Trifluoroacetone: Behind the Chemistry and the Future Ahead
Historical Development
Chemistry moves forward on the shoulders of both great discoveries and overlooked molecules, and trifluoroacetone fits into the second group for most people outside the lab. Some might think of acetone as the familiar ingredient in nail polish remover, but researchers began exploring fluorinated analogues like trifluoroacetone as early as the middle of the last century. Meeting rising interest in organofluorine compounds, chemists sought out new methods of introducing strong electron-withdrawing groups into simple molecules, chasing new reactivity. Trifluoroacetone soon ended up in many lab notebooks as researchers searched for alternatives to traditional carbonyl compounds, expanding the toolset for making pharmaceuticals and advanced materials. The journey of this compound didn't happen overnight—it reflects how synthetic chemistry adapts to the needs of industry and innovation. The compound’s progress from a rarely discussed lab curiosity to a valuable building block highlights the way the chemical industry tracks upstream and downstream effects, especially once green chemistry and process efficiency became mainstream topics.
Product Overview
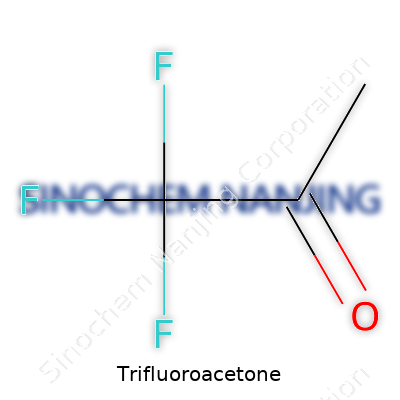
Trifluoroacetone carries the molecular formula CF3COCH3, setting it apart because that trifluoromethyl group pushes and pulls electrons in ways that ordinary acetone just can’t achieve. The presence of three fluorines reshapes its behavior both in reactions and in the way it appears physically: clear, colorless, with a sharp, biting smell that’s hard to forget once you’ve handled it. This characteristic mix of volatility and electronegativity has opened up uses in research and some limited commercial applications. Chemists see this compound as far more than a cousin of acetone—with trifluoroacetone, fluorine’s electronegativity goes toe-to-toe with the carbonyl group, making the compound uniquely reactive in specific situations. The specialty nature of this compound means it rarely sits on hardware store shelves, but demand does turn up among labs looking for strong electron-withdrawing effects in synthesis.
Physical & Chemical Properties
Handling trifluoroacetone shows why respect for chemical volatility matters. It boils easily under standard conditions, turns from a clear liquid to vapor with only a modest rise in temperature, and carries its own strong, pungent smell. The density comes in higher than water, and it mixes with organic solvents, though it does not blend well with water itself. The electron-withdrawing effect of the CF3 group shows up not just in how the molecule behaves in reactivity, but in its straightforward behavior in the bottle. The compound’s reactivity makes it less prone to self-condensation than acetone, yet its carbonyl group remains open for attack in nucleophilic addition reactions. The combination of volatility, strong odor, and sharp chemical activity makes even basic handling a job for the well-prepared.
Technical Specifications & Labeling
Packaging, labeling, and documentation for trifluoroacetone reflect the realities of working with both a volatile solvent and a potent chemical intermediate. Bottles come with robust seals, and safekeeping requires cool, well-ventilated spaces. What matters more to users than packaging are the quality guarantees like GC assay results, certificate of analysis, and compliance with applicable shipping regulations. Flammability and toxic potential stand out among the hazard ratings, meaning safety labeling carries pictograms and straightforward warnings, even for small research quantities. Storage recommendations follow expectations for volatile, reactive organics, and documentation typically makes clear not only the precautions but the real-life risks from improper handling. From my experience, those working with trifluoroacetone take documentation seriously because ignoring the hazards can turn an experiment into a dangerous event in seconds.
Preparation Method
Traditionally, making trifluoroacetone starts with precursors carrying the trifluoromethyl group, reacting them through tightly controlled hydrolysis or substitution pathways. Industrial synthesis tends towards the trifluoroacetylation of methyl organometallics or careful dehydrohalogenation of trihalogenated ketones. These processes routinely involve strong acids, bases, and sometimes specialized catalysts that need careful use to avoid unwanted side reactions or hazardous byproducts. Researchers care about yield and purity, but avoiding contamination with other fluorinated byproducts stands just as important. The efficiency and selectivity of these routes often decide which supplier ends up favored by specialized chemical firms, as the cost and purity both drive choices for labs running high-precision benchwork.
Chemical Reactions & Modifications
The true utility of trifluoroacetone shows up in its role in advanced organic synthesis, where it offers something that non-fluorinated analogues cannot provide—the electron-withdrawing power of the trifluoromethyl group tips the scales in favor of certain reaction pathways. Chemists leverage that effect for direct nucleophilic addition, enolate formation, and as a strategic handle for introducing fluorine into aromatic or heterocyclic structures. In practice, the molecule stands out during routine reactions like Mannich or aldol condensations, permitting control over selectivity or the stability of intermediates. Introducing a trifluoromethyl group using acetone as a platform means downstream molecules often benefit from increased metabolic stability or altered biological activity, which drug developers value when exploring new leads. These benefits don’t come without challenges—side reactions and sensitivity to certain bases or nucleophiles sometimes force extra caution and test-run optimization of reaction conditions.
Synonyms & Product Names
Across catalogs and scientific papers, trifluoroacetone may carry alternative names like 1,1,1-trifluoropropan-2-one or methyl trifluoromethyl ketone. Sometimes sources refer to it by its CAS number, which helps avoid confusion with related compounds. No matter the label, asking for trifluoroacetone at a specialty supplier gets you a volatile, clear liquid reserved for laboratory and synthesis work, not day-to-day use outside research or manufacturing settings. Names may shift, but the role and risks of the substance remain unchanged.
Safety & Operational Standards
Lab safety with trifluoroacetone isn’t just about hazard labels. Real-world handling of volatile, reactive compounds puts even experienced chemists on alert. Goggles, gloves, and fume hoods always show up with this liquid; nobody takes shortcuts after smelling its sharp, eye-watering vapor. Regulations focus on preventing inhalation and limiting exposure, and procedures usually require running reactions behind blast shields, with exhaust systems to clear any stray vapors. Cleanup teams get special training because a spill can fill a whole lab with overwhelming fumes. Disposal requires solvent recovery protocols and tight tracking of fluoride-containing waste. Experience in the lab teaches that minute lapses can bring headaches, or worse, so training stays thorough, documentation remains up-to-date, and storage keeps the bottle away from heat, light, and incompatible chemicals. The increasing attention to personnel safety wouldn’t mean much without solid operating procedures, and the chemicals industry has pushed up its standards to match increased scrutiny and regulatory oversight.
Application Area
Trifluoroacetone never made it into household products, but in chemical research, its niche is secure. Fluorinated building blocks attract strong interest from companies and academic groups seeking new pathways for drug development, agrochemical innovation, and the creation of specialty polymers. Medicinal chemists welcome the trifluoromethyl group for what it brings to molecular stability and activity. The compound also shows up as a diagnostic reagent for nuclear magnetic resonance work, using that unique fluorine resonance to probe the chemical environment inside test molecules. For chemists working on new catalysts or materials with non-stick or dielectric properties, trifluoroacetone serves as an indispensable intermediate. Its cost and handling challenges keep it squarely in the realm of targeted research and pilot-scale production, not routine industrial synthesis. Nevertheless, progress in medicinal chemistry and specialty materials points toward modest but continued demand.
Research & Development
The story of trifluoroacetone rides on the broader surge of interest in fluorinated molecules that reshaped organic chemistry in the last few decades. The push towards drugs and polymers with improved properties keeps researchers returning to the building block, searching for better ways to make, use, and modify it. Recent advances in green chemistry prompt questions about safer, less wasteful synthesis routes. Academic papers delve into regioselectivity and the development of new catalysts to control introduction of the trifluoromethyl group. Investment in analytical methods—like improved gas chromatography and fluorine NMR—lets chemists handle the compound with greater precision, reducing waste and improving reliability of results. Collaborative projects with industrial partners aim to adapt these research insights to scalable processes, offering small steps toward safer, cleaner manufacturing that can keep up with new regulatory limits and a demand for higher product purity in specialty applications.
Toxicity Research
Handling organofluorine compounds like trifluoroacetone means watching for toxicological risks, both familiar and unique. Animal data on this compound remains less well-known than for some related chemicals, but experts approach it with caution, considering its volatility and ability to penetrate tissues readily. Published studies show potential for irritation and central nervous system effects from inhalation or prolonged skin contact. Exposure limits reflect this risk, and occupational guidelines recommend aggressive use of personal protective equipment and engineering controls. Management of waste and air emissions falls under tight scrutiny, knowing that fluorinated compounds can have long lives in the environment. Accidents involving concentrated vapors sometimes trigger respiratory distress or eye injury, requiring immediate medical intervention. Ongoing research continues to track metabolic breakdown products and their impact, as fluorinated molecules often resist degradation and can persist in living systems for long periods. Toxicity profiles from related trifluoromethyl ketones nudge researchers toward caution, even as demand for practical toxicological data pushes laboratories to collect more real-world exposure information.
Future Prospects
Looking forward, interest in trifluoroacetone probably won’t explode, but it won’t vanish either. Its role as an intermediate in high-value, low-volume applications remains strong as synthetic and medicinal chemistry marches forward. Researchers continue searching for greener manufacturing pathways, seeing opportunity in catalytic methods that reduce waste and exposure. Regulatory pressures around fluorinated compounds—especially persistent environmental contaminants—will keep the field on its toes, driving innovation in containment, mitigation, and waste treatment. The steady pace of drug discovery guarantees ongoing demand for trifluoromethyl building blocks, as few chemical tricks offer the same ability to tune biological properties and metabolic stability. Experience in research and industry points to a bumpy road for large-scale expansion so long as safety and cost concerns dominate. Still, as chemical technology advances, trifluoroacetone stands ready to be part of the solution, connecting past insights with fresh opportunity in the chemistry labs of tomorrow.
What is trifluoroacetone used for?
Seeing Behind the Chemical Name: What Trifluoroacetone Actually Offers
Trifluoroacetone sounds like the sort of compound only a specialist might care about. But, scratch the surface, and you find a chemical that’s pushed along plenty of the modern progress we take for granted. The world of chemistry has a handful of these unsung players—compounds that hide behind names most people have never heard, but quietly shape technologies and research.
A Closer Look: From Labs to Industries
This clear, strong-smelling liquid belongs to a class of chemicals called fluoroacetones. It packs three fluorine atoms, which turns it into much more than a simple solvent. In organic chemistry labs, trifluoroacetone works as a building block. Chemists craft more complex molecules from it, often using the trifluoromethyl group as a key part. That group boosts the stability or biological activity of countless products, including pharmaceuticals and agrochemicals.
From my own time in research, I’ve seen grad students light up when their synthesis finally clicked, tracing the success back to a bottle of trifluoroacetone. It’s not flashy, but it’s fundamental. You need a way to introduce fluorine atoms into carbon-based molecules, and this is one of the cleaner, more reliable choices. Drug scientists add fluorine because it can improve how long a medicine lasts in the body, or tune how it interacts with enzymes. A review in the Journal of Medicinal Chemistry points to the growth in fluorinated drugs: over 20% of new small-molecule pharmaceuticals contain fluorine. Without a starter like trifluoroacetone, much of this innovation would lag.
Everyday Impact: Electronics and More
Moving from the lab to factories, trifluoroacetone’s utility keeps growing. In electronics, manufacturers need tricks for making semiconductors, OLED displays, and specialty coatings. Trifluoroacetone enters as a precursor—meaning it acts as a chemical stepping stone, helping add special features to circuit boards or electronic materials. These tweaks can mean the difference between a phone battery that swells up after six months and one that stays stable for years.
Making Trifluoroacetone: The Environmental Angle
With all its uses, there’s pressure to keep its manufacture efficient and safe. Traditional production methods rely on heavy-duty fluorine chemistry, which can turn nasty quickly. Nobody living near a chemical plant wants leaks, and chemists feel the same way about the fumes in their own labs. These concerns have led to new techniques using milder conditions or less toxic precursors. Some startups now use more sustainable sources of fluorine or integrate recycling loops to collect any unused byproducts. The shift is gradual—cost still drives decisions—but we’re starting to see signs that green chemistry isn’t just talk anymore.”
Risks and Responsibility
No chemical with the power to alter molecules or electronics comes without safety flags. Trifluoroacetone brings its own: inhaling its vapors burns the nose and throat, long-term exposure can irritate the skin, and disposal takes care. Regulatory bodies like the EPA and ECHA weigh in with guidelines. It’s easy to ignore these warnings—especially in a fast-moving lab—but the only way for the value of trifluoroacetone to outweigh its risks rests in responsible use, reliable containment, and a steady eye on new research for safer alternatives. The story isn’t about banning useful chemistry; it’s about demanding respect for every tool that keeps the wheels of innovation turning.
What is the chemical formula of trifluoroacetone?
Breaking Down the Chemical Formula
Trifluoroacetone’s chemical formula looks short and sweet: C3HF3O. Traditional textbooks show it as CF3COCH3. What this means is one acetone molecule has three of its hydrogen atoms swapped out for fluorine. That sounds simple, but adding fluorine atoms changes a lot about how this molecule behaves and how people can use it in labs and industry.
Fluorine’s Power in Chemistry
Speaking from a chemistry background, swapping in fluorine atoms doesn’t just change the name. Fluorine, being the most electronegative element on the periodic table, pulls electrons harder than almost anything else. That pull changes the landscape for this molecule. It stops enzymes and other molecules from cozying up the same way they do with plain old acetone. So, in reactions, trifluoroacetone doesn’t play nice with the same partners.
Real Uses in the Lab and Industry
Trifluoroacetone sometimes feels niche, but it holds a real place in research and manufacture. Synthetic chemists reach for it to build fluorinated organic compounds, which can land in medicine, agricultural chemicals, or specialty materials. Say you want to make a molecule that resists breakdown in the body or environment — adding a trifluoromethyl group often does the trick. That’s tricky, though, because these compounds can persist where people don’t want them.
Work in pharmaceutical labs puts emphasis on such molecules, since adding fluorine can create drugs that stick around in the bloodstream longer, making dosing more predictable. But this isn’t just about effectiveness; with longer life and more environmental persistence, problems show up down the line. Perfluoroalkyl substances can persist in water, the soil, and animal tissue, becoming what scientists call “forever chemicals.”
Health and Environmental Concerns
Having spent years tracking chemical contamination issues, I’ve seen the worry spurred by fluorinated compounds. Trifluoroacetone isn’t exempt from those concerns. Acute exposure risks can be managed in a controlled lab, but environmental leaks go further. Water treatment plants have trouble breaking these molecules down, which means trifluoroacetone and similar chemicals can migrate far from the original source.
The U.S. Environmental Protection Agency and agencies in other countries now move to set limits on certain fluorinated chemicals, acknowledging the long-term risks. The rules tighten, but companies still push to invent new fluorinated products, aiming for effectiveness despite the baggage.
Better Chemistry, Safer Choices
Safer chemistry starts with transparent data. Research needs to look not just at a molecule’s immediate appeal but its full life cycle — how it’s made, used, and disposed. It’s easy in the lab to focus on getting a clean reaction, but hardier, persistent molecules require scientists and companies to design with the end of life in mind. Using greener alternatives, where possible, keeps labs safer and oceans cleaner. In my experience, firms succeed best when they invest in cleanup methods upfront: carbon filtration, advanced oxidation, or incineration.
Knowing the formula for trifluoroacetone lets scientists and industry advance. Checking the downstream impact, though, means this knowledge stays anchored to both discovery and responsibility.
Is trifluoroacetone hazardous to health?
What Is Trifluoroacetone?
Trifluoroacetone shows up mostly in chemical labs, as a strong building block for making specialized molecules. People handling it rarely see it outside controlled environments. Its strong smell and volatility make it impossible to ignore once a bottle is open. I remember handling it in grad school, and our lab hood felt like the only safe place to take out the vial. It made my eyes water after a brief whiff. Right from the start, that experience shapes my view on how risky it can be.
Acute Effects and Direct Hazards
Most people don’t realize that even small exposures to chemicals like trifluoroacetone can lead to real symptoms. According to Sigma-Aldrich and similar safety sheets, a splash or strong vapor exposure may irritate the eyes, skin, or lungs. Highly volatile liquids reach the nose quickly. I’ve felt mild headaches and throat tightness after removing my mask too soon—reminders to double-check my protection protocols.
Toxicity data for trifluoroacetone is still limited compared to everyday solvents, but animal studies suggest that strong doses bring trouble quickly: nervous system symptoms, breathing problems, even more serious reactions if someone has underlying health issues. Most manufacturers urge eye protection, lab coats, and proper ventilation, which stays true across every academic or industrial setting.
Long-Term Exposure Concerns
Information about long-term health effects of trifluoroacetone lags behind other well-studied chemicals. No one wants to roll the dice with cumulative toxicity, especially when fluorinated compounds show resistance to breakdown. Other fluorinated substances, like PFOA and PFOS, linked to chronic illness and environmental persistence, push many chemists to take extra cautions. That lingering uncertainty encourages anyone regularly around trifluoroacetone to treat it with more respect, even if hard data hasn’t landed yet.
Safer Work Habits and Better Policies
I trust the basics: wear gloves, use a fume hood, keep containers tightly closed, and store materials according to fire codes. Some labs introduce mandatory training for every student, walking through real-life mishaps to help those new to tricky chemicals. Running spill drills made a huge difference in my confidence and awareness. These habits reduce the chance of exposures, while regular safety audits catch problems before someone gets hurt.
On a broader level, better tracking of workplace exposures and more open reporting among chemists and lab staff build a much clearer picture of risks over time. Sharing case studies—those stories you don’t see published—helps drive improvements faster than waiting for a textbook update. Encouraging labs to swap out high-hazard compounds with safer alternatives, when possible, further shrinks health risks over time.
Accountability and Ongoing Learning
Working with unfamiliar chemicals teaches humility. Too many researchers assume new reagents equal new risks, but trifluoroacetone proves that chemistry’s old workhorses deserve the same respect. If anything, experience should push us to keep learning—not just about the chemicals themselves, but about honest safety culture. By listening to firsthand accounts, double-checking safety data, and refusing shortcuts, labs give people the freedom to ask questions and report problems early. That’s where real progress takes root.
How should trifluoroacetone be stored?
Looking Closer at Trifluoroacetone
Trifluoroacetone doesn’t show up in most households. In labs, though, it’s a different story. Most chemists see its sharp smell, low flash point, and volatility as warning signs. I remember opening a fresh bottle in grad school — the fumes alone turned heads across the bench. This isn’t a chemical that lets you cut corners. A simple slip can mean an evacuation or a frantic shower in the safety station.
Key Facts Behind Safe Storage
Storing trifluoroacetone calls for common sense and a clear understanding of its hazards. Flammable liquids—especially those with low boiling points—never belong near heat sources or sparks. That’s easy lab safety, but it’s surprising how people still miss this. Vapors can sneak out and find their way to open flames or even a static discharge. Mixing oxygen and trifluoroacetone with a bit of heat creates problems nobody wants.
Here’s reality: temperature swings cause bottles to sweat and vent. One summer, a coworker left trifluoroacetone close to a sunny window. The bottle started leaking, and nobody noticed until that pungent odor crept down our hallway. We lost half a day cleaning up. Lesson learned. Always stash it in a flammable materials cabinet with tight seals and chemical-proof shelving, out of direct sunlight and away from temperature extremes.
Choosing the Right Container
Not all bottles stand up to aggressive chemicals. Trifluoroacetone likes to chew through plastics. Glass bottles with Teflon-lined caps work best. Swapping out any damaged or questionable lids always beats gambling with vapor leaks. Years back, I saw a cracked cap lead to a slow release in a shared fridge. Corrosive fumes played havoc with labels and rusted out shelving. Sometimes cheap containers cost more in the long run.
Know the Neighbors
Mixing strong acids, bases, or oxidizers with trifluoroacetone doesn’t end well. Organize storage so incompatible bottles don’t sit on the same shelf. I once found a bottle of trifluoroacetone too close to a drum of sodium hypochlorite— enough to make anyone nervous. Separating substances by chemical group sticks in your mind after a mistake nearly sends you to the emergency room.
Checking the Label and Logs
Labeling with clear dates, hazards, and owner information helps in busy shared labs. It’s easy to forget what’s what if bottles pile up. Spot checks—checking the bottle, shelf, and logbook—avoid surprises. Professional or home labs shouldn’t skip these steps. Everything flows smoother when people know who’s handled what, and when they last checked the stash. The buddy system works almost everywhere, but especially with volatile chemicals.
Real Solutions for Everyday Storage Problems
A few simple habits change the game. Flammable cabinets cut risks dramatically. Good ventilation whisks away stray vapors. Always wear gloves, splash goggles, and a lab coat when moving or opening the bottle. Setting up spill control kits, eyewash stations, and clear exit routes makes a difference, too. Clean storage spaces build a safety culture—newcomers pick up on the habits, old-timers stay vigilant.
Instituting lab training on chemical storage keeps everyone sharp. These rules stick with you long after school ends, shaping good decisions for years. Every time someone follows that three-point checklist—check compatibility, container, location—they nudge the odds further in their favor.
Choosing to treat trifluoroacetone with respect just makes sense. Every careful step guards people, buildings, and time—letting science unfold without drama.
Is trifluoroacetone soluble in water?
Looking at Trifluoroacetone Side by Side with Water
Ask a chemist about trifluoroacetone and you’ll probably get a mix of interest and caution. For most folks, it’s not a household name, but this compound has some interesting quirks. The heart of the question: can it truly dissolve in water? Based on decades fooling around in chemical labs, understanding why things dissolve—or don’t—starts at the molecular level.
Trifluoroacetone grabs your attention with its three fluorine atoms attached to a carbonyl structure. These fluorine atoms pull electrons toward themselves, making one end of the molecule pretty “electron-hungry.” Water molecules, being famously polar and full of ambition, want to mingle with other polar substances. Usually, “like dissolves like” is a chemistry golden rule. But trifluoroacetone tells a messier story.
The Facts: Mixing Trifluoroacetone and Water
Plenty of data points out that trifluoroacetone does dissolve in water, but not without limits. You can stir this compound into water and it won’t just float on top; it’ll sneak right in. That’s no surprise, given those fluorines, which pull those little water molecules in for hydrogen bonding. But as concentration goes up, so does the trouble: too much trifluoroacetone and the blend becomes cloudy, leading to separation rather than true mixing.
This solubility plays a role in industry, research, and environmental contexts. In lab work, you want reactions to occur as neatly and safely as possible. Water’s reputation as a solvent is hard to beat—less toxic, cheap, and easy to get rid of after you’re done. If a chemical like trifluoroacetone fits in with water, it smooths out workflows, keeps waste streams less complicated, and sometimes shaves real money from a project’s budget. These practical points matter much more than textbook theories when the goal is to solve problems at the bench.
Environmental Side of the Story
Mixing into water isn’t just a lab convenience. Once chemicals land in rivers or lakes, things get serious. Trifluoroacetone’s ability to blend with water raises its mobility; it moves with water currents and might travel far from where it was used or spilled. That increases the challenge for everyone worried about runoff or contamination. Agencies like the EPA closely track chemicals that dissolve easily, because cleanup and containment become trickier. And those fluorines? They bring unique health and environmental concerns—perfluorinated compounds can stick around for decades.
Solutions and Smarter Handling
There’s no silver bullet here. Respect for the compound begins with rigorous labeling and safe storage. In the field, researchers and engineers have pushed for more closed-loop systems, catching and recycling vapors or liquid waste so that fewer molecules escape into drains.
Pushing for water treatment advances matters just as much. Specialized filters can sometimes snatch tricky organofluorine compounds from wastewater. Close partnerships between chemists, chemical engineers, and public health experts speed this along—a lesson learned after previous messes with similar chemicals. Everyone benefits when the folks handling these materials know their risks as much as their uses.
Why Solubility Isn’t Just an Academic Exercise
The answer about trifluoroacetone’s solubility is clear in a flask, but in the real world, things get cloudier. The chemical community today stands on science, personal experience, and a pretty big sense of responsibility. If more labs and factories focus on how their chemicals move through water, we all enjoy cleaner rivers and safer cities. Sometimes, chasing answers about a single molecule lights up a much wider conversation worth having.
| Names | |
| Preferred IUPAC name | 1,1,1-Trifluoropropan-2-one |
| Pronunciation | /traɪˌflʊəroʊəˈsiːtoʊn/ |
| Identifiers | |
| CAS Number | 431-54-5 |
| Beilstein Reference | 390386 |
| ChEBI | CHEBI:38941 |
| ChEMBL | CHEMBL1230393 |
| ChemSpider | 7006 |
| DrugBank | DB04131 |
| ECHA InfoCard | 100.017.763 |
| EC Number | 206-975-3 |
| Gmelin Reference | 7549 |
| KEGG | C01773 |
| MeSH | D014258 |
| PubChem CID | 12301 |
| RTECS number | YU5950000 |
| UNII | 3X92F0YZ1B |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID3074644 |
| Properties | |
| Chemical formula | C3HF3O |
| Molar mass | 110.04 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet |
| Density | 1.367 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | 0.659 |
| Vapor pressure | 54.4 mmHg (20 °C) |
| Acidity (pKa) | 16.4 |
| Basicity (pKb) | pKb = 8.8 |
| Magnetic susceptibility (χ) | -44.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.298 |
| Viscosity | 0.456 mPa·s (25 °C) |
| Dipole moment | 2.7968 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 234.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -981 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1617.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Warning |
| Hazard statements | H225, H302, H319, H332 |
| Precautionary statements | H315, H319, H335 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 5 °C |
| Autoignition temperature | 465 °C |
| Explosive limits | Explosive limits: 2.3–17% |
| Lethal dose or concentration | LD50 oral rat 865 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1600 mg/kg (oral, rat) |
| NIOSH | TT6300000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Trifluoroacetone: 1 ppm (parts per million) |
| REL (Recommended) | 0.02 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Halogenated ketones Hexafluoroacetone Chloroacetone Fluoroacetone Bromoacetone Trifluoroacetic acid Trifluoroethanol |

