The Working Chemistry and Growing Relevance of Trimethylcyclohexylamine
Tracing the Roots: Historical Development
Trimethylcyclohexylamine didn't show up in the chemical world by accident. This compound got its foothold thanks to the hunger for better solvents, corrosion inhibitors, and specialty intermediates throughout the booming industrial expansion of the twentieth century. Researchers rolled up their sleeves and tinkered with cycloaliphatic amines because the drive to get more effective or less hazardous options never stops. Trimethylcyclohexylamine, with its unique mix of basicity and molecular bulk, began appearing in patents mid-century—catching the attention of those hoping to solve practical problems in coatings, fuels, and even perfume manufacturing. As the years rolled on, the compound’s place in research journals kept growing, reflecting a pattern I’ve often seen where incremental, tested advances outpace flashy new inventions in shaping entire sectors.
Product Overview with a Firm Grip on Reality
Trimethylcyclohexylamine isn’t some mystery ingredient—its value stands in its unusual balance of volatility, reactivity, and compatibility with a variety of organic and aqueous systems. If you open a bottle, you’ll notice the strong amine odor straight away. This compound pops up where chemical engineers and lab techs look for an amine base that resists breaking down in rough conditions and handles both organic acids and some fine-tuned metal catalysts. From giving resilience to rubber to making epoxy resins easier to work with, the product has managed to carve out a practical niche as industries edge toward more specialized solutions. To my mind, the hands-on requirements for these applications taught many to respect just how many quirks a single amine can have compared to its cousins.
Diving Deeper: Physical & Chemical Properties
Visualize a colorless or faintly yellow liquid that stays stable under ordinary lab conditions, yet carries enough volatility to demand proper storage. Melting and boiling points fall in the range of other cycloaliphatic amines, but what makes this chemical stand out comes from its strong nucleophilicity paired with relative hydrophobicity. The molecule’s three methyl groups crowd around, giving bulk and shifting electron density to the nitrogen atom. That little physical shift changes everything—from how it dissolves in common solvents to the stubborn way it refuses to participate in hydrogen bonding the same way simple amines do. In my lab days, the first time I measured its density and watched its slow reaction with water, I realized you really can’t treat all amines as interchangeable workhorses.
Technical Specifications & Labelling: Reading Between the Lines
Chemicals often come with a tangle of technical terms and certification badges. Trimethylcyclohexylamine brings labels like CAS 6763-92-6 or EINECS 229-741-8, along with clear guidelines for purity—usually upwards of 98% for specialty work. Handling guidelines focus on keeping this base away from strong oxidizers, acids, and heat. The paperwork might feel redundant at first glance, but anyone who’s managed a chemical stockroom knows how strict documentation reduces headaches. Specification sheets for this amine usually stress flash point, compatibility with metals, and even color standards, since yellowing can hint at breakdown or contamination. None of these details should end up ignored, especially since minor variables in bulk shipments can create wild swings in downstream results.
How It’s Made: A Chemist's Roadmap
Manufacturers synthesize trimethylcyclohexylamine mostly through alkylation routes. Starting with cyclohexylamine, they introduce methyl groups by reacting with methyl halides or similar reagents under basic or catalytic conditions. The challenge involves pushing methylation to the right degree without spewing out a mess of higher or lower alkylated byproducts. I've seen more than a few scrubbers and fractional distillation setups heat up while operators troubleshoot how to separate the desired trimethyl product from dimethyl or tetramethyl variants. There’s a certain methodical pride in guiding the synthesis with the right ratio of reactants, strict timing, and temperature control—the kind of hands-on work where mistakes quickly teach lessons regarding yield and purity.
Chemical Reactions & Modifications: Making a Versatile Tool
In practical settings, trimethylcyclohexylamine gets used for what it brings to the table in reaction kinetics and selectivity. You can spot it acting as a catalyst or neutralizing agent in polymer manufacturing or as part of tailor-made functional group transformations on active pharmaceutical ingredients. Its basicity—strong but not nearly as aggressive as small, unhindered alkylamines—often proves crucial. Try combining it in epoxy crosslinking or trapping acids during organic synthesis, and the results outshine many cheaper alternatives. Chemists appreciate that tweaks to its methylation pattern lead to changes in volatility or water solubility, letting them dial in formulation characteristics with unusual control. This flexibility stands out whenever I meet formulation scientists eager to beat industry benchmarks for product lifespan or resistance to breakdown.
Synonyms & Product Names: Keeping Track
In scientific papers and catalogs, trimethylcyclohexylamine picks up a host of aliases—N,N,dimethyl-1-cyclohexanamine and 1-cyclohexyl-2,2,6,6-tetramethylamine among the more technical, with others pointing to specific isomers or trade names based on producer nuances. This mix of synonyms says a lot about how widely the compound gets studied and deployed. If you’re browsing literature, keeping tabs on these names helps you avoid missing key research or mixing up similar-sounding compounds, a point hammered home by my own hours sifting chemical abstracts to confirm a reaction step in the middle of a deadline crunch.
Safety & Operational Standards: Where Lab Reality Meets Regulatory Pressure
Safety for this compound isn’t just common sense—it draws on a catalog of established practices and a clear signal from researchers who’ve seen the effects firsthand. Exposures mostly happen through inhalation or skin contact, since the amine’s volatility brings it airborne in open containers or heated baths. Guidelines from agencies like OSHA and ECHA require gloves, goggles, and effective ventilation—no shortcuts. Storage advice covers everything from spill containment to labeling, emphasizing that even moderate inhalation can cause headaches or respiratory irritation. Back in shared academic facilities, we’d double-check that fume hoods weren’t overloaded and that spill kits got restocked—small steps that prevent most panics. Company safety culture deepens fast when even minor exposures translate into discomfort or lost lab time.
Application Areas: Where It Makes a Difference
Industries make use of trimethylcyclohexylamine for jobs that need reliability over flash. Corrosion inhibition in refinery systems and pipelines gets a boost since this compound sticks to metal surfaces, forming layers that shield against acids and humidity. Resins and epoxies toughen up considerably when it enters the mix, especially in adhesives built for weathering and heavy loads. Labs tap it for pH adjustment or as a base during complex syntheses where milder alternatives fall short. It even pops up in fragrance ingredient lists—where its basic nature can stabilize volatile aromatics in a finished blend. The best perks show through when end-use products keep their edge long after others degrade, an outcome every manufacturer wants but few achieve without disciplined material selection.
Research & Development: Pushing Boundaries
New research involving trimethylcyclohexylamine rarely follows a straight line. Teams chase fresh uses—sometimes in green chemistry, sometimes in emerging battery technologies, or as part of safer fuel additives. Patents now focus on advanced surfactant systems and recyclable resins, where this compound’s special properties can drive down product failures or extend lifespan. At scientific meetups, I hear how chemists use it as a probe to study fundamental properties of amines, water solubility, and reactivity with next-generation catalysts. Long-term studies try to unlock eco-friendlier synthesis methods, while others test modifications that reduce volatility or lower toxicity without losing the amine’s chemical backbone. Skepticism and curiosity walk hand in hand here, since the limits on what this chemical can deliver depend on pushing hard at those intersection points between lab trial and full-scale production.
Toxicity Research: No Room for Complacency
Toxicological studies on trimethylcyclohexylamine underscore the need for robust risk management. Short-term symptoms—eye and throat irritation, headaches, or skin redness—stack up quickly, especially in confined workspaces. Chronic exposure research stays limited but concerning enough to prompt extra caution. Regulators and in-house safety committees often debate over exposure thresholds and cleanup measures because every chemical tells a story about unintended consequences. Case studies report on animals and in vitro systems, tracking metabolic pathways to understand where and how the compound might break down or bioaccumulate. The consensus leans toward always working with ventilation, full personal protection, and well-maintained first-aid equipment. These routines build habits and confidence, but frontline lessons matter most—spill response drills, clear labeling, and easy-to-read incident logs make the difference between routine operations and a day that slides off the rails.
Future Prospects: Building Better Solutions
Looking ahead, trimethylcyclohexylamine stands at a crossroads common to many time-tested industrial chemicals. There’s strong pressure to engineer derivatives with friendlier environmental footprints, lower volatility, or built-in safety features. Researchers look for greener routes using biocatalysis or renewable feedstocks—steps shaped not just by regulatory trends but by hands-on results that cut waste, energy use, and post-use contamination. The growing push for circular manufacturing stirs demand for compounds that don’t just perform but can be broken down or recycled after a product’s end of life. In my view, every time industry leans into chemicals with this much versatility, it helps drive a steady shift from commodity use toward high-value applications where accountability, transparency, and sustainable design carry equal weight with performance. That’s how progress in chemical manufacturing often unfolds: one thoughtful synthetic tweak at a time, fueled by live data and lived experience.
What is Trimethylcyclohexylamine used for?
What Is Trimethylcyclohexylamine Used For?
Trimethylcyclohexylamine, a name that sounds like a bit too much for a coffee shop menu, pops up in places most folks never see. If you’ve ever walked through a chemical plant, or you know your way around making plastics, you have brushed up against this compound’s world. It functions mainly as a catalyst and intermediate, but those words hide a much busier life.
Big Role in Polyurethane Foam
Sitting on a cheap office chair or a car seat, you are probably propped up by polyurethane foam. Chemists love using Trimethylcyclohexylamine to get those foams just right. The stuff helps spark the chemical reaction that turns liquid into that squishy solid. More reactivity means faster and more predictable foam production. You can actually thank this chemical for cheaper furniture, lighter car panels, and even insulating foam in your walls.
Pulling Double Duty in Corrosion Inhibition
Protecting pipelines or machines from rust isn’t flashy, but it matters in the long haul. Trimethylcyclohexylamine makes an appearance in corrosion inhibitors, especially in closed water circuits. This keeps the costly metal machinery running longer and cuts down on waste, repairs, and even energy loss from leaks. Anyone who’s fixed a hot water heating system or seen the mess of corroded radiators knows the headache avoided here.
It Doesn’t Stop There: A Chameleon in Industry
Textile businesses look for stability and lasting color in their dyes. Trimethylcyclohexylamine supports this by helping stabilize dye formulations. No one wants to fork over cash for a pair of jeans only to watch them fade after two washes. In pharmaceuticals, this amine sometimes works as a building block for complex molecules. This side rarely gets headlines, but it plays a supporting role that keeps other processes moving smoothly.
Handling the Hazards
Like plenty of amines, this one packs a strong smell and strong behavior with your eyes, skin, and lungs. Safety protocols must match the risk. I’ve seen workers wrap up tightly with gloves, goggles, and respirators around this stuff, washing off splashes right away. It reminds me of working with industrial cleaners—tools that save time, but punish anyone careless enough to skip the safety rules. Community right-to-know laws and OSHA rules offer a basic safety net, but ongoing training and transparency matter most of all.
Finding a Middle Ground
Chemicals like trimethylcyclohexylamine push the limits for manufacturers chasing lower costs, energy savings, and consumer convenience. Stricter workplace controls, environmental testing, and updated safety data keep everyone on their toes, from engineers to first responders. It’s easy to take these benefits for granted, but worker education and strong local health standards are the best bets to balance progress and protection.
Expect new forms and greener alternatives to show up, especially as more pressure lands on companies to reduce environmental hazards. Until the next generation of catalysts or corrosion blockers emerge, knowing what’s in the mix and how to deal with it makes all the difference. Experience on the ground always beats theory in the office, especially with the tough, tricky chemistry that powers modern life.
What are the safety precautions when handling Trimethylcyclohexylamine?
Why Safety Matters with This Chemical
Trimethylcyclohexylamine doesn’t pop up in everyday conversation, but it shows up in specialized industries as a catalyst, corrosion inhibitor, and intermediate for other chemicals. It comes with a chemical scent that sticks to your hands, eyes, and lungs if you’re not careful. It’s used in labs, manufacturing, and sometimes in research, so folks who handle it every day deserve clear guidance. I’ve worked around labs and industrial spaces, and the people who skip on safety gear only do so once—long-term health isn’t worth a shortcut.
Gloves, Goggles, and More: The Basics
Direct contact with skin or eyes doesn’t end well. Trimethylcyclohexylamine irritates and can burn, so a set of tight-fitting gloves remains standard. Nitrile gloves stand up well to amines and keep the liquid out. Splash-proof goggles matter even more, because eye exposure delivers sharp pain and leaves lasting damage—there aren’t do-overs when it comes to eyes. A chemical-resistant coat or apron keeps liquid from soaking into your clothes and skin. These might look like overkill on a good day, but the first splash teaches why labs stock up on them.
Ventilation Saves Lungs
Toxic vapors knock workers off balance. I remember walking into a space where chemical smells stuck to the air and gave me a headache before an hour passed. With volatile organics like this, working under a fume hood changes everything. It keeps vapors from building up and drifting into your lungs. Proper local exhaust means every breath stays cleaner and you don’t carry chemical headaches out of the lab. Facilities that skip good ventilation put staff at risk for chronic breathing problems, so reliable fume extraction always ranks as a must.
Spill Response and Cleanup
Nobody plans for a spill, but every experienced worker expects one someday. Absorbent pads and neutralizers should stand ready, along with chemical waste bags. Once, after a spill, quick action with pads and ventilation let us clear the space and limit staff exposure in minutes. Never grab standard paper towels since they can react with certain chemicals. Any material that soaks up Trimethylcyclohexylamine goes straight into labeled hazardous waste containers.
Storage and Labeling
Keep this chemical away from heat, oxidizers, and acids. A flammable storage cabinet, away from sunlight, keeps both the product and the people safe. Labels must stay readable; what you can’t read, you don’t trust. Secure storage means kids, unauthorized folks, or unprepared staff don’t stumble on dangerous surprises. At one job, a missing cap and sloppy shelf stacking led to fumes and an evacuation—nobody wants that chaos.
Training and Emergency Planning
Every facility handling Trimethylcyclohexylamine should run training refreshers. Workers need to know the symptoms of exposure—eye redness, skin irritation, breathing trouble—and how to respond right away. Quick access to emergency showers and eyewash stations makes a tough day survivable. Phone numbers for poison control and emergency response should hang in plain sight. With the right knowledge, staff can act fast and avoid panicked mistakes.
Toward Better Protection
Building a habit of proper safety gear, smart storage, and active ventilation lowers the risks tied to Trimethylcyclohexylamine. A strong culture of ongoing training keeps everyone sharp. Sharing clear methods for spill control and emergency care makes workplaces healthier and holds up against the unexpected.
Is Trimethylcyclohexylamine hazardous to health or the environment?
Understanding Trimethylcyclohexylamine
Walking through a chemical plant or reading an ingredient deck, you might spot the name trimethylcyclohexylamine. Most folks don’t keep it on hand in the garage but it turns up in industrial settings. This colorless-to-slightly-yellow liquid gets used in things like epoxy hardeners, corrosion inhibitors, and some types of lubricants. I’ve worked around industrial chemicals before, and names like this always prompt me to wonder about human and environmental risks.
What Workers Should Know
Chemicals such as trimethylcyclohexylamine deserve some healthy respect. Inhaling the vapor or getting it on the skin can cause irritation. Eyes and mucous membranes tend to react fast. NIOSH reports show that exposure, even at lower levels, leads to headaches, dizziness, and sometimes a burning feeling in the throat and nose. Over time, skin issues crop up, especially for folks who touch it regularly during manufacturing shifts.
Given these symptoms, wearing gloves, goggles, and proper respiratory protection isn’t up for debate. Workplaces that stay on top of training and PPE cut down on these risks. Having walked through operations that ignored safety basics, I’ve seen too many avoidable accidents. Companies that build solid ventilation and offer regular check-ins on equipment do much better at keeping their people healthy.
Long-Term Health Concerns
Longer-term effects create more concern. Research points to potential liver and kidney stress from heavy or repeated exposure, though there’s no full agreement on cancer links. The American Conference of Governmental Industrial Hygienists keeps conservative limits for airborne levels. Most health agencies want more long-term studies, but with what we already know, exposure should be kept low—especially for anyone with asthma or other respiratory weaknesses.
Environmental Impact
Trimethylcyclohexylamine doesn’t mix well with nature. When it escapes into streams or groundwater, it doesn’t break down quickly. Aquatic life takes a hit from the toxicity. Fish and small creatures in rivers get exposed to higher doses, leading to trouble thriving or reproducing. I’ve seen manufacturing runoff devastate waterways, and recovering those ecosystems takes years.
Airborne releases mean evaporation carries these particles into the atmosphere. From there, rain brings it down far from the original spill. Sometimes the chemical seeps into soils, where it sticks around far longer than you’d hope. We know that cleanup gets more expensive the longer a leak goes unchecked. States that monitor discharge and insist on proper treatment facilities prevent a lot of this damage before it starts.
Practical Solutions
There’s no need for panic—but there’s every reason for vigilance. At the plant, managers should install spill barriers and airtight containment areas. Regular inspections pick up leaks before they spread. Remote sensors make a big difference catching problems after hours.
Communities near factories should push for transparent reporting. Public access to information lets people know what’s getting released into their air and water. The EPA’s Toxics Release Inventory database allows anyone to check on chemicals stored or used upstream. Combining government oversight with public awareness leads to tougher standards and better health outcomes.
It helps when companies switch to safer alternatives during process upgrades. Some industries look for amines that break down easier or cause fewer health issues. Investment in green chemistry pays off in the long-run—both for workers’ health and for keeping local rivers safe for fishing, swimming, and wildlife.
How should Trimethylcyclohexylamine be stored?
Understanding the Risks
Trimethylcyclohexylamine will cause more trouble than most folks realize if you leave it in the wrong spot. This chemical gives off strong fumes, irritates skin and eyes, and reacts badly with moisture or acids. I remember working in a lab as a junior staffer, thinking “how bad could a small leak be?” until my eyes were burning and my supervisor barked at me to check the ventilation. Looking back, I know how important it is to respect the stuff you’re handling.
Keep It Cool and Dry
Heat and humidity get chemicals like this riled up. Warm storerooms usually feel fine for paperwork, but for reactive substances, temperature control becomes your best friend. Research from the National Institutes of Health shows that amines at high temperatures degrade faster, releasing more vapors and kicking off unwanted reactions.
I’ve found that a simple AC unit and proper insulation prevent most mishaps. Don’t forget about moisture—Trimethylcyclohexylamine grabs water from the air and starts to break down, releasing vapors that corrode metal or mess up electronics around it. Sealed containers matched with clear labeling keep slip-ups to a minimum.
Keeps Away From Acids and Oxidizers
Trimethylcyclohexylamine’s biggest enemies? Acids and strong oxidizers. If someone stores it near sulfuric acid or bleach, the reaction can turn dangerous fast. I’ve witnessed shelves packed tight, with everything side by side, to “save space.” News flash: you save a lot more headaches by leaving room for separation. Chemical incompatibility charts hang on my storage room wall for a reason—the reminders keep mistakes rare.
Solid Labels and Secure Containers
Clear labeling works wonders. Even workers new to a site can spot hazards if you mark them plain and bold. Many incidents trace back to “white bottle confusion”—everyone assuming they know what’s inside until it surprises them. We use sturdy, chemical-resistant containers with lids that seal tightly—never mess around with makeshift storage options.
Proper Ventilation Matters
If you smell a sharp, ammonia-like odor, ventilation likely needs work. Inadequate airflow builds up vapors, which stress everyone’s lungs and can spark fires if concentrations run high. I’ve watched staff try to push fans into aging closets. A dedicated vent system, designed for chemical storage, saves a world of hassle. The CDC recommends six air changes per hour for chemical storerooms, and that seems to keep most issues at bay.
Training and Emergency Planning
No storage rule shines without steady training. People forget what they learn, especially when turnover runs high. Every facility benefits from basic refreshers: which chemicals don’t mix, where to find spill kits, how to handle leaks. During a spill drill last year, several team members didn’t know which gloves worked best for amines—the exercise exposed a weak spot in our process, so we circled back and fine-tuned the steps.
Spill kits, splash goggles, and emergency showers belong close to where Trimethylcyclohexylamine lives. The best policies make safety materials as accessible as the chemicals themselves. By keeping attention on these basics, every lab or warehouse limits risk and guards its people.
What is the chemical structure and molecular formula of Trimethylcyclohexylamine?
Understanding the Basics
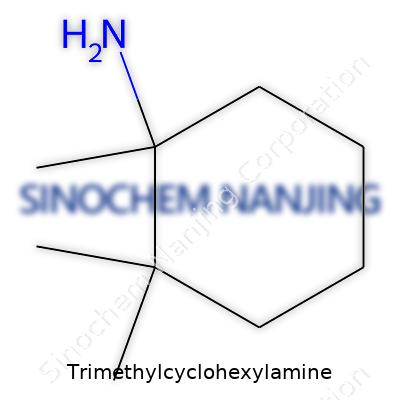
Trimethylcyclohexylamine belongs to a group of chemicals known for both their versatility and specific industrial applications. Its backbone comes from cyclohexylamine, which features a six-carbon ring—cyclohexane—with an amine group (-NH2) attached. With the addition of three methyl groups (-CH3), you get the compound that chemists call Trimethylcyclohexylamine. The name alone might intimidate those not familiar with organic chemistry, but its structure actually draws from common building blocks found in many synthetic compounds and products.
The Chemistry Behind the Name
Looking at its molecular formula, Trimethylcyclohexylamine is written as C9H19N. Here, nine carbon atoms build the bulk of the structure. Nineteen hydrogens pack around these carbons, leaving a single nitrogen atom sticking out in the molecule’s amine group. This makes the compound part of the aliphatic amine family, which means it doesn’t have aromatic rings like benzene. Instead, the ring shape is pretty flexible and can take different conformations.
If we picture the actual chemical structure, the cyclohexane ring forms the core. Chemists usually use chair or boat drawings to show all six carbons arranged in a ring. Three methyl groups attach at various positions on the ring, and an amine group attaches to one of the carbons. Methyl groups increase the bulk and change the way the molecule interacts with other chemicals. They can also affect the compound’s boiling point and solubility, details that matter in production and use.
Why Structure Matters in Practical Use
The chemical structure controls how Trimethylcyclohexylamine behaves. From my time in the chemical industry, I saw how minor tweaks—like adding those methyl groups—shift chemical reactions. The extra methyl groups on the ring significantly reduce the compound’s reactivity compared to plain cyclohexylamine. This property makes it far less likely to participate in side reactions, carving out a unique role. For example, in the rubber and plastics industry, compounds like this can provide durability to products without contributing unpleasant odors or causing chemical instability.
This compound is also considered where resistance to corrosion is vital, like in lubricants, solvents, or even specialty cleaners. The way it repels water stems from both the ring and its methyl groups. Fewer hydrogen bonds form with water, so it doesn’t mix well. This property fits right in with oil-based environments, where water resistance isn’t optional—it’s a must-have.
Finding Safer and Sustainable Options
Concerns about safety and the environment lead researchers to rethink the use of amine compounds in the lab. Inhalation or skin exposure to aliphatic amines sometimes triggers allergic reactions, headaches, or even more serious effects. In my own work, I’ve always leaned on good lab practices: strong ventilation, gloves, and limiting direct exposure. Companies now explore greener chemistry, searching for renewable feedstocks or tweaking existing structures to reduce toxicity without losing performance. Some are experimenting with bio-based amines or using processes that target waste reduction and lower emissions.
Real progress often happens by sharing knowledge and keeping up with safety data from trusted sources like the EPA or international health agencies. As industries keep pushing for cleaner and safer alternatives, staying aware of the relationship between molecular structure and real-world impact helps guide smarter decisions—not only in the lab but wherever chemicals play a role.
| Names | |
| Preferred IUPAC name | N,1,1-Trimethylcyclohexan-1-amine |
| Other names |
N,N-Dimethylcyclohexylamine N,N-Dimethylcyclohexanamine Dimethylcyclohexylamine Cycamine |
| Pronunciation | /traɪˌmɛθɪlˌsaɪkloʊˈhɛksaɪləˌmiːn/ |
| Identifiers | |
| CAS Number | 2222-01-5 |
| Beilstein Reference | 1698734 |
| ChEBI | CHEBI:197645 |
| ChEMBL | CHEMBL187869 |
| ChemSpider | 16223 |
| DrugBank | DB16661 |
| ECHA InfoCard | 100.124.439 |
| EC Number | 202-498-6 |
| Gmelin Reference | 9480 |
| KEGG | C11776 |
| MeSH | D017264 |
| PubChem CID | 85795 |
| RTECS number | BS8225000 |
| UNII | NQ2M001K34 |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID3022947 |
| Properties | |
| Chemical formula | C9H19N |
| Molar mass | 155.28 g/mol |
| Appearance | Colorless liquid |
| Odor | Ammonia-like |
| Density | 0.857 g/mL at 25 °C(lit.) |
| Solubility in water | slightly soluble |
| log P | 2.86 |
| Vapor pressure | 2.1 mmHg (at 25 °C) |
| Acidity (pKa) | 10.73 |
| Basicity (pKb) | pKb = 4.03 |
| Magnetic susceptibility (χ) | -7.74×10⁻⁶ |
| Refractive index (nD) | 1.448 |
| Viscosity | 1.14 mPa·s (20°C) |
| Dipole moment | 1.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 292.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -120.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4934.7 kJ/mol |
| Pharmacology | |
| ATC code | C01DX15 |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic in contact with skin, causes severe skin burns and eye damage, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H410 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P332+P313, P333+P313, P362+P364, P391, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 72°C |
| Autoignition temperature | 245 °C (473 °F; 518 K) |
| Explosive limits | 1 - 7.5% |
| Lethal dose or concentration | LD50 oral rat 300 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 542 mg/kg |
| NIOSH | RN8380000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Trimethylamine Cyclohexylamine Dimethylcyclohexylamine Methylcyclohexylamine |

