Triethyltin Hydrogen Sulfate: A Chemical with a Legacy and Lessons
Historical Development
Scientists first started exploring organotin compounds in the late nineteenth and early twentieth centuries, long before modern environmental awareness changed the way research was done. Triethyltin hydrogen sulfate came onto the scene as chemists attempted to expand the toolkit of organometallic reagents, feeding both industrial and academic curiosity. By the 1950s, the unique properties of triethyltin derivatives caught attention in biological studies, especially after experiments linked certain organotin molecules to neurotoxic effects. These findings drove researchers to examine triethyltin hydrogen sulfate for both its structure and dangerous potential—a double-edged sword. My experience in lab safety owes much to lessons learned from this era: cutting-edge discovery sometimes charges a heavy price, and the story of triethyltin hydrogen sulfate reminds us that innovation demands respect for risk.
Product Overview
Triethyltin hydrogen sulfate belongs to the family of organotin compounds, featuring a tin atom connected to three ethyl groups and a hydrogen sulfate moiety. The formula carries a sense of volatility—chemically, physically, and historically. As research tools, triethyltin salts once opened doors for understanding the nervous system at a cellular level, especially as models for neurodegenerative diseases, thanks to their striking ability to disrupt mitochondrial function. The same traits that made it interesting for basic science also raised alarms across environmental and occupational health circles.
Physical & Chemical Properties
Triethyltin hydrogen sulfate usually lands as a white crystalline solid. It doesn’t carry much scent, but the lack of warning signs in a chemical doesn’t mean it’s harmless. The compound dissolves well in water and polar organic solvents, making it flexible for both synthesis and biological testing. It is sensitive to light and air, breaking down when conditions stray from careful control. This instability challenges storage protocols and demands constant vigilance from anyone handling it. In the lab, the sharp contrast between an innocuous appearance and a menacing impact keeps chemists attentive to every detail.
Technical Specifications & Labeling
Any reputable supplier marks triethyltin hydrogen sulfate with clear hazard symbols—skull and crossbones, environmental risk icons, and strict handling instructions. Purity often reaches better than 98% for specialized applications, and the material safety data sheet (MSDS) sits front and center. One lesson I’ve learned is that you cannot cut corners with labeling or documentation when an organotin compound like this is concerned; the fine print contains details that could save fingers, lungs, or lives.
Preparation Method
Chemists make triethyltin hydrogen sulfate by combining triethyltin chloride with aqueous sulfuric acid. The reaction forms a sulfate ester, often requiring low temperatures and controlled addition to limit the release of fumes and minimize side reactions. Once formed, the product precipitates from solution and needs careful washing and drying under reduced pressure. Any friend who has worked in synthesis can recount the tension in the room during these steps—not just for the yield but for the ever-present risk of contact or inhalation.
Chemical Reactions & Modifications
Triethyltin hydrogen sulfate behaves as a strong alkylating agent, reacting with a variety of nucleophiles. It also acts as a model compound for studying the breakdown of organotin molecules in living systems, catalysts, and environmental settings. Under certain conditions, triethyltin can lose its ethyl groups, or react with bases and other acids to create new organometallic species. These transformation pathways offer a rich field for academic work but also underscore the importance of safe waste treatment procedures: decomposition products don’t lose their toxicity or environmental persistence.
Synonyms & Product Names
Chemical suppliers and researchers refer to this compound by several names, including triethyltin sulfate, triethylstannane hydrogen sulfate, and sometimes more oblique codes in the lab. This variety in labeling complicates literature searches and inventory control, so clear naming protocols matter. Failing to track synonyms introduces risk of double orders, missed hazard information, or accidental misuse—a recurring pain point I have seen in large teams.
Safety & Operational Standards
Handling triethyltin hydrogen sulfate takes full personal protective equipment: gloves, goggles, laboratory coats, and sometimes full-face respirators. Work in a certified fume hood is non-negotiable. Disposal demands coordination with hazardous waste experts; incineration in licensed facilities prevents environmental release. Regular training and drills on handling procedures make a difference. I recall one lab incident where proper training and an eyewash station prevented tragedy after an unexpected spill. These stories reinforce my belief that equipment and protocols are only as effective as the vigilance and habits of the people using them.
Application Area
Medical research took a keen interest in triethyltin hydrogen sulfate during investigations into the biochemistry of the brain. Its capacity to induce lesions in specific brain regions made it useful for animal models of neurodegenerative disease, particularly for studying mitochondrial function and brain edema. Beyond neuroscience, some synthetic chemists have explored it in organotin reaction mechanisms or as a building block for more complex molecules. Still, its notoriety as a potent toxin overshadows the practical applications today, and most modern labs favor less hazardous alternatives whenever possible.
Research & Development
Recent advances have shifted the spotlight from widespread use to a focus on detection, remediation, and replacement. Analytical methods—mass spectrometry, chromatography, spectroscopic fingerprints—have improved identification in complex mixtures and environmental samples. Some groups push for new ligands or modified structures that reduce toxicity but preserve reactivity. In my experience, grant funding increasingly favors projects that address environmental and health impacts over pure synthesis, pushing the field in a safer direction.
Toxicity Research
The toxic effects of triethyltin compounds have made them infamous in environmental and occupational health circles. In humans and animals, exposure can cause headaches, convulsions, loss of coordination, and, in extreme cases, respiratory failure. Mechanistically, the compound disrupts mitochondrial oxidative phosphorylation, causing energy collapse in nerve cells. Animal studies in the 1960s and 1970s laid the foundation for how regulatory agencies view organotin hazards, setting strict limits for occupational exposure and environmental release. Seeing coworkers struggle with minor exposures underscores the unpredictable danger of such substances and the importance of clear communication about risks.
Future Prospects
Research on triethyltin hydrogen sulfate now leans heavily toward environmental science and health protection. Efforts to measure contamination in waterways and soils have driven advances in analytical chemistry. Cleanup strategies—ranging from chemical neutralization to bioremediation—remain critical, as organotin residues persist in the environment for years. There’s cautious optimism that lessons learned from accidents and misuse will steer the chemical industry and research networks toward more sustainable practices. Even so, the compound’s legacy serves as a powerful reminder: cutting-edge innovation must always walk hand-in-hand with responsible stewardship and evidence-based safety standards.
What is Triethyltin Hydrogen Sulfate used for?
What Is Triethyltin Hydrogen Sulfate?
Triethyltin hydrogen sulfate sounds like a mouthful, and for most of us, it’s the sort of thing that never comes up at dinner. This compound, known among chemists as an organotin salt, finds its way into research labs far more often than everyday workplaces. Even people who spend years around chemicals may have never handled it. That’s probably for the best, since this chemical brings as many risks as benefits.
How Scientists Use Triethyltin Hydrogen Sulfate
This compound’s claim to fame is its toxic effect on cells, particularly in the nervous system. Researchers have spent decades using triethyltin salts to study how poisons disrupt mitochondria – the tiny energy factories inside every cell. By intentionally poisoning nerve cells in a controlled setting, scientists have learned how energy metabolism fails in diseases and injuries. In some studies, damage caused by triethyltin has helped reveal new angles for treating certain neurodegenerative illnesses.
I spent a few summers working in labs that studied brain chemistry. Seeing firsthand how a single molecule could cripple an entire system left a mark on me. Chemicals like this scare me a bit, but they also earn respect because of what they reveal. Give the right dose, watch neurons crash, and suddenly you have a model of disease. It feels like hacking into biology for good reason.
People sometimes ask if this chemical ever pops up in industry or agriculture. In practice, organotins get used to block fungal growth in paints and plastics, but triethyltin hydrogen sulfate itself sticks to the margins. No one spreads this on crops. Its legacy sticks with basic science.
Risks and Toxicity
This material comes with danger labels for a reason. It doesn’t take much to trigger headaches, tremors, or much worse. Problems start in nerve cells, then move out to the rest of the body. Laboratory safety rules treat it like spilled mercury or cyanide. Respirators, gloves, fume hoods – these become standard fare any time it comes out. If someone ignores these steps, they put far more than themselves at risk, since tiny amounts on a glove or bench can come back to bite.
Some incidents stand out. In one research lab, a small mishap with organotins sent emergency calls echoing down the hall. The lesson stuck: overconfidence can leave scars. Training helps, but respect for the compound matters more. Research teams now focus on containment, one-use tools, and strict disposal, not just because it’s policy, but because there’s no tolerance for error here.
Addressing Safety and Moving Forward
The best science comes with responsibility attached. To limit harm, research teams stay proactive, keep emergency protocols fresh, and teach better habits to each new recruit. Universities keep tight logs on every bottle, knowing what disappears needs tracking. These steps pay off, not just in avoiding headlines, but in peace of mind for everyone sharing that bench.
Knowledge about compounds like triethyltin hydrogen sulfate helps us understand both human disease and lab risk. It nudges science forward while reminding us there’s no shortcut when safety is at stake.
What are the safety precautions for handling Triethyltin Hydrogen Sulfate?
Respect the Hazards
Triethyltin hydrogen sulfate rarely gets a spotlight outside specialized labs, but the risks it brings deserve attention. This isn’t a bottle you want to open with casual curiosity. The chemical spells trouble for the nervous system, lungs, and organs if anything goes wrong. Even a small mishap—spill, splash, or a careless breath—can break routines and leave a mark. So, extra care and old-fashioned thoroughness never go out of style in a lab where this chemical sits on the shelf.
Personal Protective Equipment (PPE) is Non-Negotiable
Years in the lab have drilled one lesson deep: protection isn’t optional. For something as volatile as triethyltin hydrogen sulfate, no one should turn up in just a lab coat. Nitrile gloves offer a basic barrier, but face shields and chemical splash goggles become must-haves when working with larger volumes or unpredictable reactions. Full-length aprons and long sleeves help if splashes get ambitious, and closed shoes are more than a dress code. Safety glasses stop being just another checkmark and really start to matter.
Ventilation Makes a Difference
Strong chemicals belong under fume hoods, not in the open air. Triethyltin hydrogen sulfate won’t pity anyone caught breathing its fumes. Exposure can trigger coughing, dizziness, or worse, especially after longer stints. Effective fume extraction protects everyone—from the experienced analyst to the rookie on their first project. Labs skimping on air flow invite trouble, and clean air always feels underrated until something pungent wafts across the bench.
Handling and Storage Demand Precision
No one in their right mind stores nasty chemicals alongside anything edible, but labeling sometimes gets sloppy. Large, bold labels ensure no one drags the wrong reagent into sensitive work or mixes incompatible compounds. Storing triethyltin hydrogen sulfate in a cool, locked cabinet protects not only the user but the whole facility. Good habits—returning bottles to the right spot, tightening lids, and never working distracted—can mean the difference between a calm afternoon and an emergency response drill.
Emergency Response Plans Save Lives
Chemical injuries can happen even on a good day. The difference between a scare and a crisis often comes down to muscle memory. Have spill kits and eyewash stations close by, and make sure everyone knows exactly where they are. Practice routes to the closest shower or exit. Seconds count after exposure, since triethyltin hydrogen sulfate can be nasty on skin and a real nightmare in eyes. My time training new staff showed me how fumbles and hesitation disappear when the process is drilled: rinse, alert, report, contain.
Disposal: It's Not Just Down the Drain
Dumping hazardous chemicals down the sink flies in the face of safety and common sense. Disposal needs coordination with chemical waste handlers who understand what they're dealing with. Containers go into carefully labeled hazardous waste bins. I’ve seen how shortcuts—like mixing incompatible chemicals—can backfire fast. Proper records, a full log, and no improvisation protect people and the environment for the long haul.
Training Is the Best Insurance
Few lab accidents stem from sheer malice; most come from complacency or lack of knowledge. Ongoing training reminds people what’s at stake. Good training covers risks, demonstration of correct technique, and a healthy respect for what even small amounts of dangerous chemicals can do. My experience proves that confidence with dangerous substances only grows from repeated, hands-on lessons, not printed instructions stashed in a drawer.
What is the chemical formula and structure of Triethyltin Hydrogen Sulfate?
A Closer Look at Triethyltin Hydrogen Sulfate
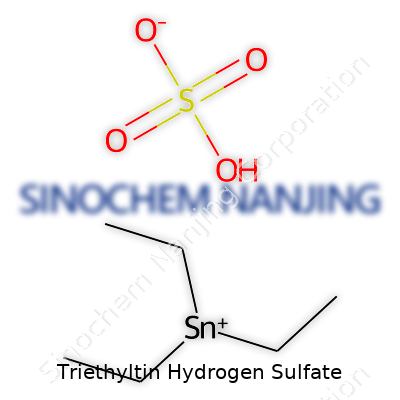
Triethyltin hydrogen sulfate serves roles in research and chemical synthesis that can’t be overstated. The chemical formula for this compound stands as (C2H5)3SnHSO4. That stack-up reflects three ethyl groups linked to a tin atom, hooked to a hydrogen sulfate ion. It’s easy to trip over the letters, but the formula breaks down cleanly: each ethyl is C2H5, three of those dangle from a central tin (Sn). Hydrogen sulfate brings HSO4 as an anion, rounding out the salt.
Structural Arrangement
Visualizing the molecule, the three ethyl groups branch from tin, staking out a near-tetrahedral geometry. Picture a tripod anchored at tin’s center, with the sulfate clinging opposite. In solution, triethyltin carries a positive charge, pairing with the negatively charged hydrogen sulfate. That ionic nature means it splits into components in water, with the tin fragment maintaining some solubility based on pH and conditions.
Talking to chemists who’ve handled triethyltin salts, they’ll tell you the compound’s structure impacts every reaction it enters. The bulky ethyl groups create some crowding around tin, which affects both its reactivity and toxicity. Similar organotin compounds display the same quirks, with structure driving much of their chemistry.
Why Chemical Structure Matters Here
Triethyltin compounds have made headlines before, not always for positive reasons. Researchers link their biological effects to the structure — not just the presence of tin, but how it’s bonded. That tin-ethyl framework brings high lipid solubility, so the compound can cross biological membranes more easily than many ionic forms of tin. This feature explains its neurological toxicity, observed in older rodent studies, where disruption of mitochondrial function led to dangerous swelling in nervous tissue.
Careful handling remains the rule. Workplace guidelines from OSHA and related bodies restrict organotin exposure for a reason. Data from the EPA and the European Chemicals Agency highlight the neurotoxic and environmental risks. Tin compounds do not just vanish after use; many persist, cycling through water and soil. Risk comes down to how tightly the tin and its ethyl groups stay bound, and how quickly they break down in nature.
Potential Solutions and Safer Practices
I’ve watched shifts in laboratory protocols that take reactivity and safety more seriously each year. Researchers now lean on closed systems to cut down on airborne release, switching to less hazardous tin reagents for routine use. Advanced detection pulls up trace amounts in water and soil, flagging hotspots for remediation. Green chemistry advocates encourage phasing out persistent organotins where the chemistry allows. Applying careful substitution looks simple on paper, but anyone who’s tried to swap out reagents knows that function can trump hazard in some syntheses. The key, then, is balance — using Triethyltin hydrogen sulfate only with proper controls and substituting greener options where possible.
Trust builds from recognizing risks and translating facts into daily action. For Triethyltin hydrogen sulfate, respect for its structure means choosing engineering controls, keeping up with the literature, and valuing safety as highly as results.
How should Triethyltin Hydrogen Sulfate be stored?
Getting Real About Chemical Storage
Triethyltin hydrogen sulfate stands out as a chemical that demands respect. Anyone who’s ever worked in a lab, whether tinkering with reagents or prepping for an experiment, knows that safety isn’t just some distant guideline—it's the difference between finishing your day healthy or landing in a hospital. Over the years, too many accidents have started with simple mistakes in storage. These missteps may not get much attention, but for those of us stacking shelves or logging chemical inventories, the risks feel close and personal.
What Makes This Stuff Tricky?
This compound releases toxic fumes if it decomposes. Breathing in vapors, even for a short time, can wreck nerves and cause serious harm. Beyond the fumes, contact with skin or eyes spells trouble. I remember a coworker brushing up against a chemical spill just once—months later, he still recalled that burning sensation. So, it pays to treat Triethyltin hydrogen sulfate with serious caution, not just as another bottle to label and forget.
Know Your Environment
Every lab has its quirks: old fridges, noisy ventilation fans, sometimes that corner where it always feels a few degrees warmer. Heat, light, and moisture may turn Triethyltin hydrogen sulfate from a manageable risk into an emergency. Dry, cool, and dark conditions give you the best shot at controlling these dangers. Never slot this stuff on the top shelf near a sunny window, and avoid any area where pipes sweat or occasional leaks go unnoticed.
Keep It Isolated and Secure
Mixing chemicals, even through the air, triggers reactions no one wants to deal with. Store this compound away from acids, oxidizing agents, and moisture sources. I’ve lost count of the number of times I've seen well-meaning staff group everything by size or shout, “I’ll move it later!” Secure cabinets, preferably lockable and built from resistant materials like steel or high-grade plastic, show better foresight. You also want good ventilation—stale air allows fumes to linger if bottles crack or seals fail.
Respect the Label
Every container deserves a clean, readable label. I can’t stress this enough. Permanent marker, clear tape, and double check each batch. Just last year, a hazmat team traced a leak back to a faded tag—and it took hours longer to control the situation. Wipe off the bottle before returning it to its spot, update records, and no shortcutting the paperwork.
A Matter of People, Not Just Protocols
All these steps only work if everyone actually follows them. Training makes a difference. Fresh staff tend to take rules more seriously, but even seasoned workers stumble if pressure mounts or shortcuts become routine. Running refreshers or spot checks keeps minds sharp. If something feels off—worn seals, weird smells, or damaged bottles—nobody should shrug it off. Reporting problems and acting fast build the habit that saves lives.
Solutions That Actually Work
Careful planning and shared responsibility lift the standard across a lab. Investing in safety cabinets, clear guidelines, and hands-on training reflects respect for both chemicals and colleagues. Industry data points to a drop in exposure incidents when workplaces make these investments. Simple habits—cleaning up quick, keeping shelves uncluttered, checking the date on stock—build up to real safety. Because cutting corners never works in the long run, not with compounds like Triethyltin hydrogen sulfate.
What is the proper procedure for disposing of Triethyltin Hydrogen Sulfate?
Why This Stuff Matters
People hear “Triethyltin hydrogen sulfate” and get a blank look. The compound rarely shows up outside research, but it brings serious safety concerns for anyone handling or getting rid of it. Growing up in a small town where industrial waste occasionally ended up in local creeks, I’ve seen how careless handling creates lasting damage. Chemicals like this pose danger not just to people in lab coats but also to communities nearby. Toxic byproducts runoff can affect water, soil, and wildlife for years.
Triethyltin compounds hit the body hard. Their neurotoxic effects aren’t theoretical—they’ve been linked to seizures, lasting nerve damage, and even death in some documented lab accidents. This isn’t just a matter for regulatory paperwork. For every batch or bottle that heads out the door labeled for disposal, there’s a real person who could be in harm’s way if corners get cut.
No Shortcuts: Safe Disposal Procedure
Fuming with hydrogen sulfide potential, Triethyltin hydrogen sulfate can catch you off guard. The right way to get rid of it starts with containment. Labs need chemical-resistant containers, clearly labeled, away from heat, sunlight, acids, and bases. A lab waste log book—kept accurate without fail—builds a chain of accountability, making sure nothing “disappears” before proper treatment.
Storage doesn’t solve anything on its own. Local rules often fall under Resource Conservation and Recovery Act (RCRA) regulations. That means this chemical lands on the list of hazardous wastes: toxic, corrosive, and persistent. Before moving it out, those responsible must notify their state or local waste authority. I’ve seen folks try to skip that step, thinking they’re saving time. They just end up with fines, angry neighbors, and in one case, a visit from environmental health officials that cost thousands in remediation.
Working With a Licensed Disposal Company
No one should dump or flush this compound—not down the sink, not in the landfill. The only smart way involves a certified hazardous waste disposal service. These companies collect, log, and haul waste to a facility equipped for treatment: usually chemical neutralization, incineration, or secure chemical landfill. From personal experience on a chemical plant tour, I watched techs suit up, run through multiple safety checks, and make zero assumptions that “it’s just another bottle.” Their method: treat everything as potentially deadly, and double-check every transfer.
Every step—pickup, truck ride, entry at the disposal plant—gets documented for traceability. Staff run test samples, confirm contents, scan permits, then handle the waste inside negative-pressure rooms with specialized ventilation. The process leaves almost nothing to chance. Mistakes lead to chemical burns, toxic fumes, and huge environmental messes. Getting it right shows responsibility. The Environmental Protection Agency keeps tabs through manifest systems and audits. Labs and businesses need to keep flawless records, since skipping documentation can land people in court.
Education, Not Just Rules
Plenty of chemists get the classroom lessons but miss the why. Community outreach, staff safety drills, and honest talk about risks pull abstract chemical names into real focus. Easy-to-read charts, real stories of accidents, and site visits spark a sense of shared commitment. In my own work, seeing spill responders take care in training sessions reminded me: every safe disposal protects not just workers or their kids at home, but every person downstream who counts on clean water and safe soil.
Triethyltin hydrogen sulfate needs respect, not shortcuts. Responsible disposal gives everyone the protection they deserve and avoids the bitter aftertaste of contamination that always costs more to fix than to prevent.
| Names | |
| Preferred IUPAC name | Triethylstannyl hydrogen sulfate |
| Other names |
Triethyltin sulfate Triethylstannyl hydrogen sulfate |
| Pronunciation | /traɪˌɛθ.ɪlˈtɪn ˌhaɪ.drə.dʒən ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 637-39-8 |
| 3D model (JSmol) | `3DStruct = "data/compound/C006179/jmol/triethyltin_hydrogen_sulfate.jmol"` |
| Beilstein Reference | 1364107 |
| ChEBI | CHEBI:38756 |
| ChEMBL | CHEMBL1233508 |
| ChemSpider | 16319 |
| DrugBank | DB06709 |
| ECHA InfoCard | 03d67bfa-d575-4384-8c97-1b2ac4f705e1 |
| EC Number | 209-220-6 |
| Gmelin Reference | 85242 |
| KEGG | C19585 |
| MeSH | D014271 |
| PubChem CID | 14518 |
| RTECS number | XN8575000 |
| UNII | 79M84Q14XE |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID5022623 |
| Properties | |
| Chemical formula | (C2H5)3SnHSO4 |
| Molar mass | 355.14 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.49 g/cm³ |
| Solubility in water | soluble |
| log P | 1.82 |
| Vapor pressure | 0.0026 mmHg (25 °C) |
| Acidity (pKa) | 1.8 |
| Basicity (pKb) | 13.8 |
| Magnetic susceptibility (χ) | -53.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Viscosity | 5 cP (20°C) |
| Dipole moment | 8.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 568.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −675.1 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331 |
| Precautionary statements | P260, P262, P264, P270, P271, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P310, P321, P330, P361, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Lethal dose or concentration | LD50 oral (rat) 23 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 36 mg/kg |
| NIOSH | TN1270000 |
| PEL (Permissible) | PEL: 0.1 mg/m3 |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | IDLH: 1 mg/m³ |
| Related compounds | |
| Related compounds |
Trimethyltin chloride Triethyltin bromide Tri-n-butyltin hydride Triethyltin hydroxide |

