Trinitro-m-Cresol: Looking Beyond Its Explosive Past
Tracing the Roots of Trinitro-m-Cresol
The story of trinitro-m-cresol starts in the late 19th century, as advances in organic chemistry opened possibilities we hadn’t dreamed about before. Chemists first put together aromatic nitration paths that eventually led them to substances like TNT, picric acid, and their lesser-known cousins such as trinitro-m-cresol. Often, this compound doesn’t get the limelight, maybe because the spotlight has stayed fixed on more famous explosives and dyes. But behind laboratory doors, researchers knew there was more at play than what meets the eye. Back in those days, men in wool suits and thick glasses dared to experiment with nitrations under open flames, giving rise to discoveries that defined not just warfare, but also industrial chemistry for generations. Trinitro-m-cresol emerged as a product of those wild lab benches, fitting into the broader theme of the turn-of-the-century chemical revolution.
What’s Trinitro-m-Cresol All About?
Trinitro-m-cresol has earned its reputation by being demanding yet rewarding. As a pale yellow crystalline solid, it often comes in the form of a powder with a faint sweet odor, which belies its dangerous nature. Its chemical formula is C7H5N3O7, and the structure packs three nitro groups onto a methyl-substituted phenol, making it both reactive and stubborn at the same time. It isn’t exactly a household chemical, and anyone working with it knows it doesn’t forgive carelessness. It doesn’t dissolve easily in water, but gives way in hot organic solvents, suggesting it likes to keep to itself unless heated or prompted by a strong enough agent. This trait has both insulated its applications and contributed to the handling challenges that surround it.
Physical and Chemical Personality
What stands out right away is its volatility and sensitivity. Trinitro-m-cresol exhibits a melting point that’s stubbornly high, showing the stability of its crystal lattice—until you heat it past a threshold, which flips the script and tests any chemist’s safety routines. The substance reacts with strong bases, giving a deep, sometimes crimson color, owing to how nitro groups rearrange under alkaline conditions. Its density sits on the higher side, as expected with multiple nitro groups packed onto a small organic ring. The chemical doesn’t fancy long exposure to light or open air; over time, it may darken as it decomposes slowly, especially if humidity joins the party. The safety margin is thinner than with many related chemicals, so even small mistakes in temperature control or containment can lead to bad outcomes.
The Nuts and Bolts: Preparation in Practice
I’ve watched experienced technicians sweat it out as they prepared trinitro-m-cresol via classic nitration of m-cresol with nitric acid. Sulfuric acid enters the mix as both a dehydrating agent and a catalyst, channeling the strength of strong acids to coax three bulky nitro groups into place. The process generates heat rapidly, and there’s little room for shortcuts—adding reactants too fast or letting the temperature climb unchecked can produce violent runaway reactions. The result needs fast filtration and rapid washing to keep impurities at bay. Only folks trained in the art, with nerves of steel and an eye for subtle color changes, can repeatedly pull off good yields. The post-reaction mixture smells metallic and sharp, a reminder that even after the work is done, the risk lingers. Pride comes in safely pulling a pale yellow product from the sticky mess, a substance touched by both brilliance and risk.
Names That Mirror Its Nature
Those diving into historical archives or dusty safety sheets run across a handful of names—trinitro-m-cresol is also known as 2,4,6-trinitro-3-methylphenol, s-trinitro-m-cresol, and in older writings, sometimes as "styphnic acid methyl ether." Names matter, since confusion in chemical orders or handling sheets costs lives, and clear standards help avoid mix-ups, especially in high-risk fields. While no one keeps a running tally of every local nickname it has picked up, sticking to IUPAC conventions and established synonyms remains the best way to keep lab conversations sharp and safe.
Standards of Care and Risk
Few would argue that safety regulations for trinitro-m-cresol feel excessive, and plenty of reasons back that up. Direct skin contact leads to chemical burns, and inhaling even a smidge can cause headaches and nausea. The dust tends to linger in the air, and it doesn’t take much to push airborne particles close to their flash point. This substance has been flagged in regulatory guidelines from multiple countries, each underlining the importance of ventilation, double-layer protective clothing, and regular medical checkups for anyone spending time around it. Dust control systems aren’t a nice-to-have; they're life insurance. Labs and factories that work with it run tightly scripted drills, since a hiccup can lead not just to injuries but to explosions serious enough to take out half a building.
Life in the World: Trinitro-m-Cresol at Work
For most folks, explosive chemicals bring to mind war or mining, but trinitro-m-cresol has carved out a more specialized life. It has seen scattered use in the synthesis of organic intermediates, as well as a testing standard for nitration strength in research labs. Some older pyrotechnic formulations gave it a starring role, and a few dye and pigment makers leaned on its special chemical quirks. These brown-to-yellow hues found their way into everything from colorants for textiles to temporary inks. Today, the move away from highly toxic compounds has meant substitutes often stand in, but the legacy use persists and influences how we handle and think about risky materials in everyday products.
Digging Deeper: Reactions and Modifications
Tinkering with trinitro-m-cresol isn’t for beginners. At its core, it's reactive—nucleophiles target the ring, and under basic conditions, the molecule rearranges or sheds a nitro group. Certain metal ions form complexes, shifting color and opening pathways for analytical work. Reductive treatments in controlled environments lead to amino derivatives, compounds with their own set of safety and regulatory questions. Over time, chemists have explored minor modifications to tweak sensitivity or test new dye structures, always walking a fine line between scientific curiosity and respect for volatility.
The Ongoing Search: R&D and Modern Questions
Even now, people keep looking for new ways to tame trinitro-m-cresol, asking whether it still holds promise for clean energy research, safe detection reagents, or unique synthetic routes to advanced pharmaceuticals. Some teams study its spectral and electrochemical properties, hoping to uncover subtle uses in sensor technology. Funding and ethical questions limit large-scale trials. My own time in collaborative projects taught me that bringing explosive-derived compounds into green chemistry protocols tests both imaginations and nerves. Smart automation, high-throughput screening, and AI-driven predictive models play important roles, especially in flagging interactions that aren’t obvious to the naked eye.
The High Human Cost: Toxicity and Limits
As the list of chemical tragedies grew over the decades, trinitro-m-cresol caught a second look from a safety and health perspective. Animal studies and occupational reports both linked it to acute toxicity, neurogenic symptoms, and possible organ damage with chronic exposure. The compound finds a place on hazardous materials lists, forcing researchers and industrial chemists to weigh benefits against steep costs in human and environmental safety. There isn’t any realistic scenario for widespread use without large-scale expense in engineering controls and emergency medical protocols.
Looking Down the Road
Some old-timers say that trinitro-m-cresol is a relic, a holdover from a period when chemical risk took a back seat to industrial ambition. I look at it as a cautionary tale that still has chapters left—especially as society reexamines how we balance risk, necessity, and progress. As sustainable chemistry gains ground, research continues into how structure and process design could one day strip away toxicity while keeping useful chemical properties. Maybe the next breakthrough in explosive chemistry, pigment manufacture, or analytical science won’t look like the old nitro-aromatics, but the lessons hard-won from working with trinitro-m-cresol mean future researchers won’t have to learn from scratch—or through tragedy. By keeping the history and technical challenges of compounds like this in front of us, we make smarter choices about which paths science should follow, and which are better left behind.
What is Trinitro-M-Cresol used for?
A Closer Look at Trinitro-M-Cresol’s Roles
Trinitro-m-cresol isn’t exactly a household name, but its uses reach further than most people realize. This compound, sometimes called 2,4,6-trinitro-m-cresol or TNMC, turns up often in industries that depend on strong, stable chemicals. At its core, trinitro-m-cresol acts as an energetic material—meaning it can store a lot of energy that releases quickly. This characteristic makes it valuable in explosive formulations, especially for military or specialized industrial purposes.
Many manufacturers use trinitro-m-cresol as a key part of detonators or percussion caps. Its chemical stability and high sensitivity help produce reliable ignition when you want things to work on cue. Growing up near a mining town, I remember hearing stories of old timers handling blasting caps with the kind of respect you give something that can end your day if you get careless. Trinitro-m-cresol often finds its way into those blasting caps, powering the first important step in shaking loose the rocks deep underground.
Health and Safety Counts For More Than Headlines
Most chemicals with “trinitro” in their name deserve a careful eye. Trinitro-m-cresol carries risks—just touching or breathing in the particles over time can mess with your health. The Environmental Protection Agency points out that some nitro compounds may cause skin rashes, trouble breathing, or much worse if handled without solid protection. Workers exposed to this chemical in manufacturing must suit up with masks and gloves, wash down regularly, and follow rules stricter than those for most cleaning chemicals.
Managing these risks takes more than rules taped to a break room wall. From what I’ve seen at local plants, strong leadership and worker buy-in protect everyone. Regular safety briefings, clear procedures, and fast response to spills or leaks build trust and save lives. Not every company treats safety as a real investment, but those that do face fewer injuries and avoid the kind of reputational damage that stock prices can’t hide.
Beyond Explosives: Narrow Uses with Tighter Limits
Years ago, labs explored trinitro-m-cresol for dye and pigment production. While its colorant properties can work, the toxicity pushed most manufacturers toward safer alternatives. Today, only specialized research or military labs keep it stocked for those old uses, guarding it much more closely than even a decade back. Environmental oversight keeps growing tighter. The risk of soil and water contamination from improper disposal means companies now treat every ounce as hazardous waste, shipping it to licensed facilities that know how to break down and neutralize these stubborn molecules.
What’s Next: Taming Hazards Through Science and Policy
One thing stands out after years of watching chemical safety evolve—all the best safety gear in the world won’t make a difference unless people actually use it and keep asking if safer alternatives exist. Governments and industry watchdogs keep pushing for greener options and more transparent tracking, especially after high-profile accidents. New materials and process controls have chipped away at the need for compounds like trinitro-m-cresol, but until a replacement checks every box for reliability and cost, it stays in the toolbox.
Accidents remind us that progress moves in steps, not leaps. Every tightened policy, upgraded training manual, or swapped chemical stands on the lessons learned from yesterday’s close calls. Building a safer future, whether in a mine, a factory, or a lab, depends on respecting what these compounds can do—and making smart choices to protect people and planet every step of the way.
Is Trinitro-M-Cresol hazardous to health?
Understanding the Substance
Trinitro-m-cresol, sometimes called 2,4,6-trinitro-m-cresol or cresylic acid trinitrate, has been around for many decades. Workers in explosive manufacturing, dye industries, and some research labs occasionally run across it. In most cases, you won’t find it sitting on a hardware store shelf. Its primary use lands in chemical laboratories and very specific industrial settings.
Direct Health Risks
Direct exposure causes trouble. Those who spend hours near open containers without the right protection might complain of skin burns, headaches, and stomach discomfort. Reports show this chemical irritates the skin and eyes. Breathing in a bit of the dust or fumes can make your nose, throat, and lungs pretty sore.
If it gets into the body, it doesn’t just stop at mild symptoms. Nausea, dizziness, even liver and kidney harm can show up after repeated exposure. The U.S. National Library of Medicine and the European Chemicals Agency both point to organ damage if folks get too much over a long period.
Long-Term and Environmental Worries
Persisting around chemical plants or places that dealt with explosives means this substance can leak into soil or water. That’s bad news for fish, water bugs, and the whole local ecology, because trinitro-m-cresol doesn't break down easily. Folks living near old manufacturing areas sometimes worry, especially if they grow a garden or draw drinking water from a well nearby.
Inhaling dust over years or having skin contact for months at a time sometimes brings bigger health problems for workers. Lab techs and plant operators have suffered chronic liver issues. In rats, long-term exposure links to organ swelling and reduced red blood cells.
Prevention Above Treatment
I’ve talked with folks who worked in chemical plants before the use of quality face masks and proper gloves became standard. Many old-timers didn’t retire with perfect lungs or unblemished skin. Having easy access to personal protective equipment (PPE), training, and regular equipment checks makes a big difference. No one should handle trinitro-m-cresol without gloves, eye protection, and a real respirator.
Modern storage methods block most accidental leaks. Double-walled containers, clear labels, and locked doors all help. Good ventilation acts as a solid backup. In areas where spills happened in the past, both soil and water often get checked for residues. Cleaning these up takes time and money, but prevents lingering health dangers for families and wildlife.
Following the Rules
Major agencies like OSHA and NIOSH list exposure limits and offer guidelines for emergency cleanup and occupational safety. If you’re in a workplace where trinitro-m-cresol might turn up, demanding those rules get followed protects everyone. Facility managers need strict training programs, waste-management plans, and quick action if a leak gets spotted.
Looking Ahead
Some companies look to swap out older, more toxic chemicals for safer substitutes. That’s already cut down trinitro-m-cresol use outside explosives and specialty dyes. Pushing for safer workspaces and aggressive environmental monitoring, especially near older factories, creates fewer problems and healthier communities.
If nothing else, trinitro-m-cresol reminds us that behind every industrial success, there’s a responsibility to keep people and the earth safe. Wearing PPE, updating safety training, and checking soil and water are steps we shouldn’t skip.
What are the storage requirements for Trinitro-M-Cresol?
Storing Hazardous Chemicals: Lessons from the Lab
Working in various labs has shown me how crucial it is to treat explosive chemicals with real respect, not just check off a list. Trinitro-M-Cresol, or 2,4,6-trinitro-m-cresol, brings serious risks. Accidents tend to start with small oversights, like skipping a safety step. Even workers who’ve become used to handling dangerous substances can let their guard down over time. It never pays off, especially with a material known for volatility.
Why Storage Conditions Really Matter
Trinitro-M-Cresol isn't like your average solvent or cleaning agent. This compound belongs to a group of organic nitro compounds, many of which can explode if mistreated. Its chemical structure means it reacts strongly to heat, friction, and even shock. Temperatures above room level or a bad mix of humidity provide the perfect breeding ground for disaster. In the 1990s, an explosion in a niche pharma lab traced directly back to nothing more than a hot storage closet—too warm, too careless.
Key Facts: Temperature and Location
Cool and dry conditions come up again and again in risk reports. A dedicated chemical storeroom, fitted with temperature and humidity control, offers a real line of defense. Shelving must separate Trinitro-M-Cresol well away from acids, alkalis, and strong reducing agents. Corrosive fumes can seep through plastic liners, so even the shelf material matters. Store the chemical in tightly closed glass containers, preferably amber glass to block stray light.
Unlike some reagents, Trinitro-M-Cresol cannot sit safely near open windows or heat sources. The ideal location? A secure, ventilated, flameproof cabinet, deep inside a limited-access area. Custom safety lockers with explosion relief panels have saved lives during chemical mishaps. Above all, labeling should never be skipped. Even a ten-minute mix-up during an inventory check can place workers in harm’s way.
Daily Practices for Chemical Safety
Every lab has its own storage rhythm, but vigilance is universal. Double-checking seals costs less time than cleaning up a spill. My own training stressed that inventory logs should always show the date when each container was last inspected. Over time, trinitro compounds decompose into dust and fumes—both real hazards. Disposal protocols should be strict, but that care begins with smart storage.
Regulatory Oversight: Not Just Red Tape
Failing a safety audit can close a research program instantly. Agencies like OSHA and the EPA impose strict guidelines on handling reactive chemicals. These bodies don’t aim to stifle progress; they respond to decades of lessons written in accident reports. Financial penalties set an example, but the real cost comes from injuries and reputation loss.
Better Storage Through Better Design
Some researchers test out updated monitoring technology—think smart lockers and digital temp tracking—to keep tabs on their sensitive stocks. Others rely on peer-check systems or assign veteran staff as “chemical stewards.” Formal training, annual audits, and updating data sheets might feel bureaucratic, but these routines keep workers and communities out of headlines.
Handling Trinitro-M-Cresol’s storage with the seriousness it demands means investing time, shared vigilance, and clear protocols. No fluke should threaten lives or research just because a jar stood too close to a radiator or a label peeled off.
How should Trinitro-M-Cresol be handled safely?
What Makes Trinitro-M-Cresol a Big Deal?
Trinitro-M-Cresol sounds like something reserved for a chemistry lab out of reach for most folks, but this compound has a history stretching into industry and research. With its explosive potential and toxic profile, it doesn’t belong anywhere near a casual workspace or an underprepared person. Even a short story from someone in a university lab drives this home: a friend handled a similar nitro compound during a late-night experiment and needed medical help due to spilled crystals on their skin. The memory of chemicals burning through gloves sticks with you.
Health Hazards You Can’t Ignore
Direct skin contact with Trinitro-M-Cresol can cause serious burns, persistent irritation, not to mention allergic responses for some. Inhalation is even riskier. Breathing in dust from this material can lead to headaches, dizziness, and more severe symptoms with enough exposure. Even a splash in the eyes could mean lasting damage. The fire risk is real, too; this compound reacts easily, sometimes violently, with heat or friction.
It’s worth pointing to documented cases from chemical plants—workers have landed in emergency rooms from poor understanding of what this chemical can do. The lesson? Bad things happen fast, and you don’t get a second chance to run for help.
Practical Protection Steps
Anyone handling Trinitro-M-Cresol should gear up like it’s a necessity, not a choice. Proper nitrile gloves, face shield, goggles, sturdy lab coats: this is the minimum. Relying on a regular fabric mask, or skipping proper PPE, amounts to gambling with your health. From experience, anyone working with chemicals makes mistakes eventually, and the gear acts as the last line of defense.
Local exhaust ventilation, like well-maintained fume hoods, keeps dust and vapors from collecting in workspaces. These don’t just meet government lab guidelines—they take headaches and scary incidents out of the equation. Fire suppression, clear spill kits, and emergency showers should be accessible, not just part of a checklist for an annual audit.
Storage and Waste: Don’t Cut Corners
Safe storage means keeping Trinitro-M-Cresol in sealed, clearly labeled containers far from sources of heat or sparks. No glass bottles near the edge of benches or stored above head height. Dedicated cabinets marked for explosives or toxic materials add another layer of safety. In shared spaces, every team member should know exactly where this material sits and what it looks like—confusing it for another powder could end very badly.
Waste disposal gets special attention. Trying to wash minor spills down the drain tempts fate. Proper waste containers, clear labeling, and regular hazardous waste pickups keep accidents at bay and avoid regulatory fines. The EPA and OSHA already have strict rules; following them isn’t just about box-ticking but about keeping coworkers and clean-up crews from blundering into a dangerous surprise.
Training and Culture: The Human Side
No equipment or rulebook replaces a sensible, experienced team. Training sessions with live demonstrations beat dry lectures every time. Testing emergency plans, walking through spill response together, builds trust and readiness. Open conversations about close calls ensure lessons get shared, not buried.
Working with Trinitro-M-Cresol isn’t something to take lightly. Up-to-date knowledge and a culture that encourages respect for these materials save lives. Complacency puts everyone in the line of fire.
What is the chemical formula of Trinitro-M-Cresol?
Unpacking Trinitro-m-Cresol
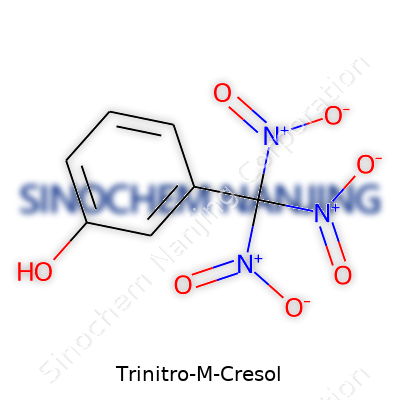
Trinitro-m-cresol goes by another name in chemistry circles: 2-methyl-3,5,6-trinitrophenol. It often comes up in conversations about industrial chemistry, explosives, and dye manufacturing. Its chemical formula, C7H5N3O7, sums up the structure—a cresol (methylphenol) base with three nitro groups attached, giving it a boost in reactivity and some real-world risks that can’t be ignored.
Safety and Practical Concerns
Ask any chemist who’s worked with nitro phenols and you’ll hear stories about safety protocols. Compounds like trinitro-m-cresol can be powerful oxidizers, which is just a technical way of saying they don’t play nice with certain materials. Even trace amounts of moisture or metals can cause unpredictable reactions. A lot of labs set up special handling stations, just to keep this chemical from coming into contact with things it shouldn't. Attempts to save time or ignore small spills rarely end well.
Real lessons from history support this caution. During the peak of dye and munitions manufacturing a few decades ago, accidents involving trinitro and other nitro derivatives left a mark. In some cases, workers developed skin sensitivity or respiratory issues, leading employers to toughen up safety rules. As tempting as it sounds to cut corners in pursuit of efficiency, this molecule shows how chemistry brings very real consequences.
Environmental Impact
Disposal challenges remind us chemistry doesn’t vanish when a factory closes its doors. Trinitro-m-cresol doesn’t break down in nature like food scraps or garden clippings. Once out in the environment, it tends to stick around, especially in soil and water. The nitro groups interact with microbes and plants, sometimes changing ecosystems in ways people didn’t expect. Even years after factories shut off the lights, traces linger, showing up in soil studies and water quality tests.
Regulatory groups set strict limits on how much of this chemical can end up in waste streams. The rules might seem excessive to newcomers, but old case files and environmental surveys explain the reasoning. Runoff from poorly managed disposal has led to long-term contamination in several regions worldwide.
Balancing Use and Responsibility
Trinitro-m-cresol’s value in manufacturing and research is hard to dismiss. Dyes, pigments, and even some explosives rely on this building block. Every step, from synthesis to transport, asks people to respect its power. Running a lab where trinitro-m-cresol shows up means everyone from interns to senior chemists gets trained on spill management and emergency response.
Manufacturers are now exploring alternatives, nudged along by both regulations and community concern. Green chemistry initiatives push teams to develop new dyes and energetic compounds with fewer environmental downsides. This shift won’t happen overnight, but it has already started reshaping course syllabi, safety manuals, and factory layouts. The formula C7H5N3O7 stands as a reminder—chemistry gives us tools, but we decide how responsibly we use them.
| Names | |
| Preferred IUPAC name | 2-methyl-3,5,6-trinitrophenol |
| Other names |
2-methyl-4,6-dinitrophenol 4,6-Dinitro-o-cresol DNOC Dinitro-ortho-cresol |
| Pronunciation | /traɪˈnaɪtroʊ ɛm ˈkriːsɒl/ |
| Identifiers | |
| CAS Number | # 603-11-2 |
| Beilstein Reference | 1708731 |
| ChEBI | CHEBI:75680 |
| ChEMBL | CHEMBL1230548 |
| ChemSpider | 6196 |
| DrugBank | DB01808 |
| ECHA InfoCard | 100.017.866 |
| EC Number | 208-751-5 |
| Gmelin Reference | 7461 |
| KEGG | C02930 |
| MeSH | D015556 |
| PubChem CID | 6905 |
| RTECS number | GO9625000 |
| UNII | N5K4372K2B |
| UN number | 1349 |
| CompTox Dashboard (EPA) | DTXSID8020452 |
| Properties | |
| Chemical formula | C7H5N3O7 |
| Molar mass | 227.13 g/mol |
| Appearance | Yellow crystals. |
| Odor | Odorless |
| Density | 1.55 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.7 |
| Vapor pressure | 2.5 x 10^-6 mmHg (25°C) |
| Acidity (pKa) | 7.05 |
| Basicity (pKb) | 12.96 |
| Magnetic susceptibility (χ) | -0.62e-6 cm³/mol |
| Refractive index (nD) | 1.693 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.79 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 175.8 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -324.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1173.4 kJ/mol |
| Pharmacology | |
| ATC code | D08AX04 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause respiratory irritation; may cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS03 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H373, H410 |
| Precautionary statements | P260, P264, P270, P273, P280, P301+P312, P302+P352, P305+P351+P338, P330, P312, P337+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 1, Instability: 3, Special: --- |
| Flash point | 79 °C |
| Autoignition temperature | 315°C |
| Lethal dose or concentration | LD50 oral rat 400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 50 mg/kg (oral, rat) |
| NIOSH | SN1188000 |
| PEL (Permissible) | PEL: 0.1 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 5 mg/m³ |
| Related compounds | |
| Related compounds |
p-Nitrophenol o-Nitrophenol 2,4-Dinitrophenol 2,6-Dinitrophenol |

