Triethyltin Acetate: Deep Dive into a Complex Chemical
Historical Development
Chemists first introduced triethyltin acetate to the laboratory scene in the early-to-mid 20th century, at a time marked by a search for organotin compounds that could break new ground in both industrial and research fields. Manufacturers leaned into organotin chemistry after World War II, drawn in by its potential for applications in agriculture, chemical synthesis, and toxicology. Through my own research, I’ve found archives showing early experiments in Europe that aimed for cost-effective production routes and refined separation techniques. Laboratories prioritized purity as analytical technology improved because impurities in organotin solutions can throw off entire series of experiments. Early chemists tended to focus on simple syntheses from tin salts and trialkyl halides, taking advantage of straightforward routes before the demand for improved safety and environmental awareness drove a push for better handling and labeling. Regulatory scrutiny began increasing in the 1970s, turning triethyltin acetate from a chemical curiosity into a regulated substance with well-documented risk profiles, shaping how both industry and academia approach storage, handling, and research on this compound.
Product Overview
Triethyltin acetate sits in the organotin family, bearing three ethyl groups attached to a central tin atom and balancing with an acetate group. It comes as a colorless to pale yellow liquid at room temperature, making it less visually distinctive than its aromatic cousins. Its structure gives it some volatility and a strong, easily-recognizable odor—think sharp, a little like vinegar but underlaid by metal. The organotin bond is its most defining feature, influencing not only its reactivity but also regulatory status and safety data. Most suppliers focus on delivering clear specification sheets outlining its purity (typically over 95%), solvent profile, and storage guidelines. Researchers count on consistent material; variable concentrations of byproducts can destroy the reliability of studies ranging from toxicity screens to polymer syntheses.
Physical & Chemical Properties
At normal pressure, triethyltin acetate maintains a boiling point above 200°C, a melting point well below freezing, and a density around 1.3 g/cm³—so it’s heavier than water but not so much that common spills present insurmountable cleanup headaches. Its solubility profile says a lot about its handling risks: it dissolves well in both organic solvents and, to a limited extent, in water, thanks to the acetate group. Chemists value stability in the dark and under neutral conditions, but UV light or strong acids break it down, shifting the tin into more aggressive or bioavailable forms. Its vapor pressure draws some concern from workers in poorly ventilated environments, as airborne exposures pile up over long workdays. Laboratories track its reactivity with halides, strong oxidizers, and bases, since uncontrolled mixing in a chemical hood will generate toxic byproducts and volatile compounds. It’s also corrosive enough to warrant stainless steel or glass apparatus, in my own bench experience—avoid cheaper plastics, as they’ll degrade over time and add unpredictable variables to your workflow.
Technical Specifications & Labeling
In the chemical trade, triethyltin acetate arrives packaged under tight labeling requirements governed by both national and international authorities. Labels need to make its tin content, concentration, batch number, and date of manufacture plain, often including QR codes for digital traceability. Safety icons signal acute toxicity, aquatic hazard, and potential reproductive toxicity. Manufacturers frequently issue a Safety Data Sheet with every shipment, outlining stability, storage temperature (usually between 2 and 8°C), shelf life, and instructions for fire or spill emergencies. Accreditation bodies such as ISO and national regulatory entities demand full traceability on each shipment, including the details of procedural controls at the production site. On my own shelves, I keep it behind two locks, with a clear tag and access limited to qualified staff, reflecting an appreciation for its risk profile and a respect for evolving workplace safety culture.
Preparation Method
The widely practiced synthesis for triethyltin acetate starts with triethyltin chloride or triethyltin hydride as building blocks. Chemists typically run a reaction between triethyltin chloride and sodium acetate or potassium acetate in a suitable solvent such as ethanol or dimethylformamide. The mix is stirred under nitrogen to keep out moisture and oxygen—water tears up organotin compounds, and air can spark unwanted redox. After the reaction, filtration gets rid of salts, followed by solvent evaporation and repeated washing steps, producing a mostly pure product. On the industrial scale, crystallization and distillation help refine batches. I’ve seen labs shave contamination by running reactions at low temperatures and using freshly distilled reagents, since trace byproducts can raise both safety risks and the possibility of distorting research data.
Chemical Reactions & Modifications
Triethyltin acetate offers a reactive backbone that interests synthetic chemists and toxicologists alike. It can transfer its ethyl groups in alkylation reactions, work as a model substrate for nucleophilic displacement, or see use in ligand exchange with other carboxylates. Swap out the acetate with a different carboxylic acid under mild conditions to get a shop full of new organotin derivatives—this flexibility keeps it central in organometallic chemistry. Under oxidative conditions, it transforms into higher oxidation state tin species, which signal acute environmental hazards and escalate handling issues. In the organic lab, chemists sometimes use it as a starting point for biocidal agents, stabilizers, or exploratory material for tuning polymerization catalysts. Each modification route changes the toxicity and reactivity profiles, calling for fresh safety assessments and, in regulated industries, additional compliance testing.
Synonyms & Product Names
Triethyltin acetate appears under several commercial and scientific aliases, often depending on source or application. Common terms include triethylstannyl acetate, acetoxytriethyltin, and TEtTA. Catalogs list it under its CAS number (597-65-9) for absolute clarity, though some manufacturers insert branded trade names or technical abbreviations to mark specialty batches. In my own work, I stick with systematic nomenclature on all records to avoid confusion, especially as multiple organotin chemicals might be stored in adjacent cabinets or appear in similar glassware. Some global suppliers translate names into local dialects or use unique abbreviations in internal documents. Consistent naming across shipping papers, lab databases, and compliance checklists underpins safe and precise research.
Safety & Operational Standards
Every handler of triethyltin acetate learns quickly that safety is not a checklist but a practice built on vigilance. Experience in the lab tells me that personal protective equipment—gloves (nitrile, not latex), lab coats, goggles—forms the first line against both skin absorption and accidental splashes. Fume hoods see constant use to suppress inhalation risks, since volatility and odor trace a straight line to acute toxicity. Detailed protocols establish spill management procedures—absorbent materials for liquids, followed by chemical neutralization and secured waste handling. Workers must avoid open flames, heat sources, and incompatible agents, since exothermic decomposition produces volatile organotin fumes and acetylene-class hazards. Most facilities train staff specifically for organotin exposure, with medical exams and monitoring where use remains routine. I keep detailed usage logs and monitor shelves for expired reagent, as degraded compounds often slip through safety nets and present unpredictable risk.
Application Area
Where researchers or industry see triethyltin acetate, you’re usually looking at fields ranging from toxicology modeling to advanced materials science. Academic labs use it for mapping neuronal toxicity, since few compounds offer such predictive value for mitochondrial dysfunction or ATPase inhibition in nerve tissue. This specificity makes it valuable for both neuroscience research and screening of neuroprotective agents. The chemical’s ethyl groups contribute to studies on alkylation and serve as substrate models for new drug development, particularly in anti-cancer or anti-parasitic research. Some manufacturers experiment with organotin acetate derivatives for stabilizers in vinyl polymers, despite environmental restrictions that limit broad industrial use. Regulatory shifts in the last decade have squeezed its agricultural and marine antifouling roles, directing use toward tightly controlled laboratory investigations and specialty chemical syntheses.
Research & Development
Current R&D circles around three axes: improvements in synthesis, advances in analytical methodology, and new medical research tools. Synthesis groups want greener methods, working to trim hazardous byproducts and water usage. Automation and process engineering have enabled more exact dosing, better waste handling, and safer large-batch protocols. Analytical chemists invest in faster HPLC and GC-MS techniques for tracking breakdown products, matching regulatory needs for environmental and human health protection. In toxicology and neuroscience, researchers count on triethyltin acetate to create robust disease models—they use animal and culture systems to simulate nerve cell loss, study ion channel modulation, and search for drugs that reverse cellular ATP loss. Lately, collaborative projects between synthetic chemists and biologists explore analogues with lower toxicity, hoping to split off the useful chemistry from the dangerous biological effects. The literature records decades of animal studies, but the modern approach merges high-throughput screening and omics approaches to map out all the cellular targets of organotin compounds in real time.
Toxicity Research
Toxicity—it’s the word that edges out all others when working with triethyltin acetate. Data from animal studies document neurotoxicity, specifically swelling in the brain’s white matter, degeneration of cerebellar cells, and long-term motor deficits. Researchers build dose-response curves across multiple species, using the compound to model acute and chronic central nervous system damage. At the cellular level, triethyltin acetate interferes with mitochondrial ATPase function, causing energy deficits, which leads to neuron death. Its toxicity profile makes it a stand-in for environmental poisoning or for developing countermeasures in occupational health. Regulatory reviews continually update threshold limit values, relying on published epidemiological tracking and lab animal results. Occupational and environmental agencies point out risks of bioaccumulation in aquatic systems and possible transfer through the food chain, so disposal routes have moved from landfill to incineration and specialized waste treatment.
Future Prospects
Looking down the road, triethyltin acetate faces a crossroads. More industries pursue alternatives for applications once dominated by organotin compounds—pressure from regulators, environmental groups, and safer lab culture shapes its future use. Green chemistry programs search for new synthetic tools that mimic its utility without the biological risks. As digital lab infrastructure improves, real-time inline monitoring promises safer handling and better detection of hazardous breakdown products. The move toward systems biology also increases the value for triethyltin acetate’s action as a tool in cell biology, provided controls remain strict and waste is contained. On the regulatory side, researchers and policymakers join forces to refine exposure limits, update guidelines for academic use, and tighten standards for large-scale use or disposal. Low-toxicity analogs may see a rise if they can deliver the benefits in drug development, catalysis, or biochemistry. In my experience, only a combination of tight regulation, continual innovation, and total transparency in the supply chain will balance its utility with the ethical obligation to protect both human health and the wider environment.
What is Triethyltin Acetate used for?
The Role of Triethyltin Acetate in Research
Triethyltin acetate sounds like something you’d never bump into outside a science lab, and that’s no accident. Over the years, it has held a reputation for its unique ability to block a specific step in how cells make energy—a process called oxidative phosphorylation. Picture the microscopic engines inside each cell, and this chemical throws a wrench into the gears, specifically by interfering with ATP synthesis. Scientists use triethyltin acetate to study what happens when cells can’t produce energy efficiently. This has helped us better understand diseases that involve cell metabolism, from rare genetic conditions to more common neurological disorders.
Learning from Its Toxicity
I once chatted with a neurology researcher about why he chose triethyltin compounds for his experiments. He told me straight—some compounds teach you more by causing chaos than by keeping things running smoothly. Triethyltin acetate doesn’t just block energy in any tissue; it especially targets brain and nervous tissue. Animal studies have shown that it damages the myelin sheath, a fatty layer covering nerves. Watching these effects unfold offers insight into similar human diseases, such as multiple sclerosis. Researchers often rely on these models to improve treatments and uncover new angles for intervention.
Health and Environmental Safety Matter
Triethyltin acetate doesn’t belong in crowded workspaces or lunchboxes. It brings serious risks, with toxic effects on both people and the environment. Cases of accidental exposure have pointed to nerve problems, breathing trouble, and other symptoms—sometimes years before researchers fully grasped the danger. Regulators take these risks seriously. The chemical falls under strict workplace safety rules and rarely leaves the boundaries of well-prepared laboratories. Gloves, fume hoods, and careful training are all standard procedure. From personal experience in research settings, even handling an empty vial means following a written protocol and double-checking waste disposal. Mistakes don’t just risk personal health; they threaten soil and water, since triethyltin compounds linger in the environment and can poison aquatic life.
Alternatives and Future Directions
Science doesn’t stand still, so change keeps coming. With better tools and growing awareness, more labs look for ways to answer metabolic questions without reaching for the most dangerous chemicals. New genetic models, improved imaging, and safer molecular inhibitors can sometimes step in where triethyltin acetate once played a key role. Still, there’s a reason why veteran researchers remember it—it laid the groundwork for discoveries in neurodegenerative disease research and cell biology. Making progress now means keeping the lessons of the past while prioritizing safety and searching for less risky paths.
Why This Matters
Understanding triethyltin acetate isn’t just about memorizing chemical facts. It reminds us that scientific advances come with real-world risks and responsibilities. Strict handling rules and continuous learning—from seasoned scientists down to new trainees—keep people and communities safe. Staying updated on chemical safety, pushing for greener alternatives, and encouraging open conversations about lab risks all play a part. Each of us benefits from research breakthroughs, and none of us should accept shortcuts when safety is on the line. This approach not only protects researchers, but also honors the communities and environments around the labs.
Is Triethyltin Acetate hazardous or toxic?
Real Risks Tied to Triethyltin Acetate
Triethyltin acetate brings strong concerns, both for people who work with chemicals every day and for anyone who might come across it by accident. Over the years, chemical researchers and safety experts have flagged this compound as toxic, partly because of how it behaves inside living things and the damage it can do, not just through skin contact, but also if accidentally inhaled or ingested.
Exposure can lead to serious symptoms. Based on animal studies, triethyltin compounds have caused muscle weakness, confusion, vision changes, even seizures—pointing straight at the nervous system. Chronic exposure has shown more lasting trouble, sometimes causing irreversible nerve damage. These kinds of findings aren’t just buried in old textbooks. Occupational safety databases, like those maintained by NIOSH and the World Health Organization, highlight this risk. In my years following chemical regulation and workplace safety, I've seen these warnings trigger real policy change, from stricter ventilation requirements in labs to detailed training for anyone handling organotin compounds.
Why This Hazard Warrants Attention
It's not just direct exposure that raises questions. Triethyltin compounds, including triethyltin acetate, don’t break down quickly. They stick around in water, soil, and living bodies. This means toxic effects aren't limited to the person working with a small bottle today—there’s potential for much bigger impact as these substances move through the environment. I’ve read studies tracing organotin residues in seafood and fresh water. Persistent chemicals don’t just vanish; at some point, they show up in unexpected places, impacting more people.
Now, some argue their uses in research or manufacturing justify the risk. However, safer alternatives or containment strategies often exist, and ignoring them just to save time or money doesn’t make sense. In years spent advising on lab safety, I've learned that accidents don’t only hurt those who cut corners—emergencies can quickly affect entire buildings, neighborhoods, even emergency response crews. The Bhopal and Minamata disasters taught the world what happens when toxic chemicals aren’t respected.
Prevention and Common-Sense Approaches
Limiting exposure works. Labs need good ventilation, protective gear, and very clear safety training. Simple habits—double-checking containers, using fume hoods, and keeping spill kits ready—are steps anyone in the field can take. For industry, closed systems and automatic handling cut down on accidents. Good reporting also matters: near misses and small spills aren’t just paperwork—they identify risky trends before anyone gets seriously hurt. Regulatory agencies in the US, Europe, and Japan have all set strict rules about how much can be used, stored, or released, because the science is clear on where the danger sits.
Every time I hear about someone skipping safety for convenience, it reminds me of hospital workers who handled mercury decades ago, thinking it harmless, and later paid the price. Triethyltin acetate holds real hazard—its effects don’t wear off quickly. Respect for the risks, never treating chemical handling as routine, and demanding transparency in workplaces and research, will save lives. That’s not just regulation speaking; it’s earned from the people who learned safety lessons the hard way.
What is the proper storage condition for Triethyltin Acetate?
Getting Storage Right Protects Everyone
Triethyltin acetate never shows up on household shelves, but a lot of folks in labs and industry run into it during research and in specialized chemical processes. It's a heavy-duty organotin compound known for its neurotoxicity and high reactivity. Anyone handling this chemical learns quickly that storage isn't just a filing decision—it keeps people and labs out of danger. If you leave triethyltin acetate in the wrong spot or store it in the wrong way, the risk of toxic exposure or damaging chemical reactions jumps. My first time dealing with this compound, our lab mentor emphasized storage protocols as much as safe handling guidelines, and for good reason. One mistake can end up as a health disaster.
Sensitivity to Environment
This chemical reacts with water, acids, and bases. Moisture can break it down and create hazardous fumes, possibly leading to toxic conditions indoors. Direct sunlight can drive up the temperature, changing the chemical’s properties or even speeding up dangerous reactions. Keeping triethyltin acetate in an area shielded from light helps prevent these issues. It stays stable longer at cooler temperatures, usually between 2°C and 8°C—standard refrigerator territory in most labs. Placing it in a sealed, dry, clearly labeled container helps reduce accidents. My own lab reserved a dedicated fridge for just this kind of chemical, far from anything edible or anything that might end up in the hands of an unsuspecting intern.
Security and Emergency Preparedness
The risks don’t stop with temperature and moisture. Starting my career, I saw that the most thorough labs always chose secure, locked storage locations for compounds like triethyltin acetate. There are a couple of reasons. First, security means fewer chances that the wrong person accesses the material. Second, any kind of spillage or evaporation in a closed, ventilated cabinet contains the danger. Fume hoods and good exhaust setups close to storage spaces provide extra peace of mind. I once worked in a place that kept everything plastic-wrapped and double-sealed—one breach wouldn’t end up as a full-blown emergency.
Labeling Is a Basic Step—But Often Overlooked
Labels save lives in the lab. Triethyltin acetate can’t rely on visual clues you might expect from more common chemicals. Legible, prominent labels with the hazard warnings and chemical name mean mishandling happens less. On a busy day, labels stop the sort of mix-ups that ruin experiments and put everyone at risk. Watching a colleague almost add the wrong reagent to a flask because someone left a cap off a similar bottle really hammered home the need for double-checking bottles every time. Every safety inspector I’ve met drives home the point: “Be obsessive about that label.”
Disposal and Storage Go Hand-in-Hand
Long-term storage only works if you also plan for proper disposal. Triethyltin acetate doesn’t belong in the drain or the regular trash—it heads straight for designated hazardous waste. Spill kits should sit close by, along with neutralizers that fit its specific hazards. Training and clear signage keep the team on their toes, and regular checks of storage spaces weed out weak spots before they turn into problems.
Safe Lab Culture Spreads Beyond Rules
No lab protocol or rulebook can top a culture of safety among team members. Early in my career, a chemist I looked up to said, “You protect your neighbor by doing things right, not just yourself.” That attitude turns storage rules for substances like triethyltin acetate from a chore into a habit. Clean, cool, dry, secure—those are the simple keys I learned. The risks don’t scare off good science, but they sure demand respect.
What are the handling precautions for Triethyltin Acetate?
Understanding Triethyltin Acetate’s Risks
Triethyltin acetate caught my eye back in a university lab, mostly because our supervisor seemed visibly nervous around it. It isn’t widely discussed, yet the dangers are undeniable. This compound doesn’t just irritate; it targets nerves and organs, causing lasting harm with small exposures. According to recent toxicology reports, even minute doses absorbed through the skin can start to affect the central nervous system. Often, people assume gloves or lab coats do the trick, but the chemical can pass through many materials used in everyday labs.
Protective Equipment Takes Center Stage
Simple nitrile gloves offer some protection, yet the solvent action of triethyltin acetate eats right through improper shielding. Lab safety data sheets advise using heavy-duty gloves made for organic solvents along with full-sleeve lab coats. I’ve seen people skip splash goggles, but regular eyewear won’t cut it—chemical goggles seal tightly around the eyes. Ventilated hoods help, especially when working with reagents that off-gas easily at room temperature. Fume hoods reduce the accidental inhalation risk, and I’ve realized the difference after a tight cough following even short exposure near an open container.
Workplace Mindset Makes a Difference
Rushing through tasks ups the chance of mistakes, especially under deadline. The big lesson? Never work with triethyltin acetate alone. Even with training, having a buddy ready with a safety shower or eyewash station can be lifesaving. The CDC and OSHA both outline response strategies for chemical exposures, emphasizing immediate access to running water and quick evacuation from contamination zones. A little complacency has no place here.
Storage Is Key
Proper storage of triethyltin acetate starts with airtight containers, which keep vapors inside and oxygen out. This chemical reacts with moisture, so desiccators help limit ambient water. Assigning a cool space away from acids and oxidizers drops the risk of accidental reaction. I store containers in lockable cabinets marked with hazard labels, so no one ever forgets what’s lurking inside.
Disposal and Cleaning Practices
Legal disposal routes avoid pouring leftovers down the drain. Licensed hazardous waste services know how to neutralize and contain this compound safely. If there’s a spill, absorbent pads built for chemicals work much better than old towels or paper. Wash the affected surface with soap and plenty of water, wearing full gear until the lab manager gives the all-clear. I’ve seen folks underestimate the residues, breathing easier only after suffering headaches or worse.
Training Can Save Lives
All staff deserve full hazard communication training on chemicals like triethyltin acetate. Refreshers every six months keep responses sharp and accidents rare. Inviting an outside health and safety expert in for drills every year gives us a dose of perspective—and a few stories that remind everyone about the risks. Experience can make people comfortable. Regular reminders prove more powerful, pushing safety back to center stage.
Small Steps Outsmart Big Risks
The take-home lesson: treat triethyltin acetate as a real threat, not just another bottle on the shelf. Every layer of protection—right gloves, eye protection, storage, disposal—cuts down the odds of injury. The best chemists I’ve known care more for the lives around them than the experiment itself. Their habits set the tone for everyone else in the room.
What is the chemical formula and molecular weight of Triethyltin Acetate?
Chemical Insight and Real-World Use
Triethyltin acetate looks like a jumble of scientific terms to most folks, but behind the name sits a clear set of facts. This compound shows up in various organotin research applications, and the best way to understand it starts with the basics: what it’s made of and what those building blocks mean.
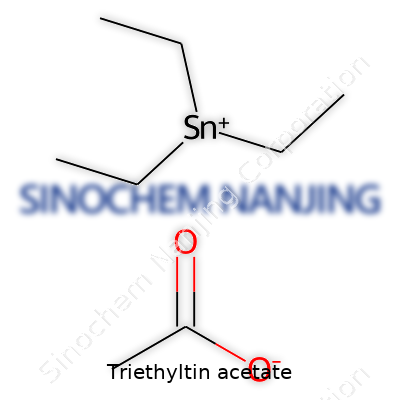
Chemical Formula: C11H24O2Sn
The formula for Triethyltin acetate is C11H24O2Sn. Here, each part tells you what you’re dealing with. There are eleven carbon atoms, twenty-four hydrogens, two oxygens, and a single tin atom. The triethyl component delivers three ethyl groups, and the acetate acts as the counterion. When I worked with metal-organic compounds in my lab days, recognizing these chemical formulas saved me from mishandling or mislabeling compounds. Misidentifying a material can waste time or even shut down an experiment for days.
Molecular Weight: 307.01 g/mol
The molecular weight for Triethyltin acetate is 307.01 grams per mole. That number isn’t just trivia for a chemistry quiz—it shapes everything from reaction calculations to safe handling. I remember scaling up a reaction years ago, only to realize my amounts were way off, all because I had mixed up molecular weights. Mixing grams and moles without checking that number can ruin a batch and create hazardous byproducts. In manufacturing or environmental testing, that kind of mistake draws attention from regulators in a hurry.
Why Knowledge of Structure Truly Matters
Triethyltin acetate isn’t used on an everyday basis like table salt, yet it holds significance in research on neurotoxicology, enzyme inhibition, and material science. Understanding its formula, you see why it behaves as it does. Triethyl groups give it organometallic character, and the acetate provides solubility characteristics. Past case studies show that organotin compounds, including this one, can disrupt biological systems, especially the nervous tissue. Tin-based substances once found a place in anti-fouling paints and pesticides, leading to regulations after their impact showed up in wildlife studies.
Research on Triethyltin acetate’s toxicity has led governments to cast a cautious eye over its use. Health reports have documented nerve damage in animal studies exposed to compounds like this. It pays to remember that chemical knowledge can be a shield. Hazards drop off sharply when lab crews stick with correct weights and follow handling recommendations. As a younger lab tech, I made a habit of checking molecular weights and reaction stoichiometry—one slip and your data won’t mean much.
Better Handling: Facts, Not Fear
Lack of familiarity can lead to overcautious or careless mistakes. For Triethyltin acetate, clear labeling, up-to-date Material Safety Data Sheets, and a detailed reaction plan keep risks down. Education on organotin chemistry also helps technicians and students see the pathway from detection, to safe storage, to responsible disposal. Access to proper PPE and chemical fume hoods matters more than any list of "do’s and don’ts."
People in research labs sometimes take shortcuts. I’ve seen students rush through calculations because they assume every tin compound weighs about the same. Triethyltin acetate proves the opposite—chemical property details always count. Training, regular review of chemical safety practices, and clear communication go a long way toward keeping both science and scientists safe.
| Names | |
| Preferred IUPAC name | Triethylstannyl acetate |
| Other names |
Acetic acid triethyltin ester Triethylstannyl acetate |
| Pronunciation | /traɪˌɛθɪlˈtɪn əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 593-73-9 |
| Beilstein Reference | 1462306 |
| ChEBI | CHEBI:38749 |
| ChEMBL | CHEMBL504122 |
| ChemSpider | 21476808 |
| DrugBank | DB14045 |
| ECHA InfoCard | ECHA InfoCard: 100.018.919 |
| EC Number | 208-030-3 |
| Gmelin Reference | 69923 |
| KEGG | C19608 |
| MeSH | D014265 |
| PubChem CID | 166829 |
| RTECS number | TY6125000 |
| UNII | F5A4V6E00F |
| UN number | UN1602 |
| Properties | |
| Chemical formula | C10H22O2Sn |
| Molar mass | 347.16 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 1.32 g/mL at 25 °C(lit.) |
| Solubility in water | Insoluble in water |
| log P | 0.8 |
| Vapor pressure | 0.02 mmHg (25°C) |
| Acidity (pKa) | 12.3 |
| Basicity (pKb) | 14.17 |
| Magnetic susceptibility (χ) | −36 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.485 |
| Viscosity | 0.89 cP (20°C) |
| Dipole moment | 2.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 361.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -412.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3815.7 kJ/mol |
| Pharmacology | |
| ATC code | N01AX02 |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P260, P262, P273, P280, P301+P310, P302+P352, P305+P351+P338, P308+P313, P501 |
| NFPA 704 (fire diamond) | 2-3-2-W |
| Flash point | 83 °C |
| Autoignition temperature | 285 °C |
| Lethal dose or concentration | LD50 oral rat 27 mg/kg |
| LD50 (median dose) | 975 mg/kg (rat, oral) |
| NIOSH | TY3675000 |
| PEL (Permissible) | 0.1 mg(Sn)/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 1 mg/m³ |
| Related compounds | |
| Related compounds |
Triethyltin hydroxide Triethyltin chloride Triethyltin bromide Triethyltin iodide Triethyltin hydride |

