The Story and Science Behind Triisobutylaluminum: An Editorial Commentary
Historical Development
Back in the 1950s, industry pioneers started chasing faster and more controlled polymerization processes. Chemists poked at stubborn old methods and dug into the world of organoaluminum compounds, and out of that era came triisobutylaluminum. This substance changed the shape of the plastics industry. Nobody pretended it was easy to handle or cheap to make, but its stubborn reactivity made it valuable. Research labs got word of its use in Ziegler-Natta catalysts, letting manufacturers turn out polyethylene and polypropylene on a whole new scale. Its discovery didn’t spring from just luck — a heap of benches, hotplates, and glassware bore the marks of years spent exploring how aluminum and hydrocarbon chemistry could fit into everyday manufacturing. Looking back, the stubborn persistence of those early labs became the backbone for plenty of what modern materials science delivers today.
Product Overview
Triisobutylaluminum crops up wherever industry engineers hunt for precision in controlling polymer growth. It’s not the household name on a bottle under your sink, but it hums behind the curtain in the making of plastics and synthetic rubber. You won’t spot it on grocery shelves, but its fingerprints end up in cereal bags, car dashboards, and heaps more. Much of industrial chemistry depends on such compounds, and few match the impact of this one in the world of catalysis. Big polymer players pay close attention to it, not least because it saves time, energy, and raw material. That said, it isn’t forgiving—sloppiness in handling invites trouble, so the story of triisobutylaluminum is also one about discipline and respect for what chemistry can do.
Physical & Chemical Properties
Triisobutylaluminum rolls out as a colorless to pale yellow liquid. It’s got a bitter scent—maybe not surprising, given its hostility toward water and air. Plenty of folks in the lab have stories about seeing an accidental puff or spark. Water vapor sends this stuff into a frenzy, whirling out hydrogen gas and heat, so closed systems and inert atmospheres like nitrogen or argon stay close at hand. The liquid clings to the lower end of the density range, floating atop water if given a reckless chance. That aversion to oxygen and moisture isn’t just a lab curiosity. Proper safety gear and airtight equipment — gloves, goggles, and a sense of caution — must go on before breaking open a bottle. Take it lightly and the compound teaches hard lessons: fast reactions and a real risk of fire.
Technical Specifications & Labeling
Labels on drums of triisobutylaluminum leap out with hazard symbols. The UN numbers aren’t there for show; disaster follows if someone fumbles a shipment. The tech teams track molarity and concentration tightly because downstream reactions hinge on consistency. No room for a clerical slip — even minor contamination nudges a catalyst batch off target, sending millions in product down the drain. Proper technical documentation spells out everything: storage temperature, inert packaging, transport conditions, and shelf life. Each point draws on tough lessons learned from mistakes, fires, or near misses over decades. For anyone working with the substance, those data sheets might read as dry as toast, but in practice, they mean someone makes it home after a shift instead of to the ER.
Preparation Method
Folks often ask, "Where does this stuff come from?" The answer leans heavily on old-fashioned chemical synthesis, with isobutylene and aluminum alkyls doing the heavy lifting. Producers set up controlled reactors, adding isobutylene to aluminum hydride or dialkylaluminum hydride. No room for guesswork — too much heat or too much pressure turns an orderly process into a hazard. Generations of process chemists have tweaked flow rates, reaction times, and purification methods, hunting for purer output and less waste. In my own training, I watched operators calibrate gas flows with trembling knuckles, knowing a slip would mean trouble. That tangible sense of vigilance passes down from supervisor to apprentice, forming an unspoken rulebook that shapes the industry’s culture.
Chemical Reactions & Modifications
Triisobutylaluminum makes its mark in organometallic chemistry due to fast, exothermic reactions with oxygen, water, and many organic compounds. Its role as an alkylation reagent lets it transfer isobutyl groups to other molecules, often creating key intermediates. The core appeal: it’s aggressive, but in the right hands, controllable. Teams often tune its structure—trading ligands, adjusting the ratios, sometimes using it to modify surfaces or craft new catalyst formulations. Industry has not run out of ways to tweak its reactivity, especially when chasing patentable processes for polymer development. Decades of patent filings and published studies keep painting new possibilities for its uses. Modification, at its core, comes from tackling a practical question — “What will make today’s run more predictable and cost-efficient than yesterday’s?”
Synonyms & Product Names
In labs and across chemical plants, nobody loves saying "triisobutylaluminum" every time. You’ll hear folks tossing around TIBA, a nicknamed shorthand for quick communication over the roar of reactors. Trade names vary by producer, but the heart of the matter stays the same. Any confusion over names in the warehouse has risk written all over it — so everyone learns the full range of synonyms by heart, crosschecking containers before every step.
Safety & Operational Standards
Safety rules didn’t come out of nowhere; they stand on a grim history of accidents. This compound reacts violently with water or moist air, so the unwritten policy is: treat every encounter like handling dynamite. Operators run scrupulous checks to make sure lines, flanges, and storage tanks stay hermetically sealed. I’ve watched old hands test for leaks with the patience of a monk, stopping a job cold if something feels amiss. Emergency training drills pop up regularly — not for vanity, but because seconds count if things go sideways. Regulatory agencies like OSHA and the EPA watch the industry, but the best practices come from seasoned workers sharing stories of close calls and the real price of carelessness.
Application Area
The star role for triisobutylaluminum is as a co-catalyst in Ziegler-Natta systems cranking out vinyl-based plastics. Thanks to its hot reactivity, it lets companies dial in precise molecular weights and structures for their polymers. Beyond that, it finds work as a scavenger, snapping up trace moisture and impurities before they throw a wrench in sensitive chemical reactions. The reach stretches out into rocket fuel research, material coatings, and even fine organic synthesis. Engineers in these spaces trade war stories about the benefits of a pure, well-behaved batch. The cost of failing to control trace water or oxygen hits hard in the bottom line—missteps here burn through money as well as inventory. At the end of the chain, what matters most: reliable products with zero tolerance for flaws.
Research & Development
Innovation around triisobutylaluminum isn’t fading. R&D teams keep searching for safer handling systems, new catalyst blends, and techniques reducing waste. Green chemistry trends push researchers to trim byproducts and make production less hazardous, but no silver bullet has emerged. Newer labs experiment with alternative solvents, more robust containment designs, and even digital controls that shut down processes before trouble escalates. The field attracts sharp minds willing to blend traditional know-how with modern analytics. Each success broadens what downstream industries can achieve — better plastics, cleaner fuel additives, and fresh compounds for the electronics world.
Toxicity Research
Toxicologists approach triisobutylaluminum with caution. Animal studies and exposure assessments guide the rules about acceptable handling, and every result prompts new questions. Inhalation, skin contact, and accidental ingestion carry acute risks. Even fumes pose a threat if extraction hoods glitch or protective gear slips. The compound doesn’t belong anywhere near food prep, medicine, or any application without strict process safeguards. Long-term health studies have not fully closed the book on its risks, but the scientific consensus stays clear: full protection, with no shortcuts. Training refreshers and lab inspections become more than bureaucratic hoops — they’re part of the wider commitment to keep everyone healthy, from the janitor sweeping up powder to the scientist running titrations.
Future Prospects
Looking ahead, the fate of triisobutylaluminum ties closely to the march of advanced materials. Industries want tougher, lighter, greener plastics — and that means better control of catalyst chemistry. The push toward environmental responsibility drives both regulatory and consumer attention. Labs keep chasing substitution strategies, low-impact synthesis paths, and closed-loop systems that sidestep legacy risks. AI-powered predictive models promise the ability to forecast how every tweak changes safety profiles and material properties, and robotics help limit human exposure on the plant floor. Despite decades in service, this compound hasn’t run out of stories to tell, or problems to solve. The same spirit that pulled it out of early postwar labs fuels new discoveries and better safeguards, year after year.
What is Triisobutylaluminum used for?
Breaking Down the Role of Triisobutylaluminum
Triisobutylaluminum doesn’t show up in everyday conversation. It matters a lot to people working in chemical manufacturing, energy, and materials science. You walk through a typical pharmacy or supermarket and don’t see it on any label, but the benefits really start to show up further upstream—long before that laundry detergent bottle hits the shelf. Triisobutylaluminum’s claim to fame comes down to its behavior as an organoaluminum compound. What stands out for a lot of chemists: it reacts quickly, makes things happen faster, and starts chemical chains that would otherwise stall out.
The Backbone of Polymer Production
Polyethylene and polypropylene are everywhere, from food packaging to car parts. To create these plastics efficiently, factories rely on something called Ziegler-Natta catalysis. Triisobutylaluminum acts as a co-catalyst in this process. It helps kick polymers into gear, turning simple molecules into tough, flexible chains. Before these breakthroughs, folks depended on slow, expensive methods that produced lower-quality material. Today’s production wouldn’t keep up with global demand without this compound in the toolkit.
Crafting Pharmaceuticals and Fine Chemicals
If you’ve ever worked in a drug lab or pharmaceuticals plant, you get used to the complex steps behind medicines. Triisobutylaluminum often comes into play as a reducing agent. In simple terms, it helps add atoms to molecules in a predictable way. In drug chemistry, control means safety—one wrong step and you lose whole batches or risk impurities. Organometallic reactions using this reagent add building blocks cleanly. Sometimes, that makes the difference between a safe pain reliever and a rejected shipment.
Finding Use in Specialty Materials
People working with advanced electronics and coatings rely on high-purity materials. Triisobutylaluminum steps in as a precursor to alumina layers, which help protect circuits or reduce friction in moving parts. The compound provides an efficient starting point for making thin films through methods like chemical vapor deposition. This translates into better performing smartphones, energy-saving LEDs, and advanced medical devices.
Handling Comes with Real Challenges
Anyone who’s handled triisobutylaluminum remembers the strong reactions it has with both water and air. This chemical isn’t for amateurs. Water causes explosive fires, so strict safety procedures come into play. Working in industry for years, I’ve watched colleagues go through extensive training before they even open a drum. Special storage systems, dry glove boxes, and constant hazard checks end up becoming daily routines. The industry could use smarter packaging and simple sensors to make early detection easier and incidents less likely.
Looking Forward with Safer Processes and Alternatives
Some researchers hunt for less hazardous substitutes, but triisobutylaluminum’s performance keeps it in the lineup for now. Training and process control have made a huge difference in accident rates over the past decade. At the same time, better communication between chemical producers and users will boost transparency about risks and safety best practices. Seeing progress on automation tools feels encouraging—auto-injectors and smarter feed systems cut down the chance for leaks or splashes in busy plants.
The Real-World Takeaway
Though invisible to most people, triisobutylaluminum plays a steady, powerful role in manufacturing goods that shape modern life. Whether you think about medical breakthroughs, lighter vehicles, or longer-lasting electronics, the chemistry behind these advances can’t be taken for granted. Respect for safety, constant training, and new technology will decide how industry continues to rely on this material into the future.
What is the chemical formula of Triisobutylaluminum?
The Formula Unpacked
Triisobutylaluminum comes with the chemical formula C12H27Al. This compound features a central aluminum atom bound to three isobutyl groups. Picture it like a hub at the center of a wheel, with spokes shooting out. Each arm carries an isobutyl chain, which means three of them curl out from the metal. Not only does this give the compound its name, but it also defines how it reacts and behaves in all sorts of industrial situations.
Real-World Impact of a Formula
The structure written down by chemists isn’t just for showing off in textbooks. In my days researching industrial chemicals, I saw firsthand how a change in a formula can make or break a manufacturing process. Triisobutylaluminum’s formula gives it high reactivity. Failing to respect that causes fires, explosions, and costly mishaps. Most of the time, plant workers learn fast to fear and respect this clear, highly flammable liquid.
Industries dealing with plastics production—especially polyolefins—have relied on triisobutylaluminum since the mid-20th century, back when the Ziegler-Natta process changed how people viewed plastics. Folks who work on polyethylene and polypropylene probably recognize the connection: the aluminum compound lines up molecules in a way that can be controlled, tweaking strength, flexibility, and other important properties in the end product.
Getting the Formula Right
There’s no room for error mixing chemicals like C12H27Al into a reactor. Lab mistakes lead to real headaches, with wasted materials, lost production hours, and sometimes much worse. A friend working in a Gulf Coast plant told me that their safety protocol hinges on everyone knowing these details without second-guessing. Material mislabeling brought a close call during a scheduled polymer batch. All it took was someone not double-checking the compound used.
Chemical safety data sheets exist because ignoring the right formula sometimes leads to injury or worse. Proper ventilation, specialized handling gear, and careful storage keep everyone on site safe. Companies struggle with accident records, sometimes facing investigations and huge fines. In this environment, just knowing chemical shorthand keeps lives on course and reputations intact.
The Bigger Picture: Confidence and Accountability
People trust certain materials to do what they promise. Engineers and product designers need accurate info to scale up a pilot process to full production. Mistaking triisobutylaluminum for another similar-sounding compound can lead to runaway reactions or ruined product. Mistakes ripple through the supply chain, burning up trust quickly.
Sometimes, teams train together over spilled grains, splash guards, and fire suppression drills, all because one compound’s formula is the difference between safety and headlines about disaster. This isn’t abstract—every chemical plant manager I’ve interviewed knows the story of the “right” formula better than any textbook author does.
Moving Toward Better Practices
People in the chemical industry can cut down on most risks by sticking to clear, standardized training. Regular drills, tighter labeling systems, and automated checks for raw materials bring peace of mind to big and small operators alike. A formula like C12H27Al isn’t just a set of symbols; it carries weight with every fill-level check and every decision to open a drum. Only attention to these everyday details keeps industry wheels turning without costly or painful lessons.
How should Triisobutylaluminum be stored and handled?
A Chemical Demanding Respect
Triisobutylaluminum isn’t your average liquid. It reacts violently with water and can catch fire if exposed to air. People who work around this material often talk about it with a kind of wary respect. In a chemical plant or a research lab, even a small mistake can lead to a scary situation, so preparation matters.
Clear Rules Beat Shortcuts
One mistake people make is getting comfortable after a few safe transfers. This stuff can catch you off guard even if you’ve worked with flammable liquids your whole career. It makes sense to use storage tanks made of stainless steel or other compatible metals since containers need to seal tightly. Any leak, and you might end up with a fire hazard or worse. No one wants to relive the stories people tell about sudden flare-ups from a single drop hitting a damp glove.
Keep Water Out—Always
Moisture is the biggest enemy. Triisobutylaluminum reacts so fast that even humidity in the air can be dangerous. Transfer lines and storage equipment should stay bone-dry. Desiccant systems, nitrogen purging, and regular inspections help keep water away. From experience, the biggest trouble seems to come from ignoring the details—cracked gaskets, old seals, a loose bolt on a transfer hose. These small oversights bring real risk.
Trained Eyes on the Job
Training sticks with you the first time you watch a simulated spill video. Paying attention to your gear—like goggles, gloves, face shields, antistatic suits—feels a lot less annoying when you consider the burns this compound can inflict. Handling always involves two people. Fresh eyes catch mistakes that tired ones miss, especially late in a shift.
Why Ventilation Isn't Optional
Good ventilation means safer air. Some fumes can build up without much warning. Direct vent hoods, explosion-proof fans, and clear air pathways matter every single day. Labs and facilities without proper exhaust systems run the risk of dangerous concentrations or accidental ignition.
Routine Checks, Not Afterthoughts
No one likes paperwork, but logbooks and maintenance charts tell a story. Leaks start small. Building a habit of checking valves, seals, and containers every time pays off. Comparing notes with past inspections can catch a trend before it becomes a disaster.
How to React if Things Go Wrong
If there’s ever a spill, smothering with dry sand—not water—keeps things from escalating. Fire extinguishers rated for flammable metals and special spill kits belong close by. Quick response plans help everyone act fast without freezing up. Convening regular drills builds confidence and avoids panic.
Health Matters—Don’t Shrug Off Symptoms
A whiff of this compound can irritate the lungs. Any splash in the eyes or on the skin means an immediate trip to the eyewash station and then straight to the medical office. Ignoring symptoms isn’t tough-guy behavior—it’s gambling with your future health.
Learning From Every Incident
Every time someone shares a near-miss or documents a spill, the rest of us get another chance to do better. Triisobutylaluminum rewards careful, respectful work. Rushing or skipping steps risks lives and equipment. Safety isn’t a burden—it’s the true cost of keeping things running well.
Is Triisobutylaluminum hazardous or flammable?
What’s At Stake with Triisobutylaluminum
Triisobutylaluminum pops up in news stories lately due to its use in chemical manufacturing, especially in plastics and catalysts. The science behind it seems a world apart for most people, but what matters most isn’t technical jargon—it’s the everyday reality for workers, emergency responders, or anyone living near a site that handles this substance. My own work in a lab drove home how quickly lab safety can turn from a boring checklist into a make-or-break factor. This chemical sits at the crossroads of opportunity and risk.
No Room for Carelessness
Triisobutylaluminum ranks as one of the more dangerous chemicals you can store in a facility. It burns instantly on contact with air, sometimes erupting in flames nobody expects. Splashes reach further than you’d guess, and the fumes poke right through skin and clothing unless you have the right gear. NIOSH and OSHA categorize it as a "pyrophoric" liquid—meaning, more or less, it sets itself alight in regular air.
The danger lurks in small things: a leaky valve, a loose gasket, or even humidity in the air. Some labs swap stories about a single drop sparking a fire, and those aren’t tall tales. In my own safety training, we watched footage nobody would want to see twice—a tech forgot to fully tighten a cap, and a small leak shot out a jet of fire. Triisobutylaluminum burns with a blinding intensity, raising temperatures above what most fire extinguishers can manage. That fire took a specialist team and left scorched equipment as a warning to everyone else. The lesson etched itself in my memory.
Why the Hazard Matters
Chemicals like this don’t just threaten a single lab or plant. Accidents can drift miles downwind. Safety mishaps have forced towns to evacuate. In Texas, chemical plant incidents have led to hospitalizations and expensive cleanups. Triisobutylaluminum reacts with moisture, letting off flammable gas. Water, the usual hero in fires, turns into an enemy here—it actually makes the flames worse.
The stakes reach beyond fires. If the chemical gets on skin, it burns deep and leaves lasting scars. You need a liquid-tight suit, face shield, and gloves that cover to the elbow or higher. Most people outside of industry don't realize how simple oversights—a tired worker, a skipped checklist—can snowball into disasters.
Preventing Trouble Before It Starts
No one wants a tragedy. I spent countless hours as a rookie inventorying our emergency equipment and double-checking every seam in every storage drum. That kind of nitpicking isn’t just busywork; it’s what separates a safe plant from tomorrow’s news headline. The Chemical Safety Board tracks lessons learned: specialized transfer tools, thorough training, clear evacuation routes, and continuous leak monitoring. The best-run facilities drill like firefighters—prepared for every “what if” long before the alarm sounds.
Neighbors should know what chemicals rest behind those fences. Public transparency can push companies to upgrade safety practices. Regulators have a role, but real change comes from workers who talk frankly about mistakes and keep a sharp eye out for shortcuts. Tech advances like real-time sensors and smart valves help, but nothing replaces a workplace culture where everyone feels part of the safety net.
Triisobutylaluminum reminds us that progress doesn’t always arrive quietly. It demands respect—and the discipline to handle it as if every day could be the day the margin for error vanishes.
What are the safety precautions when using Triisobutylaluminum?
The Reality of Working with Triisobutylaluminum
Triisobutylaluminum isn’t a household chemical. People using it usually work in the thick of specialty manufacturing or research. The first thing most folks notice is how this liquid can burst into flames just from touching the air. Handling it without much thought quickly turns into an accident.
Understanding the Fire Hazard
I once saw a new lab tech reach for a bottle without knowing the risk. The cap came off and a golf ball-sized fireball leapt up. No one picked up burns, but it burnt a hole through the foil hood filter—quick reminder that fire isn’t the sort of “maybe” consequence here.
Triisobutylaluminum reacts fiercely with moisture too, whether that's humidity or skin. You splash a few drops and they'll light up on a sweaty glove. So folks need real preparation and well-planned steps.
Practical Safety Habits
Always use a glovebox filled with dry, inert gas. Those gloves need to seal tight, without any pinholes. Even small leaks spell trouble. Those who don't have fancy equipment have to use a Schlenk line and serious patience. Common nitrile gloves alone offer almost no protection. Add flame-resistant clothing and wear splash-proof goggles, then throw a face shield on top.
Before work starts, people need to run through a checklist: Are all the fire extinguishers charged? Is there a Class D extinguisher on hand? Are the local exhaust hoods working? If just one of these pieces isn’t in place, it’s smart to hit pause. Water-based extinguishers backfire—literally. Dry sand or powder only.
Storage Smarts
Triisobutylaluminum demands real respect. Store it in containers designed for pyrophoric substances, sealed with airtight caps. A dry, temperature-controlled storage cabinet away from main walkways beats shoving it on any convenient shelf. Label everything, right down to the outer protective drum. I remember a neighbor lab swapping cans to save space; ten minutes later their bench was a mess of scorched glass.
E-E-A-T in Practice: Fact and Experience
Documented incidents show poor planning leads to hospital visits. The U.S. Chemical Safety Board lists cases where even brief exposure caused severe burns. The chemical’s reactivity is well documented by the manufacturers—read their data sheets like gospel.
A single spill can go from nothing to disaster in seconds. The right training goes beyond written protocols—it’s about drill and experience. I worked with a mentor who’d set up dummy drills every month. That practice built instincts to stay calm if something did go sideways.
Emergency Response
If a leak or fire breaks out, don’t reach for water. Dry chemical extinguishers only. Evacuate the room, hit the alarms, and let trained responders take charge. Skin contact isn’t something to brush off—flush the area with mineral oil, not water, and seek help right away.
For disposal, neutralizing agents and remote handling keep people out of harm’s way. Follow local rules for hazardous waste. Cutting corners to save a few minutes turns into days of paperwork or much worse.
Keeping Everyone Safer
Making a habit of preparation, PPE, and respect for the substance keeps people safe. Stories and scars teach us—Triisobutylaluminum rewards the cautious, not the careless. No one wants to learn the hard way.
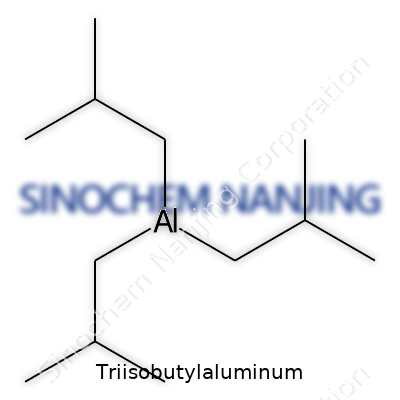
| Names | |
| Preferred IUPAC name | Tris(2-methylpropyl)aluminium |
| Other names |
TIBA Triisobutyl aluminium Aluminum, triisobutyl- Triisobutylaluminium |
| Pronunciation | /traɪˌaɪsəˌbjuːtɪl.əˈluːmɪnəm/ |
| Identifiers | |
| CAS Number | 100-99-2 |
| Beilstein Reference | 3581730 |
| ChEBI | CHEBI:33368 |
| ChEMBL | CHEMBL1335145 |
| ChemSpider | 57672 |
| DrugBank | DB11492 |
| ECHA InfoCard | 100.184.499 |
| EC Number | 213-690-5 |
| Gmelin Reference | 75892 |
| KEGG | C18621 |
| MeSH | D014270 |
| PubChem CID | 119098 |
| RTECS number | AT4370000 |
| UNII | H0P54P1L5E |
| UN number | UN2872 |
| Properties | |
| Chemical formula | C12H27Al |
| Molar mass | 186.36 g/mol |
| Appearance | Colorless to slightly yellow liquid |
| Density | 0.81 g/mL at 25 °C |
| Solubility in water | Reacts violently |
| log P | 4.3 |
| Vapor pressure | 9.3 hPa (20 °C) |
| Acidity (pKa) | 48.6 |
| Basicity (pKb) | 28.50 |
| Magnetic susceptibility (χ) | −74×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Viscosity | 1.13 mPa·s (20 °C) |
| Dipole moment | 0.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -325 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4419.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H260, H314, H411 |
| Precautionary statements | P210, P222, P231+P232, P280, P303+P361+P353, P305+P351+P338, P377, P422 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Flash point | 10 °C |
| Autoignition temperature | 225 °C |
| Explosive limits | 3.5% - 18% (in air) |
| Lethal dose or concentration | LD50 (oral, rat): 2,000 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 2,500 mg/kg |
| NIOSH | WA2625000 |
| PEL (Permissible) | PEL (Permissible) for Triisobutylaluminum: Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of Triisobutylaluminum: "2 mg/m³ (as Al), 8-hr TWA |
| IDLH (Immediate danger) | IDLH: 2 mg/m³ |
| Related compounds | |
| Related compounds |
Trimethylaluminum Triethylaluminum Tripropylaluminum Tri-n-butylaluminum Diisobutylaluminum hydride |

