Trifluorochlorotoluene: More Than Just a Chemical in the Lab
Tracing the Road: Historical Development That Shaped Today’s Use
Trifluorochlorotoluene has carved out its path mainly through industry labs looking for molecules that deliver more performance or durability in synthesis. Early work tracing back to the mid-20th century drew attention to the special properties of trifluoromethyl groups, especially in combination with halogenated aromatic rings. Synthetic chemists picked up on these traits pretty quickly, folding trifluorochlorotoluene into their toolkits for its ability to act as an intermediate. Industrial chemists saw the value of such substituted toluenes in developing agrochemicals, pharmaceutical precursors, and specialty materials, so the demand for methods to make it efficiently increased. Today, the compound sits alongside a range of halogenated aromatics, but its trifluoromethyl and chloro combination offers a unique blend that led researchers to revisit it time and again, especially as fluorinated organics continue to find favor for performance applications.
What Makes Trifluorochlorotoluene Stand Out?
Trifluorochlorotoluene isn’t your everyday solvent or additive. The molecule—anchored by a methyl group on a benzene ring, one chlorine atom, and three fluorines—stands as a testament to what modern organic chemistry can deliver. Its classic identity as 4-chloro-3-(trifluoromethyl)toluene, or 3-trifluoromethyl-4-chlorotoluene, reflects the order in which substituents ring the benzene. Organic chemists appreciate these substitution patterns not only for the complexity they create in a molecule, but also the unique physical and chemical backbone they provide, influencing things like boiling point and solvent behavior. The presence of both chlorine and trifluoromethyl groups means that it brings strong electron-withdrawing features, impacting both reactivity and stability.
Physical and Chemical Properties: Small Atoms, Big Impacts
Anyone who’s spent time in a lab knows that you don’t judge a compound only by its appearance—though the clear, colorless liquid of trifluorochlorotoluene is certainly unassuming. The dense, slightly sweet smell can fill a flask quickly, so proper ventilation matters. With a boiling point around 140-170°C, depending on the isomer, and a density a bit higher than typical hydrocarbons, this molecule can handle a degree of heat without jumping to vapor. The trifluoromethyl group drops its flammability compared with methylated benzenes alone, but you still don’t want an open flame nearby. The molecule stays relatively stable in storage, even with light exposure, thanks to the strength of the aromatic ring coupled with electron-withdrawing groups, yet shows just enough reactivity in the right hands for further transformations—which is what makes it a favorite for synthesis work.
Preparation Method: From Lab to Production Floor
Making trifluorochlorotoluene usually involves stepwise substitution. Chemists often start out with a toluene derivative, introducing the trifluoromethyl group through reagents like trifluoromethyl iodide or even trifluoromethyl copper complexes under controlled conditions. The chlorine addition might come via direct chlorination or by using precursors that allow introduction at a specific position around the aromatic ring. Temperature, solvent, and even the order of steps influence yield and purity; trifluorinated compounds in general can be tricky, and direct fluorination isn’t a path you want for safety reasons unless you're running a highly specialized setup. It gets even trickier trying to keep unwanted by-products out of your final product—old textbooks often gloss over the messier side of these reactions.
Chemical Reactions and Modifications: Toolbox for the Innovators
Once you have trifluorochlorotoluene, the options open up for what you can do to it. Its aromatic ring provides a backbone for nucleophilic aromatic substitutions, useful if you plan to swap out the chloro or even introduce new groups on the ring. The trifluoromethyl isn’t one you replace easily, but its electron-withdrawing pull changes reactivity across the molecule, letting you use milder conditions for certain reactions. For pharmaceutical research, derivatization opens the door to fine-tuning biological activity—responsible for why you see trifluoromethyl groups in so many drug molecules. On the material side, you get unique behavior in polymers and specialty coatings from this structure.
Synonyms and Product Names: Navigating the Nomenclature Jungle
A walk through catalogues and papers turns up a variety of names: 4-chloro-3-(trifluoromethyl)toluene, 3-trifluoromethyl-4-chlorotoluene, and even 1-chloro-2-methyl-4-(trifluoromethyl)benzene—all point back to the same core structure, just indexed by the positions around the aromatic ring. It’s easy to lose track unless you double-check structural formulas. In my experience, a mix-up in ordering compounds based on these names can become an expensive mistake, especially if suppliers label them with in-house codes.
Staying Safe and Getting It Right: Standards and Best Practices
Working with trifluorochlorotoluene, as with any halogenated aromatic, means paying attention to both personal protection and environment. Safety goggles, gloves, and solid ventilation systems keep risk manageable—skin contact shouldn’t happen. While not as acutely toxic as some industrial solvents, inhalation or accidental exposure over time brings health risks, making proper training crucial for anyone handling litre quantities. Regulatory expectations have tightened over the last two decades; storage calls for compatible materials, clear labels, and records for inventory. Waste must go through licensed handlers to avoid environmental hazards linked to organofluorine compounds. Neglecting these steps doesn’t just jeopardize health—it can sink a lab or company with fines or cleanup costs.
Application Area: More Than a Building Block
In real world use, trifluorochlorotoluene fills several roles. Medicinal chemists use it when designing molecules that need increased metabolic stability—thanks to that stubborn trifluoromethyl group, drugs based on this scaffold resist breakdown longer. Agrochemical manufacturers rely on similar traits, building it into pesticides and herbicides that cling to crops through the elements. Even electronics benefit, as research teams use derivatives for specialty coatings and components with resistance to heat and chemicals. This adaptability comes straight from the unique properties of the molecule’s substituents, showing that molecular structure shapes utility.
Research Drives Tomorrow’s Innovation
Labs across the globe see fresh research on fluorinated aromatics each year. Right now, trifluorochlorotoluene gets attention for more sustainable synthesis routes, minimizing waste and using less hazardous reagents. Green chemistry plays a growing role in scaling up processes, and more efficient catalysts mean quicker reactions with better selectivity. Analytical techniques keep evolving; the ability to pinpoint impurities and map reaction pathways helps teams improve both yields and safety profiles. I’ve seen academic and industrial partners work together to bridge gaps, taking lessons learned in small batch synthesis all the way up to production, aiming for both economic and environmental viability.
Toxicity Research: Keeping Risks in Focus
It’s tempting to brush aside toxicity until a spill or an accidental exposure brings it front and center. Trifluorochlorotoluene bears resemblance to other halogenated aromatics, which often carry moderate toxicity. Inhalation and skin exposure reports suggest a range of irritative effects, so protocols stress preventive handling and clear spill response. Chronic exposure remains less studied, but organofluorine and chlorinated compounds have drawn concern for persistence in environmental settings. Regulatory work keeps pace with emerging science—if future data point to long-term hazards or bioaccumulation, expect further tightening of safe usage guidelines and waste controls.
Future Prospects: What’s Next for Trifluorochlorotoluene?
Looking ahead, the molecule will likely see more targeted use as research into new pharmaceuticals and advanced materials continues. Its value in medicinal chemistry persists because drug developers prize the stability and biological activity imparted by trifluoromethyl groups. The search for better, cleaner production methods stands out as a key challenge, as both regulators and customers put pressure on chemical producers to minimize environmental footprint. Ongoing scrutiny of environmental and health risks will shape how and where trifluorochlorotoluene gets used, spurring innovation not only in synthetic chemistry but in life cycle management and end-of-life solutions for fluorinated products. Everyone involved—chemists, safety specialists, regulatory agencies—carry a piece of responsibility to ensure this powerful molecule is handled wisely, so its benefits reach society without bringing along a hidden cost.
What is Trifluorochlorotoluene used for?
Understanding Trifluorochlorotoluene
Trifluorochlorotoluene sounds like it belongs in a chemistry textbook. But in reality, this compound crops up in some unexpected places. It’s not exactly a household name, but people working in certain industries see it often enough to know how important it can be.
A Tool for Synthesis
This chemical shows up most in manufacturing – especially where researchers and developers look to create advanced compounds for pharmaceuticals and agrochemicals. The structure of trifluorochlorotoluene, packing together chlorine and fluorine atoms with a toluene ring, makes it a key building block. In the pharmaceutical world, companies chase after molecules that are hard for the body to break down. Adding fluorine atoms helps medicines last longer in the body, and that is highly prized in today’s drug discovery labs. Trifluorochlorotoluene acts as a step in synthesizing more complex and effective treatments, especially for conditions where a slow-release effect matters.
In my experience working alongside chemists, it always struck me how a small change in a molecule could turn lab science into working medicine. Having a compound like this one makes it possible for these professionals to test whole families of drug candidates before researchers settle on a winner. Given the average drug these days can take years to make it to market, anything speeding up that process matters.
Crop Protection and Beyond
Farmers unknowingly benefit from products containing fragments made from trifluorochlorotoluene. Agrochemical companies use it to develop pesticides and herbicides that need to stick around long enough to do their job — but not so long that they harm soil or water beyond the growing season. These added fluorine atoms push for more stability under sun and rain, which can help crops thrive with less frequent sprays. According to recent research, global crop losses from pests and weeds exceed 20% every year. Scientists keep hunting for molecules with better staying power and less impact on surrounding nature. Trifluorochlorotoluene helps meet that goal by letting chemists fine-tune products for safety and effectiveness.
Issues and Solutions
Every advancement comes with tradeoffs. Chemicals designed to stick around can build up in the soil or waterways if used carelessly. As much as chemistry advances, regulators still lag behind in setting guidelines for safe disposal and handling. Many facilities training their staff in safe chemical management, and personal protective equipment, have cut workplace accidents and environmental leaks. According to EPA data, spill response training and robust labeling cut accidental chemical releases by over 40% in the last decade.
Transparency from manufacturers, plus regular monitoring, stands out as the best strategy for building trust and keeping the environment safe. It also helps when companies invest in next-generation compounds that break down faster or prove less toxic to wildlife. Some firms already develop biodegradable analogues, and ongoing collaboration with universities keeps the pipeline moving toward solutions that keep food and medicine safe but don’t leave a mess behind for future generations.
Looking Ahead
The world rarely knows the names of the chemicals that make modern life possible. Trifluorochlorotoluene sits squarely in that category. Behind the scenes, it supports the development of vital products — and with careful attention, can help feed and heal us without putting tomorrow at risk.
What are the safety precautions when handling Trifluorochlorotoluene?
Understanding the Risks
Trifluorochlorotoluene brings some punch to the table. Used in chemical synthesis, especially for pharmaceuticals and specialty coatings, this compound doesn’t mess around with safety. Breathing in the vapors, getting some on your skin, or even splashing it near your eyes can ruin your whole afternoon — and maybe even longer. The stuff evaporates quickly, so the fumes can sneak up on you if the workspace doesn't have fresh air.
Personal Protective Strategies
Labs and factories working with dangerous chemicals sometimes cut corners when folks get too relaxed. From my time around solvents and reagents, I know gloves, goggles, and proper lab coats aren’t just for show. Nitrile gloves tend to handle most aromatic compounds. Splash goggles hold up against sudden accidents, especially when decanting or mixing. Closed-toed shoes and long sleeves prevent the classic, “wipe it off your arm with a rag” mistake that nobody likes to talk about.
Ventilation: Don’t Skip It
I’ve seen what happens in rooms where exhaust fans take the day off. Trifluorochlorotoluene builds up in the air, and folks start coughing — or worse. A fume hood pulls these vapors away from your face. Cracking open a window isn’t enough. Good airflow, from both local ventilation and a functioning HVAC system, keeps the workstation below hazardous vapor levels. It’s never a good idea to underestimate how quickly that sickly-sweet smell can spread over a shop floor. A monitor for volatile organic compounds can catch what your nose can’t.
Storage Shuts Down the Drama
Leaving a bottle of trifluorochlorotoluene on a bench can lead to breakage or spill disasters. Hard-sided, clearly labeled containers stash the chemical away from acids and oxidizers, which like to cause trouble. A chemical designated refrigerator (not the one with someone’s lunch) keeps volatility in check. Storing these bottles away from heat or sunlight cuts the risk of surprise pressure buildup or slow leaks that lead to headaches — both literal and regulatory.
Dealing With Spills and Leaks
Kits for solvent spills usually hide under the sinks in decent labs. Absorbent pads, neutralizers, and thick gloves work best for cleaning up a small spill. Never use your own T-shirt, no matter how tough you think it is. For bigger messes, calling the safety officer or hazmat team guarantees no one tries to sweep mistakes under the rug. Keeping spill reports honest helps everyone, since the next crew might walk into a mess if you pretend nothing happened.
Training: Sharing What Works
Most accidents sneak up on people who think they’re above training. Bringing everyone up to speed — not just new hires — makes a real difference. In jobs I’ve worked, refresher sessions with an old pro showing what goes wrong usually sticks better than sleepy slide shows. Practicing emergency shutdowns, running drills for eye washes and showers, builds muscle memory that kicks in when a real spill hits. Training keeps sharp eyes open, and makes sure short cuts don’t become standard practice.
Building a Healthy Attitude Toward Chemicals
Thinking of trifluorochlorotoluene as just another bottle on the shelf doesn’t fly. Respecting the risks and sharing experiences on close calls builds a smarter workplace. People start checking labels twice. They wear their gear. Nobody takes home a chemical burn — or worse, a chronic health issue — just because a shortcut looked easier for five minutes. A safety culture pays off, not just for balance sheets, but for getting everyone home in one piece each day.
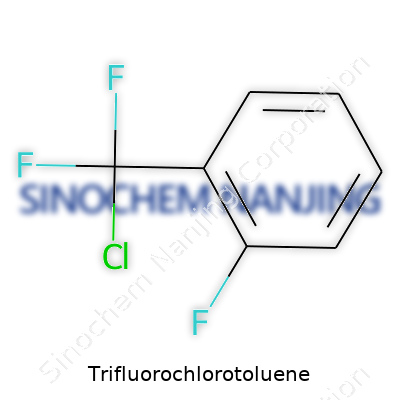
What is the chemical formula and structure of Trifluorochlorotoluene?
Getting to Know the Compound
Trifluorochlorotoluene walks a line between everyday organic chemistry and the more specialized work that shapes everything from crop protection to high-performance polymers. The real meat of its identity comes down to its molecular formula and the shape its atoms take in space. Chemically, it’s written as C7H4ClF3. To break this up, you're dealing with a seven-carbon frame—straight from toluene—where a chlorine atom and three fluorine atoms step in for some of those usual positions on the benzene ring.
The Structure Shows Its Character
Toluene brings a methyl group (–CH3) attached to a benzene ring. Trifluorochlorotoluene comes from this family, but it swaps hydrogen neighbors on the ring for three fluorine atoms and one chlorine atom. Put yourself back in your chemistry class, picturing a hexagonal benzene ring. Now, pick four spots on the ring and imagine one gets the methyl group, another picks up a chlorine atom, and three others invite fluorine atoms to the table.
The exact location of each atom matters. One common arrangement is 4-chloro-2,3,5-trifluorotoluene, where the methyl group sits at position 1, fluorines spread out at positions 2, 3, and 5, while chlorine lands at position 4. Each subtle change in this arrangement tweaks its properties and potential uses. The structure looks simple, but these choices ripple throughout chemistry, changing the way it behaves and interacts with other substances.
Why This Structure Matters in Real Life
Living and working around industrial labs made it clear that even tiny changes to a molecule’s shape or the atoms attached can send effects all the way through a chemical process. Fluorine, for instance, shrinks a molecule’s reactivity, makes it tougher, and often thwarts degradation by heat or strong chemicals. Chlorine brings in its own quirks, pulling on electrons and changing how the molecule links to others. These effects help explain why chemists turn to chemicals like trifluorochlorotoluene in the upstream design of pharmaceuticals and electronic materials.
It’s also hard to overlook what this chemical means for the environment. Adding fluorine and chlorine ups the stability, but raises new challenges. Some compounds with these atoms last a long time out in the world or resist traditional cleanup methods. Working with them in a lab or factory means wearing more layers of safety, closely tracking their movement, and keeping runoff in check. You can’t ignore waste management because compounds like this can hang around in waterways or get into soil if things go sideways.
Finding Smarter Paths Forward
Watching changes in chemical manufacturing over the past ten years, I’ve seen more teams push for “greener” approaches. That means recycling solvents where possible, using closed-loop systems, and looking for bio-based or less hazardous alternatives. Regulatory standards today expect traceability and environmental monitoring as a given, not a bonus. For trifluorochlorotoluene, progress might mean designing routes that use milder reagents, make less waste, and cut down risk to workers.
In short, the formula C7H4ClF3 and its specific benzene ring substitutions do far more than fill out a label—they steer the molecule’s uses, benefits, and challenges. Science keeps pushing for structures that balance technical performance and safety, and that’s not slowing down any time soon.
How should Trifluorochlorotoluene be stored?
Most people outside a lab barely give chemicals a thought. In research and manufacturing, though, safe chemical storage is about more than just ticking a box on a safety checklist. Trifluorochlorotoluene, a compound used in many specialty applications, sits in the “handle with care” category. I’ve seen too many storage rooms where someone shrugged and shelved a chemical, only to regret that shortcut later. Accidental mix-ups, labeling errors, or ignoring compatibility charts—it always seems like small oversights until something goes wrong.
The Challenge: Volatility and Health Risks
This compound gives off vapors—chemists often catch the scent before they see the hazard label. Trifluorochlorotoluene isn’t just another flask on the shelf; it acts much more like a volatile organic solvent than people assume. Inhalation, skin absorption, and accidental ingestion all spell out trouble, with mild to moderate toxicity that can irritate the skin, eyes, and lungs. Cumulative effects might not show up right away, but nobody looks back wishing they’d skimped on safety measures.
Safe Storage: Not Just About the Rules
Temperature swings and humidity affect stability. From experience, letting this substance sit out at room temperature for too long, especially during a summer heat wave, increases the odds of pressure build-up or decomposition. I always stressed using a chemical storage refrigerator or cool, well-ventilated storeroom set below 25°C. Remember that regular office fridges rarely meet lab safety standards and don’t control for hazardous vapor buildup.
Exposure to light can degrade trifluorochlorotoluene. That’s why I always opt for amber glass bottles—clear bottles invite trouble. Keep each container tightly sealed, as loose caps let vapors leak into the storage area. If labeling wears off, confusion starts, so waterproof labels kept legible and up-to-date save time and prevent disaster later.
Compatibility Matters: No Mixing Chemicals
Lining a shelf with incompatible chemicals is like setting a trap that springs when least expected. I separate trifluorochlorotoluene from strong oxidizers, acids, alkalis, and flammables. Flammable storage cabinets built from metal, vented to the outside, and grounded for static control work best. I've seen what happens when incompatible chemicals end up together: at best, a ruined experiment; at worst, a pricey cleanup, lab evacuation, or emergency response call.
Ventilation and Containment
Closed storage cabinets only go so far. Chemical fumes can sneak out, especially if seals age and crack. Labs that use local exhaust ventilation over storage units (such as chemical fume hoods or vented cabinets) avoid surprise vapor clouds. Inhalation isn’t just a minor inconvenience—dizziness or shortness of breath can lead to accidents or errors elsewhere in the lab.
Simple Protocols, Real Results
Spill kits at the ready, absorbent materials close at hand, and clearly posted emergency contacts make a difference. I push coworkers not to use open flames or eat and drink where volatile chemicals sit, and for good reason. No packed shelves, no crowding bottles—space in storage cuts down accidents when someone reaches for a container in a rush.
Training matters. Nobody picks up safety through osmosis. Hands-on lessons, walking folks through proper handling, and routine checks of shelved chemicals reinforce habits that last long after an initial safety seminar.
In this business, storing trifluorochlorotoluene safely isn’t about following rules blindly—it’s about caring for colleagues and valuing the time, money, and health of everyone in the room.
What are the possible health hazards of Trifluorochlorotoluene exposure?
What is Trifluorochlorotoluene?
Trifluorochlorotoluene, known in labs for its chemical toughness, shows up in manufacturing paints, coatings, and specialty chemicals. With its sharp smell and clear appearance, this solvent rarely makes the headline, but those working around it notice the fumes quickly.
Breathing Problems
No one forgets the blast of harsh air in the lungs when walking into a room where solvents fill the air. This stuff gives off vapors that hit the nose and throat hard. Workers in confined spaces without good ventilation can end up coughing, with tight chests, or headaches. Long-term exposure stirs up chronic bronchitis and carries risks for those already dealing with asthma or sensitive airways. Based on data from NIOSH, repeated breathing in of similar fluorinated chemicals eventually chips away at lung function.
Skin Contact: Not Just an Itch
There’s nothing minor about splashing this chemical on bare skin. In a hot shop or paint booth, sweat opens pores, turning a quick splash into a deeper burn. Rashes, redness, and sometimes blisters have shown up in folks working without gloves. Some cases linger, with eczema flaring up even after protective routines improve. Direct skin contact can lead to chemical burns over time, especially without soap and clean water close by.
Eyes and Accidents
Even the most careful worker fumbles sometimes. Trifluorochlorotoluene splashed in the eye stings more than soap. Annoying at the least, damaging at worst, it can scrape the surface, start up swelling, or even cause blurred vision if ignored. Under factory lights, these mistakes get costly fast.
Nervous System Risks
Solvents like this don’t just stick to skin—they pass into the bloodstream. With enough exposure, some workers report dizziness, lightheadedness, and nagging headaches that don’t shake loose. Chronic exposure carries the risk of impacting memory or slowing coordination. Studies from OSHA highlight that repeated solvent handling can add up, burdening the central nervous system over months or years.
Potential for Toxic Build-Up
While less common, the possibility of trifluorochlorotoluene accumulating in body tissues sparks concern. Nobody wants to end up screening for liver or kidney problems down the line. Long shifts handling drums or cleaning up spills without masks put workers especially at risk. As seen with other halogenated solvents, even moderate exposure can tax detox organs over time.
Solutions That Work in Real Shops
My own stint working around chemical baths taught me quick that good ventilation beats any fancy gadget for keeping fumes low. Tradespeople should have fresh air moving—fans and open doors do more than any “low odor” label promises. Gloves rated for chemical resistance, splash goggles, and fitted respirators turn accidents into minor annoyances instead of medical emergencies. Regular safety meetings help; swapping stories about close calls sticks in the mind more than reading labels ever will.
Companies should run exposure testing and encourage workers to rotate roles, cutting down on long, day-after-day exposure. Showers and clean work clothes matter too. Pushing for better awareness, from supervisors to shop floor hands, keeps everyone looking out for signs of trouble—red eyes, bad coughs, headaches—and not ignoring them until someone’s down for the count.
Responsibility and Constant Vigilance
Working with trifluorochlorotoluene means owning responsibility for personal safety and watching out for coworkers. Staying informed, pushing your boss for better gear, and reporting symptoms early changes more than any rulebook. Over time, health and peace of mind always outrank cutting corners on a tough job.
| Names | |
| Preferred IUPAC name | 1-chloro-2-(trifluoromethyl)benzene |
| Other names |
4-Chlorobenzotrifluoride p-Chlorobenzotrifluoride para-Chlorobenzotrifluoride PCBTF 1-Chloro-4-(trifluoromethyl)benzene |
| Pronunciation | /traɪˌflʊəroʊˌklɔːroʊtəˈluːiːn/ |
| Identifiers | |
| CAS Number | 98-17-9 |
| Beilstein Reference | **1718733** |
| ChEBI | CHEBI:87428 |
| ChEMBL | CHEMBL373965 |
| ChemSpider | 21585881 |
| DrugBank | DB14004 |
| ECHA InfoCard | 100.011.036 |
| EC Number | 602-048-00-5 |
| Gmelin Reference | 85447 |
| KEGG | C18799 |
| MeSH | D014276 |
| PubChem CID | 69702 |
| RTECS number | XO8575000 |
| UNII | 4M87A1T02D |
| UN number | UN3334 |
| Properties | |
| Chemical formula | C7H4ClF3 |
| Molar mass | 180.56 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic |
| Density | 1.375 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 3.7 |
| Vapor pressure | 3.6 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ -0.7 |
| Magnetic susceptibility (χ) | -61.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.463 |
| Viscosity | 0.889 cP (25 °C) |
| Dipole moment | 3.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 286.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | Trifluorochlorotoluene |
| Std enthalpy of combustion (ΔcH⦵298) | -2937.3 kJ/mol |
| Pharmacology | |
| ATC code | D08AX04 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 41 °C |
| Autoignition temperature | 535°C |
| Explosive limits | Lower: 1.1% Upper: 9.5% |
| Lethal dose or concentration | LD50 oral rat 13 g/kg |
| LD50 (median dose) | LD50 (median dose): 4,000 mg/kg (oral, rat) |
| NIOSH | GR4810000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | No REL recommended |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Benzotrichloride Benzotrifluoride Dichlorotoluene Trifluorotoluene Chlorotoluene |

