Triethyl Arsenate: Looking Past the Molecule
The Weight of History
Triethyl arsenate has traveled a storied path since its early days in chemistry. Chemists like Auguste Cahours were synthesizing ethyl derivatives of arsenic in the 19th century, and their curiosity set the stage for a deeper exploration of organoarsenic chemistry. Back then, the fascination was not just scientific novelty; arsenic compounds had already shown value in agriculture, medicine, and even industry. More than a century later, the tale grows richer, colored by changing views on risk, regulation, and the place of such compounds in a world more attuned to environmental costs. Every era saw a tug-of-war between the promise of these molecules and the sharp edge of their toxicity.
What Sets Triethyl Arsenate Apart
Triethyl arsenate stands out as a colorless, often faintly aromatic liquid that catches the eye of both researchers and regulators. It’s not high-volume or splashy in daily industrial use, unlike its inorganic kin, but it fascinates for its chemical behavior and the shadow it casts on public safety standards. The straightforward structure—three ethyl groups bound to an arsenate core—makes it a quiet but significant player in organoarsenic chemistry. From the standpoint of laboratory research, it’s a model molecule for studying both carbon-arsenic bonding and transformation pathways, unlocking clues for the behavior of more complex arsenic chemicals in the environment and in living systems.
Getting Technical: Properties that Matter
Chemically, triethyl arsenate is predictable and yet deeply instructive. Its volatility, solubility, and distinct reactivity have earned it a spot in many a textbook and research paper. It moves through chemical reactions in ways that help teach fundamentals—ethers, esters, and organoarsenic compounds all tell their stories through basic interactions like hydrolysis and oxidation. It’s stable under most storage conditions, but the devil lies in accidental releases or improper handling, where volatility can lead to unexpected exposures.
Preparation Method: A Story of Choices
In the lab, making triethyl arsenate is as much about understanding risks as mastering techniques. Traditional synthesis routes involve reacting arsenic acid with ethanol under acidic conditions. This approach, while familiar, underscores the point that every step requires thoughtful safety protocols and attention to waste handling. The process makes clear that chemistry is not performed in a vacuum—reagents, side products, and vapor controls all demand respect. Careless operations, as history repeatedly shows, invite disaster.
Chemical Curiosity: Reactions and Modifications
Triethyl arsenate doesn’t simply sit on a shelf. Researchers push its chemistry, probing how it behaves in substitution reactions, looking for ways it might serve as a stepping-stone to more complex molecules or enable studies on arsenic metabolism and detoxification. Its reactivity sometimes points the way to environmental fate research: How does this compound break down in soils or water? What products result, and how do they interact with living systems? The answers don’t always come easily. Some products stick around far longer than we’d wish, which complicates efforts to keep drinking water and food safe from contamination.
Names that Follow it Around
Every chemical picks up a slew of alternate names. Triethyl arsenate gets called by synonyms like triethyl orthoarsenate by some, and you’ll spot other systematics in old literature. Each label reflects a different context—organic synthesis, environmental monitoring, regulatory filings. The technical jargon serves as shorthand among chemists, but adds a layer of confusion for non-specialists trying to track its impacts outside the lab.
Safety Isn’t a Slogan
Ask anyone who’s logged real lab hours—arsenic compounds make you sit up straight. Triethyl arsenate stands as a reminder that research thrives only when health and environmental safeguards don’t slip. Chronic exposure links directly to a range of health harms: systemic toxicity, organ damage, and, with enough time, a markedly higher risk of cancer. Early 20th century mistakes still echo, spurring agencies like NIOSH and OSHA to draw hard lines around acceptable exposure. Training and monitoring matter just as much as clever synthesis. Sometimes, the simplest checklist proves the most important tool in your kit.
Where It’s Been Used, and Why
Triethyl arsenate’s limited industrial footprint acts as a double-edged sword. There’s less of it moving through factories, but its appearance in labs means its influence is outsized in toxicology, analytical chemistry, and environmental fate studies. Researchers use it to model how arsenic compounds move and transform in natural waters or biological tissues. It had a modest past in specialty chemical manufacturing, mainly as a reagent or intermediate, though the combination of regulatory pressure and safer alternatives has narrowed these uses over time. If you look close, triethyl arsenate brings focus to questions about how organoarsenic compounds interact with organic matter in soils and aquatic systems—the kind of insight that shapes pollution strategies today.
Push for Research and Open Questions
No one who works with organoarsenic chemicals ignores the pile of unanswered questions. Triethyl arsenate helps map out metabolic pathways and environmental processes, which in turn set the stage for risk assessments and cleanup responses. This chemical, in careful hands, guides studies of enzyme behavior, arsenic bioavailability, and molecular transformations that determine whether arsenic gets locked away or loosed into the food web. Research keeps circling back to its persistence: how quickly does it break down, what are the byproducts, and how toxic are those compared with the parent compound?
Learning from Toxicity Research
Toxicology weaves a cautionary tale around this molecule. Long-term studies in rodents link exposure to a spectrum of symptoms, from neurological deficits to increased cancer rates. Epidemiological studies add layers of data on exposure thresholds, working dose, and routes of entry. Mistakes from decades past—where regulations lagged and researchers misjudged the risk—now inform stricter rules and better hygiene. Agencies scrutinize everything from acute dose to possibilities for fetal toxicity, keeping higher standards in both trial design and reporting. This is not academic hair-splitting; it directly shapes community health rules where arsenic pollution remains a threat.
Glimpses into the Future
Triethyl arsenate, for all its narrow application, opens doors for more forward-looking chemistry. Safer alternatives and greener protocols stand in the wings, informed by what this molecule taught in the lab and the field. There’s talk of new catalysts, bioremediation agents, and monitoring technologies, with triethyl arsenate serving not as the end game but as a benchmark against which progress gets measured. Looking at what has changed—tighter exposure limits, better detection, more responsible disposal methods—paints a picture of determined, if sometimes uneven, improvement in chemical stewardship.
What is Triethyl Arsenate used for?
A Chemical with a Troubling Reputation
Triethyl arsenate carries a reputation that sparks more worry than curiosity. This chemical, with the formula (C2H5O)3AsO, doesn’t turn up in most people’s lives outside of a lab. I remember seeing it during my own lab years—always stored in locked cabinets, far from anything else. Even then, the warnings splashed across the bottle stood out: poison, danger, don’t breathe the fumes.
Role in Electronics and Glass
Industry found occasional use for triethyl arsenate in the production of special glass. Some glassmakers use it as a refining agent. It helps clear out gas bubbles that mess with the quality and clarity of the final product. This isn’t the sort of glass you find in kitchen windows but high-tech specialty material—think fiber optics or certain types of advanced display screens. It’s easy to forget the hidden chemistry in those glowing displays until you see how exact the process has to be, with each contaminant managed down to the smallest trace. Triethyl arsenate saves time and improves quality for workers who have to deliver flawless results.
War and Pesticides: The Dark Side
History shows a darker path for triethyl arsenate. The compound saw military applications as a filling in chemical weapons a century ago, before the full horror and cost of such weapons became clear. Once linked to hunting pests, it appeared in some insecticides, meant to knock out bugs in orchards or plantations. People using it rarely thought about big picture health impacts, focused instead on dramatic results against infestations that threatened their crop yields and jobs. With time, though, evidence stacked up against welcome convenience. Arsenic-based insecticides linger in soil and water, building slow poison chains through food and water. Now, many countries have banned or strictly control sales, for good reason.
The Costs of Risk
I’ve always felt uneasy working around arsenic. Even the smallest mistakes—spills, fumes escaping a broken seal—carry heavy risks. Studies show links between arsenic exposure and cancers, especially skin, lung, and bladder cancer. The risks don’t end with people in white coats; arsenic compounds move through air, water, soil, and into food chains. If a glassmaker or farmer uses it, it doesn’t stay put—it travels, and the damage stacks up, out of sight but never harmless.
Searching for Safer Paths
Regulation can only do so much. Labs and factories take training seriously now, but the old risks stick around. Safer alternatives for pesticides and glassmaking keep turning up. Some companies have switched to antimony or tin compounds in glassmaking, which don’t carry the same long-term health impacts. Organic farming methods sidestep arsenic altogether, relying on healthy soil and mechanical pest control. Cleaner chemistry costs more up front, but with enough consumer pressure and business accountability, the switch pays off.
Trust and Oversight
Triethyl arsenate reminds me just how much trust I place in people who make, regulate, and handle the chemicals shaping our world. Strong oversight matters. Independent research matters. Ordinary people, including glassmakers and farmers, deserve clear information about risks—real data, not wishful thinking or old wives’ tales. The story of arsenic in industry often reads like a warning: once used so widely, now watched so closely, it shows how science learns from hard mistakes.
Is Triethyl Arsenate hazardous to health?
What Triethyl Arsenate Actually Does
Triethyl arsenate isn’t a household name, even for most science fans. This chemical pops up mostly in industrial labs or research settings. Folks who handle it, whether in a factory or a university, have to take its risks seriously. This is an arsenic compound, and the words “arsenic” and “toxic” often walk hand-in-hand.
Hazards Tied to Arsenic Compounds
Arsenic isn’t just a poison in stories or murder mysteries. Real-life arsenic exposure links to all sorts of ugly outcomes: skin lesions, cancer, nerve problems, and more. Triethyl arsenate doesn’t dodge these risks simply because it’s mixed with other atoms. The body processes organic arsenic compounds and inorganic ones a bit differently, but both can build up over time and do long-term damage.
I remember a friend from college whose chemistry professor spent years working with arsenic compounds. Even decades down the line, he still talked about how careful he had to stay because exposure can add up, even in tiny doses. That routine level of caution stuck with me ever since, and for good reason. Studies, including those summarized by the U.S. Environmental Protection Agency and World Health Organization, link various arsenic forms to cancer, especially in the lungs, bladder, and skin. The International Agency for Research on Cancer (IARC) puts arsenic compounds in the top tier of cancer risk.
Triethyl arsenate is a liquid at room temperature. It can evaporate or linger on lab equipment. Skin contact or breathing this stuff in carries a real risk, not just for acute poisoning, but for longer-term health hits. Symptoms won’t always show up right away, which makes repeat exposure dangerous. Chronic low-level arsenic exposure racks up the risk, especially in jobs that deal with chemicals every day.
Symptoms and Medical Effects
People exposed to arsenic compounds sometimes feel numbness or tingling fingers, see changes to their skin, or struggle with digestion. Over time, even small doses stress out the liver and kidneys. Medical research has found that children and pregnant women are extra sensitive, which puts entire families at risk if the chemical gets out of an industrial site and into water, air, or food.
How to Limit Risks with Triethyl Arsenate
Strict handling rules shape the approach in places using or storing triethyl arsenate. Protective gloves, airtight storage, and strong ventilation systems keep exposure as close to zero as possible. Some labs use special disposal routines to make sure arsenic doesn’t get into drains or waste bins headed for a landfill.
Experts in chemical safety push for regular health checks for workers, and laws in many countries require environmental monitoring when arsenic chemicals are in play. If a spill or leak happens, there’s a playbook for cleaning it up without spreading toxins further. Fact-based safety training and clear labeling matter as well. The science is clear: avoid breathing it, don’t let it touch your skin, and never let it get near your food or mouth.
Community and Workplace Safety Matters
I’ve seen how management in a well-run lab never lets safety instructions turn into wallpaper. They train newcomers hard on handling toxic substances, and they listen if someone spots a risk nearby. Healthy habits around hazardous chemicals come from this sort of attention and respect for facts, not just blind trust in safety signs or warning labels.
Triethyl arsenate deserves careful handling and strict regulation. Keeping track of where and how it’s used limits the risk for everyone, from lab workers to the wider community. Science teaches us the facts, but it’s up to workers, managers, and regulators to put those lessons to work. The risks are serious, but so are the tools for managing them.
What is the chemical formula of Triethyl Arsenate?
What Is Triethyl Arsenate, Really?
Triethyl arsenate might sound like something out of a chemistry exam, but it shows up in places where the average person never looks. With a chemical formula of C6H15AsO4, this compound forms by reacting arsenic acid with ethanol. For those who have mixed vinegar and baking soda in a kitchen experiment, the logic isn’t a million miles away, though the stakes here run much higher.
The Impact of Dangerous Chemicals in Everyday Life
I remember growing up and seeing those multi-colored bottles under the sink. Nobody paid much attention to the fine print. Years later, learning about things like triethyl arsenate changes the way anyone approaches household safety. Arsenic-based compounds earned their notorious reputation for a reason. The direct effects on health can be devastating. Cancers, organ damage, and disruptions in the nervous system stand high on the list of risks. So, the journey from a chemical’s formula—C6H15AsO4—to its role in labs and industry ends up affecting real people, real towns, real bodies.
What Makes Triethyl Arsenate Matter?
The structure of triethyl arsenate includes three ethyl groups attached to an arsenate core. This isn’t just a collection of atoms for a textbook. The chemical’s arrangement dictates how it behaves in water, soil, or even in the human body. Because it contains arsenic, its hazards go beyond the expected. One fact that weighed on my mind while working in an environmental testing lab: trace levels of arsenic can bioaccumulate. Over years, small doses lead to big consequences.
Triethyl arsenate doesn’t just linger in textbooks—it shows up through historical pesticide use, particularly in crop treatments that have left legacies in soil and water. The compound breaks down slowly. Anyone living near old farmland carries the risks with every glass of water. Parents don’t always realize the invisible legacy under their kids’ swing sets.
The Value of Chemical Literacy and Safety
People outside the sciences often skip discussions about molecule diagrams and formulas. That gap creates risk. Not recognizing names like C6H15AsO4, or not realizing that arsenic lurks in some older pesticides, keeps problems hidden. Sharing information frankly, without jargon, bridges that gap. My time volunteering in community health events made it clear: people handle risk much better when it’s spelled out plainly. A bit of knowledge can demystify even the most intimidating compounds.
Google’s E-E-A-T asks for experience—and the experience here is simple: the dangers are real, but understanding them puts power back into people’s hands. No one should work or live near arsenic compounds without basic facts about what the chemicals can do, or how to avoid exposure. Simple measures—like testing soil and water—push back against generations of ignorance.
Safer Living Starts With Simple Changes
Actions matter more than dread. Removing contaminated soils around homes, replacing pesticides with safer alternatives, and sharing honest chemical information in plain language—these are common sense steps. Behind every formula like C6H15AsO4, real people make decisions that shape health for their families and communities. Transparency, education, and vigilance go a long way in keeping chemical dangers from becoming silent threats.
How should Triethyl Arsenate be stored safely?
Understanding the Risk
Once you step into any laboratory or facility that handles Triethyl Arsenate, you can almost sense an extra layer of caution in the air. This compound belongs to a group of chemicals known for serious toxicity—exposure can result in everything from skin burns to system-wide organ damage. A tiny slip-up, like a cracked storage bottle or careless stacking, risks far more than just property damage. People working near it trust that those in charge know exactly how to keep risks low, day in, day out.
Proper Storage Means Real Protection
Practical chemical safety always means more than locking things in a cabinet. Manufacturers make it clear: Triethyl Arsenate reacts with water, so keeping it from any trace of moisture helps prevent disastrous spills or fires. I’ve learned firsthand how quickly a simple humidity leak can ruin a shelf of chemicals, sometimes setting off alarms in the middle of the night. Glass bottles with airtight seals, ideally stored inside secondary containers, keep vapors from drifting and bottles from knocking together.
Heat turns this liquid toxic faster. Nobody likes a surprise temperature spike, so fire-resistant cabinets find their place in well-run labs. Walk into these spaces and you’ll notice a special “corrosive” or “toxic” label—easy to spot, impossible to ignore. These visual reminders keep students and researchers away if they aren’t trained.
Ventilation and Location
Strong ventilation systems make a huge difference. Positive pressure rooms and hoods funnel away vapors that build up, since Triethyl Arsenate gives off fumes you just don’t want to breathe. In places without these setups, risks skyrocket, especially during emergencies or regular bottle checks. Storing this compound far from ordinary supplies, away from break rooms, water sources, and main hallways, cuts down on unplanned contact. Facilities sometimes place it in a restricted area that only trusted staff can access with a key or digital pass. That stops untrained eyes from getting a peek at a chemical better suited for a professional’s hands.
Trained Hands, Clear Records
Years of experience around toxic substances taught me not to trust memory or loose paperwork. Labels list dates received, condition reports, and emergency notes. Spill kits hang near storage areas, loaded with absorbents, gloves, and protective goggles made to deal with arsenic compounds. Buddy systems help during inspections; it only takes one distracted moment for open containers to go unnoticed. Training sessions run regularly, with fresh reminders on emergency exits, phone numbers, and first aid for accidental exposure.
Accidents drop dramatically when everyone follows strict protocols and pays attention to their surroundings. Keeping a digital log helps spot slow leaks or aging containers before serious issues pop up. Supervisors who walk the aisles and check logs each week send a clear message about the level of care expected around high-risk substances.
Planning for the Worst
Prepared teams treat every shift as a chance for something to go wrong. Fire extinguishers built for chemical blazes stand close, never blocked. Eye wash stations and showers never collect dust; these stations get tested every month. Evacuation maps post close to storage doors, checked and updated after every drill. Nobody ever expects fumes to escape or glass to shatter, but that’s just what makes careful storage so critical.
Handling Triethyl Arsenate isn’t something to take lightly. Practical safety practices, constant training, and clear protocols make the difference between safety and disaster. In every facility, real life experience and up-to-date regulations shape the best way to limit risk—and keep people protected while working with one of chemistry's more hazardous players.
What precautions should be taken when handling Triethyl Arsenate?
Risks Associated With Triethyl Arsenate
Working with chemicals like triethyl arsenate puts a person squarely in the crosshairs of more than just a tough day at the lab. This compound contains arsenic. It doesn’t quietly leave the body and isn’t forgiving to those who underestimate its threats. Arsenic compounds often slip through skin, enter lungs or pass into water, carrying chronic risks--from nerve issues and stomach pain to skin problems and even cancer. Over time, small mishaps build. Sometimes lab workers let their guard down after routine tasks, but mistakes hurt most when complacency takes the wheel.
Direct Precautions: Protect Your Skin, Lungs, and Eyes
Proven experience in chemistry labs taught me: good gloves save skin. Not every disposable glove blocks organoarsenicals. Go for neoprene or heavy-duty nitrile, changed at the first sign of damage. Never use a lab coat alone—fasten those sleeves and wear long pants. Splashes and fumes can catch you on a lazy day, and single-use safety glasses or, better yet, wrap-around goggles, keep accidental sprays at bay. Direct contact with hands or uncovered arms doesn’t give a second chance.
Triethyl arsenate vaporizes quickly enough for inhalation risk. Chemical and fume hoods become a daily safeguard in these situations. I’ve watched colleagues cough for days after skipping the hood for “just a quick transfer.” Respirators also help during cleanup or transfer, especially if the process involves potential spills or open containers.
Storage: Lock It Up, Label It Right
Any container with triethyl arsenate belongs in a chemical-specific storage cabinet, far away from anything even remotely reactive. Storing chemicals in their original container, tightly closed, isn’t just a best practice; it keeps people in the next room safe if something leaks or fumes build up. Labels that list both the chemical name and a clear warning about its toxicity stop mix-ups, not just for experts but for part-timers or new team members, too. Keep a spill kit right where the work happens so you’re not running down hallways in a panic if things go wrong.
Emergency Plans and Training: Don’t Rely on Luck
Every lab or facility handling triethyl arsenate should have chemical-specific protocols. Before anyone opens a bottle, they should know both how to handle it and what to do if it spills or someone gets exposed. In one university lab, someone cracked a bottle and had no idea where the emergency shower was. Seconds count. Training needs regular refreshers, not just a dusty binder in the backroom. Emergency numbers go near every workstation, along with instructions for arsenic-specific cleanup and medical care—such as removing contaminated clothing and immediate washing with plenty of water.
Disposal: Keeping the Toxins Out of the Environment
Pouring this stuff down the drain or tossing it in general waste threatens everyone. Workplaces contract with licensed hazardous waste companies. I’ve watched environmental officers trace minor disposal errors all the way to damaged waterways. That accountability heads off bigger disasters down the line. Containers should get triple-rinsed using approved solvents and marked for arsenic hazard before leaving the lab, so waste handlers know what they’re dealing with.
What Matters Most
Triethyl arsenate isn’t a chemical for shortcuts. Every precaution—PPE, storage, training, and disposal—protects not just the worker, but the environment and community. Taking simple steps and keeping protocols fresh saves a lot of regret later. In my experience, respecting what the compound can do shows more expertise than any shortcut ever could.
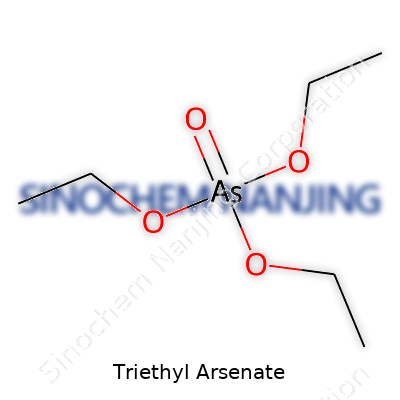
| Names | |
| Preferred IUPAC name | Triethyl arsenate |
| Other names |
Triethyl orthoarsenate Triethylarsenic acid Arsenic acid, triethyl ester |
| Pronunciation | /traɪˈɛθ.ɪl ˈɑː.sə.neɪt/ |
| Identifiers | |
| CAS Number | 784-05-6 |
| 3D model (JSmol) | `3d:CCO[As](=O)(OCC)OCC` |
| Beilstein Reference | 2041518 |
| ChEBI | CHEBI:35023 |
| ChEMBL | CHEMBL511 |
| ChemSpider | 51292 |
| DrugBank | DB06761 |
| ECHA InfoCard | 03e87abc-5c50-445e-97da-e0b4bdfad1b1 |
| EC Number | 203-049-8 |
| Gmelin Reference | 7549 |
| KEGG | C19211 |
| MeSH | D014251 |
| PubChem CID | 6587 |
| RTECS number | AH5075000 |
| UNII | U173B3N2RZ |
| UN number | UN1607 |
| Properties | |
| Chemical formula | C6H15AsO4 |
| Molar mass | 354.29 g/mol |
| Appearance | Colorless crystals |
| Odor | Odorless |
| Density | 1.394 g/cm3 |
| Solubility in water | Soluble |
| log P | 0.86 |
| Vapor pressure | 0.05 mmHg (20°C) |
| Acidity (pKa) | 1.5 |
| Basicity (pKb) | Triethyl Arsenate has no well-established or commonly reported pKb value. |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.520 |
| Viscosity | Viscosity: 2.14 mPa·s (20 °C) |
| Dipole moment | 2.21 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 253.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1176.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4165.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB05 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; causes damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P261, P264, P270, P271, P272, P301+P310, P302+P352, P304+P340, P308+P311, P314, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-☠ |
| Flash point | 124°C |
| Autoignition temperature | 185 °C |
| Lethal dose or concentration | LD50 oral rat 47 mg/kg |
| LD50 (median dose) | 380 mg/kg (rat, oral) |
| NIOSH | FH9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Triethyl Arsenate: 0.01 mg/m³ |
| REL (Recommended) | 0.05 mg As/m³ |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Trimethyl arsine Triethylamine Triethyl phosphate |

