Trichlorotrifluoroacetone: Chemistry, Progress, and Where We Go From Here
Tracing the History of Trichlorotrifluoroacetone
Every chemical compound tells a story, and Trichlorotrifluoroacetone's roots reach back to a time when chemists craved new tools for synthesis and analysis. The rise of organofluorine chemistry took off in the mid-20th century after researchers noticed the unique behaviors that halogenated ketones offered for reactivity and selectivity. Old industrial processes left their mark, but the focus soon shifted to more specialized syntheses as organic science matured. In my experience, the slow but steady documentation in open literature points to a pattern: early usage rarely wins headlines, true value emerges as the scientific community figures out how a quirky molecule fills a gap. This has certainly applied to Trichlorotrifluoroacetone as folks started to recognize its ability to act as a building block and analytical reagent.
Looking Over the Product and Its Unique Traits
Trichlorotrifluoroacetone stands out. It offers a structure where three chlorine atoms balance the flash and fire of a trifluorinated acetyl group. That combination crafts a molecule that does more than just look interesting on paper. The physical characteristics matter to those of us who handled chemicals in both the classroom and the lab: a clear, sometimes pale liquid, whose volatility delivers both hazard and advantage. The sharp odor can make you respect its power. A high density and notable boiling point give you cues before you even run a reaction. What fascinates me is the way its chemical backbone shrugs off many traditional reagents, yet dives headlong into specific transformations, reinforcing that some molecules really can be both robust and responsive.
Dissecting Key Properties and Behavior
For people used to mixing and matching solvents and reagents, the physical properties drive your choices. Trichlorotrifluoroacetone won't dissolve everything under the sun, but it slips into organic layers with ease while keeping its main reactive sites shielded until needed. A triple halogen substitution (three chlorines, three fluorines) by itself turns a molecule into a tightly wound spring of chemical potential. The keto group adds another layer — making this compound more than just a novelty, but a player in enolate chemistry, halogen metathesis, and selective nucleophilic attack. As temperatures shift, the behavior changes too: in my work, overheating leads to breakdown or unwanted side reactions, but—handled mindfully—the compound stays stable enough for most anticipated processes. Its resistance to hydrolysis became a selling point in analytical circles, where persistent reagents open doors to consistent measurements.
Technical Standards and Essential Labeling
Accurate labeling becomes more than just a paperwork matter with compounds like Trichlorotrifluoroacetone. Every shipment, every bottle, ought to map back to specific purity discounts and known impurities. Precise reporting for percentage purity, trace elements, and solvent residues makes a difference both for reproducibility in research and for safety. Anyone who has worked in industry will admit that a slip-up — even a 0.5% unknown contaminant — can shift the results, especially in catalyst systems and sensitive synthetic routes. Label transparency isn't an arbitrary requirement; it's a reflection of hard-learned lessons from years of scale-up accidents and mysterious batch failures that nobody wants to re-live.
Synthesizing Trichlorotrifluoroacetone: Practice and Progress
Commercial preparations often trace their lineage to old chlorination and fluorination protocols, sometimes involving hazardous intermediates. The healthiest progress comes when manufacturers embrace cleaner, less hazardous reagents and find a way to reduce byproducts and waste. Early syntheses left scars on both the environment and the operators, reminding us that improvement is both a technical and ethical obligation. The common synthetic approaches now lean on carefully controlled halogen exchange reactions, rigorous temperature management, and select catalysts that limit unwanted rearrangements. From my discussions with process chemists, control remains everything—from reaction time to the purity of starting materials, from vigilant monitoring to strict containment. Small mistakes can lead to runaway reactions, leaks, or hazardous exposure.
Chemical Reactions and Modifications: Room to Explore
What gets the synthetic chemist excited is Trichlorotrifluoroacetone's nature as a reactive platform. The molecule finds a seat in both nucleophilic and electrophilic transformations. Common lab techniques make use of its keto group for condensation reactions, where its electronic structure helps steer selectivity rarely matched by simpler ketones. The presence of strong electron-withdrawing substituents — three chlorines next to three fluorines — pulls electron density hard, making this chemical a prime candidate for enolate generation and subsequent coupling reactions. The beauty lies in how easily small modifications shift reactivity trends—one chlorine gone, one functional group switched, and you might get a drastically different outcome. Researchers have made headway into using it as a ligand precursor, an intermediate for custom fluorinated organics, and even as a trap for elusive nucleophiles that standard ketones miss.
Names by Which Trichlorotrifluoroacetone Hides
The story of a chemical's aliases sometimes reads like a global scavenger hunt. In journals and catalogs, you'll see labels such as 1,1,1-Trichloro-3,3,3-trifluoroacetone, or by its CAS number, and sometimes as trifluorotrichloroacetone or by simplified acronyms in process documentation. Multiple names can trip up even the experienced chemist and muddy the waters for purchasing and compliance, so a careful cross-check of nomenclature and molecular structure saves more than a little embarrassment or overhead in both ordering and research conversations.
Keeping Safety at the Forefront
Everyone who steps foot in a chemical lab needs a gut sense of how hazardous substances behave — and Trichlorotrifluoroacetone asks for respect. The blend of volatility, halogen content, and potent reactivity gathers risks on several fronts. Direct skin or inhalation exposure isn't just unpleasant; it can rapidly provoke symptoms or long-term effects that catch unprepared hands off guard. I remember people who underestimated this class of halogenated ketones and paid for it with acute discomfort and weeks of paperwork. Toxicological records emphasize careful ventilation, barriers like gloves and face shields, and highly practiced storage routines—no exceptions, no shortcuts.
Where the Compound Makes Its Home: Applications from Lab to Industry
The real-world utility of Trichlorotrifluoroacetone shows up as much on the benchtop as it does in specialty production lines. Analytical chemists value the compound as a chelating agent and a derivatization reagent. It enhances sensitivity or changes the volatility of target molecules, making identification easier and more reliable in spectrometric assays. In the private sector, I've seen it used for fine-tuning pharmaceuticals and agrochemicals — forming new carbon–carbon bonds or introducing halogenated motifs for improved bioactivity or metabolic stability. Custom organic syntheses rely on its dual halogen content for fine control in multi-step routes. Its reputation in coordination chemistry also pays dividends, with some researchers chasing after unique metal complexes utilizing the ketone's backbone as a scaffold.
Pushing Forward: Ongoing Research and Creative Uses
Researchers continue poking at the edges of what Trichlorotrifluoroacetone can do. Its unusual electronic structure has triggered interest in asymmetric synthesis, especially among teams aiming to construct fluorinated frameworks relevant to new medicines or materials. There’s a push for greener methods — both in how the compound is made and in how it’s used — since awareness of its persistence and potential environmental burden grows. Teams are now working on tweakable analogues that keep beneficial features while lowering toxicity or simplifying the work-up. Applications in advanced electronics, through incorporation in specialty polymers or as a functional monomer, get more real as organic electronics expands. Monitoring these threads from publications and conferences, I see a cycle: niche uses spawn further innovation, which then trickles out to broader applications as prices fall and tools improve.
Toxicity and Health: Knowledge Driving Protection
Toxicology data crafts the rulebook for safe handling. Trichlorotrifluoroacetone, like its halogenated kin, can be unforgiving when inhaled or spilled across unprotected skin. Early tests, often in animals or cell cultures, flagged acute toxicity and the possibility of cumulative organ effects, particularly from long-term exposure. Having spoken with industrial hygienists, I know they keep an eye on both acute and chronic endpoints, plus tricky byproducts that might form under heat or UV exposure. Waste disposal protocols have moved past the bad old days of simple incineration or uncontrolled landfill. Today’s standards call for complete destruction using well-baffled scrubbers and proper tracking. There’s plenty of room for additional research, especially as new applications raise the chance of unintended contact.
The Road Ahead: Promise and Practical Hurdles
Looking toward what's next for Trichlorotrifluoroacetone, much hangs on the balance between chemical ingenuity and responsible stewardship. On one hand, the molecule promises significant advances for custom synthesis, screening libraries, and specialized analytical work. On the other, regulatory pressures and a changing environmental ethos demand better lifecycle management and substitute development when possible. Future progress asks not just for smarter chemistry, but for wider conversation between academic labs, industry players, and public advocates. Training the next generation of chemists now includes both technical skill and risk literacy, especially as new uses for compounds like this emerge. Building a sustainable roadmap often comes down to honest data sharing, continuous monitoring, and open-eyed adaptation to both opportunities and the lessons of past mistakes.
What is Trichlorotrifluoroacetone used for?
The Chemistry Behind the Name
Trichlorotrifluoroacetone sounds like a mouthful, but for those who spend time in the chemistry lab, the compound has a clear role. With its unique set of chlorine and fluorine atoms, it stands out both for how it behaves and what it can do. People working in chemical synthesis value this molecule because it offers a set of reactive properties that can’t be found in standard ketones.
Trusted Tool in Coordination Chemistry
Most lab veterans remember the first time they saw this stuff in action. Its main value comes into play with metal complexes. Chemists use trichlorotrifluoroacetone as a chelating agent. That just means it helps “grab” onto certain metal ions, like lanthanides and transition metals, and holds them close. This technique lets researchers pull out specific metals or stabilize them in a certain state.
Without reliable chelating agents, work with these metals gets messy. That can slow down projects across pharmaceutical labs, material science benches, and even nuclear fuel research, since many of these areas need separation or stabilization of rare metals. Having a compound that reliably does that job saves time and avoids wasting precious resources, and nobody working on a grant deadline sneezes at that.
Role in Analytical Chemistry
Straightforward analysis depends on useful reagents. Trichlorotrifluoroacetone helps out in spectroscopy—an essential lab technique worldwide. Analytical chemists use it to create strongly fluorescent complexes that register at low detection thresholds. Detecting trace amounts of heavy metals becomes easier, which supports not just fancy research, but routine water and soil testing too. Municipal labs and environmental agencies often rely on this technology to spot toxins before they reach the food chain.
Specialized Organic Synthesis Shortcut
Many advanced molecules start with difficult transformations. The presence of both chlorine and fluorine atoms in trichlorotrifluoroacetone lets it spark reactions most other reagents miss. During retrosynthesis planning, organic chemists often note what functional groups a molecule brings. Trichlorotrifluoroacetone delivers both electron-withdrawing power and space for modifications. It’s picked to make specialized building blocks in drug design, crop-protection agents, or fluorine-containing polymers known for their resilience.
Handling Real-World Pitfalls
With all these uses, the compound comes with proper handling warnings. Chlorine- and fluorine-rich molecules tend to carry reactivity risks. Anyone who’s prepped a complex with it remembers the need for good gloves, eye protection, and a solid fume hood. Safe lab routines shape how widely these chemicals get adopted outside of specialized centers. Mistakes can lead to costly exposures—an outcome that can shut down a project or risk lives.
Looking for Solutions Going Forward
Some researchers see green chemistry as the next step. They’re working on ways to get the benefits of trichlorotrifluoroacetone without the hazards. Teams look for biodegradable alternatives or ways to recover it after use, cutting down hazardous waste. Sharing these best practices in journals and at conferences opens the field to more scientists who might otherwise steer clear of compounds with safety baggage.
We use trichlorotrifluoroacetone because the benefits remain clear in certain corners of chemistry. Its role as a metal complexing agent, sensitive analytical tool, and synthesis aid can't be ignored—especially when precision and sensitivity make the difference between a breakthrough and a setback.
What are the safety precautions when handling Trichlorotrifluoroacetone?
Introduction: Reality in the Lab
Most chemicals don’t announce their hazards before you pop the lid. Trichlorotrifluoroacetone might sound like just another tongue-twisting compound, but experience tells a different story. It’s not only about its tongue-twisting name—this stuff can pack health risks for those not careful. Over the years, I’ve learned through direct handling, not just book instructions, that organizing safety starts before you ever open the container.
Personal Protective Equipment Isn’t Optional
If you’ve ever worked with halogenated organics, you get it—nitrile gloves prevent skin from stinging, and splash-resistant goggles keep vapors out of eyes. Long-sleeved lab coats and closed-toe shoes aren’t just formalities. Even the most seasoned techs I know learned the hard way by skipping a glove or ducking a face shield for a “quick” task. I’ve seen irritation and coughing after short contact. Respecting those barriers has saved me more than one trip to the medical office.
Ventilation Counts—Fume Hoods Are Worth Their Weight
I’ve worked in rooms that felt air-tight and stuffy and in spaces with a humming fume hood. Trichlorotrifluoroacetone isn’t a fragrance—its volatility and reactive nature belong behind glass shields. Routine transfers, storage, and waste disposal take place under local extraction. No exceptions if you want to avoid headaches or worse. It’s a lesson many learn after a whiff gone wrong or after a day with upper airway pain.
Spill Response Starts Before the Spill
Several years back, I saw a small bottle slip. Instead of panic, the team reached for a pre-stocked spill kit. Absorbent pads, baking soda for neutralization, dedicated respirators—the basics were all there. I can’t emphasize enough that clean-up plans and drills let everyone sleep better at night. Alkali neutralizers keep things safer thanks to the compound’s acid chloride nature, which could otherwise react badly with water.
Label Everything, Even If Only You Handle It
Substituting containers or leaving a flask unmarked creates confusion and breeds accidents. I once found an unlabelled reagent and later learned it contained concentrated Trichlorotrifluoroacetone. That close call stuck with me. Labeling prevents unintended mixing, cross-contamination, or accidental disposal—especially when temporary storage becomes much longer than planned.
Storage Decisions Matter
Temperature and humidity fluctuate in most older labs. Storing Trichlorotrifluoroacetone away from heat, strong bases, oxidizers, and sunlight reduces the chance of decomposition or unexpected reactivity. I always advocate for secondary containment and keeping incompatible materials well separated. Too many incidents I’ve read about—and a few I’ve seen—began with bottles resting side by side on overcrowded shelves.
Training Makes All the Difference
No one walks into a new chemical environment and handles things perfectly. I’ve mentored new lab techs and insisted on practical safety drills. Digital safety data sheets are good, but hands-on instruction on virtual tours or mock incidents makes procedures stick. Sharing both near-misses and best practices shapes a safety culture that protects everyone.
Better Safety Through Shared Responsibility
Handling Trichlorotrifluoroacetone safely isn’t just about rules set by a supervisor. It grows from treating every step with respect, drawing on lessons from others, and never getting complacent. Real vigilance and a healthy dose of humility seem old-fashioned to some, but they keep workspaces safe and productive. The bottom line: don’t cut corners, and always look out for your team along the way.
What is the chemical formula of Trichlorotrifluoroacetone?
The Science Behind Trichlorotrifluoroacetone
Trichlorotrifluoroacetone might sound complicated, but its chemical formula gives a clear picture: C3Cl3F3O. This isn’t some abstract mix of letters and numbers. It’s a blueprint detailing exactly what goes into this molecule. Each atom in the formula matters—three carbons, three chlorines, three fluorines, and one oxygen. Chemists often piece together formulas like these, not just to show off their knowledge, but to explain how molecules react and interact, which really determines their value in the real world.
What Makes C3Cl3F3O Stand Out?
C3Cl3F3O belongs to a category known as beta-diketones. In my own days at the lab bench, I found that these compounds often pop up in high-impact research—from coordination chemistry to material science. Trichlorotrifluoroacetone, with both chlorine and fluorine atoms stacked onto the framework, isn't just some theoretical structure. Those halogen atoms—chlorine, fluorine—give the molecule unique traits. For instance, the strong electron-withdrawing nature of fluorine makes the compound more reactive in some situations, helping researchers fine-tune chemical reactions or develop advanced materials.
Why Accurate Formulas Matter
Mixing up a chemical name or formula isn’t just an error on paper. Getting the formula right can save time, money, and sometimes even lives. In the pharmaceutical industry, for example, a misplaced atom changes everything—a medicine’s effects, interactions, or even its safety. Every batch starts with an accurate formula. In my own experience, labs double-check these details long before products move out the door. Having rigorous attention to detail keeps research credible and products reliable.
Everyday Impact & Responsible Handling
Most people will never see trichlorotrifluoroacetone outside a lab, but its uses reach far. Chemists use compounds like this for making specialized ligands, extracting metals, or synthesizing complex molecules. Still, this substance also raises safety issues—remember, halogenated organics bring health and environmental challenges. Chlorine- and fluorine-heavy compounds don’t break down easily. If waste isn’t treated right, it can stick around in the soil or water for years. I’ve seen first-hand how labs now use tighter controls when handling materials with strong halogens, pushing for safer disposal and recycling. Green chemistry isn’t just a buzzword—it matters every time we open a new reagent bottle.
Finding Solutions and Moving Forward
It’s not enough for chemistry to stay in the lab. Industry has a responsibility to use powerful chemicals like trichlorotrifluoroacetone wisely. That means building better processes for both making and disposing of them. Researchers now hunt for alternative compounds with lower toxicity or turn to closed-loop systems to keep hazardous materials out of the waste stream. Companies invest in new filtration tech and purification protocols. These steps don’t just protect the environment—they protect workers and communities too.
Understanding formulas and their wider impact helps scientists, manufacturers, and regulators make smart decisions. The best practices in chemistry come from keeping a close watch on both what goes into a reaction and what comes out. That sort of thinking leads to safer products, cleaner processes, and a stronger trust between the science world and everyone who relies on its discoveries.
How should Trichlorotrifluoroacetone be stored?
Why Proper Storage Matters
Trichlorotrifluoroacetone sounds like something you don’t want in your morning coffee. At first glance, it’s another lab chemical with a long name and a complicated formula, but mishandling this substance doesn’t only ruin a research project; it puts people at genuine risk. Volatile compounds such as this one highlight a truth those in science learn fast—small missteps can turn an everyday lab into a danger zone. In college, one careless cap left unsecured exposed our whole building to an unexpected stench and a safety scramble. It taught us firsthand that chemical storage shapes the safety of everyone around.
Keep It Dry, Keep It Cool
Moisture does no favors to trichlorotrifluoroacetone. Water gets involved, and suddenly you’re looking at corrosive byproducts and unpredictable reactions. Dry storage goes beyond tossing a container on a shelf. It means finding a place away from sinks, steam, or anything that might invite humidity. In some labs, that might call for a dedicated desiccator or a tightly sealed container with silica packets. The difference between a functional chemical and a contaminated one may just come down to a dry cabinet.
Heat turns this chemical into a problem quickly. Low temperatures slow decomposition and evaporation, and the risk of accidental inhalation drops with the vapors locked down. My old lab stuck to refrigerators designed for hazardous materials, rather than the food-stuffed fridges at home. Let’s be honest: nobody wants toxic fumes mingling with last night’s takeout.
Ventilation and Isolation
Storage doesn’t just mean “put out of sight.” Good air movement keeps any accidental vapors from collecting, so a chemical storage cabinet with built-in ventilation makes a big difference. Fume hoods are standard fare in chemistry departments, and they do serious work containing leaks. Sealing everything in proper containers, made from materials the chemical won’t eat through, keeps exposure away from the people who just walked in for their shift.
Isolation matters more than new lab workers sometimes realize. Mixing incompatible chemicals leads to release of hazardous fumes or even fires. Trichlorotrifluoroacetone doesn’t play well with strong bases or oxidizers, so tossing it on a shelf with general supplies isn’t playing it safe—it’s asking for an emergency. At our facility, labels go on every side, and incompatible chemicals don’t share a cabinet.
Access and Responsibility
Not everyone should have free access to hazardous materials. Restricting entry to those with real training and a need for the material keeps accidents in check and ensures that storage rules get followed every time. Locked storage cabinets and tight inventory lists protect everyone, including the folks who might not realize what risk they’re walking into.
Protecting Health with Simple Steps
Gloves, goggles, and lab coats sound like basic stuff, but these barriers keep skin and eyes safe from accidental spills. Routine checks for leaks or damaged containers cut down surprises. It’s tempting to rush, especially with a crowded lab schedule, but every time protocols slip, another chance for danger pops up.
Solving safety issues with trichlorotrifluoroacetone isn’t about new technology. It’s about consistent respect for hazards, routines that everyone follows, and communication when things change. Real safety never happens by accident.
Is Trichlorotrifluoroacetone hazardous to health or the environment?
Chemicals in Real Life: What We Deal With
People who work in labs, factories, and even university classrooms bump into chemicals with long, tongue-twisting names. Trichlorotrifluoroacetone, for example, sounds like something out of a chemistry textbook, but it shows up in real products and research. It can serve as a building block for other compounds or as a specialized solvent. Some see it only as a tool; others wonder what risks come with it.
How Trichlorotrifluoroacetone Affects Health
Exposure routes count: inhalation, skin contact, even ingestion, whether by accident or poor handling. People in research and manufacturing have the front-row seats. Available safety data—though not exhaustive—raises eyebrows. Evidence points toward skin and eye irritation. Inhaling vapors leads to coughing, watery eyes, sore throat, and sometimes chest tightness. Long-term exposure information remains sparse, which leaves a big blind spot. Manufacturers classify chemicals like this as harmful if swallowed or inhaled, and warnings about possible organ effects after repeated exposure keep health officers on alert.
Personal experience in chemical work teaches quick respect for anything with halogen atoms. Similar compounds can disrupt liver or kidney function. In the late 2000s, I remember a colleague developing a rash after a minor spill with a related solvent—no gloves, just a short moment, and he had to see the company doctor. Not all chemicals act the same, but it's better to overestimate risk than pay the price. If your work brings you near trichlorotrifluoroacetone, reliable gloves and fume hoods make a big difference.
Environmental Impact: Beyond the Lab
Trichlorotrifluoroacetone carries both chlorine and fluorine, which can spell trouble for the environment. Chlorinated organic compounds often hang around in soil and water longer than most people expect. They resist natural breakdown, and that lets them spread far from the original source. Once in the water, small aquatic life can pick up the contamination, and it works its way up the food web. This is not a chemical that disappears overnight.
Experience from industrial spill cleanups shows that even small leaks from storage tanks need quick action. In regions with heavy chemical production, groundwater boards regularly hunt for halogenated solvent traces. Even low concentrations can take years to clear out. Scientists tracking ozone depletion look for substances similar to trichlorotrifluoroacetone, since halogenated chemicals historically damaged the ozone layer—especially those with chlorine and fluorine.
Solutions: Practical Steps Forward
Anybody handling this substance can cut down risks by sticking to clear rules: use closed systems, manage spills right away, and dispose of waste in certified treatment plants. Workers should know and trust their personal protective gear. Management needs to keep material safety data sheets updated and easy to access, not locked away in clunky binders. In my experience, nothing beats regular safety drills and a culture where people actually speak up if they notice a leak or spill.
Regulators keep a close eye on chemicals in this family. They require reporting of usage and disposal and push for alternatives if health or environmental risks overtake the benefits. No single chemical wins a permanent pass. As new information comes to light, rules and work practices must shift. That keeps both people and ecosystems safer, day by day.
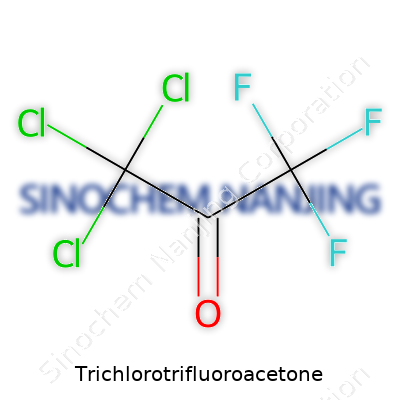
| Names | |
| Preferred IUPAC name | 1,1,1-Trichloro-3,3,3-trifluoropropan-2-one |
| Pronunciation | /ˌtrɪk.lɔː.roʊˌtraɪ.flʊə.rəˈæs.ɪˌtoʊn/ |
| Identifiers | |
| CAS Number | 421-50-1 |
| Beilstein Reference | Beilstein Reference 1701143 |
| ChEBI | CHEBI:51519 |
| ChEMBL | CHEMBL3917878 |
| ChemSpider | 26769 |
| DrugBank | DB13755 |
| ECHA InfoCard | 100.011.142 |
| EC Number | 206-999-8 |
| Gmelin Reference | 82255 |
| KEGG | C19205 |
| MeSH | D014248 |
| PubChem CID | 12521 |
| RTECS number | YO8575000 |
| UNII | 4A3S8L3DZT |
| UN number | UN3272 |
| Properties | |
| Chemical formula | C3Cl3F3O |
| Molar mass | 252.37 g/mol |
| Appearance | White crystalline powder |
| Odor | Sharp |
| Density | 1.616 g/cm3 |
| Solubility in water | Insoluble |
| log P | 0.9 |
| Vapor pressure | 14.2 mmHg (25 °C) |
| Acidity (pKa) | 5.18 |
| Basicity (pKb) | 15.64 |
| Magnetic susceptibility (χ) | -44.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.395 |
| Viscosity | 0.89 cP (25°C) |
| Dipole moment | 2.54 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 395.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −1000 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2072.7 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Warning |
| Hazard statements | H301+H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P210, P261, P280, P304+P340, P305+P351+P338, P312 |
| Flash point | 44 °C (111 °F; 317 K) |
| Autoignition temperature | 268 °C (514 °F; 541 K) |
| Lethal dose or concentration | Lethal dose or concentration (LD50/LC50): "LD50 (oral, rat): 470 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2000 mg/kg (oral, rat) |
| NIOSH | GZ3850000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.24 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Hexafluoroacetylacetone Trifluoroacetyl chloride Trichloroacetic acid |

