Trimethylethoxysilane: Past, Present, and Future in the World of Chemistry
The Story Behind Trimethylethoxysilane
Trimethylethoxysilane didn’t just show up one day in a lab. Its roots go back over half a century, weaving through the evolution of organosilicon chemistry. Silanes, in general, came into play as researchers looked for new materials for glass treatment, electronics, and high-performance coatings. The development of trimethylethoxysilane came from this push—scientists tinkering with various silyl compounds found that adding an ethoxy group to the silicon atom opens up a range of new uses. Over time, industries began to recognize the value of silane coupling agents, both for their ability to bind organic and inorganic materials and for their remarkable stability under various conditions. Trimethylethoxysilane secured its place thanks to its consistency in performance, demonstrated in research papers and through the needs of specialized manufacturing lines.
Understanding What You’re Dealing With
Anybody who’s worked in a chemistry lab recognizes the value of knowing a material inside and out. Trimethylethoxysilane appears as a clear, relatively low-viscosity liquid with a distinct, ether-like odor. It boasts a fairly high vapor pressure at room temperature—enough to command respect during handling. The three methyl groups attached directly to the silicon atom provide hydrophobic character, while the ethoxy group is the source of its chemical flexibility. This balance gives it just the right level of reactivity for several silicon-based reactions, especially where a quick alkoxysilane is needed for surface treatments or building tailored molecules. The liquid itself is flammable and volatile, so it’s not something you want splattered around, and its chemical stability allows for safe storage under dry conditions, yet it’s ready to react with water or alcohols under the right circumstances.
Details Behind the Label
Chemical specifications tend to stretch across a page, but for me, what stands out about trimethylethoxysilane is its simplicity. Its molecular formula, C5H14OSi, explains almost all its behavior in practical terms. On chemical shipment drums or small reagent bottles, you’ll come across names like "ethoxytrimethylsilane" or "trimethylsilylethoxy," and sometimes even the shorthand TMES. Labeling regulations across regions focus on its physical hazard state—it’s a flammable liquid, and you need proper ventilation and grounding to avoid static discharge accidents. The most critical detail for the label: storage in tightly sealed containers away from moisture because water starts the hydrolysis process.
From Basic Ingredients to Finished Product
Mixing up a batch of trimethylethoxysilane rarely makes it into the spotlight, but in industry, the route generally starts with trimethylchlorosilane and ethanol, carried out under conditions that keep water at bay. The goal is to encourage substitution of the chlorine atom for the ethoxy group and to drive off byproducts like HCl, sometimes using a base to help mop up the acid as the reaction runs. This process needs careful temperature control—a lesson I learned after watching a much-too-enthusiastic reaction bubble over, driving home just how exothermic things get. Labs usually use anhydrous techniques, with inert gas like nitrogen, to prevent unwanted reactions. After synthesis, distillation refines the product to the necessary purity for high-tech use.
The Reactions That Make It Valuable
One reason trimethylethoxysilane earned its keep is the way it can act as a silylating agent for alcohols, acids, and amines. For chemists trying to protect reactive groups during a multistep synthesis, this compound provides a useful shield. You can see its true nature during hydrolysis—mix it with water and the ethoxy group falls off, leaving behind trimethylsilanol, which can further condense to silicon-oxygen-silicon linkages. In practice, researchers turn to it to modify glass surfaces, add hydrophobic coatings to mineral fillers, or introduce the trimethylsilyl group for analytical chemistry applications. The reactivity with nucleophiles such as alcohols and amines extends its role far beyond just a surface treatment chemical.
Synonyms Crop Up Everywhere
Over time, producers and researchers haven’t let one name stick. You’ll find trimethylethoxysilane listed as ethoxytrimethylsilane or TMES across suppliers and papers. These aliases make life interesting when hunting for reference material or ordering reagents, sometimes forcing a double-check to avoid confusion with similar silanes. The variety of names speaks to how widely it’s used—in everything from chemistry labs to industrial plants focused on surface engineering and electronic devices.
Standard Practices on the Floor
In practice, handling this compound can’t be taken lightly. Regulations focus on eye and skin protection and ensuring ventilation—anyone who’s gotten a whiff of it knows why. Spills create a fire hazard, so grounded containers and strict protocols for transferring the liquid matter a lot. In my experience, ignoring the advice about inerting storage containers or letting air into a bottle early leads to headaches, if not worse. National and international standards echo this reality, emphasizing local exhaust ventilation, flameproof equipment, and rigorous training for anyone with access.
Who Needs Trimethylethoxysilane?
Its reach stretches well beyond the chemistry bench. Polymer manufacturers use it to modify resins for enhanced durability and water resistance, while electronics producers turn to silanes like TMES for delicate semiconductor surface passivation, shielding circuits from humidity. Even in construction, it finds roles as an additive to adhesives and sealants. Analytical chemists value it as a reagent for preparing volatile derivatives for gas chromatography analysis, streamlining work with compounds that might otherwise decompose or stick to surfaces. This cross-sector demand keeps research funding incoming and encourages suppliers to offer high-purity grades in increasing amounts.
Pushing the Research Envelope
Those who’ve spent time in research labs know that organosilicon chemistry is still evolving. Universities keep probing into new uses for trimethylethoxysilane in nanotechnology, particularly as a precursor for superhydrophobic coatings and as a building block for complex polymers. Green chemistry initiatives push for alternatives to solvents and processing methods that reduce byproducts and waste, putting more focus on finding milder, less energy-intensive synthesis steps. Over the past decade, collaborative projects between academia and industry have aimed to stretch the boundaries, exploring catalytic pathways for milder, more selective reactions while prioritizing worker safety and environmental impact.
Looking at Toxicity and Safety Data
Safety never comes second, especially with chemicals that volatilize easily. Toxicology studies point to moderate acute toxicity by inhalation or ingestion. Short-term exposure often irritates mucous membranes, and splashes in the eyes or on skin bring about burning or redness. Chronic exposure data remain limited, highlighting the need for conservative safety margins. Responsible labs rely on closed systems when feasible, supported by fume hoods and personal protective equipment. Proactive health monitoring and a culture of immediate spill response go a long way toward preventing harm. Government agencies prioritize ongoing review of worker exposure limits and encourage substituting less hazardous alternatives when available.
Down the Road: Shifting Goals and Expanding Uses
What the future holds for trimethylethoxysilane depends heavily on advances in materials science and tighter regulations on chemical manufacturing. Researchers look for ways to harness its reactivity for greener surface treatments in the textile and packaging industries, while electronics continue to demand purity and reliability at every stage. As manufacturing processes trend toward automation and lower-energy syntheses, the silane sector is pressured to supply products with reduced environmental footprints. The search for less toxic alternatives isn’t likely to stop, but trimethylethoxysilane’s versatility, honed by decades of feedback from chemists and manufacturers, will keep it in play whenever a reliable, easily handled alkoxysilane fits the bill.
What is Trimethylethoxysilane used for?
Why Trimethylethoxysilane Matters
Trimethylethoxysilane doesn’t headline flashy tech conferences or get much attention outside the walls of a lab, but for anyone involved with coatings, adhesives, or advanced materials, this chemical quietly shapes our products. Drawing from experience in labs and manufacturing floors, I’ve seen this silicon-based compound play key roles that often get overlooked. While it might blend into ingredient lists, its impact stands clear once you look under the surface.
Building Better Surfaces
A big part of Trimethylethoxysilane’s power comes from its ability to treat and modify surfaces. Once applied, it creates a thin, water-repellent layer. Glass suppliers use this protection in windows, electronics, and lab equipment to keep moisture, fingerprints, and grime from sticking. In the world of electronics, a single silane treatment adds longevity and boosts insulation—helpful in chips and displays where stray currents mean trouble.
Construction and automotive coatings gain strength from this chemistry. Car windshields keep their clarity longer, and tile walls in showers shed water with less effort. The product helps keep dirt off, which saves cleaning time and supports durability. These everyday quality-of-life upgrades depend on molecules like Trimethylethoxysilane doing their work behind the scenes.
Sticking Power for Adhesives and Sealants
Offices and families both rely on glues and sealants that don’t easily crack or peel. Many modern adhesives owe extra grip and flexibility to this same compound. By bonding at a chemical level with glass, metal, and ceramics, these agents strengthen the connections they form. Construction, electronics assembly, and packaging lines need these strong bonds to resist heat, water, and pressure. Without this level of support, expensive repairs and product failures would pile up much faster.
Cleaner Chemistry for Modern Manufacturing
Environmental safety is a growing question in every industry. Working with silane-based chemicals, I’ve noticed a gradual switch toward options with lower emissions and greater stability. Trimethylethoxysilane stands out by helping manufacturers both lower their use of hazardous solvents and avoid extra waste due to its efficiency in small doses. Chronic exposure still calls for safety gear and good ventilation, but compared to other water-proofing agents, the compound shows lower toxicity in real-world shop conditions.
What Needs Improvement
Even as Trimethylethoxysilane improves coatings and connections, it comes with challenges. Reliable handling and transport need training, and spills can pose flammability risks. The chemical’s shelf-life gives a clock to manufacturing timelines—stale stock brings waste and extra costs. Some countries now push for responsible disposal and recycling, so companies look for biodegradable options or chemicals made from renewable sources.
Better labeling and real-time monitoring in factories could reduce risks further. Open communication between chemical suppliers, end users, and safety experts gives everyone a chance to make smarter moves. Switching to more sustainable silanes or adding recovery systems for used chemicals would help.
Final Thoughts
Trimethylethoxysilane often works outside the spotlight, but anyone involved in practical science, engineering, or manufacturing can point to places where small molecular tweaks bring big results. Improving how we work with these building blocks—using facts and real-world habits—means stronger products, safer jobs, and less waste.
What are the safety precautions when handling Trimethylethoxysilane?
Looking at the Risks Up Close
Trimethylethoxysilane isn’t a chemical you just splash around in a lab. The liquid gives off vapors that irritate the nose and throat almost instantly, so a little recklessness comes back to bite you fast. I’ve seen folks get lazy about goggles or gloves and end up with nasty rashes or red eyes no one wants. Handling this stuff teaches respect. As soon as you open a bottle, a sharp odor stings your lungs, and you remember right away—take it seriously every single time.
Personal Protective Equipment Is Basic Survival
Chemical splash goggles stay on, no excuses. Regular safety glasses leave skin exposed, and that’s not a gamble anyone should make. Laboratory coats and chemical-resistant gloves should form your uniform. Nitrile gloves resist chemicals well and I always opt for a full forearm cover rather than just palm protection. Skin exposure to this chemical burns and itches hours after the fact. All it takes is one careless movement to regret skipping protection.
Ventilation Keeps Things Breathing
Many small labs trust fume hoods, and for good reason. I’ve worked in both cramped and well-planned labs, and areas with actively moving air lower the risk of inhaling fumes. Leaving doors open or relying on a window isn’t enough; only true ventilation really moves the vapors out. In some places, we use local exhaust ventilation as a backup. Once, someone tried working in a closed room, and in minutes, the air got sharp and hard to breathe. A fume hood keeps those risks far lower.
Fire Risks and the Right Storage
Trimethylethoxysilane catches fire more easily than many solvents. Static spark or simple friction from containers can start a fire. Solvent-resistant storage cabinets make all the difference here. Keeping the bottles away from direct light, heat sources, and oxidizers cuts down risks. In my time working with it, I’ve always kept the original container tightly shut. Decanting or transferring should happen slowly and only under controlled conditions to avoid fumes and spills.
Spill Response and Clean Up—Preparedness Pays
Spills create panic if you’re not ready. From personal experience, proper containment kits often make the difference between a minor hiccup and a real hazard. Absorbent pads, soda ash, or sand soak up small spills before they spread. Wiping up with paper towels spreads the chemical and makes the situation worse. Once, I watched a minor splash on a benchtop turn into a long cleanup because the right absorbents were missing. Chemical waste disposal needs sealed, labeled containers. That step cuts down on confusion during inspections or emergencies.
Respect Training and Clear Procedures
No one becomes safe through written protocols alone—practice matters. I stress hands-on training with new team members every time. People often forget a step, like checking a glove for holes or tying back hair near the work area. Refresher demonstrations catch carelessness before small mistakes grow into something irreparable.
Health Monitoring and Responsible Practices
Anyone working with this chemical should have access to medical checkups. Chronic headaches, skin redness, or respiratory issues can signal overexposure. From what I’ve seen, incidents drop fast when teams treat symptoms and near-misses seriously. Experienced mentors watching for lazy shortcuts help more than any poster or manual. A strong safety culture means everyone watches out for each other and doesn’t skip the basics, even during busy deadlines.
Conclusion
Working with Trimethylethoxysilane calls for dedicated attention to detail, real-life experience, and open communication between everyone in the room. It’s not about avoiding blame but preventing harm, using the tools and knowledge each team member builds over time. Respect for the chemical and each other keeps labs safer for everyone who steps inside.
What is the chemical formula and structure of Trimethylethoxysilane?
Getting to Know Trimethylethoxysilane
Every now and then, a compound pops up in chemistry labs and on factory floors for how easily it changes the character of a surface or catalyzes a high-stakes reaction. Trimethylethoxysilane fits this bill, thanks to its distinct silicon-based makeup and reactive functional groups. This colorless liquid makes coatings more water-repellent, helps prevent unwanted sticking in adhesives, and even streamlines life in the electronics world. Still, the first step to working with any chemical is understanding its DNA—both formula and structure.
The Chemical Formula
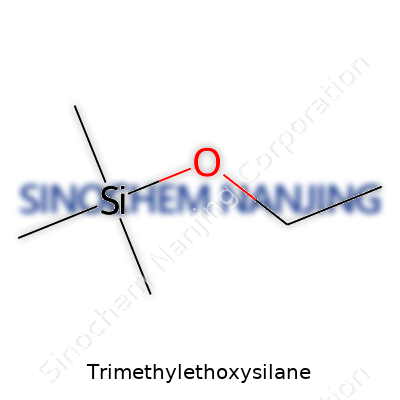
Trimethylethoxysilane carries the formula C5H14OSi. This may look like alphabet soup at first, but each letter speaks to its blend of silicon, carbon, hydrogen, and oxygen. Each molecule holds one silicon atom at its core. Three methyl groups (–CH3) and one ethoxy group (–OCH2CH3) attach to this silicon. That arrangement gives the substance its unique properties, setting it apart from other silanes.
Chemical Structure and Why It Matters
Take a closer look at the skeleton: three methyl groups cluster around the silicon atom, with one ethoxy group branching away. If you were to draw it, you’d spot the silicon sitting center stage, juggling three –CH3 groups and one –OCH2CH3 group. The chemical shorthand for the molecule is (CH3)3SiOC2H5.
This structure fuels the way it interacts with water, glass, and other materials. Three methyl groups act as a shield, fending off moisture and building slick, non-stick surfaces. The ethoxy group brings a touch of flexibility—breaking off under the right conditions, it can leave silicon behind in just the right spot. On glass, that means forming tough, water-hating layers. On electronic components, it adds stability and safeguards sensitive bits from corrosion.
Safety, Handling, and Industry Use
Trimethylethoxysilane often shows up in labs where researchers chase better coatings for solar panels, or in industrial plants pumping out advanced adhesives and sealants. Of course, handling any organosilicon agent calls for respect and care—this one releases flammable vapors, and contact with moisture starts a chain of reactions best kept in check with good ventilation and the right gloves. Storing it away from water and open flames keeps risks down.
Silicone chemistry opens doors across industries, but safety keeps those doors open for the next team through. Simple habits—good labeling, sealed containers, and swift cleanup for any spills—mean everyone stays healthy in the push to discover what these molecules can do. And the more surface science leans on these flexible, water-resistant coatings, the more people need to know both the opportunities and the pitfalls.
Looking Forward: Making the Most of Trimethylethoxysilane
As cleaner manufacturing gains ground and new electronic devices hit the market, demand for surface-active agents only grows. Companies look to silanes to build smarter, greener materials. Improvements in waste management, fume extraction, and less hazardous alternatives help push the field in a safer direction. Regulation and continuing research each play a part—making sure benefits flow without sidelining worker safety or the environment. Chemistry never stands still, and neither do the industries using these tools. Understanding both the formula and the way the molecule fits together unlocks new chances to solve old problems, with more reliable data and sharper innovation as the end goal.
How should Trimethylethoxysilane be stored?
Understanding the Risks
A lot of people working with chemicals get comfortable around them after a while. Over time, it’s easy to start thinking, “Nothing’s ever blown up on my shift.” Then someone misses a step, and the near-miss stories surface in the break room. Trimethylethoxysilane may not sound as troubling as hydrofluoric acid or liquid ammonia, but it brings plenty of risk to a workplace that doesn’t give it respect. Direct contact can burn, inhaling fumes hurts lungs, and letting it mix with water or air sets off a domino line of problems, ending in fires or worse. OSHA’s chemical database includes this substance for a reason.
Common Sense and Practical Steps
Often, safe storage for any volatile organosilicon compound comes down to a handful of simple habits. People who have handled bottles of Trimethylethoxysilane know leaked fumes aren’t just unpleasant—they create a real emergency. So, keep containers tightly sealed. Even new staff on their first day can understand why a broken lid spells trouble. In my years spent in labs and small-scale production spaces, I’ve learned never to trust old glass bottles or rusty metal drums. You want certified containers; nothing bought second-hand off surplus sites or found in a forgotten corner.
Temperature, Light, and Air—Not Just Checkboxes
Put bluntly, Trimethylethoxysilane hates extreme heat, sunlight, and moisture. Leave it on a sunny shelf, and don’t be surprised by violent reactions later. Store it cool, but not freezing—think standard chemical storage, around 15°C to 25°C. Somebody I know once stashed it under a vent to “keep it cool,” then spent the afternoon fighting a chemical spill. Sunlight keeps breaking down the stuff, and over months, even a little exposure shortens shelf life.
Keep this substance away from any sources of water. One careless splash, or even humid air, reacts with Trimethylethoxysilane to release flammable gases. More than one fire started from something as simple as an air conditioning leak dripping where it shouldn’t. It doesn’t take a PhD to know that flammable means flames. At a minimum, aim for a dry, dedicated storage cabinet labeled with clear hazard warnings.
People and Process Matter More Than Policies
Training trumps anything written on a wall poster. Every person near the storage area needs to know what to do if things go wrong. In larger companies, yearly refresher sessions work. For small teams, I’ve found informal weekly toolbox talks drive the point home. Even a quick five-minute run-through of “what not to touch” and “who to call” keeps complacency away.
Routine Counts (And Saves a Lot of Headaches)
Don’t trust memory or appearances—keep an up-to-date inventory and perform weekly checks. Spotting corrosion, warped caps, or even dust means it’s time to address issues, not shuffle them aside. Assign a specific person to these walkthroughs. Letting everyone do it often means nobody does.
Solutions for Reducing Mistakes
Upgrade old cabinets and buy new spill kits before accidents push the issue. Good labels and accurate logs help, but finding the right fire extinguisher nearby and training staff to use it matters more. Some facilities invest in alarm systems to flag leaks or rising temperatures. Keeping only the amount you need on hand reduces risk and waste. Lock up bulk supplies far from people, not just out of sight behind a thin door.
Treating Trimethylethoxysilane with this level of seriousness makes for a safer workplace, few surprises, and plenty of peace of mind at the end of a shift.
Is Trimethylethoxysilane compatible with other chemicals?
How This Silicon Compound Mixes With the Lab World
Trimethylethoxysilane has a spot on my shelf that I treat with respect. Seen in many coatings, sealants, and even in pharma, its role looks simple at first glance—the molecule shows up to protect or modify surfaces by leaving a thin silicon layer. The question many ask before using it in any real-world process: Is it likely to play along with other chemicals in the mix? The answer’s more complicated than a generic yes or no.
A Quick Dive Into What It Does
Take a moment with this compound, and you notice its trait: it’s a silyl ether. The ethoxy group wants to react with water and alcohols, which can spark concerns about unintended byproducts if you’re not careful. You get not just a surface treatment, but also ethanol as a byproduct, which isn’t always ideal. Think about any electronic device that needs a dry environment—moisture can spoil the end result.
I’ve worked with teams who tossed Trimethylethoxysilane into organic synthesis to hide certain reactive sites. The moment water crept in or trace acids hung around, side reactions flared up—leaving folks with less product than they hoped for and sometimes messy purification steps. Safety data shows its tendency to hydrolyze, so the advice gets simple: avoid excess water, strong acids, and bases unless you know exactly what outcome you’re chasing. Even atmospheric humidity has a way of sneaking into glassware and throwing a wrench into careful plans.
Minding Your Lab Neighbors
Lab work’s a team sport. Whether you’re at a university or in industry, one lesson sticks out: don’t guess when you mix chemicals, especially with things like Trimethylethoxysilane. The molecule partners up with non-polar solvents like hexane, toluene, or even dry dichloromethane. Most chlorinated solvents keep things calm if you’re running organic synthesis, though it can foul up instruments if run through without proper cleaning afterward.
Go down the list, and you run into nitrate salts, concentrated acids, or bases—Trimethylethoxysilane wants no part in those reactions unless tightly controlled. Databases like PubChem and PubMed document reports on exothermic reactions with strong oxidizers. Mix-ups can mean heat, pressure, or even glassware failure. The stakes get even higher when scaling up from milligrams to kilograms, as I’ve seen during pilot production runs where improper cleanup led to damage worth thousands of dollars.
Why Compatibility Checks Matter
It’s not just about wasted product or short-term risks. Regulations demand proper hazard evaluations. Manufacturers and workers have suffered unwanted exposures simply because someone assumed a silyl ether would "probably" not react with another factory chemical. Global data from agencies like OSHA and the ECHA back up those stories—thorough compatibility checks have prevented needless harm, lost batches, and legal headaches.
What Works in Practice
Dry, inert gas conditions—nitrogen or argon—reduce most surprises. Teams I’ve worked with rely on split samples and gloveboxes to keep things reliable. Clear labels and material safety data sheets are non-negotiable on any bench or production floor. There’s also wisdom in regular reviews of protocols since new literature comes out every month describing new use-cases or failures in compatibility with common catalysts or process additives.
If you want to avoid wasted effort and bad surprises, respect the boundaries of each chemical. Know your solvent system, avoid water and reactive reagents unless the reaction scheme says so, and check the latest safety reports. Open communication makes a bigger impact than any single technical fix. Skip the shortcuts, and reliability follows.
| Names | |
| Preferred IUPAC name | trimethylethoxysilane |
| Other names |
Ethyldimethylsiloxytrimethylsilane Ethoxytrimethylsilane Trimethylsilylethoxide Trimethylethoxysilane TMES |
| Pronunciation | /traɪˌmɛθ.ɪl.ɪˌθɒk.siˈsaɪ.leɪn/ |
| Identifiers | |
| CAS Number | [7693-27-8] |
| Beilstein Reference | 3858732 |
| ChEBI | CHEBI:85398 |
| ChEMBL | CHEMBL506964 |
| ChemSpider | 62289 |
| DrugBank | DB11273 |
| ECHA InfoCard | 03-2119928497-34-0000 |
| EC Number | 213-685-5 |
| Gmelin Reference | 84804 |
| KEGG | C19693 |
| MeSH | D013994 |
| PubChem CID | 11436 |
| RTECS number | VV7325000 |
| UNII | F7IYTIW6YN |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C5H14OSi |
| Molar mass | 120.24 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Odorless |
| Density | 0.745 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 0.8 |
| Vapor pressure | 13.3 kPa (20 °C) |
| Acidity (pKa) | 12.7 |
| Basicity (pKb) | Trimethylethoxysilane has a pKb of approximately 25 (extremely weak base) |
| Magnetic susceptibility (χ) | -62.7 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.372 |
| Viscosity | 0.38 mPa·s (25 °C) |
| Dipole moment | 0.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 241.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -322.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1868.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H319 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P370+P378 |
| NFPA 704 (fire diamond) | 1-4-1 |
| Flash point | 17 °C |
| Autoignition temperature | 287 °C |
| Explosive limits | LEL: 1.5%, UEL: 13.5% |
| Lethal dose or concentration | LD50 Oral Rat 7400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 7600 mg/kg |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | rel: 2-8°C |
| IDLH (Immediate danger) | IDLH: 1500 ppm |
| Related compounds | |
| Related compounds |
Trimethylchlorosilane Trimethylsilyl ether Triethoxysilane |

