Triiodoacetic Acid: A Practical View From the Lab Bench
Historical Development
Triiodoacetic acid brings a particular fascination to anyone interested in halogen chemistry or practical organic synthesis. This compound started making appearances in research journals in the 20th century, riding the wave of increasing interest in iodinated organics. Early work with triiodinated molecules found much of its initial momentum from medical imaging research, especially as practitioners tried to find safer, more effective contrast agents for X-ray and CT scans. Many researchers sitting at lab benches around the world started exploring not just the physical properties, but also the reactivity and possibilities for customization that triiodoacetic acid offered. In my own university days, reading about the history of these halogenated compounds made clear how tightly chemistry and real-world problems are linked—new materials often emerge from the cross-section of curiosity and need. Triiodoacetic acid never got as much attention as its cousin triiodobenzoic acid, but it holds a particular space in the conversation about versatility and chemical power.
Product Overview
Triiodoacetic acid, with the formula C2HI3O2, looks rather simple in structure but brings impressive complexity in how it behaves. Its chemical skeleton consists of a simple acetic acid backbone, with all three hydrogens on the methyl group swapped out for iodine atoms. Anyone who has worked with this sort of substitution recognizes that introducing three bulky iodines onto a molecule truly changes everything about how it interacts with solvents, how it recrystallizes, and how it participates in further chemical reactions. There's an undeniable sense of purpose underlying any decision to order or synthesize triiodoacetic acid—chemists reach for it because they want the heavy, reactive punch that only triiodination delivers.
Physical & Chemical Properties
In handling triiodoacetic acid, you get a heft both literal and figurative: it crystallizes out as a dense, sometimes off-white solid, bearing little similarity to common laboratory acids like acetic or benzoic. Trying to dissolve it in water or organic solvents offers mixed results, thanks to the three bulky iodines. Solubility always stays lower than you hope, and the substance remains fairly stable at room temperature with a melting point higher than its unsubstituted relatives. Those who have weighed out these crystals notice a certain grainy, heavy texture in the scoop, and anyone heating the sample recognizes the release of iodine vapors long before decomposition kicks in—a safety reminder not lost on any attentive chemist. Dense halogenation means that triiodoacetic acid exhibits unique behavior in UV-Vis and NMR spectroscopy, revealing clues about its structure and the ways electrons shift under the shadow of all those iodines.
Technical Specifications & Labeling
Triiodoacetic acid does not sit on shelves in grocery store quantities, and specifications matter a lot to anyone who orders or uses it. Even minor impurities—residual mono- or diiodoacetic acids, unreacted starting materials, and traces of iodine—can influence experiments. Certificates of analysis list purity, melting range, and the outcome of titration assessments. Labeling speaks to the hazards proudly: handle under a fume hood, store away from light, and keep containers tightly sealed. In my experience, detailed labeling also tells you how the batch was dried, which can make a big difference to a reaction. Most chemists sizing up a new bottle quickly glance at these details before planning any work.
Preparation Method
Synthesizing triiodoacetic acid never comes easy, especially on any scale above a few grams. The classic route starts with acetic acid or a methyl-substituted substrate, which is reacted with iodine in the presence of an oxidizing agent like nitric acid. This substitution requires both patience and a certain respect for the hazard, as the reaction mixture can liberate iodine vapors and heat up quickly. Keeping temperatures low and mixing thoroughly becomes more about safety than efficiency. Filtering the crude product, scrubbing it with appropriate solvents, then drying under vacuum gets you to a respectable purity, though a repeat crystallization often proves necessary. Every chemist worth their salt knows to watch out for side reactions—over-oxidation, unwanted byproducts, lingering color residues. Only careful control and willingness to monitor progress under a hood keep the operation safe and worthwhile.
Chemical Reactions & Modifications
Triiodoacetic acid invites further manipulation. The three iodine atoms serve as tempting handles for nucleophilic substitution, making the molecule useful for introducing iodine into other frameworks or as an intermediate in more complicated organic syntheses. One popular direction points toward radiochemical applications, using the acid as a scaffold for iodine-125 labeling. In laboratory settings, reactions under basic or acidic conditions reveal how the heavily iodinated compound can lose iodines or undergo decarboxylation. Modifying the acid group or replacing one or two iodines with other substituents leads researchers into new territory, sometimes looking for better contrast agents, sometimes for drugs that ride into the body on the back of this dense atom cluster. Any student who runs these reactions soon develops an appreciation for the quirks and practical challenges lying behind every step.
Synonyms & Product Names
Chemists sometimes refer to triiodoacetic acid by alternate names—sometimes “TIAA” comes up, especially in shorthand in lab notebooks. Other times, the names echo the systematic approach: “acetic acid, triiodo-,” “triiodoethanoic acid,” or “2,2,2-triiodoacetic acid.” Looking across catalogs, one might see it listed under these variations, each name a nod to the same structure but reflecting different conventions. Nobody wants to confuse it, because the consequences of mixing up halogenated acetic acids can range from failed experiments to genuine safety problems.
Safety & Operational Standards
Safe handling of triiodoacetic acid walks a narrow line. The iodine content brings concerns about both acute and chronic toxicity, with possible impacts on thyroid health—a lesson drilled into any chemist who starts working with heavy halogen compounds. Direct contact can irritate the skin, while dust or vapor inhalation brings a persistent, harsh medicinal odor along with risks to the respiratory system. Proper procedures mean always running reactions in ventilated hoods, wearing gloves, and planning for spills. Waste disposal rules call for segregating halogenated organics, typically keeping them far from non-halogenated waste streams because iodine remains difficult to treat in standard organic solvent disposal systems. Laboratories with high safety scores run routine training and keep careful logs for every order and use.
Application Area
Most people outside the chemistry world only see triiodoacetic acid as a curiosity, but certain fields put it to real use. Radiopharmaceutical manufacturing stands near the top, where triiodoacetic acid’s dense iodine atoms become the platform for building imaging agents. In analytical chemistry, it helps as a derivatizing agent—modifying molecules to improve detection or identification in complex biological samples. Some routes even explore its presence in specialty reagents for organic synthesis, especially when researchers wish to test the influence of heavy atom substitution on reaction pathways or molecular dynamics. Its role in life sciences always draws careful scrutiny due to potential toxicity, yet the compound's utility sometimes outweighs these downsides if managed with proper controls.
Research & Development
New studies using triiodoacetic acid push into directions ranging from imaging contrast improvements to targeted drug delivery. Researchers try to understand whether swapping in alternative halogen patterns or tweaking the acid group delivers advantages for solubility or bioactivity. Many studies measure exact distribution throughout tissues, studying both benefits and risks—particularly relating to iodine’s well-known role in thyroid regulation. In the world of organic methodology, the acid serves as an instructive case for the introduction of large, electron-withdrawing groups into simple backbones. This compound allows scientists to probe how such changes affect reactivity, nucleophilicity, and other essential chemical traits. As new analytical tools become available, more fine-grained understanding emerges for each subtle modification, though challenges in synthesis and purification persist even as technology improves.
Toxicity Research
Concerns about toxicity run deep with triiodoacetic acid. The dense iodine content triggers worry about acute poisoning, chronic thyroid disruption, and general cytotoxicity. Animal studies and cell assays usually show that higher iodinated compounds accumulate more in tissues, with possible implications for both efficacy and unwanted effects. In real-life lab settings, the acid can act as an irritant, and accidental exposure gets flagged immediately given the risk profile. Over the years, my own experience and review of literature suggest caution always trumps convenience here: even small spills get documented, and medical monitoring sometimes becomes necessary for frequent-handling personnel. Any move toward broader medical use requires rigorous, transparent studies on metabolism, excretion, and possible longer-term systemic impacts.
Future Prospects
Looking ahead, triiodoacetic acid seems likely to remain a specialty chemical, finding life at the interface between synthetic chemistry, medical imaging, and targeted therapy development. The increasing demand for precise imaging agents and the ongoing search for moieties capable of robust radio-labeling keeps triiodoacetic acid in the research spotlight. My conversations with researchers lead me to believe that future improvements will turn on new preparation methods yielding higher purities with less waste, as well as chemical modifications that mitigate toxicity without sacrificing utility. Debates about environmental and human health risks continue to shape whether such compounds enter wider markets, but the trend toward precision medicine and tighter control over drug distribution patterns creates opportunities for molecules just like this one. As ever, advances depend on a close link between bench scientists, safety professionals, and clinicians willing to keep questions alive about both chemistry and consequence.
What is Triiodoacetic Acid used for?
Unpacking a Chemical You Rarely Hear About
Triiodoacetic acid doesn’t exactly pop up in everyday talk — I’ve spent plenty of time around labs, yet this compound still gets sideways glances from seasoned chemists. It’s not the household name you find on the back of a cleaning spray, or in medicine cabinets. So what is it, and what do people actually do with it?
How Chemistry Pushes Boundaries
Triiodoacetic acid appears as a specialty chemical, and it’s typically sized up for its role as a reagent. Anyone who’s taken college-level chemistry can recall the endless line-up of acids and reagents stacked on classroom shelves. You may wonder, does every one of these odd-sounding chemicals really serve a purpose outside the textbook?
This one actually does. Triiodoacetic acid acts mainly as a building block for exploring complex organic reactions. In research settings, scientists use it to add iodine atoms in a targeted way to organic molecules. Iodination can seem esoteric at first, but in practical terms, it gives scientists product options in medicinal chemistry, pharmaceutical research, and imaging technology. For instance, tagging certain molecules with iodine sees use in the development of diagnostic agents — some radiolabelled compounds used for thyroid diagnostics actually rely on similar chemistry.
Why Research Labs Value Iodinated Compounds
In labs where I’ve worked, the demand for selective halogenation always comes up. Introducing iodine atoms into molecules gets tricky without the right tools. Triiodoacetic acid brings three iodine atoms specifically arranged around an acetic acid backbone. That gives chemists a level of predictability and control — especially when looking for high yield, fewer byproducts, and clear data for reactions. This control isn’t just about laboratory neatness: getting clean results saves time and reduces costs in the race to publish or develop new drugs. If you’ve tried running a messy reaction, you already know the pain of endless column purification and wasted weekends.
Concerns and Safe Handling
No commentary on niche chemicals gets complete without talking about safety. Chemists know that dealing with compounds loaded with iodine needs careful handling. Iodinated materials can be reactive, affect biological systems, and pose waste disposal headaches. I’ve seen folks lose weeks’ worth of samples to improper storage or face hasty evacuations over what seemed like a trivial spill. Above all, observing proper protocols for personal protection and chemical waste isn’t red tape — it’s how you keep people safe and research projects on track.
Looking Toward Smarter, Safer Use
One eye keeps watching for better ways to manage specialty reagents. Waste treatment remains a sore spot with halogenated compounds, and green chemistry circles push for less hazardous alternatives or improved recycling. Those solutions don’t materialize overnight, and in the meantime, knowledge transfer and staff training help prevent expensive mistakes. In my experience, most chemical mishaps get traced to skipped steps or overlooked warnings rather than the chemicals themselves. Culture shift toward more proactive education delivers real gains — fewer spills, higher yields, and better insights for everyone involved in chemical research.
What are the safety precautions when handling Triiodoacetic Acid?
Facing Hazards Head-On
Triiodoacetic acid isn’t your garden-variety household chemical. This isn’t something you grab without thinking. The first time I opened a container in a lab, you could almost feel the tension in the air. Gloves weren’t just optional accessories—they were the only thing standing between my skin and a nasty chemical burn. Direct contact leaves red patches, and the fumes stick with you long after you leave the room. If you breathe in too much, expect your throat to burn and your lungs to ache. I watched someone skip eye protection once—he spent hours flushing out his eyes. The warning wasn’t lost on anyone in the room.
Real Protection, Not Just Good Intentions
There’s nothing fancy about proper preparation. Grab a thick lab coat, not a flimsy apron. You want splash goggles that fit close and gloves made for acids, so Nitrile or Neoprene usually top the list. And don’t work alone. Every lab that respects its people pairs up workers whenever acids like this get involved. If something spills, nobody wants to be searching the lab for help. Washing stations and safety showers stay clear and ready—no clutter, no locked doors. It’s all too common to find eyewash stations blocked with boxes; in a good lab, this stuff stays within arm’s reach.
Airflow and Storage
Triiodoacetic acid doesn’t get along with bad ventilation. Fume hoods aren’t just for show, they suck away vapors before they drift up your nose. If you store the acid in a stuffy cupboard or near heat, you’re looking for trouble. I once saw an unmarked drawer filled with acids after hours—a silent disaster waiting for a curious hand. The right spot stays cool and dry, never in sunlight, and always among chemicals that won’t react if something leaks.
Spill Response in Real Life
Most spills don’t look dramatic, but that doesn’t mean they’re harmless. Grab a spill kit—an old towel won’t cut it. Neutralizing agents do the work, not water or soap. If something splashes, you start at the eyewash or shower for at least fifteen minutes, counting every second while cursing under your breath. Reporting the incident right away helps everyone learn, and nobody plays hero by skipping medical checks. I’ve seen small cuts turn into serious injuries when people tried toughing it out.
Training and Keeping Up With Facts
Knowing what Triiodoacetic acid does isn’t enough. Workers review the latest material safety data sheets (SDS) before shifting a single drop. Training isn’t a boring checkbox—it’s real-world survival. I remember the first time I sat through a chemical safety course, barely awake, thinking it was just another hurdle. Later, I saw a new team member freeze up during a small spill. Training kept the panic down, and every refresher since reinforces that cool heads—and the right information—keep everyone safe.
Walking the Walk
Staying safe around Triiodoacetic acid means doing the small things right, every time. This isn’t about checking off a list, it’s about respecting a chemical with teeth. From gloves to ventilation, teamwork to training, the risks don’t reward shortcuts. You walk away with unburned skin and clear lungs because you paid attention and took no chances. That’s what real safety looks like.
What is the chemical structure of Triiodoacetic Acid?
The Science Behind Triiodoacetic Acid
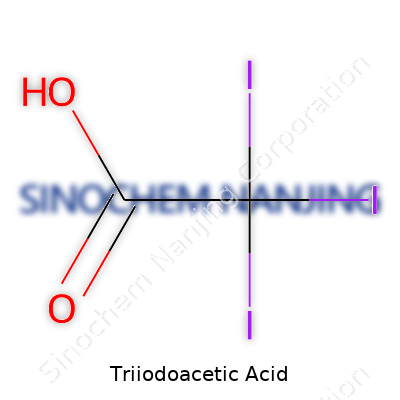
Triiodoacetic acid carries an intimidating name, but at its core, the chemistry comes down to a simple structure. It starts with the backbone of acetic acid—think vinegar, without the water. In triiodoacetic acid, three iodine atoms replace three hydrogen atoms on the methyl group, so the familiar CH3 becomes CI3. The rest stays the same: a carboxyl group (COOH) hanging at the end. Chemically, people write it as C2HI3O2. Looking at its actual composition: Iodine atoms hug one end of the chain, while the acid group anchors the other. There’s no complex branching or twisty molecule here. The structure gives it unique properties, mostly because of those hefty iodine atoms.
Why the Structure Matters
Iodine doesn’t just add weight to the molecule. In triiodoacetic acid, it changes how the compound behaves. Those three iodine atoms suck electron density away, making the carboxyl group even more acidic than regular acetic acid. That’s not a small change. Strong acidity in chemical terms often leads to increased reactivity. The molecule becomes more likely to participate in exchange reactions, especially with other charged substances. This sort of behavior makes triiodoacetic acid useful in laboratory settings. For instance, its heavy halogen load and high reactivity can lend itself to synthesis routes that involve halogen transfer, or as a tracer in certain types of radiation detection or metabolic studies.
Environmental and Safety Concerns
Carrying around three iodine atoms isn’t just interesting; it changes how triiodoacetic acid behaves outside the lab. Compounds with heavy halogens don’t break down easily. They often hang around in soil and water after they escape the lab or hospital. Worries grow over these sorts of chemicals because persistent halogenated molecules can cause problems for living things, from bacteria up to humans. Iodine, needed in small doses for our thyroid, can disrupt hormonal balance if released in odd chemical forms or concentrations. So, storing and handling triiodoacetic acid calls for real care. Any spills or leaks demand prompt clean-up, keeping the compound out of drains and groundwater.
How the Science Community Responds
Labs that use triiodoacetic acid typically put strict procedures in place. They check ventilation, train staff on the risks, and invest in airtight containers. The chemical industry has worked to switch toward less persistent alternatives whenever possible, cutting down the environmental load. Disposal protocols now focus on neutralization and incineration under controlled conditions, which helps keep iodine out of the water table. Ongoing research keeps an eye on how these halogenated acids move through ecosystems, drawing on findings from environmental chemistry and public health. Regulatory agencies track compounds like triiodoacetic acid, setting legal limits, reviewing toxicity, and updating best-practice guidelines to match new evidence.
Paths Toward Safer Chemistry
Substituting heavy halogens with lighter ones, or using non-halogenated acids in the same role, cuts the risk for both people and the planet. Industries testing alternatives weigh not only the chemical work but also persistence and bioaccumulation effects. Many new synthetic methods now favor greener solvents and milder reaction conditions. Adopting less hazardous compounds improves workplace safety and reduces the cost of waste disposal. The work continues, backed by real-world research and practical solutions that shed light on every part of a molecule's journey, from the beaker to the broader environment.
How should Triiodoacetic Acid be stored?
Safety in the Lab: Why Storage Matters
Working in chemistry labs early in my career, I learned pretty fast that mistakes with chemicals rarely bring a second chance. Triiodoacetic acid stands out as a compound demanding respect. It sits on chemical shelves thanks to its usefulness in synthesizing other compounds and testing reactions. Yet, its halogen substitutes make it react unpredictably if handled carelessly. Taking shortcuts with storage does more than waste money—it can put people at risk.
Storing Triiodoacetic Acid: Getting the Basics Right
Triiodoacetic acid belongs to the family of iodoacetic acids, which often give off fumes and can trigger strong reactions with water or light. My own experience has shown that strong acids want quiet, stable environments away from daily commotion. I kept these compounds sealed in airtight glass containers, stored inside dedicated, clearly labeled cabinets. This method cuts down risks of accidental exposure or mixing, especially in crowded spaces where multitasking can lead to slips.
Industry guidance and chemical suppliers agree: cool, dry, and well-ventilated spots keep triiodoacetic acid stable. Any damp corner invites gradual breakdown or leaks, even slow corrosion of storage containers. Glass never reacts with triiodoacetic acid, making it a practical, trusted choice—plastic or metal containers only increase chances of unwanted chemical reactions down the line. Police your shelves for cracks in containers and ensure chemical names stay visible and bold, easy for anyone to spot on a rushed day.
Light and Ventilation: Shielding Against Trouble
Strong light can degrade triiodoacetic acid. I’ve watched colleagues make the mistake of leaving acids near sunny windows or under cheap, cracked bulbs—fumes build up, temperatures spike, and before you know it, the air stings to breathe. Extra insulation and dark cabinets solve that problem fast, and good ventilation keeps lingering vapors from collecting. This not only protects those working with the acid but also reduces the risk of corrosive damage to shelving and other nearby equipment.
Why Labels and Training Mean More Than Fine Print
In every lab I've worked, mistakes came up less often when storage rules weren’t just on paper. We’d review chemical storage routines monthly, not because rules changed, but because having the habit meant younger staff saw safety as normal, not extra effort. Every chemical—triiodoacetic acid included—gets a clear hazard label and an emergency spill kit close by. Personal experience taught me to always check expiration dates and signs of leaks before each use, since even the best storage plan means nothing if the crew takes shortcuts.
Practical Solutions For Real Labs
Simple steps go a long way. Lock up acids and track who uses what and when. Set routines for checking condition of containers every week. Make emergency wash stations easy to reach. Any lab, from school basements to research hubs, benefits by holding regular briefings and refreshers—familiarity and practice make all the printed protocols actually work in the moment.
Nothing compares to hands-on experience. Handling acids like triiodoacetic acid without cutting corners shows you care not just for your own safety, but for everyone sharing the workspace. Clear storage rules, the right equipment, and real attention to detail lower stress, prevent hiccups, and let science move forward safely.
What is the shelf life of Triiodoacetic Acid?
Why Shelf Life Matters
Chemicals rarely offer the luxury of sitting on a shelf forever. Triiodoacetic acid, a compound seen in research labs and industrial setups, reminds us of that with its own time limits. In daily work with fine chemicals, I’ve learned the difference between active, reliable material and something that’s quietly degraded over months. Triiodoacetic acid keeps its punch only when handled right.
Shelf Life Basics
Triiodoacetic acid doesn’t age gracefully under poor conditions. The typical unopened shelf life stays around two years at room temperature, based on guidance from reputable chemical suppliers and literature. Moisture, sunlight, fluctuating temperatures—exposure to any of these shortens its lifespan. Some lots last a little longer if handled with care, but degradation creeps in if storage isn’t tight.
Keeping It Stable
An airtight container, a dark cupboard, and a stable, cool room do more for triiodoacetic acid than any complex preservation method. Desiccators help, especially in humid regions. Anyone who’s opened an old reagent and caught a whiff of acid or spotted clumpy crystals knows what happens with careless storage. Small routine steps—checking seals, minimizing opening time, keeping containers clean—stretch out usable life.
What Happens As It Ages
Chemical reliability drops off before complete spoilage. Unseen at first, small changes like gradual discoloration, caking, or a weaker aroma mean it’s no longer trustworthy. Over time, exposure triggers slow breakdown. That can create impurities, which skew results and waste time for anyone chasing precise data or trying for a reliable synthesis. An expired acid won’t announce its decay, so routine checks matter. Lab audits flagged old stocks in my own experience, forcing hard choices about tossing out expensive material versus risking experimental accuracy.
Safety Concerns
Old chemicals do more than let a process fizzle—they can turn dangerous. Decomposition sometimes leaves behind volatile byproducts or less predictable reactivity. Working with any out-of-date acid, especially one with halogens like iodine, can raise the odds of an accident. Safety data sheets warn about possible hazardous fumes or unpredictable compounds. At one facility, a forgotten bottle with degraded triiodoacetic acid had to be handled as hazardous waste. Disposal costs climb when material’s no longer in peak shape.
How To Choose and Store
Anyone using triiodoacetic acid should check manufacturing and expiration dates on every label. Regular inventory reviews can save money in the long run. Buying smaller amounts reduces waste and makes for less hazardous disposal. Requesting certificates of analysis at the time of purchase shows confidence in product quality. If a lab or facility struggles with frequent waste or loss, reviewing ordering routines or storage locations often helps.
Supporting Data and Solutions
Leading manufacturers outline proper storage in their documentation. Sigma-Aldrich, for example, highlights two years at room temperature, away from moisture and light. Studies in analytical chemistry journals reinforce how water uptake and heat mean loss of activity. Education and ongoing reminders about these points keep both research and production teams safer and more efficient.
Periodic refresher training, inventory scheduling, and safer disposal options offer sensible solutions. These simple shifts guard both pocketbooks and people.
| Names | |
| Preferred IUPAC name | 2,2,2-Triiodoacetic acid |
| Other names |
Triiodoacetic acid Trijodessigsäure Acetic acid, triiodo- Triiodoethanoic acid |
| Pronunciation | /traɪˌaɪ.əʊ.dəʊ.əˈsiː.tɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 594-60-5 |
| 3D model (JSmol) | `3D model (JSmol)` string for **Triiodoacetic Acid**: ``` C(C(=O)O)(I)(I)I ``` This is the **SMILES** string, which can be used to visualize the 3D model in JSmol or other molecular viewers. |
| Beilstein Reference | 1249488 |
| ChEBI | CHEBI:52718 |
| ChEMBL | CHEMBL408089 |
| ChemSpider | 5312949 |
| DrugBank | DB14689 |
| ECHA InfoCard | 100.011.039 |
| EC Number | 607-468-7 |
| Gmelin Reference | 54067 |
| KEGG | C19308 |
| MeSH | D014264 |
| PubChem CID | 656633 |
| RTECS number | AJ8050000 |
| UNII | C138XV25F9 |
| UN number | Un2469 |
| Properties | |
| Chemical formula | C2HCl3O2 |
| Molar mass | 493.718 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.867 g/cm3 |
| Solubility in water | Slightly soluble in water |
| log P | 0.523 |
| Vapor pressure | 0.000016 hPa (25 °C) |
| Acidity (pKa) | 0.7 |
| Basicity (pKb) | 2.65 |
| Magnetic susceptibility (χ) | -89.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.779 |
| Dipole moment | 3.3732 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 377.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -19.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -730.1 kJ/mol |
| Pharmacology | |
| ATC code | D08BX02 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS05 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P260, P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-1 |
| Flash point | Flash point: 113 °C |
| Autoignition temperature | 150 °C |
| Lethal dose or concentration | LD50 (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 960 mg/kg (rat, oral) |
| NIOSH | NA |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Iodoacetic acid Diiodoacetic acid Trifluoroacetic acid Acetic acid Monochloroacetic acid Dichloroacetic acid Trichloroacetic acid |

