Trifluoroacetyl Chloride: Understanding Its Role and Relevance
Historical Development of Trifluoroacetyl Chloride
Trifluoroacetyl chloride showed up in the research labs during the strong push for new fluorinated chemicals in the mid-20th century, right when companies and researchers hunted for compounds with unique reactivity and serious volatility. My own early reading in academic archives turned up articles from the post-war era, where chemists experimented with fluorinated building blocks for specialty syntheses and high-tech applications. Its introduction into commercial catalogs followed patterns seen with many tailored halogenated reagents: at first, only large manufacturers or well-funded labs could obtain it, but interest from the pharmaceutical and agrochemical industries expanded its production and distribution steadily. This history reflects the broader shift in industrial chemistry as demand grew for compounds that could offer extreme properties—chemical toughness, high reactivity, and compatibility with modern organic transformations.
Product Overview and Importance
Trifluoroacetyl chloride, known for its distinctive chemical formula CF3COCl, stands out due to its ability to introduce the trifluoroacetyl group into a variety of organic molecules. Chemists like myself have seen just how useful this reagent can be when the goal is to make molecules that need a trifluoromethyl group in very specific spots. Some popular names you’ll hear in the lab or see in papers include trifluoroethanoyl chloride and perfluoroacetyl chloride. These synonyms hint at a shared chemical role: providing strong electron-withdrawing power and delivering a chlorine atom for selective functionalization. Its sharp, pungent smell makes its presence obvious in the lab, yet, its industrial appeal does not rest in comfort — it brings a potent set of reactions for those who know how to handle it.
Physical and Chemical Properties
A bottle of trifluoroacetyl chloride doesn’t look especially dangerous, but its colorless, volatile nature demands respect. You open that bottle, and even at room temperature, fumes will escape—the boiling point sits low, and its high vapor pressure means it evaporates rapidly. That volatility, coupled with strong corrosivity, has made it both valuable and tricky to work with. The molecule itself combines the trifluoromethyl group (–CF3), known for fierce electron withdrawal, with the highly reactive acid chloride function. These features shape much of its chemical behavior. Coming from my own experience in organofluorine labs, trifluoroacetyl chloride’s reactions can get out of hand easily if you don’t plan every step and control your setup rigorously. It hydrolyzes quickly on contact with water or even moist air, pumping out nasty fumes of hydrogen chloride and trifluoroacetic acid, so working under strictly anhydrous conditions isn’t just best practice—it’s a line between success and a ruined day.
Technical Specifications and Labeling
Those of us handling trifluoroacetyl chloride must rely on clear labels and documentation, both for safety and for technical accuracy. Its packaging always warns about corrosivity, toxicity, severe risk of burns, and respiratory hazards, not just to meet regulations, but because the dangers are really that severe. Chemists check purity (often above 99 percent for scientific uses), water content, and methods of verification like NMR or GC analysis. Reliable suppliers provide all that information, letting labs avoid surprises. If you’ve spent time restocking chemical shelves, you know the relief of seeing straightforward hazard symbols and storage guidance: keep it cool, dry, and separate from bases or nucleophiles that could trigger a violent reaction.
Preparation Methods and Synthesis Insights
Making trifluoroacetyl chloride involves the interaction of trifluoroacetic acid with chemical chlorinating agents. In labs and factories, reagents like phosphorus pentachloride, thionyl chloride, or oxalyl chloride serve well for this purpose. Each chlorinating method aims to remove water and substitute the hydroxyl group with a chlorine atom, but all options churn out hazardous byproducts requiring strict waste control. My own attempts at small-scale prep under expert supervision always reinforced how careful dosing and temperature control matters. Even seasoned chemists respect procedures—an exothermic surge or failure to capture corrosive vapors can endanger staff and equipment alike.
Chemical Reactions and Modifications
Once in the flask, trifluoroacetyl chloride acts as a powerful electrophile. Chemists, myself included, have relied on it to install trifluoroacetyl groups onto diverse starting materials—amines, alcohols, and thiols—for everything from pharmaceuticals to crop-protection chemicals. The trifluoromethyl group shifts the electronic landscape of any molecule it joins, and the resulting products resist metabolic breakdown, which is precisely why pharmaceutical developers eye this chemistry for next-generation drugs with improved stability. Catalysts and controlled conditions can tweak the selectivity or reaction rate, but experience still counts a great deal in getting clean, predictable results. Quench your reaction improperly, and you risk unleashing a storm of acidic vapors no matter how fancy your fume hood.
Synonyms and Naming Nuances
Chemical nomenclature, for all its formal rules, often creates confusion for those not steeped in the literature. Ask for trifluoroacetyl chloride in a research lab, and some colleagues will recognize perfluoroacetyl chloride or even trifluoroethanoyl chloride as the same thing. These names spring from different traditions—some focus on the acyl group, others on the perfluoro substitution. For regulatory or safety paperwork, the correct label makes all the difference, both in procurement and in training new lab members to match what’s on the bottle to the protocol on the bench.
Safety Concerns and Operational Standards
If you spend time with trifluoroacetyl chloride, you don’t forget the health risks. The substance doesn’t just irritate; it can burn on contact. That danger extends to the air: vapor exposure triggers coughing, airway constriction, and even long-term lung injury if handling is sloppy. Strict storage away from bases, water, or incompatible metals is just standard practice—it comes from hard lessons learned by earlier generations. Modern labs rely on gloves rated for acid chlorides, chemically resistant goggles, and thorough ventilation. Spill kits and emergency showers belong nearby. Training matters as much as the gear. I once watched a postdoc splashed by an unnoticed drop, leading to hours flushing the affected skin under running water. Now, that incident has a place in every training session I run on hazardous reagents.
Application Areas Driving Demand
Most trifluoroacetyl chloride ends up in synthetic organic chemistry, especially in the creation of pharmaceuticals and crop-protection agents where resistance to breakdown in tough biological environments gives products a commercial edge. Laboratories use it to introduce trifluoromethyl groups that boost stability, influence reactivity, and fine-tune molecular profiles for the final product. Specialty polymers and advanced materials push its use further, especially in fields seeking membranes, coatings, or specialty plastics with extreme chemical or thermal resistance. In my own lab stints, reactions with trifluoroacetyl chloride often formed the backbone of multi-step syntheses. New drugs and diagnostic agents frequently trace their lineage back to a simple acylation step powered by halogenated reagents like this one.
Current Research and Development Prospects
Research communities have not exhausted the potential of trifluoroacetyl chloride. I’ve seen growing interest in methods that cut down hazardous waste and reduce energy input for large-scale reactions, especially as environmental regulations get tougher. Recent literature highlights catalytic alternatives to classic acid chloride chemistry, aiming to lessen corrosive byproducts and streamline downstream processing. Teams continue to push for better process safety as well, using in-line monitoring, sealed reaction vessels, and more intelligent automation. Some groups now explore efforts to recycle byproducts from trifluoroacetyl chloride synthesis, linking chemistry development to corporate sustainability goals. Academic curiosity still drives basic studies on reactivity and mechanism—details that sharpen future applications or lead to new classes of functionalized molecules.
Toxicity Studies and Workers’ Rights
Toxicity has always shadowed the widespread adoption of substances like trifluoroacetyl chloride. Government agencies and industry watchdogs persistently highlight the acute dangers, and published animal studies confirm that both short-term inhalation and direct contact can inflict real harm. Chronic exposure has not been studied as deeply, but case reports and personal accounts from older chemists portray a risk that nobody wants to ignore. Training, clear communication, and rigorous enforcement of PPE are now the baseline in most places, with research labs and manufacturing sites routinely reviewing updated studies on long-term effects. My own habits reflect those lessons—a splash guard, a well-sealed waste bottle, and a willingness to slow down and check every step twice.
Looking Ahead: Future Prospects
The chemical world rarely stands still, especially with regulatory pressures and technical ambitions always at cross-purposes. For trifluoroacetyl chloride, the future will be shaped by two forces—the continuing need for powerful, reliable fluorinated reagents and the rising barriers to handling hazardous chemicals responsibly. Research keeps shifting toward safer, greener chemistry, and new trifluoromethylation techniques may eventually sidestep acid chlorides entirely. Still, its versatility and proven record in high-value synthesis will hold its place on chemical shelves for some time yet, even as teams innovate around its limitations. Any young chemist entering this field should know exactly why it demands respect, and how handling it safely opens doors in complex, life-changing molecular science.
What is Trifluoroacetyl Chloride used for?
What Brings Trifluoroacetyl Chloride to the Table?
Trifluoroacetyl chloride holds a steady place on chemical shopping lists, especially among those working on new pharmaceuticals and specialty materials. The presence of three fluorine atoms tightly bound to a carbon skeleton gives it qualities other reagents just can’t match. The compound acts as a toolkit item for chemists who need to add a trifluoroacetyl group—this changes the behavior of molecules in ways that often mean better stability or different reactions down the line.
Where It Shows Up: Real World Uses
Not every chemical leaves the laboratory and ends up in a product you touch every day. Trifluoroacetyl chloride, though, plays a clear role behind the scenes. During my time shadowing a process development team, I watched how this compound factored into steps toward antiviral and anticancer agents. Many modern drugs have special fluorinated parts that help them stick around in the body longer or reach their target more reliably. Trifluoroacetyl chloride adds these parts in a single step. Chemists value this efficiency.
Crop science also leans on it. Modern pesticides and herbicides often need to last through rain and sunlight, but not linger in soil for years. Putting a trifluoro group into those molecules often finds the right balance, and trifluoroacetyl chloride makes that possible at scale. It often goes unseen, but these transformations shape the safety and effectiveness of the products we rely on for food production.
What Makes Handling Trifluoroacetyl Chloride Tricky?
Even with its clear benefits, this compound demands respect. The gas it gives off has a punchy, biting smell, and routine lab gloves offer little protection. I learned early on that only well-ventilated spaces and specialized gear keep people safe. If spilled, it reacts fast with water, generating fumes nobody wants in their lungs.
This risk limits where small teams or teaching labs can use it. Large chemical plants install scrubbers and sealed systems, often backed by real-time air monitors and emergency plans. These steps call for investment, but cutting corners with materials like this one never ends well.
Troubleshooting Environmental Impact
Concerns over the long-term fate of trifluoro compounds continue to grow. The same stability that makes these chemicals valuable for drugs and farm products also means they break down slowly in nature. Some breakdown products can travel far from their origin and resist treatment at water plants.
People are looking for cleaner ways to work with these fluorinated building blocks. Alternative reagents that have a smaller environmental footprint draw interest. I’ve seen research teams push for recycling schemes where leftover material gets captured and reused. Tighter regulations, especially in the EU and parts of Asia, nudge companies toward greener chemistry—and create an incentive to consider every drop and vapor.
Better Approaches for Safer Chemistry
Education makes a difference. Any lab worker handling trifluoroacetyl chloride benefits from informed mentors and routine safety drills. Newer students sometimes rush, but strong training slows them down and saves trouble later. Transparency about hazards and honest reporting of accidents build trust across organizations and protect everyone.
Industry partners have started to share better protocols and collaborate with regulators. Open discussions about waste handling and air quality help set standards high. Changing habits isn’t always comfortable, but it tends to deliver long-run savings and higher confidence for everyone down the supply chain.
Looking Forward
Trifluoroacetyl chloride will likely stick around in advanced chemistry, especially where performance and precision outweigh easy alternatives. The challenge now comes down to making every reaction count, designing syntheses that leave fewer byproducts, and keeping both people and the environment safer along the way.
What are the safety precautions when handling Trifluoroacetyl Chloride?
Understanding What You’re Dealing With
Trifluoroacetyl chloride stands out in the lab. You know that sharp, pungent smell signals danger. It reacts with water, even moisture in the air, to unleash toxic gases such as hydrogen chloride and trifluoroacetic acid. Without gloves or goggles, blinking or breathing near this chemical can leave you in bad shape. Eyes start burning, skin itches or blisters, and lungs fill with stabbing pain. I’ve seen labs grind to a halt because someone overlooked these risks. Don’t trust your senses. By the time you smell it, you’re exposed.
Reliable Personal Protection
Start with your gear. Nitrile gloves alone won’t block everything. I grab two layers—chemical-grade gloves under nitrile. My face shield goes on before the bottle comes out. Splash goggles protect where safety glasses fail. Every coat I wear gets buttoned at the sleeve and collar. No bare patches. I met a coworker who got a few drops on his wrist. Two hours later, he’s looking at blisters and an ER bill. Thick lab coats won’t guarantee safety, but they buy you time when accidents happen.
Ventilation Isn’t Optional
A working fume hood is my lifeline. Even in rooms with fans blowing, vapors sneak out and hit unprepared staff. I keep the sash low and never reach above open fasks. It’s tempting to skip the hood for “just a quick transfer,” but I’ve seen shortcuts end with fire alarms and emergency cleanups. Managers should never let outdated hoods slide. Airflow meters keep everyone honest—you need to check those often. In a real emergency, the fume hood buys precious seconds that regular fans can’t.
Correct Storage Changes Everything
Trifluoroacetyl chloride belongs alone, sealed up, and away from moisture. I never put it near bases, solvents or oxidizers. Even a forgotten bottle left in a shared fridge spells disaster. Clear, tight labeling on a bottle saves lives—no one should guess at its contents. Silicone stoppers and Teflon-lined lids resist corrosion. Anything else risks leaks that can eat through shelves or send vapors drifting through the building. I’ve seen what happens when labels rub off and folks mistake one clear liquid for another. A locked, vented cabinet with a hazard sign isn’t overkill.
Staying Ahead of Accidents
Everyone in the room learns where the safety shower and eyewash sit. Practice drills matter. I remember fumbling with stiff levers in a real spill—those seconds can mean real harm. Folks who wait to read the safety sheet after an incident endanger everyone. Spills call for calcium carbonate or other neutralizers, not a handful of paper towels. Full-face respirators stand ready for a real leak. Supervisors and chemists should review emergency procedures face-to-face, not just on paper.
Growing a Culture That Cares
Chemicals like trifluoroacetyl chloride pull no punches. Respect for the risk can’t just live in a binder. People need regular stories and reminders. Peers should check each other’s protection before every use—no shame in a double-take. I trust folks who ask questions and share close calls. Lessons learned from past incidents grow sharper with each conversation and hands-on session. Safety lives in the habits of every lab worker and can’t thrive unless management values clear communication over speed.
How should Trifluoroacetyl Chloride be stored?
Why Storage Gets Personal
Spending years in chemical laboratories, some things stick with you. The acrid smell of poorly stored reagents. The frantic dash for the eyewash when a bottle handle gives out. Among all the substances handled, trifluoroacetyl chloride really demands respect. Not so much for its reputation as an industrial staple, but for its aggressive reaction to moisture and its impact on safety.
What Makes Trifluoroacetyl Chloride a Challenge?
It reacts hard with water — even humidity in the air. Opening the container in a muggy storeroom leads to fuming, choking gas clouds that spell instant trouble. Breathing that vapor irritates the lungs. Contact with skin can burn and blister. For me, memories of a coworker’s hand turning red after a spill left a mark. This isn’t just about chemistry. It’s about protecting people from expensive mistakes and real harm.
Solid Steps to Safer Storage
So, let’s skip complicated jargon and get straight to what keeps everyone out of the emergency room. Trifluoroacetyl chloride belongs in a tightly sealed glass or Teflon bottle — never metal, since it will corrode almost anything in a hurry. That container stays inside a desiccator or a dry box filled with silica gel or phosphorus pentoxide. Forget cardboard boxes or those chipped old jars in the storeroom; those let water in, and water sets off dramatic reactions.
A cool spot, away from direct sunlight or heat sources, protects the chemical from breaking down or expanding, which means less chance of leaks and spills. Never place it up high or in busy passageways; one bump could drop the bottle and unleash a nasty chemical cloud. In my labs, the best spot proved to be a locked, ventilated cabinet marked with a hazard sign — clear enough that nobody accidentally grabs the wrong bottle.
Don’t Let Labels Collect Dust
Relying on memory courts disaster. Labels on containers should always include the name, concentration, and date received. Sometimes, chemicals outstay their welcome. An old container of trifluoroacetyl chloride can produce acidic fumes and pressure, making old, forgotten bottles a real hazard. Regular inventory checks catch these before they become grenades waiting in storage.
Personal Protection Goes Hand in Hand
Even perfect storage won’t fix careless handling. Safety goggles, gloves, and a heavy-duty lab coat block accidental splashes. Simple habits — like keeping fume hoods on, opening only one bottle at a time, and never pipetting by mouth (that’s a lesson nobody forgets) — turn safe storage into a routine instead of a chore.
Everyone Can Raise the Standard
The responsibility belongs to more than just lab managers. Open training, real-world drills, and easy-to-read safety charts take the mystery out of chemical safety. Anyone walking into a storeroom should know what that yellow hazard diamond means.
Better policies pay off. University labs publish incident rates, while companies lose fewer days to accidents in shops that enforce real storage rules. Proper storage doesn’t just help avoid fines and surprise inspections; it keeps people healthy and work flowing without interruption.
Moving Forward: Culture Over Compliance
In the end, storing trifluoroacetyl chloride safely goes beyond ticking boxes for auditors. It’s about trust among coworkers, respect for health, and a sharp eye for the unexpected. Setting a higher bar for storage conditions means not just following rules, but protecting each other and getting home safely, every time.
What is the chemical formula of Trifluoroacetyl Chloride?
Understanding What We’re Working With
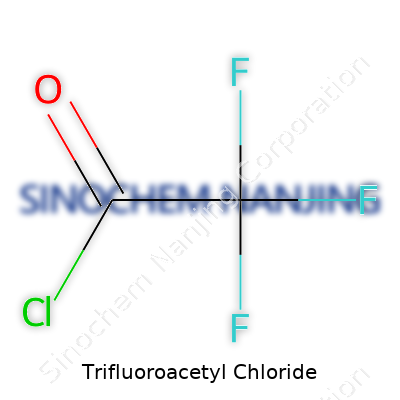
Everyday lab life throws dozens of chemicals at us, each with its own shorthand. When someone brings up trifluoroacetyl chloride, accuracy isn’t just a suggestion—it shapes the way we handle, transport, and think about risk. The chemical formula for trifluoroacetyl chloride is CF3COCl. Breaking it down, the molecule contains a trifluoromethyl group (CF3), a carbonyl (CO), and a chlorine atom attached. Nothing fancy, but it hides behind that tidy arrangement some real bite and real value.
Why Chemists and Industry Care About the Formula
I’ve worked in a busy analytical lab and learned pretty fast that mixing up chemical formulas messes with both safety and results. Trifluoroacetyl chloride stands out because of its reactivity. In the hands of pharmaceutical chemists, it helps build complex molecules, often acting as an acylating agent to introduce the trifluoroacetyl group into organic compounds. Drug discovery leans on tiny variations in structure for big jumps in activity. So knowing that there’s a trifluoromethyl group and a chloride sitting on the same molecule hints at its intense behavior.
Outside the lab, industries making specialty polymers or agrochemicals keep this compound close by for the same reasons. The strong electron-withdrawing effect of the trifluoromethyl group changes the way target molecules perform, adding stability or ramping up a product’s disease-fighting strength. Getting the formula right means the correct raw materials arrive, contracts get fulfilled, and nobody risks safety over a paperwork slip-up.
Safety Is Not Just Fine Print
I remember a spill drill—trifluoroacetyl chloride was mentioned in the briefing. Its formula pointed us straight to what gear we grabbed and how we treated exposure. The molecule can hydrolyze quickly, producing hydrogen chloride (HCl) and trifluoroacetic acid (TFA), both hazardous. The very presence of both the trifluoromethyl and the acyl chloride groups means the reaction on contact with water or skin is more aggressive than most everyday chemicals.
Ignoring these details can lead to injuries. Clear labeling and strict adherence to handling procedures, all driven by an accurate understanding of the formula, set the tone for safe workspaces. Emergency decontamination stations, good ventilation, and up-to-date training don’t happen by accident. Safety data sheets rely on formulas that somebody double-checked.
Where Precision Pays Off
Mistakes around formulas can lead straight into regulatory trouble. The European Chemicals Agency and the US Environmental Protection Agency track trifluoroacetyl chloride because its use connects directly to environmental risks. Leaks or improper disposal raise the stakes. When regulators arrive, they look for correct paperwork, hazard communication documents, and proof that teams are working with the right data.
Standardization starts with the right formula on the label, on digital records, and in every logbook. Young chemists get drilled on formulas for a reason—they set off a series of decisions about protective equipment, ventilation, and waste management. If the chemical formula confuses someone or ends up wrong on a container, penalties can follow. Facts matter in the real world, and a slip on formula accuracy leads straight to problems you really want to avoid.
What are the potential hazards of Trifluoroacetyl Chloride exposure?
Unpacking the Risks
Trifluoroacetyl chloride carries a reputation in chemical circles as a harsh substance. Its official use looks specialized—common in making pharmaceuticals, agrochemicals, or advanced chemical intermediates. Spend enough time in a lab or industrial setting and you’ll hear rumors about its reactivity and what can happen if things go wrong. My own time in a research facility made it clear: safety protocols are non-negotiable. One leaky fume hood or careless mistake, and you’re dealing with more than paperwork.
Harsh on the Body
Even small amounts can trigger trouble. Inhalation means trouble breathing. The gas version irritates mucous membranes and can go straight for the lungs, leading to coughing, chest tightness, and sometimes chemical pneumonia. Liquid splashes burn on contact. Eyes sting and water, or worse, lose vision. Get it on skin and you’re in for a painful inflammation that doesn’t take long to show up. Long sleeves and goggles matter because trifluoroacetyl chloride asks for respect. No shortcuts.
Why Safety Can’t Take a Backseat
I’ve watched coworkers—some new to the field—underestimate fumigants like this one. They wore gloves, maybe skipped the splash shield, didn’t check ventilation. A quick task can turn serious with this stuff, especially due to its fast-release hydrogen chloride fumes. In a poorly-ventilated space, gases collect quickly. Exposure can build up before anyone notices. Data from the CDC and chemical safety boards show respiratory issues rank as the most common symptom in accidental releases, followed by eye and skin burns.
Environmental Damage Runs Deep
A spill in the lab or at a production site has consequences beyond the building. Trifluoroacetyl chloride reacts strongly with water, forming hazardous byproducts. Hydrochloric acid isn’t something anyone wants leaking into drains or soil. Sensitive aquatic life and plants can’t fight back—the result is often mass die-off and long-term contamination. Incidents in places with poor emergency preparation tend to grab local news headlines fast.
Practical Safety Steps
It takes more than a binder of SDS sheets sitting on a shelf to get safety culture right. Real protection comes from gear that fits, ventilation strong enough to keep the air moving, and training that sticks. Teams need regular drills, not only theoretical rules. Safety showers, eyewash stations, and acid-resistant gloves can make the difference in a worst-case scenario. Small labs can collaborate with safety consultants for tailored risk assessments, building redundancy into systems for storing or handling volatile chemicals.
Accountability Isn’t Optional
It’s tempting to believe experience itself offers a shield. I’ve seen even seasoned professionals get overconfident, but trifluoroacetyl chloride doesn’t care how many years you’ve worked in chemistry. Accountability helps everyone stay sharp. Supervisors and lab managers serve as a backstop, but peers can reinforce safe habits, too. Open reporting of near-misses gives valuable lessons for the future.
Moving Toward Safer Workplaces
Safer alternatives to trifluoroacetyl chloride exist for some processes. R&D teams can prioritize less volatile, less toxic reagents in protocol development. Meanwhile, investment in new containment technology and automated chemical handling trims exposure for everyone on site.
Everyday Vigilance Saves Lives
My own career sharpened my sense that real safety grows from habits, not just rules. Trifluoroacetyl chloride stands as a reminder that even one misstep can upend lives—inside and outside the lab. Each person handling it deserves both the equipment and the culture that keeps them safe.
| Names | |
| Preferred IUPAC name | Trifluoroacetyl chloride |
| Other names |
Trifluoroacetyl chloride Trifluoroacetic acid chloride Perfluoroacetyl chloride TFA chloride Acetyl trichloride fluoride |
| Pronunciation | /traɪˌflʊəroʊ.əˈsiːtɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 354-32-5 |
| Beilstein Reference | 1209225 |
| ChEBI | CHEBI:35596 |
| ChEMBL | CHEMBL141360 |
| ChemSpider | 12635 |
| DrugBank | DB13524 |
| ECHA InfoCard | 100.003.965 |
| EC Number | 205-204-8 |
| Gmelin Reference | 80040 |
| KEGG | C18703 |
| MeSH | D014264 |
| PubChem CID | 66034 |
| RTECS number | AJ3675000 |
| UNII | R8QQZO8R6O |
| UN number | UN3252 |
| CompTox Dashboard (EPA) | DTXSID8031637 |
| Properties | |
| Chemical formula | C2ClF3O |
| Molar mass | 114.47 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.419 g/mL at 25 °C (lit.) |
| Solubility in water | Reacts slowly |
| log P | 0.9 |
| Vapor pressure | 400 mmHg (20 °C) |
| Acidity (pKa) | -0.26 |
| Basicity (pKb) | 15.06 |
| Magnetic susceptibility (χ) | -37.5e-6 cm³/mol |
| Refractive index (nD) | 1.298 |
| Viscosity | 0.539 cP (20°C) |
| Dipole moment | 2.414 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 297.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -727.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -726.8 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H250, H301, H314, H330 |
| Precautionary statements | P210, P261, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P311, P312, P363 |
| NFPA 704 (fire diamond) | 3-0-1-W |
| Flash point | Not below -18°C (0°F) (closed cup) |
| Autoignition temperature | 400 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 250 mg/kg |
| LD50 (median dose) | LD50 (median dose): 300 mg/kg (rat, oral) |
| NIOSH | AC7285000 |
| PEL (Permissible) | PEL: 2 ppm |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 10 ppm |
| Related compounds | |
| Related compounds |
Acetyl chloride Trifluoroacetic acid Trifluoroacetamide Trifluoroethanol Trifluoroacetic anhydride |

