Trimethylhexamethylene Diisocyanate: Navigating Innovation, Risk, and Responsibility
Tracing the Journey: Historical Context and Shifting Demands
Chasing new building blocks for industry has never been a simple affair. Trimethylhexamethylene diisocyanate, the sort of chemical that rarely makes headlines outside technical circles, tells a story of problem solving and curiosity. Early in the twentieth century, isocyanates like TDI and MDI grabbed most of the attention, but corners of the chemistry world kept exploring structure-property relationships—especially for applications like specialty coatings. Diisocyanates offer a high degree of reactivity. Over time, industrial chemists noticed that the addition of methyl branches, as with isomers of trimethylhexamethylene diisocyanate, shifted volatility, toughness, and reactivity in interesting ways, giving manufacturers more control over final product properties in coatings, adhesives, and sealants. This direction led to adoption in the automotive, electronics, and construction sectors, each looking for coatings that could handle tough environments without compromising appearance or adhesion.
Understanding the Product: Chemistry, Labels, and Presentation
Trimethylhexamethylene diisocyanate appears in chemical catalogs under a patchwork of names—TMDI or TMXDI, depending on suppliers or labeling conventions. The molecules lurk as colorless to pale yellow liquids, often sporting a sharp acrid odor that betrays their reactivity. With boiling points pushing beyond 250°C and relatively low viscosities, industrial users value these characteristics for how they influence application and cure rates. Their chemical formula, old-fashioned as that may sound, doesn't change: C11H18N2O2. Quality-conscious buyers scan certificates listing purity, NCO content, water content, and color (by Hazen scale). Out in the open, strict regulations demand clear hazardous material labels warning about possible respiratory and skin hazards. Packaging never feels casual; from transport to storage, everything signals caution—not because manufacturers are fussy, but because the risks have proven real, both in the lab and on the shop floor.
The Process Behind the Product: Synthesis and Modifications
Talking about making TMDI goes beyond following recipes. The process starts with careful selection of trimethylhexamethylene diamines, followed by phosgenation. Each step pushes plant operators and safety engineers to manage heat, toxic byproducts, and purity. It’s no secret phosgene remains a potent toxicant, adding layers of containment and emissions control that drive up costs and complexity. Once produced, this type of diisocyanate often undergoes further tweaking—blocking, for instance, with alcohols or other nucleophiles, allowing users to control reactivity and storage stability. This menu of possible chemical modifications supports differentiation; tweaks let producers finetune working time and mechanical characteristics after application. Handling and transferring this stuff into usable intermediates never gets routine; those on the front lines see genuine danger in every splash or cloud, always weighing risk against the demands of the jobs ahead.
Names, Synonyms, and Misunderstandings
People working with isocyanates know how quickly a slip in nomenclature sparks confusion. In different papers or on different continents, trimethylhexamethylene diisocyanate may be called TMXDI, 2,2,4- and 2,4,4- isomers, or even specialty labels aligned with manufacturers’ branding. For newcomers, the jumble of abbreviations creates headaches during sourcing, literature searches, or regulatory filings. Regulatory lists insist on sharp definitions, but on the ground, teams need to put in extra diligence to guarantee they’re working with the right material and not a substitute with unexpectedly different reactivity or volatility.
Navigating Safety: Beyond Warnings and Data Sheets
Nobody working in environments like spray coating lines or resin mixing rooms glosses over the dangers of isocyanates. Acute exposure scrapes throats, stings eyes, and triggers asthma-like reactions. Chronic exposure, even under levels allowed by law, can result in long-term respiratory issues or sensitization. Ventilation, personal protective equipment, strict access controls, and routine air monitoring become part of daily ritual—less about compliance than trust in shared responsibility. Older generations in these industries sometimes downplayed these issues, but after decades of health cases and tightening liability, attitudes have changed. Risk management requires repeat training, clear communication, and relentless attention. Every incident—no matter how small—teaches something new about human error, equipment failure, or accidental cross-contamination. The stakes stay high because mistakes don’t just mean lost product; they mean compromised health.
Where It Winds Up: Applications Across Industries
Few end users realize how many surfaces or circuits rely on specialty diisocyanates. Trimethylhexamethylene types end up in tough, flexible coatings on cars, bridges, and electronics, and in high-performance adhesives for composite materials and medical devices. The appeal is about more than chemical curiosity: brands demand scratch resistance, flexibility, and clarity, while contractors value coatings that cure quickly and endure abuse from weather, chemical spills, or UV exposure. The electronics world relies on these materials not for glamour but for protecting sensitive circuitry from moisture, dust, and thermal cycling. Every new application brings pressure to use less hazardous inputs, to squeeze out more performance, and to minimize downtime. That demand pushes R&D labs hard, forcing constant reevaluation of raw materials, catalysts, and even alternative chemistries. It is a space shaped by trial, error, and the drive to solve immediate problems on factory floors.
Under the Microscope: Ongoing Research and Toxicity Findings
Over several decades, research on isocyanate toxicity has shifted from singular focus on acute reactions to a broader view of long-term risk. Respiratory sensitization stands as the most talked-about risk; scientific panels agree on this, yet specifics depend on formulations, use patterns, and workplace conditions. Some animal studies link chronic exposure to immune system disturbances and rare cancers, though epidemiological signals in humans remain less clear outside occupational asthma. In the lab, modifying diisocyanates to attach “blocking” groups or swapping in less volatile precursors helps to reduce airborne concentrations and thus worker risk—though no solution achieves perfect safety. Recent government and academic reports urge caution, supporting lower permissible concentrations in workplace air and promoting better exposure assessment techniques. The data always spark debate in industry, where balancing product performance and health risk turns into a moving target every time a new study lands.
Technical Challenges and Solutions on the Horizon
It’s easy to call for safer chemicals from the comfort of a conference room; out in the field, engineers run into stubborn limits. TMDI’s toughness and versatility in high-performance coatings keep them in demand, but the push to innovate never stops. Some labs experiment with non-isocyanate polyurethanes, adjusting aliphatic chains or looking at fully bio-based feedstocks. Others hunt for process changes to trap or neutralize stray emissions—adding scrubbers, improving seals, setting stricter batch records, and auditing the lifecycle of each drum. Researchers emphasize working with end users to understand true environmental loads, not just theoretical exposure. The slow adoption of green chemistry principles in this area isn’t about reluctance but about matching technical requirements that alternatives sometimes can’t yet meet. Meanwhile, tight collaboration among materials scientists, toxicologists, and regulatory watchdogs drives incremental progress—faster than in decades past, but never quite as fast as urgency might demand.
Future Prospects: Adjusting Course With Lessons Learned
The future for trimethylhexamethylene diisocyanate won’t look like the past. Global regulations around industrial chemicals stand poised to tighten, especially in developed economies where chronic disease links prompt faster rule changes. Industries pushing the edge of electronics, automotive, and new construction keep seeking better, safer, stronger coatings—pressures that drive both innovation and hard reflection about health tradeoffs. Some chemical firms pivot their R&D to alternatives such as blocked isocyanates, waterborne systems, or even next-generation biopolymers, hoping to maintain performance while sidestepping legacy hazards. The cycle repeats: new materials bring new unknowns, and regulators, scientists, and end users all learn together. Long-term, the winners in this space won’t just offer resilient chemistry—they’ll earn trust from workers, safety regulators, and the communities where their plants run. Tackling these challenges means balancing ambition with humility, learning from hard-won experience, and keeping both eyes on health and environment at every stage.
What is Trimethylhexamethylene Diisocyanate used for?
What’s Behind This Chemical Name?
Trimethylhexamethylene diisocyanate—usually called TMHDI—doesn’t roll off the tongue, but it shows up in more places than most people realize. This compound belongs to a group known as aliphatic diisocyanates. These ingredients have a big impact on the world of specialty coatings, sealants, and elastomers. What makes TMHDI stand out? It adds resilience, heat resistance, and longevity to products that take a real beating in daily life.
Where Do We Find TMHDI?
TMHDI goes straight into making coatings that need to last—especially outside, under sun and rain. If you drive a car, some of the clearcoat on its surface owes part of its toughness to this chemical. Products like outdoor furniture, wind turbines, and heavy-duty industrial flooring see a boost from TMHDI-based coatings because they resist yellowing, cracking, and the slow breakdown from the environment.
Manufacturers also lean on TMHDI for specialty adhesives and sealants that face tough conditions. Pipes in water treatment plants, marine components, aircraft interiors—engineering choices come down to chemistry that holds up and won’t fail early. That’s where this compound earns its keep. TMHDI-based polyurethane elastomers get molded into wheels, grips, and flexible parts that need to bend without getting brittle or sticky with age.
Health and Safety Concerns
Working with TMHDI requires real caution. Years of research and workplace stories point out that diisocyanates can cause breathing trouble, skin irritation, and wheezing for people exposed on the job. High-profile cases have led to tighter regulation. The European Chemicals Agency added TMHDI to lists calling for better risk management. In the US, the Occupational Safety and Health Administration (OSHA) reminds employers to keep exposure as low as possible. People doing this work need full training, good ventilation, and the right protective gear. The facts show that even a small exposure may lead to sensitization, making future contact risky.
Communities near factories sometimes worry about air and water emissions tied to chemicals like TMHDI. I’ve witnessed local residents struggle to get answers after odd odors drifted into their neighborhoods. Public health researchers want more transparency and better monitoring. One solution that stands out is the use of closed systems and scrubbers to keep chemicals contained inside the plants—keeping both workers and bystanders safer.
Striving Toward Safer Industry Practices
Industries using TMHDI keep searching for greener alternatives, but the balance between durability and lower toxicity isn’t always easy to achieve. Some researchers work on waterborne coating options, hoping to phase out more toxic ingredients without sacrificing toughness. These efforts need support from policymakers and company leaders, not just scientists.
For now, the key lies in education and ongoing monitoring. Real-world incidents, from employee health issues to accidental leaks, teach hard lessons. Listening to workers and affected communities gives companies a clearer idea of what safer practices should look like. Regulators push for recordkeeping and transparent reporting. The more we use data and technology to track risks, the better chance we have at preventing harm while keeping the benefits of tough, long-lasting materials.
What are the health hazards of Trimethylhexamethylene Diisocyanate?
What Workers Face on the Job
I’ve met folks who handle chemicals like Trimethylhexamethylene Diisocyanate (TMDI) every day. They mix coatings in paint shops, work with resins in factories, or help design new plastics. Most just want a safe workplace and a steady paycheck. But TMDI can turn routine tasks into health gambles if safety slides or information gets lost in translation.
TMDI is a powerful isocyanate used mostly in making specialty polyurethanes. Breathing in even low amounts can spark allergies that never really go away. Some people find themselves short of breath just walking into a facility where TMDI floats in the air. Isocyanates are notorious for this—sensitization shows up as asthma, wheezing, or coughing. The tricky part is once sensitivity develops, even tiny exposures can trigger symptoms. It doesn’t always take years of contact, either. A big dose all at once or repeated, lower exposures both put workers at risk.
Short-Term Risks and Long-Term Damage
Spills, splashes, or sloppy air filtration aren’t just annoyances. Direct skin contact with TMDI can cause rashes, chemical burns, or eye irritation. Fumes irritate noses and throats quickly. Folks may shrug off irritation as “part of the job,” but that would be a mistake. My experience watching colleagues in the auto industry taught me that serious reactions sometimes hit the healthiest people hardest. In a busy shop, you don’t want to discover your gloves or goggles are old or cracked after an accident happens.
TMDI’s biggest danger lies in repeat or ongoing exposure. Chronic effects show up over time and cheat folks out of healthy years. Studies back this up: workers with long-term isocyanate exposure are at higher risk for chronic lung problems, even cancer. The risk doesn’t stop at the factory gate. Isocyanates can linger on clothing, spreading home; I’ve seen families affected indirectly, with loved ones picking up traces on laundry or skin.
Making Safety Real, Not Just Policy
Regulators know the numbers. OSHA and NIOSH set TMDI exposure limits for a reason—exceeding even small amounts at work can change lives. I remember seeing a plant manager overhaul an entire ventilation system after a near-miss—a rare boss who listened when technicians reported headaches and chest tightness after a change in the formula. In my view, every company should follow that lead. Real air monitoring, routine equipment checks, and actual buy-in from every supervisor matter far more than posters on the wall. Respirators and gloves are only useful if workers have the training and trust to use them every shift.
Health Should Come Before Production
Alternatives exist. Newer resins sometimes skip hazardous isocyanates, and automation can keep human hands out of the most dangerous processes. When that’s not possible, enforcing strict hygiene and monitoring takes over: air extractors, enclosed systems, generous paid sick leave for anyone showing symptoms. Healthcare access has to stay strong, so workers can report symptoms early without fear.
TMDI isn’t a household word, but the folks who use it deserve more than a footnote in a safety manual. Workers bring skill and pride to their jobs; asking them to risk their lungs or skin for a wage is just too high a price. Every employer and engineer owes it to themselves and their team to cut risk wherever possible. That’s a lesson learned best through action, not slogans.
How should Trimethylhexamethylene Diisocyanate be safely handled and stored?
Taking Chemical Safety Seriously
Anyone who’s spent time in a paint factory or a polyurethane production line knows about the strong smell that bites at the back of your nose. Trimethylhexamethylene diisocyanate (TMDI) is one of those pungent chemicals with a reputation for powerful reactivity and real health risks. Exposure can bring on asthma-like symptoms, skin irritation, or even more severe respiratory damage. I remember the first time I caught a whiff near an open drum—I learned fast that two layers of gloves and a fitted mask weren’t optional extras.
The Real Risks: Why Ventilation Matters
TMDI vapors rise quickly at normal room temperature. Without steady air exchange, vapor concentrations sneak up fast. Relying on open windows won’t cut it. Workers should make use of purpose-built fume hoods and exhaust fans. Regular air monitoring with detection badges or sensors gives everyone peace of mind, especially during bulk transfers or spills.
Everyday Protections Make the Difference
No one wants to develop a cough or rash from showing up to work. Well-fitting goggles, nitrile gloves, and chemical-resistant coveralls are part of the daily uniform. In shops I’ve managed, we keep a clear separation between where people suit up and the spots where TMDI gets handled. Showers and changing areas help keep the chemical out of cars and off kitchen tables at home.
Smart Storage Cuts Down on Emergencies
TMDI reacts with water in the air, producing carbon dioxide and heat—sometimes enough to compromise a drum. Avoid storing it in humid basements or next to doors that see outside air. Drums and totes last longer sealed tightly, tucked away from direct sunlight. Block off storage spots from forklift traffic or anything with a spark, since this is a flammable substance.
Inspection routines prevent leaks and surprise pressure buildups. Labels showing hazards and emergency contacts trim confusion when something spills or someone gets exposed. I’ve noticed the difference after switching to color-coded labels; nobody wonders which chemical they’re grabbing.
Response Preparedness: Small Steps, Big Payoff
Emergency plans should never collect dust. Even short drills—simulated spills, first aid react outs—make everyone sharper when the real thing hits. Stocking up spill kits with neutral sorbents, extra gloves, and appropriately-sized waste drums means crews don’t scramble if a hose cracks or a barrel tips. Training goes beyond a yearly video; hands-on, repeated demos let everyone respond confidently.
Building Habits That Stick
Safety doesn’t come from a heavy binder or a locked cabinet—it comes from habits set at every level of the team. New hires shadow experienced staff. Regular reminders replace boring lectures with short, practical pointers. Little actions—wiping down gear, snapping closed a lid, double-checking a gauge—add up. I’ve seen the tone change from “extra work” to everyone looking out for each other.
A Culture of Respect for Chemicals
TMDI’s dangers never fully disappear, but working with care and respect, crews can ramp up safety without slowing down. Healthier workspaces, fewer lost-time incidents, and smoother shifts prove that smart handling and storage pay off well beyond the bottom line.
What personal protective equipment is required when working with Trimethylhexamethylene Diisocyanate?
Facing Hard Truths on Chemical Safety
Trimethylhexamethylene diisocyanate, often popping up in polyurethane coatings or specialty adhesives, lands workers in a tough spot. These jobs look straightforward, but one careless day can haunt someone down the line. Speaking from years around construction and industrial sites, safety rules around chemicals tend to get ignored or rushed, either from pressure to finish or simply not knowing the real risks.
This chemical opens the door to big health threats. Inhaling its vapor makes breathing tough, triggers asthma-like symptoms, and can even sensitize the lungs permanently. Spills or splashes on skin might trigger rashes, burns, or allergies. Without question, proper personal protective equipment (PPE) means a lot more than a dust mask and old gloves.
The Real Gear You Need—Not Just to Check a Box
Questioning big claims, I asked old friends in manufacturing what works. Across the board, everyone starts with respirators. Not surgical masks from a home supply, but full facepiece or half-mask air-purifying respirators with organic vapor cartridges. For heavy concentrations, supplied-air respirators fit the job. Disposable masks simply don’t stand a chance against these fumes. The data backs this up, too. NIOSH tracks occupational asthma cases, finding isocyanates like this one as a leading trigger.
For hands, no hardware-store cotton gloves can suffice. Nitrile or butyl rubber gloves block skin contact best, replaced often at the first signs of weakness. Thick neoprene suits or Tychem garments protect arms and bodies; thin coveralls or street clothes never cut it. Anything splashed on a sleeve, even for a second, can lead to skin issues for months. It's an uncomfortable truth too many discover after the fact.
Face shields and splash goggles matter nearly as much as breathing protection. Vapors irritate eyes swiftly, so sealed goggles stay on until the last tool is put away. Regular safety glasses miss too many gaps where vapor creeps in, and most folks end up rubbing itchy eyes, which only deepens exposure.
No Substitute for Training, No Excuses for Skipping Checks
PPE won’t work without training or when the habit is to “just try to be careful.” I’ve watched people collapse routines—gloves tossed, hoods left down, or respirators reused past their safe window. Supervisors have a duty to retrain, push for fit testing, and keep fresh stock on hand. Workers themselves should ask for equipment that fits right—not just size, but also the actual task at hand. Leaks can form if a hood is loose or gloves don’t seal tight at the wrist.
Maintenance sounds dull, but it keeps PPE from dying on the job. Rinsing goggles, double-checking cartridges, swapping out gloves after each session—these tasks save skin and lungs. Emergency eyewash stations or showers look out of place until a spill turns them into a lifeline. In my work life, folks who push for drills and regular equipment checks go years without incidents, while those who don’t start racking up worker’s comp cases fast.
Push for a Culture, Not Just Compliance
Wearing proper PPE for trimethylhexamethylene diisocyanate isn’t just a box to tick so inspectors back off. Everyone sharing the worksite should take ownership, holding each other to standard. One time, a coworker’s cracked mask led to a major exposure, and others noticed it too late. That memory sticks with the team today, shaping habits on every shift. Nobody wants to cut corners knowing the cost isn't just monetary, but also a burden on personal health for years ahead. A culture of calling out hazards, bringing up equipment flaws, and replacing worn gear does far more than any written policy ever could.
Is Trimethylhexamethylene Diisocyanate regulated or restricted by any authorities?
Knowing the Chemical
Trimethylhexamethylene diisocyanate (TMDI) packs a punch in manufacturing circles. Producers use it for coatings, adhesives, and elastomers, where durability or flexibility matters. It’s not a household word, but this stuff turns up in places that count—factories, workshops, and job sites dealing with high-performance plastics and paints. Most folks never notice it, but the workers and engineers staring at safety data sheets sure do.
The Regulatory Landscape
Government agencies aren’t asleep at the wheel with chemicals like TMDI. In the United States, the Environmental Protection Agency (EPA) includes diisocyanates in their chemical oversight programs. This isn’t red tape for the sake of hassle. Isocyanates as a group cause skin and respiratory irritation, and asthma cases linked to workplace exposure keep cropping up in reports. The Occupational Safety and Health Administration (OSHA) enforces strict rules for companies using isocyanates, requiring ventilation, protective equipment, and medical surveillance.
Europe does not mess around, either. The European Chemicals Agency (ECHA) lists TMDI on databases tied to REACH regulations. Under REACH, manufacturers and importers submit safety data, and employees handling the chemical get training on its dangers. It’s not about banning products just because their names sound intimidating; it’s about tracking risk and keeping workers healthy.
In Asia, growing workforces in chemical manufacturing pay attention to global trends. Japan and China have both expanded regulations about workplace exposure, largely echoing rules established elsewhere. So, people making and handling TMDI can’t say no one warned them.
Why Controls Matter
A poorly ventilated plant can turn a useful chemical into a ticket to the emergency room. I once toured a facility where a new batch of workers didn’t fit their respirators correctly. The air didn’t smell unusual, but two guys coughed for weeks. Their boss later shared that isocyanate exposure caused their symptoms. Training and oversight saved the rest of the crew from breathing that risk every shift.
Health effects with diisocyanates deserve respect, not just careful paperwork. Chronic asthma isn’t just an itchy throat—a friend working in coatings told me every job site handled these drum containers with kid gloves because the wrong move could shut down a small plant with sick calls. These stories drive home why it matters for authorities to step in.
Room for Better Practices
Strong rules help, but they only count if followed. Plants that provide steady training and invest in better gear keep their injury numbers down. Digital monitoring systems track air quality in real-time nowadays—much better than guessing by smell or gut feeling. Small shops or sub-contractors sometimes lag behind on these steps, relying on old routines and luck, which isn’t a safe bet.
Clear communication sits at the heart of real safety efforts. Labels, data sheets, and easy-to-read warning signs shouldn’t turn into background noise. Management that listens when folks report symptoms or suggest safety tweaks set the tone for safer work. The pressure to cut corners in competitive industries stays strong, but the long costs of sick workers and legal blowback add up quickly.
Bureaucracy can frustrate, but the intention remains rooted in common sense: keep people healthy, hold businesses to account, and prevent tomorrow’s headlines from echoing yesterday’s mistakes.
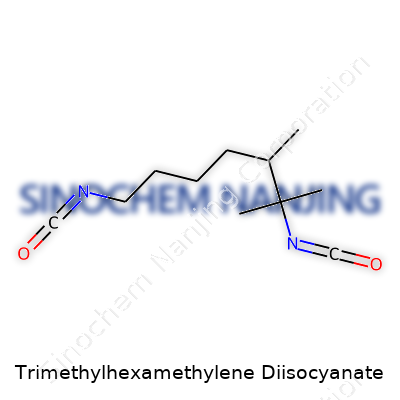
| Names | |
| Preferred IUPAC name | 2,2,4-Trimethylhexane-1,6-diisocyanate |
| Other names |
TMDI Isocyanic acid, trimethylhexamethylene ester 1,6-Bis(isocyanatomethyl)-2,2,4-trimethylhexane Hexamethylene diisocyanate, trimethyl derivative 2,2,4-Trimethyl-1,6-hexamethylene diisocyanate |
| Pronunciation | /traɪˌmɛθ.ɪlˌhɛk.səˈmiːl.iːn diˌaɪ.ˌaɪ.səˈsaɪ.əˌneɪt/ |
| Identifiers | |
| CAS Number | 2566-54-3 |
| Beilstein Reference | 1718731 |
| ChEBI | CHEBI:53288 |
| ChEMBL | CHEMBL4306653 |
| ChemSpider | 123641 |
| DrugBank | DB16955 |
| ECHA InfoCard | 100.067.319 |
| EC Number | 248-207-6 |
| Gmelin Reference | 80777 |
| KEGG | C18745 |
| MeSH | D016439 |
| PubChem CID | 85754 |
| RTECS number | OM2975000 |
| UNII | OVV8M43R8K |
| UN number | 2483 |
| CompTox Dashboard (EPA) | DJ8R6D9V6C |
| Properties | |
| Chemical formula | C12H18N2O2 |
| Molar mass | 258.34 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent odor |
| Density | 0.990 g/cm3 |
| Solubility in water | Insoluble |
| log P | 3.2 |
| Vapor pressure | 0.01 mmHg @ 25°C |
| Acidity (pKa) | 14.9 |
| Basicity (pKb) | 13.61 |
| Magnetic susceptibility (χ) | -5.97×10⁻⁶ |
| Refractive index (nD) | 1.4510 |
| Viscosity | 22 mPa·s (at 25 °C) |
| Dipole moment | 4.02 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 438.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -743.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –3976 kJ/mol |
| Hazards | |
| Main hazards | Harmful if inhaled. May cause allergy or asthma symptoms or breathing difficulties if inhaled. Causes skin irritation. May cause an allergic skin reaction. Causes serious eye irritation. Suspected of causing cancer. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H331, H334, H317, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P284, P302+P352, P304+P340, P305+P351+P338, P308+P311, P312, P333+P313, P337+P313, P342+P311, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-2-W |
| Flash point | 163 °C (325 °F) |
| Autoignition temperature | 225 °C |
| Explosive limits | 2.5% - 10.2% |
| Lethal dose or concentration | Lethal dose or concentration (LD50/LC50): LD50 oral (rat): > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 5,000 mg/kg |
| NIOSH | NIOSH: **NQ6475000** |
| PEL (Permissible) | 0.005 ppm |
| REL (Recommended) | 0.035 mg/m3 |
| IDLH (Immediate danger) | IDLH: 75 mg/m³ |
| Related compounds | |
| Related compounds |
Hexamethylene diisocyanate Isophorone diisocyanate Methylenediphenyl diisocyanate Toluene diisocyanate |

