Triethyl Orthopropionate: The Backbone of Versatile Chemistry
Historical Development
Chemistry tells its own story through changing needs, shifting priorities, and experimental breakthroughs. Triethyl orthopropionate came into play back in the era when chemists sought tools that would open up routes for both industrial and laboratory synthesis. People in labs and factories looked for more stable orthoesters compared to orthonitriles or esters with shorter carbon chains. Their patience paid off. By the 1950s, recipes involving simple alcohols and propionic acid derivatives allowed steady production. This compound became a staple solvent and a reactant that never disappeared from the shelves. Over time, industries realized that its reactivity and reliability made it a regular in discussions about protecting groups and rearrangement reactions.
Product Overview
Triethyl orthopropionate is no household name, yet it pops up in a surprisingly wide range of chemical conversations. It appears as a colorless to pale yellow liquid, carrying a mild odor reminiscent of ethers and strong solvents. The ability of this compound to form and react with a broad list of functional groups makes it indispensable for chemists aiming for controlled transformations. Many modern catalogues still list it as a specialty reagent, always in demand among synthetic organic chemists and process engineers.
Physical and Chemical Properties
This orthoester carries a molecular formula of C9H20O3 and shows a molecular weight just above 176 g/mol. A boiling point often settles around 163–165°C—handy for distillation and for any protocol demanding separation. Solubility prefers the embrace of alcohols, ethers, and most organic solvents, while it stays away from water due to easy hydrolysis. Its density falls near 0.87 g/cm³, so it floats slightly on water’s surface. Heat, strong acids, and bases break it into propionic acid and ethanol, a fact that workers in the plant or lab learn quickly.
Technical Specifications and Labeling
Chemically pure grades maintain a purity above 97%, usually confirmed by gas chromatography or NMR. Labels on containers list not just the chemical name and purity, but also batch information and appropriate storage guidance. Factories move it in steel drums or glass bottles lined with polytetrafluoroethylene to avoid corrosion and contamination. Regulatory numbers such as CAS and UN codes always appear on shipping documentation, meeting requirements for traceability as dictated by safety authorities. Companies keep strict control over shelf life, exposure limits, and recommended handling instructions to protect both the product and the staff.
Preparation Method
Production often starts with propionic acid and ethanol, both common chemicals, and an acid catalyst, typically sulfuric acid or p-toluenesulfonic acid. The process kicks off with esterification, then moves to transesterification where excess ethanol pushes the reaction toward the orthoester. Modern facilities rely on continuous removal of water, often through azeotropic distillation, to drive conversion. Spent acid gets neutralized, and solvents recycled, making the process efficient and eco-friendly where possible. Skilled operators know that balancing temperature, reactant ratios, and removal of byproducts crafts a final product pure enough for sensitive reactions.
Chemical Reactions and Modifications
Triethyl orthopropionate acts as more than a bystander in organic synthesis. Labs turn to it for introducing protective groups, particularly in carbohydrate chemistry where sugars need shielding during stepwise synthesis. A key strength comes from its hydrolyzable nature: under acidic or basic conditions, it cleaves to give propionic acid and ethanol. This step provides strategic flexibility in multi-step syntheses. Beyond protection, it also supports alkylation, condensation, and rearrangement reactions. Its structure lets chemists fine-tune selectivity and reaction rates by swapping in different catalysts or reaction partners. As research advances, the subtle tweaks in the structure or use in tandem with other reagents continue to push possibilities.
Synonyms and Product Names
This compound collects names from both old texts and modern chemical catalogues. Most commonly, you’ll see “triethyl orthopropionate” but sometimes suppliers label it simply as “propionic acid, triethyl orthoester” or “propanoic acid triethyl orthoester.” In research circles, abbreviations like TEOP or TriEtOP slip in for convenience, though regulatory paperwork always insists on full IUPAC labeling to prevent confusion. One thing remains consistent: any chemist scanning the shelves recognizes it no matter which name ends up on the bottle.
Safety and Operational Standards
Working with triethyl orthopropionate demands care and an understanding of risks. Inhalation or skin exposure irritates mucous membranes, so proper gloves, goggles, and fume hood operation become standard practice. Safety data sheets lay out first aid steps, fire fighting guidance, and spill handling advice. Since the compound reacts swiftly with acids, bases, and water, chemists double-check storage containers stay dry and tightly sealed. Regulations in the EU, US, and Asia assign hazard codes linked mostly to flammability and health effects. Fire extinguishers, easy access to eyewash stations, and routine training on chemical handling round out daily safety practices.
Application Area
Industries favor triethyl orthopropionate for its performance as a reagent in pharmaceutical and agrochemical synthesis. Medicinal chemists appreciate how it slides into reaction schemes powering the creation of complex intermediates for drug molecules. Agricultural labs draw on its reactivity to build protective groups or tailor pesticide molecules. Outside pure chemical synthesis, it occasionally finds use as a flavoring agent, though always at carefully regulated concentrations. Some specialty polymers and resins benefit from its inclusion during manufacturing, tailoring properties like flexibility or chemical resistance in final consumer products. No matter the field, its ability to guide selective chemical changes guarantees frequent use in research and manufacturing settings.
Research and Development
Modern research doesn’t leave this orthoester behind. Groups focused on new drug discovery run experiments to see whether it can protect tricky functional groups or improve yields in long synthesis sequences. Green chemistry initiatives check for more sustainable methods of production and look for biodegradable replacements. Thanks to its accessible structure, it acts as a testbed for reactivity studies, helping researchers model similar reactions with costlier or less stable reagents. Companies partner with universities to explore new modifications or applications, seeing the compound not as mature technology, but as an evolving toolbox. Increasingly, R&D teams check ways to make its manufacturing safer, reduce waste, or discover derivatives that broaden its usefulness across industries.
Toxicity Research
Toxicologists have kept a steady eye on triethyl orthopropionate, sharing findings across chemical and pharmaceutical communities. Reports highlight that ingestion or inhalation in large amounts causes central nervous system effects, like headache and dizziness, along with liver or kidney stress after sustained exposure. Animal studies outline clear dose thresholds above which the substance causes harm, leading organizations like the EPA and ECHA to set exposure limits for both acute and chronic contact. Routine workplace monitoring, coupled with health screening for those handling the chemical daily, marks a responsible approach in factories and laboratories. Newer studies aim to clarify possible environmental impacts, especially if waste streams from manufacturing reach soil or water supply.
Future Prospects
Looking ahead, demand for triethyl orthopropionate appears steady, especially as pharmaceutical and specialty chemical industries grow in Asia, Europe, and North America. Researchers predict ongoing interest thanks to its balance of chemical versatility and relative safety compared to more volatile orthoesters. Technological improvements in catalyst design and process automation may lower costs or improve product quality, opening up applications in fields where precision matters. Academic and industrial R&D is projected to deepen exploration into alternative synthetic pathways, less toxic analogs, or routes that harness bio-based feedstocks for sustainability. With regulatory bodies emphasizing worker safety and waste reduction, manufacturers keep pushing the science of both production and application, keeping this reliable workhorse in play for years to come.
What is Triethyl Orthopropionate used for?
A Real Player in Organic Synthesis
Triethyl orthopropionate isn’t a chemical you’ll find sitting around on grocery store shelves, yet it pulls a lot of weight in laboratories across the world. People in the pharmaceutical and chemical industries reach for this compound when they want to build something new or protect fragile bits and pieces in complicated molecules. In my own chemistry classes, we always looked at reagents like this as workhorses—unassuming until you need a specific job done.
Supporting Pharmaceutical Development
The pharmaceutical industry runs on innovation. Drug researchers constantly explore new routes to synthesize medicines, and triethyl orthopropionate serves as a handy tool in their kits. Its main draw sits in its role as a reagent for creating esters—essential building blocks for many drugs. During the formation of active pharmaceutical ingredients, controlling side reactions makes all the difference. This compound helps shield certain groups from unwanted change, letting chemists steer a reaction precisely where they need it to go. In antihypertensive agents or antiviral drugs, that level of control affects both success and safety.
Making Life Easier for Chemical Manufacturers
Beyond medicine, specialty chemical makers also value triethyl orthopropionate for its efficiency. Its ability to transfer ethoxy groups lets folks modify alcohols or acids right at the bench. I remember a production-scale plant tour during my university years, where an engineer quietly remarked how reagents like this kept their processes slick, cutting down on waste or expensive side reactions. Each streamlined step saves money, cuts emissions, and keeps dangerous leftovers out of the air and water. In bulk chemical synthesis, such details often decide a project’s fate.
Cleaner, Smarter Choices
Environmental safety should never fall by the wayside, especially with specialty reagents. Proper storage and handling of triethyl orthopropionate keep risks in check, but researchers are always pushing for cleaner options. Replacing harsh chemicals with safer or greener alternatives takes time, yet this drive shapes much of today’s research. Working with chemical suppliers, manufacturers can phase in processes that use less hazardous inputs or produce less troubling waste. Regulatory oversight, such as reach and EPA guidelines, nudges companies in the right direction. Responsible sourcing and transparent material data sheets help buyers make smarter decisions anywhere along the supply chain.
Practical Steps Toward Safer Use
Training and a culture of care make all the difference in routine lab work. Before anyone pours triethyl orthopropionate into a flask, clear protocols must be in place. Emergency eyewash stations, fume hoods, proper gloves—these basics can’t be skipped. Regular audits, whether by internal staff or outside professionals, catch overlooked practices and old habits that could lead to trouble. In my early laboratory jobs, peer checks caught more potential accidents than any sign on the wall ever did. Shared responsibility fosters a safer environment for everyone.
Room for Innovation
The hunt for more sustainable materials never stops. Green chemistry holds promise, and improving legacy reagents like triethyl orthopropionate will rely on continued research, feedback from industry partners, and steady information sharing. Building on today’s tools, tomorrow’s chemists will craft safer, smarter, and cleaner methods that improve lives both inside the laboratory and out in the wider world.
What is the chemical formula of Triethyl Orthopropionate?
Getting Familiar With Triethyl Orthopropionate
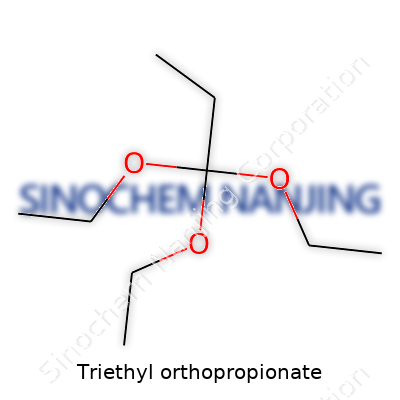
Triethyl orthopropionate isn’t a chemical that most people hear about every day, but it definitely finds its place in the world of chemistry and manufacturing. The chemical formula stands as C9H20O3. Behind these letters and numbers lies a structure made from three ethyl groups bonded to a central propionate molecule—three chunks of carbon and hydrogen, each attached to an oxygen atom, then all clinging to a three-carbon backbone. This formula spells out exactly how the atoms arrange themselves, which tells scientists and anyone in the chemical supply chain what to expect.
Why Is the Chemical Formula Important?
Knowing the formula matters. Each atom counts when labs look for purity, calculate how much material to use, or predict how the substance behaves. For someone like me who spent years in a production facility, a mix-up in a formula causes more than confusion—it wastes time, money, and creates safety risks. Triethyl orthopropionate’s precise structure sets it apart from similar-sounding chemicals and helps avoid those costly blunders.
Real-World Uses and Applications
With C9H20O3 on its label, this compound comes into play as an intermediate in pharmaceuticals and fine chemicals. It works well as a reagent for making new esters or adjusting the profile of a solvent blend. A process engineer or research chemist reaches for triethyl orthopropionate when a reaction calls for gentle, predictable performance instead of some volatile wildcard. Known for being less reactive than some other orthoesters, it offers a practical route in synthesizing molecules with specific properties or purity needs.
Where Missteps Happen
Chemistry textbooks sometimes lump together orthoesters, but precise formula recognition keeps misunderstandings to a minimum. C9H20O3 only fits this compound. If someone confuses it with, say, triethyl orthoformate (C7H16O3), they risk throwing synthetic pathways off track, causing reactions that either stall or go wild. In my experience, clear labeling and regular double-checks—especially before scaling up a batch—has saved more than one team from expensive recalls or cleanup efforts.
Advance Planning and Storage
Triethyl orthopropionate, like most specialty chemicals, needs careful handling and storage. Its chemical formula hints that it doesn’t mix well with water or extreme pH conditions. Good ventilation and sealed storage keep both the material and the workplace safe. Over the years, I’ve seen companies invest in simple solutions, such as color-coded inventory or digital tracking, to make sure what arrives from the supplier actually matches what’s needed in the recipe.
Education and Clear Communication
Teaching new hires about formulas like C9H20O3 sometimes seems tedious, but clarity upfront pays off. Guided hands-on training gives them a sense of the material’s behavior and real risks. Clear material safety data built around the formula helps teams know what’s coming out of those barrels and how it fits in the process. Even experienced technicians learn new things about subtle differences between compounds through continuous training.
Looking Ahead
Accurate scientific communication rests on details like chemical formulas. Sharing these details builds trust from the lab bench to the boardroom. Companies investing in ongoing education and better documentation wind up with safer teams, fewer accidents, and more reliable finished products. Each time you see a formula like C9H20O3, it’s more than numbers and letters—it’s a foundation for smart, safe, and innovative work.
Is Triethyl Orthopropionate hazardous or toxic?
Getting Clear on the Safety of Triethyl Orthopropionate
Triethyl orthopropionate shows up in some chemical labs and industrial processes. It carries the kind of name that makes people wonder what sort of hazards it might bring along for the ride. Over the years, I’ve seen just about every kind of chemical warning, from obvious dangers to hidden risks that show up long after use. A compound like this deserves real scrutiny.
What Does Science Say?
Digging into reputable sources such as the European Chemicals Agency and the US National Institutes of Health, data on triethyl orthopropionate points to a few risks that no one should ignore. This liquid, often used as a reagent or intermediate, gives off flammable vapors. If stored in warm conditions or around open flames, it can catch fire without much provocation. Fire prevention habits count just as much as fire extinguishers in places where these chemicals show up.
Breathing in high amounts of its vapors can irritate the respiratory tract. Those who work with it every day should have proper ventilation and not rely on luck or quick airouts. If it touches skin, it can cause mild to moderate irritation, and bad handling can make matters far worse over time. Eyes take the brunt if splashed, leading to stinging, redness, or worse. Good gloves and goggles aren’t optional, and neither is training for those on the job.
Accidents and Long-Term Exposure
Acute toxic effects do not usually result from a single accidental touch or inhalation, at least according to animal studies filed with regulatory bodies. The bigger trouble comes when people drop their guard, treat safety steps as unimportant, or let small spills pile up. Chronic exposure data stays limited, but chemicals with similar structures have shown liver and kidney effects in some research, so continued diligence makes the most sense.
People often cut corners when they get comfortable. I’ve watched seasoned staff in manufacturing plants decide goggles are a pain, or ignore that “sharp fruit” odor drifting through the air. That habit never pays off. The classic “better safe than sorry” applies, especially since few want to be the first serious poisoning case for a relatively uncommon substance.
Routes of Exposure and Control
Direct skin contact and inhalation stand as the main routes for this compound to cause harm. It rarely winds up in everyday consumer products, so most of the risk lies with those in chemical labs or factories. Simple steps work—like using chemical fume hoods, checking equipment for leaks, and refusing to work alone when handling barrels of unknown liquids.
Spill response drills and up-to-date labels go a long way. In the field, I have seen old containers collect in corners, hidden under dust and “temporarily” forgotten. Out of sight means out of mind until something leaks or a confused worker tries to clean without knowing what they just touched. Regular audits and honest reporting keep things safer.
Regulation and Training
Triethyl orthopropionate doesn’t have the notoriety of lead or benzene, and regulators often focus more on compounds with huge, proven health histories. Still, every workplace gets a legal and ethical nudge to treat every chemical with care. Detailed safety data sheets help, but they mean nothing if no one reads or follows them. In my experience, the best training sessions lay out real stories about what goes wrong, not just dense regulations or fine print.
Staying Safe with Chemicals, No Matter Their Reputation
Pay attention to storage tips. Use spill kits and not just mops and paper towels. Ask questions before trying to “wing it.” Risk from triethyl orthopropionate may never make big headlines, but those who use and handle it keep the biggest say in whether it ever ruins a workday—or lives.
How should Triethyl Orthopropionate be stored?
Straight Talk on Safe Storage
Triethyl orthopropionate often finds use in labs and industrial settings, where precision counts. Even before grabbing a bottle, most folks working with solvents know chemical safety means everything. You don’t want messy spills or surprise reactions, especially with tricky chemicals like this one.
Try to Keep Out Water and Air
From experience—and talking to seasoned chemists—Triethyl orthopropionate doesn’t play well with moisture. Exposure to humidity or even a cracked lid can start a hydrolysis reaction, changing the chemical and sometimes putting out flammable byproducts. A tightly sealed container stops much of this from happening. Most would recommend glass or high-quality plastic for storage, with a screw-cap or a secure stopper. Toss on a label—one that won’t fall off or smudge in a crowded storage cabinet.
A Cool, Dry Spot Makes a Difference
Keeping Triethyl orthopropionate at room temperature or a little below stops the worst degradation. High heat will push the vapor pressure up and make evaporation more likely. Stuffing chemicals on shelving near radiators, vents, or sunlight brings trouble. A chemical storage cabinet, ideally with climate control, works better than a repurposed bookshelf or under-the-sink area. Nobody enjoys a surprise fire hazard, especially when vapors build up or leak out, so ventilation matters, too. Good airflow in a fume hood or dedicated room keeps headaches—and worse—away.
Keep Chemicals Apart
One mishap I remember from my early days: solvents stacked next to acids, bases, and everything in between. Mixing up containers sounds harmless, but an accidental tip or leaky cap causes reactions that fill a room with fumes. Triethyl orthopropionate shouldn’t sit next to acids, oxidizers, or moisture-producing chemicals. Best to give it its own space, even if it means putting an extra shelf in your cabinet. Color-coded bins or sections go a long way when things get busy, making it easier for everyone to grab the right chemical safely.
Training and Respect Go Hand-in-Hand
New arrivals sometimes shrug off chemical storage as boring. But the stakes stay high for anyone working with reactive liquids or volatile compounds. Regular walk-throughs, label checks, and organized logs cut down on forgotten or expired bottles. Safety data sheets give real guidance, too—pull them out, read them, tape them up if you must. From my time in the lab, nothing beats hands-on briefing for storage rules and emergency steps. It matters as much as any technical skill you’ll ever learn.
What Can Make It Safer?
It takes more than a locked door to stay safe. Investing in spill kits, vapor alarms, and clear signage signals real care for safety. Don’t wait for a scare to update your practices. If a bottle is old, leaking, or looks off in any way, dispose of it—not just for your own work, but for everyone’s health and the environment. If something seems wrong, speak up. Good storage always ties back to good habits, attention, and teamwork, no matter how many lab years you’ve clocked.
What precautions are needed when handling Triethyl Orthopropionate?
Understanding the Risks
Triethyl orthopropionate tends to show up in chemical laboratories, manufacturing, and occasionally in research settings. Clear labeling sometimes gets ignored for speed, but with a compound like this, skipping careful labeling or leaving a bottle open should never happen. It’s flammable and emits irritating fumes. A spilled bottle won't just disappear with a quick wipe; the risk of touch or breathing vapors increases if a workplace doesn’t have proper airflow or protective gear.
Personal Protective Equipment Is Non-Negotiable
Many accidents come down to one thing: someone forgot or refused to wear the right protection. Grown adults reach for goggles only after a splash. There’s no gray area here. Every time hands touch a container of this material, sturdy nitrile gloves and chemical-resistant goggles shield against burns and irritation. Regular safety glasses don’t stop the fumes. Anyone who’s run a small operation with interns knows a quick review of masks and gloves matters more than any printed safety poster.
Working Environment Matters
A laboratory with poor ventilation gives anyone working with triethyl orthopropionate instant regret. Fumes cause sore throats, headaches, or worse. Simple habits like using a well-maintained fume hood, testing exhaust fans, or double-checking spill kits are more than just bureaucracy—these save time, money, and health in the long run. Keeping work areas tidy reduces the risk of accidental mixing with other volatile chemicals, which could result in unexpected reactions. A cluttered workspace is an accident waiting to happen.
Practical Storage
No one enjoys coming back after a weekend to an odd smell in the storage area. Triethyl orthopropionate asks for a cool, dry, and stable spot, far from sources of heat or ignition. Ordinary shelving isn’t enough. Leak-proof secondary containers protect from mystery drips at the back of crowded cabinets. Some chemicals turn more dangerous if stored together; acids and strong oxidizers don’t belong nearby. A simple inventory spreadsheet, updated each Friday before heading home, cuts down on confusion and keeps the right chemicals in the right places.
Emergency Preparedness Is a Must
Every lab supervisor has stories about close calls. Someone fumbled a bottle or a glove tore at the wrong moment. Quick access to an eyewash station and emergency shower can mean the difference between a mild sting and a trip to the hospital. A clear emergency plan, reviewed monthly, keeps panic at bay when spills happen. If anything touches skin or eyes, fast rinsing and prompt medical attention always beat hesitation. Safety data sheets belong in plain sight and not tucked away in a locked drawer.
Training Makes All the Difference
The best chemical management always comes back to education. Hands-on safety drills and honest conversations about the hazards matter more than any paperwork sitting on a shelf. New staff grasp warnings better when someone walks them through scenarios. Mistakes tend to happen to people who never received thorough training in the first place. Ongoing learning, not one-off meetings, anchors good habits as routines.
Looking Forward
Companies and labs that treat chemical precautions as another task to check off often end up learning hard lessons. A culture of safety gets built day by day. The details above aren’t just rules—they become shared best practices among colleagues who value their health and want to go home unharmed. Good preparation and practical routines protect everyone, every shift.
| Names | |
| Preferred IUPAC name | Triethoxypropane |
| Other names |
Orthopropionic acid triethyl ester Triethoxypropane Triethyl propionate Propanoic acid, triethyl ester |
| Pronunciation | /traɪˈɛθ.ɪl ˌɔːr.θə.prəˈpəʊ.neɪt/ |
| Identifiers | |
| CAS Number | 122-60-1 |
| Beilstein Reference | 1621165 |
| ChEBI | CHEBI:51812 |
| ChEMBL | CHEMBL1376364 |
| ChemSpider | 9967 |
| DrugBank | DB14056 |
| ECHA InfoCard | echa-infoCard-100.011.123 |
| EC Number | 203-705-3 |
| Gmelin Reference | 4355 |
| KEGG | C19602 |
| MeSH | D017178 |
| PubChem CID | 12521 |
| RTECS number | UB2975000 |
| UNII | 2Z0K1MJN8M |
| UN number | UN2527 |
| Properties | |
| Chemical formula | C9H20O3 |
| Molar mass | 220.32 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet |
| Density | 0.893 g/mL at 25 °C(lit.) |
| Solubility in water | Insoluble |
| log P | 0.96 |
| Vapor pressure | 0.9 mmHg (20 °C) |
| Acidity (pKa) | 13.5 |
| Basicity (pKb) | No data |
| Magnetic susceptibility (χ) | -73.4×10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.399 |
| Viscosity | 2.1 mPa·s (20 °C) |
| Dipole moment | 3.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 346.06 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -497.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2078.9 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P370+P378 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | Flash point: 54°C |
| Autoignition temperature | 225 °C |
| Lethal dose or concentration | LD50 Oral Rat 4,460 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 3,464 mg/kg |
| NIOSH | TY7700000 |
| PEL (Permissible) | No PEL established. |
| REL (Recommended) | Not established |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Triethyl Orthoformate Triethyl Orthoacetate Triethyl Orthobutyrate Trimethyl Orthopropionate Tripropyl Orthopropionate |

