decomposition analyses, or as a template for safer, more efficient new compounds. Some developers rely on it to test detonators, booster compositions, or measure the impact of various additives on energetic output. It isn’t a household name, but for academic labs and select defense projects, it offers a blend of practical power and theoretical flexibility.
Research & Development
The research map for trinitroanisole branches widely. Universities focus on changing electronic properties, looking for ways to push detonation power up or drop toxicity down. Industrial labs run tests on aging, long-term storage, and ways to convert bench synthesis into scalable production. Analytical chemists pick trinitroanisole to study new detection technologies, hoping for breakthroughs in forensic identification or environmental trace analysis. Over the last decade, new derivatives have been proposed and tested, some offering promise as lower-sensitivity alternatives to classic explosives, others as steppingstones to even safer compounds. My time in the lab highlighted just how much patience and diligence this field demands—safe progress comes one experiment at a time.
Toxicity Research
Toxicity studies around trinitroanisole show a mixed picture: not as infamous as dinitrocompounds, but no one calls it benign. Short-term exposure can irritate skin, eyes, and respiratory tracts, and repeated long-term contact could trigger allergic reactions or systemic toxicity. Animal studies in certain research settings point to moderate acute toxicity. Chronic impacts don’t outrank those of classic industrial pollutants, but knowledge gaps remain because few extended human exposures get reported or studied. Researchers push for more robust testing, hoping to define safe limits for use and cleanup, since energetic chemical spills can create health and ecological risks downstream. An honest approach means labeling hazards clearly, using safe handling methods, and supporting medical teams with real data if exposure happens.
Future Prospects
Trinitroanisole won’t vanish from shelves or journals any time soon. It presents ongoing opportunities in both chemistry and applied material science. Tighter regulations and pressure to develop “greener” explosives keep pushing researchers toward compounds like this that offer safer handling profiles without sacrificing necessary performance. Innovations in catalyst design, green chemistry, and computational modeling help map the next generation of derived compounds. If eco-friendly explosives or less hazardous mining agents hit the market over the next decade, chemistry at its core—often sparked by studies involving trinitroanisole—will likely play a foundational role. Anyone looking for breakthroughs in clean-up technology, real-time detection, or low-waste energy release probably keeps an eye on this unassuming compound and its family, as the world looks for new answers on the safety, sustainability, and responsibility front.
What is Trinitroanisole used for?
Understanding Trinitroanisole in the Real World
Trinitroanisole usually sparks curiosity among people who work with specialty chemicals, especially those in the defense sector. It’s a compound designed to meet a specific set of demands, and its story ties into bigger issues about safety, security, and technological advancement.
Role in Explosives
Most people haven’t come across trinitroanisole at the hardware store. Its main job happens behind closed doors in military labs and defense plants. Experts know trinitroanisole as a booster explosive. It’s not just another chemical powder; it’s the kind that bridges the gap between less sensitive ingredients and the ones that pack a punch. Think of it as the spark that safely triggers a controlled explosion rather than an unpredictable bang.
This niche use comes from its qualities. The molecule has three nitro groups, giving it the punch people in the field rely on. It brings stability, behaving better during transport and storage compared to more notorious compounds like TNT. For engineers building safer munitions or trying to prevent accidents, this sort of stability isn’t optional—it's the only way to work responsibly.
Why It Matters Beyond the Laboratory
Growing up near a factory town, I learned early how people in these industries stay up at night thinking about safety. All it takes is one mistake, and things can change forever. So, when chemists select ingredients like trinitroanisole, they’re looking for ways to do their jobs without putting lives on the line. The people manufacturing or transporting explosives—whether for mining, construction, or defense—rely on the predictability and control it brings.
The way trinitroanisole handles heat and shock means fewer stories on the evening news about workplace disasters. It’s not just about numbers in a safety manual. It’s about parents coming home at the end of a shift.
Environment and Security: Two Sides of the Same Coin
Not all explosives are created equal when it comes to environmental impact. The residue left behind after detonations can seep into the soil and water; chemicals hang around long after a job finishes. Industry experts spend years studying how compounds behave after use. Trinitroanisole, despite its strength, creates fewer persistent byproducts than some older formulas.
Security circles also keep an eye on these ingredients. The same properties that make trinitroanisole valuable to professionals make it attractive to people with less noble goals. That’s why regulations around its handling run deep. You need real credentials, oversight, and a clear chain of custody to get anywhere close to it. Any steps skipped mean more risk of it landing in the wrong hands.
Looking for Better Solutions
People working with this compound face tough questions every day. Engineers and chemists ask: Can we find a formula with less environmental fallout? Is there an even safer compound that doesn’t compromise performance? Governments often fund research projects looking to answer these questions, and teams from around the world share what they find.
Having watched neighbors whose jobs depended on chemical plants, I’ve seen that changes in policy and science directly affect real people. Whenever a safer or cleaner alternative moves from the lab to the field, it’s a small victory—one that matters just as much to the families of those working as to the people benefiting from finished projects.
Final Thoughts
Trinitroanisole’s role speaks to bigger stories—about risk, progress, and the relentless search for something better. Whether in explosive engineering or environmental stewardship, it’s proof that even quiet chemical formulas play a part in the safety nets we depend on.
What are the safety precautions for handling Trinitroanisole?
Staying Safe with Trinitroanisole
Dealing with explosives like trinitroanisole demands respect, patience, and experience. Trinitroanisole, often used as a component in explosive formulations, can surprise even seasoned handlers with its sensitivity and reactivity. Keeping both people and property safe depends on following grounded safety practices, not just relying on protocols written in a binder.
Personal Protective Equipment Matters
No matter how confident you feel, gloves and safety goggles are standard for a reason. Trinitroanisole dust stings the eyes and irritates the skin. Nitrile gloves and chemical-resistant face shields carry a lot more value than they seem. Personal experience has shown me that people tend to get complacent, especially if they’ve never seen an accident firsthand. Real protection starts at the very first step, before a single scoop hits the scale.
Controlling the Work Environment
It takes more than just keeping a workspace clean; it calls for a calm, focused environment. Static electricity builds up fast on plastics, synthetic brushes, or even the soles of certain shoes. A grounded workbench stops that spark before it starts. Using antistatic tools isn’t an option—it’s the least you can do. Good airflow matters but you can’t rely on a regular office fan. Only explosion-proof fume hoods and exhaust systems keep vapors and dust from building up. Plenty of labs try to cut corners here, but one spark is one too many.
Storing Trinitroanisole Responsibly
Leaving trinitroanisole in just any cabinet is asking for trouble. Heat, sunlight, shock, and friction all play a role in how dangerous it gets. Put it in a temperature-controlled storage, preferably away from acids and reducing agents. Never keep large quantities on hand; break it into small, labeled containers, and log every gram. History shows that disasters tend to hit those who forget what’s sitting in the back of the storage room.
Mixing and Handling Dos and Don’ts
It’s easy to get distracted when weighing or mixing chemicals, but a distracted mind leads to mistakes. Never use metal tools to handle trinitroanisole, since scraping and impact can trigger a reaction. Instead, use soft plastic or Teflon spatulas. Mixing powders? Always add trinitroanisole gently and slowly—dumping it in once and stirring fast risks uneven distribution and hotspots. Keeping the batch small and manageable has saved people from bigger trouble than most would believe. Documentation matters, too: write everything down, from lot number to the tiniest spill.
Emergency Preparedness
Accidents happen fast. Have a safety shower and eyewash station within arm’s reach. Fire extinguishers and first aid kits aren’t just for show; hang them up in plain sight—not locked away in the boss’s office. Knowing the evacuation route and doing real drills, not just signing a training sheet, can make the difference in an emergency. Rely on knowledge, not luck. More than once, a practiced response has saved entire teams from disaster.
Continuous Education and Training
No singular guideline covers every surprise that comes with trinitroanisole. Years working in chemical labs prove that regular training pays off, especially training grounded in real events and taught by people who have faced emergencies themselves. Collect stories, ask for written case studies, and never treat safety as a one-time box to check. Trinitroanisole isn’t forgiving, and that’s a fact anyone using it needs to keep in mind every day.
What is the chemical structure of Trinitroanisole?
Looking Closer at Trinitroanisole
Trinitroanisole doesn’t show up in everyday conversation, but its structure says a lot about how chemistry shapes technology. This organic molecule, known to chemists as 2,4,6-trinitroanisole, stands out because it mixes a basic aromatic ring with some of the most influential groups in explosives chemistry: nitro groups.
Breaking Down the Structure
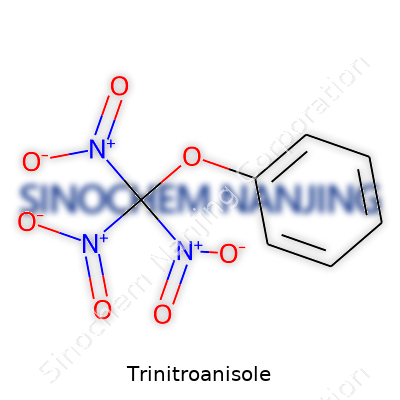
Let’s keep it straightforward. Trinitroanisole starts with a benzene ring, which forms the backbone. A methoxy group (-OCH3) hooks onto the ring, right at the first carbon. Spread out on the ring, you then have three nitro groups (-NO2) attached at the 2, 4, and 6 positions.
The arrangement matters. The methoxy group, being on the first carbon, affects how the molecule reacts with its environment. The nitro groups, bold and energetic, bring a level of instability but also the energy needed for the molecule’s main applications—primarily as an explosive or in explosive mixtures. Structurally, it’s a simple swap of hydrogen atoms for nitro and methoxy, but the impact is far from minor.
Real-World Relevance
My old research team once handled trinitroanisole during a study on alternatives to common explosives. Compared to trinitrotoluene (TNT), which runs the show in most military and construction settings, trinitroanisole has distinct advantages. It’s less sensitive to shock, which cuts the risk of accidental detonation on the job.
The chemical structure does more than look complicated on paper. Those three nitro groups charge the molecule up with chemical energy. The methoxy group tames the beast a bit, making trinitroanisole a more stable compound than some of its cousins. In practice, workers and researchers care less about flashy names and more about safety and reliability. A molecule that’s packed with energy, but slow to blow unless triggered, makes for safer handling and better storage.
Keeping Safety in Mind
Working with nitroaromatic compounds takes focus and respect for the risks. Trinitroanisole shares the same health and environmental concerns as other explosives. Mishandling spills or dust can spark emergencies, and breakdown products from these materials threaten soil and water if folks don’t follow strict cleanup routines. During my time in the lab, good ventilation, up-to-date safety gear, and constant equipment checks made the difference between routine research and a trip to the emergency room.
Finding Better Solutions
Chemists keep experimenting with substitutions on the benzene ring, adjusting sensitivity and performance. Trinitroanisole’s structure gives it a unique spot in this lineup, bridging the gap between safety and power. Investments in training, modern safety protocols, and greener synthesis methods help minimize the risks and keep the science going strong.
While it might sound like just another mouthful of chemical jargon, trinitroanisole’s structure has helped move explosives technology in a safer direction. Combining basic building blocks—nitro groups for energy, a methoxy group for moderation—gets us technology built for the long haul, without losing track of safety and environmental responsibility.
How should Trinitroanisole be stored?
Understanding the Risks and What Real Safety Looks Like
Anyone working with explosives knows the margin for error hovers close to zero. Trinitroanisole, recognized for its explosive potential, brings specific storage demands that can’t be swept under the rug. I remember a chemical plant I toured in my early twenties, where a single misplaced barrel spelled weeks of lost sleep for the staff. That experience stuck with me. Nothing sharpens attention quite like hearing about a close call—especially with materials as sensitive as trinitroanisole.
Explosives like trinitroanisole respond quickly to heat, shock, friction, and sparks. Even taking the slightest chance can spark a disaster. Keeping it dry ranks up there with the most basic safety principles. Moisture prompts deterioration, which leads to instability. A rock-solid roof and leak-proof floors serve as nonnegotiables. I’ve seen rusted ceiling beams threaten entire inventories—the result never matches the risk.
Ventilation belongs on the must-have list. Toxic fumes pose a constant, invisible threat. Years ago, someone shared their experience of a closed, stuffy storage shed where fumes lingered and protective gear barely kept up. Fresh air swaps and a steady temperature go a long way in keeping storage conditions stable. Trinitroanisole handles temperature swings poorly. Anything outside a moderate range increases sensitivity, and freezing temperatures risk crystal growth—a recipe for trouble.
Security Counts Just as Much as Science
I’ve never met a security expert who treats a storage facility for energetic materials like just another warehouse. Fencing, surveillance cameras, and access logs form the backbone of any responsible operation. Doors lock behind heavy bolts and badges track who's inside. Stories float around the industry about missing records and open gates—mistakes that invite theft or worse.
Legally, trinitroanisole sits under explosive regulations. In my experience reading regulatory bulletins, missing paperwork earns attention from inspectors faster than almost anything else. Labels must stay clear and up to date. Every drum, even if half-empty, needs its contents and hazard information displayed in language the team understands. One old-timer I met recounted how a faded label led to wasted hours and dangerous confusion. He said, “Don’t cut corners on what goes on the outside—it protects what’s inside.”
Training and Planning Make a Real Difference
No storage protocol works if people don’t know how to follow it. New employees sometimes underestimate explosives, comparing them to more forgiving compounds. Proper training, repeated routinely, helps the rules become instinct rather than an afterthought. In my early days, the best lessons came from seasoned workers walking me through “what-if” scenarios. If that drum leaks, or if something knocks into a stack, every step after matters.
Emergency procedures need to live in the open, not stuffed in a dusty manual. Alarms, spill kits, fire suppression systems, and regular drills push teams to react properly under pressure. I’ve seen places where every worker carries a laminated card outlining next steps in an emergency—simple tools often make the difference between a scare and a tragedy.
Looking Forward: Improvements and Accountability
Technology keeps moving. Digital temperature and humidity monitors send real-time alerts before tiny swings grow into larger risks. More operators track every container’s journey, cutting down on mix-ups. Still, following best practices stays a matter of responsibility, not just rules. Expert oversight, peer reviews, and regular inspections anchor safe storage in a culture of diligence.
Trinitroanisole storage boils down to respect—for the material and for the safety of the people nearby. Every real-world accident winds up as a reminder: shortcuts belong nowhere near an explosive. Proper storage rewards careful habits, and nobody ever regrets keeping respect front and center.
Is Trinitroanisole explosive or hazardous?
Looking Beyond the Formula
Trinitroanisole pops up in some chemistry circles as a topic for explosive potential. The name sounds technical, but it shows up in discussions about explosives, lab safety, and industrial manufacturing. If you’ve ever worked with chemicals or handled science curriculum, you know names don’t tell the full story. Years of reading Material Safety Data Sheets (MSDS) have taught me: the label on the bottle never covers all the risks.
Hazard in the Lab and Beyond
Trinitroanisole has roots in organic chemistry. Its chemical structure includes three nitro groups attached to an anisole ring. From hands-on lab days, chemicals with multiple nitro groups have an established record of instability and sensitivity. Nitroglycerin and TNT have similar features, which instantly raises a red flag.
The real risk of trinitroanisole lies in its energetic properties. It’s used mostly for research, synthesis, and sometimes considered as a component of explosives. It doesn’t earn a spot in the “safe” chemicals drawer. The presence of several nitro groups means it stores a lot of energy. History shows that storage and transport of compounds like this, especially under heat, shock, or friction, create opportunities for energetic release.
The science isn’t all theoretical. NIJ (National Institute of Justice) records point to uses in energetics—a polite way to describe explosives and propellants. The sensitivity can vary based on how pure the sample is and the environment in which it’s handled. Crystallized samples can shock more easily than liquids or mixtures.
Practical Hazards in the Real World
During time spent in teaching labs and talking with safety officers, I’ve seen people gravitate toward downplaying these risks, especially when a compound isn’t as famous as TNT. But trinitroanisole brings its own set of headaches. Inhalation, skin contact, and fire risk all deserve attention. Even small spills require attention. Some accidents stem from well-meaning workers who tried to measure just a pinch more or clean up in a hurry.
The Environmental Protection Agency includes related nitroaromatic compounds on lists of hazardous pollutants. That’s not surprising. Chemically, they stick around in soil and water for a long time, affecting ecosystems. Improper storage causes leaks, and those leaks travel downstream, literally or figuratively.
Solutions Rely on Knowledge and Discipline
People who work with trinitroanisole, or anything like it, need solid training and up-to-date information. Relying strictly on labels can backfire—MSDS details and peer-reviewed journals give the whole story. Double-checking procedures saves lives and resources. I remember a story from a university lab that kept explosives logs locked and reviewed them twice a week, because history had taught them what complacency costs. That level of care sounds exhausting, but experience shows it pays off.
There’s no single barrier that eliminates risk with hazardous compounds. Using blast shields, chemical fume hoods, and non-sparking tools makes a big difference. It’s not just the big mistakes that cause problems—the small, everyday lapses pile up. Regular drills, reminders, and audits force us to stop and think before a routine job turns dangerous.
Schools and industry need ongoing communication between safety officers, chemists, and emergency responders. The right kind of language—clear, direct, and honest—helps everybody from the new hire to the senior researcher. No one benefits from downplaying the explosive risk.
Trinitroanisole has earned a place on the hazardous list for solid reasons. Whether working with grams or barrels, real safety starts with understanding exactly what’s in front of you, and respecting the risks with action instead of assumption.
| Names | |
| Preferred IUPAC name | 1-methoxy-2,4,6-trinitrobenzene |
| Other names |
1-Methoxy-2,4,6-trinitrobenzene TNA Trinitroanisol |
| Pronunciation | /traɪˌnaɪtroʊˈænɪsoʊl/ |
| Identifiers | |
| CAS Number | 606-35-9 |
| Beilstein Reference | 607251 |
| ChEBI | CHEBI:132965 |
| ChEMBL | CHEMBL197210 |
| ChemSpider | 57013 |
| DrugBank | DB06411 |
| ECHA InfoCard | 100.007.490 |
| EC Number | 208-749-0 |
| Gmelin Reference | 6260 |
| KEGG | C19160 |
| MeSH | D019373 |
| PubChem CID | 120822 |
| RTECS number | SY8575000 |
| UNII | N5R4J36B4J |
| UN number | UN1336 |
| CompTox Dashboard (EPA) | DTXSID6010319 |
| Properties | |
| Chemical formula | C7H5N3O7 |
| Molar mass | 227.14 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.69 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.93 |
| Vapor pressure | 0.00022 mmHg (25 °C) |
| Acidity (pKa) | 12.86 |
| Basicity (pKb) | 11.92 |
| Magnetic susceptibility (χ) | -51.0·10⁻⁶ cm³/mol |
| Dipole moment | 4.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 248.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –119.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1725 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Explosive; harmful if swallowed, inhaled, or absorbed through skin; causes eye, skin, and respiratory tract irritation. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS02,GHS01 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P240, P250, P280, P370+P378, P401, P501 |
| NFPA 704 (fire diamond) | 1-4-2 |
| Flash point | 150 °C |
| Autoignition temperature | 280 °C |
| Lethal dose or concentration | Lethal dose or concentration (LD50, oral, rat): **655 mg/kg** |
| LD50 (median dose) | LD50 (median dose): 250 mg/kg (oral, rat) |
| NIOSH | SN 2275000 |
| PEL (Permissible) | PEL for Trinitroanisole is not specifically established by OSHA. |
| REL (Recommended) | 0.01 ppm |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
2,4,6-Trinitrotoluene Trinitrobenzoic acid Trinitrophenol Trinitroresorcinol |

