Trimethyltin Acetate: A Ground-Level Look at a Notorious Organotin Compound
Historical Development
Trimethyltin compounds have been floating around chemical labs since organotin chemistry carved its path in the early twentieth century. Researchers poked into the field searching for new catalysts and stumbled on dozens of oddities. Tin, once mostly a solder and alloy player, slowly entered more sophisticated territory. Trimethyltin acetate, less famous than its cousins tri-n-butyltin or dibutyltin dichloride, drew attention not for how it helps, but for the trouble it causes. Many early experiments with this compound focused on the damaging effects it could have on the nervous systems of lab animals. These studies cracked open the tin-organic field, shining a light where chemical curiosities met hard realities about toxicity. Today, historical records and dusty papers spell out all sorts of mishaps and discoveries. Researchers learned to respect it, treat it as both a useful chemical tool and a stark warning—one that shaped modern handling codes for lots of organotin substances.
Product Overview
Chemists know trimethyltin acetate by its white, crystalline appearance and sharp, almost biting odor. Unlike bulk industrial chemicals handled on freight scales, this compound usually pops up in flasks and vials, handled cautiously with good ventilation. Sometimes folks use it to make new organotin derivatives, sometimes as a quirky reagent in synthesis work. It never took on the mass-market life that other organotins did in PVC stabilizers or biocides, but its pop-up appearances around neurological research, and as a precursor in organometallic chemistry, offer enough reasons to keep it in the supplier catalogues.
Physical & Chemical Properties
Trimethyltin acetate doesn’t hide its nature. At room temperature, it takes on a solid form, usually white to off-white crystals or a powder. This compound melts at moderate temperatures, generally above 160°C, and puts off a pungent smell that can clear a room—nobody forgets that first whiff. Its molecular structure features the trimethyltin moiety—three methyl groups bonded to a tin center—paired with an acetate ion. It dissolves in organic solvents like chloroform and ether, steers clear of water, and tends to break down in contact with acids or bases. Its volatility is moderate, so labs keep the lids tight or risk contamination that travels not just through air but on skin and clothing, tracking trouble from a spill site to breakroom tables.
Technical Specifications & Labeling
Every shipment of trimethyltin acetate runs through a gauntlet of regulatory checks. Suppliers stamp drums and jars with symbols: acute toxicity, environmental hazard, skull and crossbones for good measure. Safety Data Sheets hammer home the main message—don’t touch, breathe, or allow into drains. Country-by-country, the labels keep growing, not shrinking. Chemical purity values play a big role for research buyers. Specifications often circle 98% to 99% minimum purity, with tests for impurities like tetramethyltin or other organometallic crosstalk. Packing leans heavily toward glass or high-integrity plastics. Temperature control helps, but it’s the human element—careful handling, proper gloves, fume hoods—that draws the line between routine and disaster.
Preparation Method
Synthetic routes to trimethyltin acetate have seen little wild innovation lately. The go-to approach starts with trimethyltin chloride meeting sodium acetate or silver acetate in a solvent like chloroform or ether. Chemists mix, stir, remove the salt by-products, and evaporate off the solvent to reveal product crystals. It’s one of those reactions that racks up as many stories about what not to do as tips about yields and purity. Impurities like moisture or unreacted tin salts can wreck the end product, or worse, set off reactions the next time someone tries using the material as a precursor. Correct storage, often in an inert atmosphere, dodges many headaches—for those who learn from their mentors’ mistakes.
Chemical Reactions & Modifications
Trimethyltin acetate seldom sits alone—chemists apply it to make all sorts of tin-based compounds and organometallic hybrids. Toss it in with halide donors, and it might swap out its acetate for something more reactive. Pull it into cross-coupling chemistry with transition metal catalysts, and it can help stitch together carbon frameworks that start new drug candidates or functional materials. Its knack for transferring methyl groups in organic synthesis gave it some fans, though nobody ignores the residue risks or the high clean-up standards demanded when tin-based reagents play a part. Careless reactions or improper conditions risk forming toxic by-products, highlighting why good technique and clear documentation stay at the center of any protocol.
Synonyms & Product Names
Trimethyltin acetate travels under a set of pseudonyms—trimethylstannanium acetate, stannane, trimethyl-, acetate, or simply TMT acetate. Contractors and catalogues swap these names around, but every chemist who works with organotins knows the shorthand, trims the syllables in the lab, and automatically pairs the name with the protocols they grew up learning. Some suppliers drop the metal’s oxidation state or include the CAS number as a badge of identity, making sure shipments match up with lab inventories that rely on strict naming conventions.
Safety & Operational Standards
Safety never becomes a box-ticking exercise around trimethyltin acetate. Just a few milligrams mishandled can throw a whole building’s air out of safe range, and researchers swap plenty of war stories about accidental exposures, lightheadedness, or months spent trying to track a persistent odor. Gloves alone fail to guarantee safe handling—a full suit, tight-fitting goggles, and a double-checked ventilation system rise up as the minimum set of barriers. Waste must travel straight to special hazardous disposal, kept away from ordinary sinks or trash cans. Training often takes the form of mentorship, with senior staff walking new hands through the critical steps until caution becomes automatic. It’s not just about ticking rules, but about respect for a compound whose bite extends far beyond the molecule itself.
Application Area
Trimethyltin acetate doesn’t show up much in wide-reaching industries. Instead, its place belongs to corners of research and chemical synthesis where precision matters. Neuroscience claims it as a tool for modeling demyelination and neurotoxicity, allowing researchers to dig into mechanisms of brain damage and search for protective strategies in conditions like multiple sclerosis. Some research groups walk the edge by using the compound to generate other tin-based building blocks for specialty materials, or as an intermediate in organometallic transformations. Realistically, its commercial use stays limited because of its fierce toxicity and challenging waste streams. Regulation often keeps it at arm’s length outside of carefully controlled academic or government labs.
Research & Development
Studies keep turning trimethyltin acetate into a lens that brings unwanted effects into focus. Research chases down exactly how the compound damages neurons, seeking out the fine line between doses that open new knowledge and those that cause irreversible damage in animal models. Researchers publish studies exploring treatments that could block, reverse, or limit tin-induced neuropathologies. Animal studies, cell cultures, and detailed chemical analysis all pull together to build a picture—sometimes it’s ugly, but it’s real. Every year, these investigations add to a slow-growing body of regulations around permissible exposures, storage limits, and cleanup methods. Industry and academia work in tandem here, often driven by both curiosity and a sense of caution that didn’t exist, say, four decades back.
Toxicity Research
No discussion of trimethyltin acetate ducks its reputation. The compound attacks the nervous system with ruthless efficiency: even minor doses can cause tremors, movement disorders, and brain swelling in rodents. For humans, symptoms can include vomiting, headaches, delirium, and, in rare cases, lasting neurological damage. Researchers have tracked the underlying mechanics to mitochondrial pathways and calcium metabolism, linking the compound to oxidative stress and cell death. Studies document environmental persistence, too—tin doesn’t break down easily and can ride in water, soil, or air, posing risks to wildlife. Regulations have ramped up worldwide not just for this compound, but for the whole organotin family. Warnings echo not just in the lab, but in environmental protection circles, where the long-term impacts remain a concern.
Future Prospects
The road ahead for trimethyltin acetate runs straight into questions of risk and necessity. On one hand, a growing focus on safer alternatives nudges chemists away from hazardous tin compounds in chemistry and neuroscience research. Many funding agencies press for greener reagents, and industry shifts toward substances with less baggage. On the other hand, specific research questions—especially related to neurotoxicity and demyelination—keep some demand alive, connecting this compound to rare but vital scientific progress. The future could hold more restrictions, tighter controls, or even outright bans outside strict research settings. Some labs explore ways to treat or neutralize the compound’s risks, aiming for smarter clean-up or less-toxic chemical cousins. The pressures build, but so does the understanding. It’s not about eliminating risk—more about managing it, learning from the past, and building forward with eyes open, not shut.
What is Trimethyltin Acetate used for?
The Lab World of Trimethyltin Acetate
In the chemical world, there’s always a quiet hubbub about substances you don’t hear about on the evening news. Trimethyltin acetate is one of those. Folks working in organic and organometallic chemistry use it as a tool for research. Researchers use compounds like this to understand molecular behavior, design new reactions, and figure out how tin-based chemicals interact with others. It’s not a household name for regular consumers, but it has a steady spot on many laboratory shelves.
Breaking Down Its Practical Value
Working with chemistry for a few years, I’ve seen that specialty chemicals such as trimethyltin acetate show up most in places chasing niche reactions. In organic chemistry, it’s part of the toolbox for making compounds that regular tin chemicals just can’t manage. Researchers have used it to create carbon-tin bonds. These bonds end up in pharmaceuticals, advanced materials, and all sorts of chemical intermediates. Trimethyltin groups have unique reactivity, making them a choice for preparing certain building blocks in synthetic chemistry. It’s a little bit like having a special wrench for a stubborn bolt, and sometimes, that’s the only tool that gets the job done.
Concerns and Hazards
The flip side of this specialty comes with real-world headaches. Handling trimethyltin acetate means wrestling with a substance that’s both toxic and difficult to get rid of safely. Organotin compounds in general have a pretty bad reputation for their effects on nerves and other body systems. One accidental spill can make a big mess, as this stuff isn’t just hard on lab benches—it’s dangerous for people. Scientists understand these risks well. Universities and research facilities keep strict rules for storing and using anything organotin-based. Safety training isn’t just a box to tick; it’s the recipe for keeping people healthy.
Environmental and Ethical Considerations
The way trimethyltin acetate lands in the environment matters just as much as how it works in the lab. These chemicals don’t break down easily. They can linger in waterways and soil, risking harm to wildlife. I’ve seen chemists argue about the right ways to dispose of tiny sample vials, knowing a careless pour down the drain could stick around for years. Respect for the environment doesn’t just mean regulations—it starts with the decisions made at every lab bench.
Looking for Better Solutions
New generations of scientists and industry leaders keep searching for less harmful options. Research into “greener” chemistry means substituting dangerous reagents with safer ones wherever possible. In some areas, chemists have found alternative tin-free routes for making important molecules, though not every reaction has an easy substitute. Strict safety data sheets, clear training, and investment in proper ventilation and disposal systems cut down on problems before they start. For every bottle of trimethyltin acetate on the shelf, there’s a growing push to find ways that don’t put people or the planet at risk.
Responsible Progress, Fact-Based Decisions
Trimethyltin acetate keeps its place as a specialty chemical, important for cracking tough scientific problems. But smart use always means weighing risks, keeping up with the latest research, and never losing sight of safety or environmental health. Progress comes from asking good questions in the lab and making sure every answer stands on careful science—no shortcuts, no blind spots.
What are the safety precautions when handling Trimethyltin Acetate?
Understanding the Risks
Trimethyltin acetate shows up in specialized labs more than on typical factory floors. It’s not something folks encounter every day, but those who work with this compound know it has a dangerous side. Inhaling or touching it can lead to headaches, nausea, and even severe neurological problems. The stories from the eighties, when workers handled trimethyltin compounds without enough protection, linger in industrial safety circles. Some suffered lasting nerve issues. That’s a hard lesson to ignore.
Personal Protective Equipment Isn’t Just a Suggestion
Nobody walks into a lab with trimethyltin acetate wearing only their regular street clothes. Lab coats, chemical splash goggles, and face shields become non-negotiable. Close-fitting nitrile gloves beat out latex, which offers less chemical resistance. If you ever caught a whiff of something sweet when opening a bottle, you likely realized the fumes sneak up fast. That’s why wearing a respirator, rated for organic vapors, offers one of the best defenses.
Ventilation and Engineering Controls
Fume hoods rank among the best friends of any chemist handling this stuff. Running an experiment with trimethyltin acetate on an open bench almost always brings trouble. Good airflow pulls stray vapors away, not toward your nose. A fume hood, kept uncluttered and well-maintained, does its job better than trying to air out a room later. Local exhaust and closed systems keep the risk down for everyone working nearby.
Careful Containers and Clear Labels
People who forget to label jars often pay for it down the road. Trimethyltin acetate reacts with moisture and breaks down over time, so glass containers with tight seals work best. A faded marker or mystery flask invites accidents. Regular inspections spot leaks, cracks, or residue buildup before problems start spreading.
Spill Response and Cleanup Plans
Nobody wants to scramble in a panic after a spill. I’ve seen labs lose hours from one broken vial because people didn’t have proper protocols or materials nearby. Absorbent pads, a spill kit rated for organotin compounds, and buckets of sodium thiosulfate for neutralizing help. Everyone working in the lab needs a walk-through of the plan at least monthly. Practicing a scenario helps people react level-headedly when something actually happens.
Training and Culture Matter
What kept me safe wasn’t just rules taped to a wall—it was older colleagues stepping in when someone worked carelessly or got too confident. Regular training and strong mentorship make the difference. No one should handle trimethyltin acetate alone until they prove caution in routine tasks. If people offer advice or corrections, they usually do it because years in the lab taught them hard lessons.
Medical Monitoring and Communication
Labs working with toxic chemicals do well to offer regular medical checkups. Tests tracking nerve health pick up early warning signs before symptoms get bad. Quick communication if something feels off saves trouble later. If gloves tear or a spill happens, reporting it right away beats waiting or hoping a rash fades.
Improving Safety as a Team
No single piece of advice solves every hazard. Each lab needs regular reviews of its safety practices, and open conversations about what’s working and what isn’t. Sometimes, updating a protocol or swapping gear makes life easier and safer for everyone. People protect each other as much as they protect themselves. That’s the real strength behind safe chemical handling.
What is the chemical formula of Trimethyltin Acetate?
Getting to Know Trimethyltin Acetate
Trimethyltin acetate doesn’t show up in everyday conversations, but the compound pops up in labs and research papers often enough for anyone involved with organotin chemistry. This chemical goes by the formula C5H12O2Sn. That means every molecule packs three methyl groups connected to a tin atom, sitting alongside an acetate group. Breaking it down, it holds three –CH3 groups and one –OOCCH3 group, all bound to tin, so you get a molecule that keeps things simple in its structure but complex in behavior.
Why the Formula Matters in Science
For chemists, the formula isn’t just a string of letters and numbers. It’s a code that hints at how the compound acts in experiments or the environment. Organotin compounds like trimethyltin acetate make headlines for a couple of reasons. Some researchers reach for them when developing catalysts or probing the effects of organometallics in biological systems. Others raise red flags because organotin compounds have a reputation for disturbing ecosystems and showing toxicity that forces industry and academia to handle them carefully.
In my own run-ins with tin-based reagents, having a precise formula on hand made all the difference when trying to balance out a reaction. You want to know exactly what elements and functional groups come to the table because mistakes in this area can end up wasting time, setting off unwanted reactions, or even risking personal safety. For anyone stepping into a chemistry lab, the formula isn’t trivia. It sits at the center of any safe and successful workflow involving organotins.
Risks Linked with Trimethyltin Acetate
Every chemist faces trade-offs between usefulness and toxicity. Trimethyltin compounds, acetate included, get flagged for neurotoxicity. That means even small amounts can harm animals and potentially people. Studies show organotins disrupt cellular processes, even at low concentrations. European regulations and U.S. oversight groups both watch these chemicals closely, precisely because of these risks. For anyone using them, solid knowledge of what’s inside the bottle—down to the formula—acts as the first step toward careful handling and disposal.
I remember a graduate project that involved tracking organotin byproducts across several reactions. Precise labels made cleanup much smoother. There’s peace of mind in knowing exactly which formula you’re working with—especially on lab benches crowded with similar-looking compounds.
Building a Safer Future with Accurate Information
Safety starts with good information. Listing the formula, C5H12O2Sn, clearly on containers keeps everyone on the same page. Proper training in chemical handling expects folks to remember more than just the structure. It’s about understanding reactivity, knowing what protective gear matches the hazard, and never cutting corners with waste. Schools and universities play an important part by teaching students to spot these formulas early, so the next wave of researchers steps into careers with safety built in.
The sustainability side doesn’t always get talked about, but it matters. Developing greener alternatives means understanding today’s compounds right down to the formula. Each piece of data surrounding trimethyltin acetate—composition, reactivity, health effects—builds the foundation for tomorrow’s safer chemicals.
How should Trimethyltin Acetate be stored?
Why Storing Trimethyltin Acetate Demands Care
Trimethyltin acetate falls into the category of organotin compounds that call for a lot of respect and careful handling. A compound like this, used sometimes in organic synthesis and research labs, can bring trouble if someone stores it like household cleaning supplies. Toxicity isn’t something to wave off—organotin compounds have brought painful lessons in chemical safety over the years. Trimethyltin in particular has a reputation for neurotoxicity in people. Improper storage or careless handling can cause contamination, health problems, and expensive cleanup.
From my time in chemistry labs, people who treat toxic substances casually end up with stories nobody wants to repeat. The sharp, particular smell of tin compounds lingers as a warning long after a spill. I remember one incident where a neglected lid led to fumes that cleared a room in minutes. Local authorities required a decontamination service, and the entire lab lost a week of work. That sort of risk—both financial and personal—isn’t theoretical.
Getting Storage Right: Steps and Solutions
Storing trimethyltin acetate right starts with keeping it away from heat, spark, and light sources. Direct sunlight, warm rooms, or open shelves don’t cut it. A ventilated, temperature-controlled chemical cabinet, shielded from UV exposure, gives much better protection. With highly toxic compounds, storing chemicals at a consistent 20-25°C helps reduce pressure buildup or breakdown risks.
Humidity control stands out almost as much as temperature. Keep the container tightly closed and stored in a dry place. Moisture can react with the contents, cause hazardous leaks, or trigger slow decomposition. Laboratories that follow best practices use desiccators or keep humidity below 50% in chemical storage rooms. Storing each container inside a secondary tray or bin adds another layer of insurance against accidental leaks.
Trimethyltin acetate doesn’t work well with acids or oxidizers. Storage requires segregation from acids (including everyday acids like hydrochloric or sulfuric) and strong oxidizing agents. Mixing these chemicals, even by accident, can produce toxic gases or aggressive reactions. Good chemical storage never puts incompatible compounds on the same shelf or even the same secondary container.
Clear labeling matters more than fancy lockboxes. Every bottle should display the chemical name, hazard symbols, date received, and responsible person’s initials. Emergency teams, coworkers, or even a bystander deserve to know what sits on those shelves—especially after an accident. My own experience taught me never to trust faded or handwritten labels. Print new labels, and check them quarterly.
Protecting People, Not Just Product
Personal protective equipment stands between a simple storage task and lasting injury. Lab workers shouldn’t move or handle trimethyltin acetate without gloves, lab coats, and splash goggles. Many labs keep a face shield handy for extra protection during transfer. Think of it this way—basic PPE costs cents per use, but accidents ring up medical bills in the thousands.
Training for anyone who touches the bottle should stay current. A freshly hired staff member or visiting researcher needs a walkthrough before ever opening a cabinet. Training should cover risks, cleanup procedures, and what to do after accidental exposure. Keeping kit like spill containment materials and eye wash stations nearby can turn a crisis into a routine cleanup. My own labs practiced emergency walkthroughs every six months, making sure even the clumsiest chemist knew the steps.
Trimethyltin acetate deserves respect, not fear. With simple planning, sturdy labeling, and a bit of daily vigilance, labs keep people safe and protect their research investment. That’s the payoff of good storage—a workplace that never sees a toxic tin disaster on the evening news.
Is Trimethyltin Acetate toxic or hazardous?
Understanding the Threat
Big words and chemical names sometimes pop up in headlines, but the story often gets lost behind the jargon. Trimethyltin acetate sounds harmless, maybe like something you’d find in a lab jar tucked at the back of a shelf. This substance actually packs a dangerous punch. People working with chemicals know that organotin compounds, trimethyltin varieties included, can be more than just a handful—they inflict real harm.
Toxicity You Can’t Ignore
It’s not only scientists who point out the dangers of trimethyltin acetate. Real evidence comes from case studies and workplace incidents around the world. Exposure usually happens by breathing in dust or vapors, swallowing contaminated material, or through skin. Trimethyltin itself has a notorious history for tearing up nerves. Public health records from metal recycling centers and chemical plants link confusion, violent headaches, and seizures to exposure. Deaths and lasting brain damage have cast a long shadow over its story.
How Poisoning Happens
Trimethyltin compounds slip past the protective fences our cells build. They harm the nervous system first, but organs like the kidneys and liver take a hit too. Imagine memory loss and behavior changes after a single accident in a plant; stories like this aren’t rare in countries with relaxed safety rules. One case recorded in The Lancet told about workers who inhaled organotin dust and ended up hospitalized with muscle weakness and psychotic symptoms. The toxic effects sneak up on the body, so people can get very sick before anyone realizes the real cause.
The Bigger Health Picture
It’s hard to ignore that most cases happen in places where chemical controls lag behind. Tom, a safety tech I met, talked about a plant that once stored drums of trimethyltin acetate, then shifted its stock quietly after a coworker collapsed. Stories from the field keep circling back to the same point: basic training, proper storage, and real vigilance prevent tragedy. In places without rigorous oversight, trimethyltin acetate slips through the cracks, and so does public health.
Environmental Cost
Organotins drift into air and water. When they do, fish and wildlife start showing strange behavior and reproductive changes. Not only people pay the price. Runoff from factories reaching streams and rivers hurts entire ecosystems. Places near poorly regulated facilities end up risking contaminated water, bringing hidden harm to nearby communities. It’s happened in countries pushing for fast industrial growth, where environmental watchdogs struggle to keep up with illegal dumping and leaks.
Reducing the Risk
The fix starts with labeling and strict storage rules. In labs where hazardous chemicals stay locked away, injuries drop. Factory owners who invest in solid ventilation systems keep their people safer. Simple steps—like gloves, eye protection, and regular air monitoring—save lives. Public databases let people check if dangerous chemicals are being used or transported nearby. Some countries push for substitutions, urging companies to roll out safer compounds in place of trimethyltin acetate routines. It’s not rocket science, just common sense and accountability.
Moving Forward with Awareness
Nobody wants to face a crisis in silence, so real change calls for awareness at every level. Teaching young technicians, enforcing local safety plans, and listening when workers speak up can keep trimethyltin acetate from becoming tomorrow’s headline. The hazardous label sticks for a reason—the facts and the stories back it up. In our drive to innovate and build, safeguarding life and health always deserves a front seat.
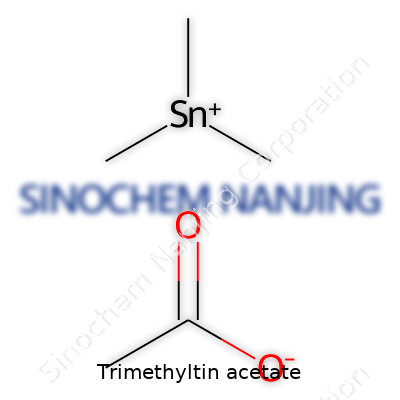
| Names | |
| Preferred IUPAC name | acetyloxy(trimethyl)stannane |
| Other names |
Acetic acid, trimethyltin salt Trimethyltin(IV) acetate Trimethylstannyl acetate |
| Pronunciation | /traɪˌmɛθɪlˈtɪn əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | [1066-45-1] |
| Beilstein Reference | 1541792 |
| ChEBI | CHEBI:135564 |
| ChEMBL | CHEMBL4299601 |
| ChemSpider | 161100 |
| DrugBank | DB02145 |
| ECHA InfoCard | 100.044.212 |
| EC Number | 213-856-6 |
| Gmelin Reference | 85372 |
| KEGG | C19153 |
| MeSH | D014259 |
| PubChem CID | 2734168 |
| RTECS number | WH2625000 |
| UNII | 9M8K127T1W |
| UN number | UN2354 |
| Properties | |
| Chemical formula | C7H16O2Sn |
| Molar mass | 206.96 g/mol |
| Appearance | Colorless liquid |
| Odor | pungent |
| Density | 1.32 g/cm³ |
| Solubility in water | decomposes in water |
| log P | 1.72 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 3.5 |
| Basicity (pKb) | pKb = 6.48 |
| Magnetic susceptibility (χ) | -23.0e-6 cm³/mol |
| Refractive index (nD) | 1.488 |
| Viscosity | 290 cP (20°C) |
| Dipole moment | 1.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 344.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -393.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1623.5 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and eye irritation, may cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H311: Toxic in contact with skin. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-3-2-W |
| Flash point | Flash point: 110°C |
| Lethal dose or concentration | LD50 (oral, rat): 47 mg/kg |
| LD50 (median dose) | 40 mg/kg (rat, oral) |
| NIOSH | UN2810; RTECS: WH8575000 |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | IDLH: 4 mg/m³ |
| Related compounds | |
| Related compounds |
Trimethyltin chloride Trimethyltin hydroxide Trimethyltin bromide Trimethyltin iodide Trimethyltin fluoride |

