Monomethyltrichlorosilane: An Editorial View on an Overlooked Industrial Linchpin
Historical Development
Long before we had touchscreens and smartphones in every pocket, chemists started poking around with silicon compounds. Monomethyltrichlorosilane, with the formula CH3SiCl3, emerged from the early push to unlock organosilicon chemistry in the 1940s. This era saw big changes in the way chemistry shaped our surroundings. It wasn’t about chasing trendy applications; it was about building a foundation. Over the decades, researchers shed their lab coats for safety glasses and eventually handed off processes to engineers. It took industry a while to stop treating this compound as a mere intermediate, and to think harder about its full range of possibilities. With each technical leap from German, American, and Japanese laboratories, the applications multiplied.
Product Overview
Monomethyltrichlorosilane comes as a clear, colorless liquid with a strong, acrid odor that lets you know right away it means business. I’ve handled plenty of bottles that had sweaty labels—some folks keep it too close to water—so it’s not just a character you meet in textbooks. Used mainly in silicone production, it bridges simple silicon chemistry and some truly complex applications. This compound carves its mark in coatings, adhesives, sealants, and chemical vapor deposition. The real magic lies in the methyl group and trichlorosilane backbone, letting it graft onto molecules or surfaces in unexpected ways. Plenty of chemists reach for it as a precursor, but more imaginative folks see it as a material waiting for a story.
Physical & Chemical Properties
Handling monomethyltrichlorosilane in the lab feels like dealing with a clever but temperamental colleague. It boils at about 66°C, has a density a little heavier than water, and doesn’t waste time hydrolyzing in the presence of moisture. Expose it to air, and you get a loud hiss of hydrochloric acid gas—no substitute for proper ventilation and gloves. The compound loves to react with water, throwing off heat and making a mess if you don’t control the pace. For physical chemists, the structure blends organic and inorganic, combining silicon’s stable core with a reactive trio of chlorines and a single methyl group. This split personality lets it act as both crosslinker and functionalizing agent.
Technical Specifications & Labeling
Bottles and drums of monomethyltrichlorosilane usually come labeled as CH3SiCl3, with tightly regulated purity levels because trace water and alcohol contamination spoil reactions fast. Before any shipment, suppliers check for clarity, proper chemical composition, and the absence of unwanted isomers. Lower-grade product can mean headaches for everyone down the line, and seasoned buyers insist on specifications matching their exacting needs—from reagent quality labs to massive polymerization reactors. Regulatory rules in places like the EU and the US aim to keep handlers safe, listing it as a dangerous good for both domestic and international transport.
Preparation Method
Old-school chemists took silicon tetrachloride and methylated it using aluminum methyls. These days, industrial producers turn to direct processes, feeding methyl chloride and silicon-based precursors over copper catalysts at elevated temperatures. The process might sound simple on paper, but feed purity, reaction temperature, and catalyst tuning have a giant impact on yield and byproduct profile. I learned the hard way that a rushed synthesis brings in tetramethylsilane and other contaminants nobody welcomes in downstream chemistry. At scale, every percent of conversion and selectivity matters, and it’s never just a question of pushing more raw materials through the reactor.
Chemical Reactions & Modifications
If you want reactive silicon on a budget, you don’t need to look any further. Monomethyltrichlorosilane opens up a cloud of possibilities. It reacts sharply with alcohols, amines, even weak nucleophiles. Controlled hydrolysis yields siloxane chains, which form the backbone of silicone materials. Under dry conditions, the chlorines exchange for alkoxy groups, letting you build networks and bridges between molecules or surfaces. In surface modification, I’ve seen it used for making glass water-repellent and introducing organic functionality onto metal oxides. Look at academic journals, and you’ll find every sort of trick using this compound, from cross-linking agents in advanced polymers to building blocks for hybrid organic–inorganic nanomaterials. The range of possibilities keeps growing as techniques improve.
Synonyms & Product Names
This compound turns up under various names, sometimes adding confusion for newcomers. Some call it methyltrichlorosilane, others use monomethyltrichlorosilane or even abbreviate it as MTS. In product catalogs and research papers, you might spot terms like trichloromethylsilane or compare it to its close cousin trimethylchlorosilane. Each name points to slight variations or commercial branding, but most professionals know to check the chemical formula to avoid costly mistakes.
Safety & Operational Standards
Anyone who treats monomethyltrichlorosilane casually is asking for an accident. The vapor stings the eyes and lungs, and skin contact goes from irritating to dangerous. Hydrolysis releases hydrogen chloride gas and a sticky resin, leading to ruined equipment or worse, a trip to the hospital. Labs and factories working with this compound use closed systems and high-quality personal protection, including chemical splash goggles and gloves resistant to chlorinated compounds. Emergency response plans need to stay up-to-date. Once, I watched someone clean a spill without a respirator—the aftermath reminded everyone why safety protocols have teeth. Waste streams demand careful neutralization, and storage near water or heat sources only multiplies the risks.
Application Area
Despite its age, monomethyltrichlorosilane still attracts fresh uses. The core application sits in silicone polymer production, acting as a cross-linker and functional group source. In coatings, it modifies glass and metals, turning plain surfaces into water-beading wonders. Electronics fabricators use it for chemical vapor deposition, crafting dielectric layers and protective coatings. The semiconductor industry leverages its reactivity to build thin films with specific characteristics. More recently, this stuff turns up in nanotechnology, where surface chemistry controls everything from biosensors to energy conversion. End users rarely see it labeled on a product, but its fingerprints show up all over modern materials.
Research & Development
The research world never stops tinkering. Scientists worldwide focus on greener production routes, hoping to replace high-energy processes with milder methods that cut down on chlorinated waste. Academic teams dive into functionalization techniques, exploring how monomethyltrichlorosilane can graft unusual groups onto polymer backbones, build complex assemblies, or guide nanoparticle synthesis. Advances in computational chemistry bring better predictions about reactivity and help flag unwanted side reactions. I’ve seen industry-academic partnerships chase more sustainable pathways, like finding new catalysts or ways to reuse byproducts. The field stays dynamic, reflecting shifting priorities around the world—less chlorinated waste, more complex architectures, and tighter control over purity and polymer properties.
Toxicity Research
The hazards of monomethyltrichlorosilane are real. Toxicity studies show high inhalation risks, and acute exposure threatens human health fast. Animal studies link repeated exposure to organ damage and respiratory issues. Most of the risk lies in hydrolysis products, especially hydrogen chloride, which causes burns on contact. Regulatory agencies insist on transparency in labeling and reporting to help prevent mishaps. The push for greener chemistry has sparked efforts to limit emissions from production and end use. Despite advances in safety protocols, accidental releases still turn up in industrial reports, and research on long-term, low-level impacts continues. For workers and communities near manufacturing plants, these studies translate into real-world changes—better ventilation, scrubbers, and improved medical screening.
Future Prospects
The story of monomethyltrichlorosilane is far from over. The quest for sustainable materials keeps researchers hunting for safer production routes, with lower energy demands and less hazardous byproduct streams. Market demand for high-performance silicones and nanomaterials feeds investment in new downstream chemistry. Several companies and academic labs are tweaking catalysts and modifying equipment to boost yields and purity without ratcheting up environmental burdens. Environmental regulations keep tightening, forcing every link in the supply chain to rethink water use, emissions, and waste. If the industry succeeds, we’ll see products made with fewer resources and less impact. Meanwhile, emerging fields like flexible electronics and next-generation coatings will pull this molecule into unexpected new corners. Monomethyltrichlorosilane’s reactivity and adaptability still make it a valuable tool, but anyone working with it will need to balance potential with responsibility, and strategy with safety. For chemists, manufacturers, and users alike, engaging with this compound means facing both opportunity and accountability, and that challenge isn’t going away soon.
What is Monomethyltrichlorosilane used for?
What Monomethyltrichlorosilane Brings to the Table
Walk through a production lab stocked with glassware, or step into a silicon wafer facility, and you might pick up a whiff of something sharp in the air. That could be monomethyltrichlorosilane. Sporting a chemical structure that builds off of silicon and chlorine, it’s no household name, but this chemical shapes products people use every day.
One of its strongest calling cards shows up in the world of coatings. Manufacturers looking to protect surfaces against water or extend longevity turn to this compound for its ability to form tough, water-repellent barriers. These barriers matter for electronics, medical devices, and construction materials. Without them, corrosion eats away at expensive gear, or water weakens building components a lot faster. Hydrophobic coatings help gadgets survive accidental spills, keep solar panels cleaner for longer, and make glass easier to wash. Monomethyltrichlorosilane makes these benefits possible because it bonds tightly to glass, ceramics, and metal.
Making Silicone Polymers
Most people interact with silicone way more often than they realize—think bakeware, medical tubing, electronics seals. The backbone of these materials relies on key building blocks like monomethyltrichlorosilane. Creating versatile silicone polymers starts with reactions involving this compound. Silicon atoms join with oxygen, then get customized by groups like methyl and chlorine. That’s chemistry at its most practical. A lot rides on the quality and purity, so any batch misstep risks expensive downtime for a processing plant.
From Electronics to Everyday Goods
Chips need to stay clean, dry, and insulated to work right. During the manufacturing process, engineers deploy monomethyltrichlorosilane to coat silicon wafers. It serves as a precursor in making siloxane films, which help control the surface properties of semiconductors. Building up layers atom by atom in a controlled way, this approach impacts chip speed, yield, and ultimately, that smartphone or computer on someone’s desk.
Safety, Environmental, and What Must Change
Anyone who’s worked around monomethyltrichlorosilane knows the risks firsthand. The stuff reacts sharply with moisture—including what’s in the air—to form hydrochloric acid. Protective gear and proper ventilation are non-negotiable if you want to avoid mishaps and injuries. Trained chemists and operators stress the handling hazards for good reason. When a leak happens, it’s not just about a chemistry lesson; it's a visit from hazardous materials crews and a long afternoon cleaning up.
Bigger picture: the drive toward greener chemistry pushes industries to reconsider old habits. Some companies invest in closed-loop systems and advanced scrubbers to capture emissions before they hit the air. These new protocols add cost, but they cut impact on the environment and the health of plant workers. Groups like the Occupational Safety and Health Administration (OSHA) and European Chemicals Agency provide detailed safety guidelines; those rules save lives and keep livelihoods intact.
Building a Safer and Smarter Future
Research teams and industrial chemists keep searching for alternatives that do the same job with less risk. As the demand for high-performance coatings and electronics grows, alternatives with less environmental baggage will gain traction. In the meantime, every responsible lab and factory keeps a close eye on their processes, making sure the tools that make modern life possible don’t come at too high a cost for people or the planet.
What are the safety precautions when handling Monomethyltrichlorosilane?
Why Safety Matters With Monomethyltrichlorosilane
Plenty of folks don’t hear about monomethyltrichlorosilane unless they work in a lab or chemical plant. It shows up in specialty coatings and advanced electronics. Small vials or barrels of this clear liquid carry some big risks. My first time in a lab that used the stuff, the supervisor told me upfront: “One mistake with this, and you’ll never forget it.” That stuck with me. The chemical stings, eats through skin, and spits out fumes that can stop your breathing. No fancy degrees are needed to know you’ve got one tough customer on your hands.
The Real Hazards
Monomethyltrichlorosilane reacts fast with water, even with moisture in the air. That means it turns into hydrochloric acid gas right there in the room—bad news for your lungs. Skin contact doesn't just irritate; it burns. Eyes turn red and painful, and long exposure can set off asthma or leave lasting scars. I’ve seen a coworker lose his sense of smell for days. To top it off, the vapors can trigger alarms and draw emergency response, interrupting the whole workday. Nobody wants to test the ER staff’s skill with chemical burns.
Basic Steps to Handle It Right
One of the best ways to keep safe is to work in a proper fume hood. Hoods with strong airflow grab those harsh vapors and dump them far from your breathing zone. I always double-check the airflow before opening any container. Gloves make sense—nitrile or neoprene, not basic latex. If any patch of skin feels cool or tingly after a splash, wash right away, don’t wait “just a second.” Splash goggles aren’t optional; regular glasses miss too much.
Before the first use, have a real spill kit on hand. Neutralizers only help if you can reach them, so stash a kit at arm’s length. Facility training pushed this lesson: store monomethyltrichlorosilane tight and sealed, with drying agents, away from any accidental water source. Never dump leftover liquid down the drain or toss it as regular trash. Follow the hazardous waste rules—almost every place has them. Keeping chemical labels crisp and easy to read can save lives. I've learned to read those labels before reaching for anything.
What Can Go Wrong and How to Stop It
Sometimes, using old gloves or forgetting to check the hood leads to trouble. In one case, a rookie stored a bottle near a sink; humid air built up enough pressure to blow the cap straight off. After that day, our team made it a rule: every bottle gets checked at the start of each shift, and none goes closer than five feet to water sources. Running regular safety drills works better than any lecture. The best solution comes from making safe choices automatic—grab fresh gloves, test the hood, double-seal containers, and make sure spill response happens in seconds, not minutes.
Protecting Health for the Long Haul
People who take shortcuts around monomethyltrichlorosilane get hurt. Respirators only work if you fit them right and change cartridges often. Telling a supervisor about leaks or weak airflow helps everyone. Some bosses don’t like hearing about problems, but I’ve watched a strong safety culture keep good people healthy and happy. Reading material safety data sheets, before and after handling the chemical, saved me from bad mistakes. Safety isn’t a chore—it's the difference between injury and going home with a clear head and steady hands.
Staying alert, preparing for trouble, and taking the small steps—these choices turn a risky job into a safe routine.What are the storage requirements for Monomethyltrichlorosilane?
Understanding the Hazards
Monomethyltrichlorosilane sounds technical, but what matters most to anyone handling this chemical is safety. This compound reacts fiercely with water and air moisture, giving off dangerous hydrochloric acid fumes. Imagine spilling something and having the room fill up with acidic vapor. If something goes wrong, workers and the nearby environment pay the price. Even small doses of careless storage can trigger corrosion or respiratory risks. That’s why preparation is more than a checklist: it’s about keeping people and property out of the danger zone.
Safe Storage Conditions
Storing this chemical isn’t about tossing containers on a shelf and hoping for the best. It asks for a dry, tightly sealed space, away from even tiny leaks of humidity. Rusty pipes, leaking ceilings, or condensation on the walls can lead to disaster here. In the past, I’ve seen facilities try to cut corners by using ordinary steel drums. Not a good move. Monomethyltrichlorosilane eats through metals that don’t resist corrosion, and the right answer is always lined or special-grade containers designed to hold up against the bite of aggressive chemicals.
Temperature swings matter, too. Warm rooms can build up pressure in sealed drums—and nobody wants a chemical to start hissing out. Best storage practice means keeping the area cool, dry, and monitored for leaks. Even lights and electrical circuits in the storage room should be rated for use around volatile chemicals, to avoid sparking a bigger crisis.
Real-World Precautions
From experience, good signage always helps. Labels showing hazard class and emergency action steps don’t just tick the box for compliance—they remind workers there’s no wiggle room for mistakes. Anyone accessing the storage needs training in what PPE to grab and which ventilation switches to flip before opening anything.
Ventilation isn’t just a technical spec. Without fresh airflow, vapor hangs around and finds its way into lungs. If your chemical room smells even a little bit sharp or acidic, that’s a warning to upgrade ventilation or patch up your storage seal. Leak detection equipment helps a lot, and alarms connected to high-traffic areas can prevent a slow fume leak from turning into an accident.
Steps Toward Safer Handling
Fires with Monomethyltrichlorosilane aren’t just rare possibilities. This chemical reacts with water-based fire sprays, making traditional firefighting impossible. That’s why a CO2 or dry chemical extinguisher stands closest to locked cabinets, and responders need training before stepping into the room in the first place. Keeping phone numbers for chemical response teams posted on the wall is no small thing.
Good policy means checking inventory often for leaks, bulging drums, or rusty seams. Smaller transfers done inside ventilated hoods, with everyone wearing goggles and heavy gloves, lower the risk even more. Spill kits with absorbent materials, neutralizers, and clear instructions make cleanup fast—speed matters with hazardous vapors in the air.
Pushing Storage Standards Further
The world keeps learning from past incidents. Facilities handling Monomethyltrichlorosilane borrow safeguards from best-in-class chemical storage, auditing suppliers and storage routines before a single drum leaves the shipping dock. Digital monitoring tracks temperatures and humidity round the clock now, a step up from just peeking through the window. Training gets updated with real accident reports, so new staff hear more than dry lectures—they hear stories and scenarios that stick with them past orientation day.
Between regulatory agencies, serious suppliers, and front-line workers, chemical safety becomes shared ground. Storage rules stay strict for a reason: there’s no substitute for a healthy workforce and a safe community next door.
What is the chemical formula of Monomethyltrichlorosilane?
Monomethyltrichlorosilane: Knowing More Than Just the Letters
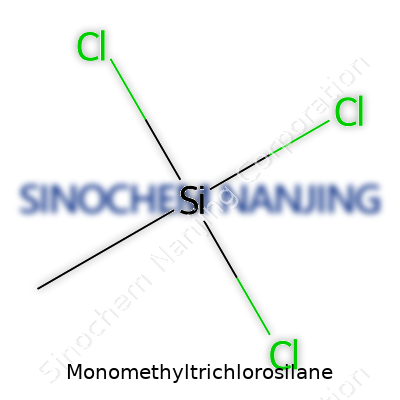
Working in a lab sparked my respect for chemical names that sound more like tongue twisters. Monomethyltrichlorosilane is one of those. It reads like a dare for a spelling bee contestant, but it tells a simple story once you break it down. The chemical formula for Monomethyltrichlorosilane is CH3SiCl3. Behind those few letters and numbers stands a compound with both promise and responsibility.
Demystifying the Formula
This compound comes from methyl group chemistry. The “mono” points to a single methyl group (CH3) linked to silicon (Si). Silicon then gets hitched to three chlorine atoms (Cl), leading to SiCl3. Together they give us CH3SiCl3. Every time I see chemicals built like this, I think of the periodic table’s quiet logic. If methylchlorosilanes were music, this one plays somewhere near the top of the scale—simple, sharp, and ready for transformation.
Why Monomethyltrichlorosilane Matters
I remember the first time I handled a bottle of Monomethyltrichlorosilane in a process safety class, the MSDS sheet taped right to the glass. The fumes reminded everyone that chemistry isn’t always friendly. Still, industry chases this stuff. Manufacturers use it for making water-repellent coatings, adhesives, and even for preparing surfaces inside electronics. The tech world leans on these modified silanes to protect circuit boards and manage the delicate dance of insulation versus conductivity.
Go past the surface, and you see risks. Monomethyltrichlorosilane reacts fiercely with water, giving off hydrogen chloride. I’ve seen old glassware etched from a stray drop left in a sink. The formula serves as a warning and a promise. Handle wrong, and it punishes carelessness. Managed well, it brings benefits across electronics and protective coatings. Our daily lives start to depend on obscure compounds like this—coating glass that never fogs, making smartphone screens tougher, or prepping composite materials for aerospace projects.
Back Up Claims with Facts
Data from the EPA and the National Library of Medicine show Monomethyltrichlorosilane gets regulated tightly. This policy doesn’t come out of paranoia: one sniff of its vapors can try to burn your lungs. Historical incidents of overexposure underline why safety rails matter in both legal regulations and shop-floor routines. Tech industries spent years refining cleanroom protocols partly because of hazardous chemistry involving silanes like this.
Global production now runs in the thousands of tons per year, supporting everything from waterproof electronics to composite materials meant for wind turbines. Scientists, including colleagues from my graduate work, spend months tweaking industrial recipes to cut emissions and limit exposure. Safety training holds a real place, not just lip service.
Ideas for Better Practice
There are paths toward safer and cleaner usage. Closed-loop systems cut down on leaks. Real-time air sensors, now cheaper, can catch vapor spikes before workers catch a whiff. Ongoing research aims to swap toxic silanes for alternatives with a friendlier safety profile or add scavenger chemicals that mop up any hydrogen chloride formed in accidents. Practical steps in the workspace—simple fume hoods, gloves that truly hold up, and digital recordkeeping for spills—give everyone a fighting chance to manage tricky chemicals.
Every formula in chemistry tells a story. CH3SiCl3 stands as more than random pieces stitched together—it’s a reminder that new technologies need real respect for science, not just a search for the next big product.
How should Monomethyltrichlorosilane be disposed of safely?
Understanding the Dangers
A bottle of Monomethyltrichlorosilane won’t look menacing on a shelf. Clear liquid, sharp, almost sweet odor – it could be mistaken for dozens of other chemicals from a distance. But as anyone who’s worked in a lab or on a manufacturing line learns, this stuff means business. Contact with water unleashes fumes of hydrochloric acid. Eyes start to burn, lungs catch fire with just a whiff, and skin develops an instant, angry rash. Spills and careless disposal send the risks up several notches—not just for workers, but also for folks living downstream, for the water supply, for the air outside.
The Chain Reaction of Bad Disposal
Tipping a beaker into a drain or tossing a container in the regular trash isn’t just lazy—it starts a chain that can end with toxic gas spreading through a facility or leaching into the ground. Sewer lines weren't built for chemicals like this. If water touches Monomethyltrichlorosilane, that hydrochloric acid doesn’t stop at the lab door. Public wastewater plants can’t neutralize or filter it without big risk to workers and equipment. The same goes for landfills: these sites mix all kinds of waste, and even a small leak can spell trouble for years in the water table. Real-world accidents have led to big evacuations, fines, and tragic injuries.
PPE and Containment — Basics with No Negotiation
Personal experience has taught me that cutting corners, even just once, brings regret. In glove, apron, and goggles, you realize quickly how little stands between safety and catastrophe. Every container must remain tightly sealed, far from moisture or heat. Materials labeled as “incompatible” like strong bases, acids, or water sources—never store them nearby. Double-checking labels and storage logs could save hours, dollars, and medical bills down the road.
Regulatory Guidance and Professional Help
EPA and OSHA leave little room for guesswork here. These agencies require labeling, training, and clear protocols because Monomethyltrichlorosilane isn’t forgiving. Many states add their own extra layers of scrutiny—California’s strict with air and water monitoring. It helps to review local laws, not just federal rules. Consulting with a certified hazardous waste manager never feels like overkill. These folks have experience with unexpected leaks or container failures, and know which procedures keep people out of danger.
Transport and Neutralization — No Shortcuts
Disposal companies use dedicated barrels, not just off-the-shelf drums. Every step gets tracked: labeling, secure loading, paperwork, and a final destination that treats rather than just “stores” these byproducts. Controlled hydrolysis in specialized treatment plants neutralizes the chemical, converting dangerous acid into something safer under strict ventilation and with constant monitoring. Workers keep neutralizing agents on hand in case anything splashes or goes wrong.
Education, Routine, and Accountability
Complacency shows up in the news all the time. Old bottles found in forgotten corners, labels faded, and no one sure who bought them. Every lab or factory storing Monomethyltrichlorosilane ought to run regular trainings, update inventory logs, and set a disposal schedule. Peer checks and digital tracking help. Managers who walk the floor and ask direct questions find gaps before accidents can appear.
Building Real Safety Culture
Disposing of Monomethyltrichlorosilane safely takes more than paperwork and good intentions. It calls for a culture that treats routine as sacred and values the smallest details over speed or convenience. That culture keeps people healthy, protects the environment, and makes sure “cleanup” never becomes the headline.
| Names | |
| Preferred IUPAC name | Trichloro(methyl)silane |
| Other names |
Methyltrichlorosilane Trichloromethylsilane Trimethyltrichlorosilane |
| Pronunciation | /ˌmɒnoʊˈmɛθɪlˌtraɪˌklɔːroʊˈsaɪleɪn/ |
| Identifiers | |
| CAS Number | 7550-45-0 |
| Beilstein Reference | 1750804 |
| ChEBI | CHEBI:85157 |
| ChEMBL | CHEMBL154122 |
| ChemSpider | 86768 |
| DrugBank | DB14106 |
| ECHA InfoCard | The ECHA InfoCard of Monomethyltrichlorosilane is "100.004.309". |
| EC Number | 200-895-8 |
| Gmelin Reference | 9037 |
| KEGG | C19438 |
| MeSH | D008899 |
| PubChem CID | 66205 |
| RTECS number | VV9275000 |
| UNII | 6UY020341D |
| UN number | UN1243 |
| CompTox Dashboard (EPA) | DTXSID5020707 |
| Properties | |
| Chemical formula | CH3SiCl3 |
| Molar mass | 180.49 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Pungent |
| Density | 1.17 g/cm³ |
| Solubility in water | Reacts violently |
| log P | 1.9 |
| Vapor pressure | 75 mmHg (20 °C) |
| Acidity (pKa) | 1.7 |
| Basicity (pKb) | 12.0 (25°C) |
| Magnetic susceptibility (χ) | -62.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.403 |
| Viscosity | 0.46 mPa·s (25 °C) |
| Dipole moment | 1.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 289.2 J⁄(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -542.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1058.8 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | **GHS02, GHS05, GHS06** |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H335, H410 |
| Precautionary statements | P210, P220, P261, P280, P305+P351+P338, P310, P303+P361+P353, P304+P340, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Flash point | 2 °C (35.6 °F) - closed cup |
| Explosive limits | 4.7% - 32.2% |
| Lethal dose or concentration | LD50 oral rat 1800 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1600 mg/kg |
| NIOSH | YT2450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Monomethyltrichlorosilane: "1 ppm (5 mg/m³) (OSHA) |
| REL (Recommended) | 0.5 ppm (2 mg/m³) |
| IDLH (Immediate danger) | IDLH: 20 ppm |
| Related compounds | |
| Related compounds |
Trimethylsilyl chloride Chlorodimethylsilane Dimethyldichlorosilane Methyltrichlorosilane |

