Trichloronitromethane: A Chemical Through the Lens of Progress, Risk, and Responsibility
Historical Development
Trichloronitromethane began its journey in the late 19th and early 20th centuries, at a time when chemical synthesis marked the birth of countless new compounds, many beyond the wildest dreams of early chemists. The context of its discovery tells a story about industrial ambition and the relentless chase for innovation. Chemists sought these organochlorine molecules to advance everything from dye manufacture to new explosives. Back then, safety took a backseat. The priorities tipped toward yield and novelty, with less regard for the possible consequences or downstream effects on health and nature. The continued research on trichloronitromethane soon revealed properties worth both celebration and concern, setting a stage for decades of debate over how to harness, control, and safeguard its use.
Product Overview
Today, trichloronitromethane gets a range of labels—chloropicrin being one you’ll likely find in literature or commercial catalogues. Its main uses extend across fumigation, chemical synthesis, and certain laboratory-based research. Its volatility and strong odor alone trigger instant recognition among chemists and field workers. What separates it from other common chemicals is not just the punch of its reactivity, but the ping-pong of benefits and hazards that crop up wherever it goes. Nobody, from agricultural workers to environmental scientists, can simply dismiss its power, and anybody who handles it knows they’re dealing with something that can both drive progress and demand caution in equal measure.
Physical & Chemical Properties
A clear, heavy liquid with a pungent scent, trichloronitromethane looks harmless enough but delivers a potent punch as both an irritant and a fumigant. Its molecular structure—one carbon holding tightly to three chlorines and a nitro group—gives it a unique fingerprint, both in spectral analysis and in its environmental interactions. This chemical boils quickly for its density, making accidental vapors a problem anytime temperatures rise or containment fails. Its behavior in water, soil, and air never stops at simple presence; its reactions and transformations rip through biological and chemical systems, creating byproducts that demand a closer look. Physical properties alone only tell part of the tale; the presence of those chlorines bonded to a nitromethane core makes for reactions that can be both explosively energetic and environmentally persistent.
Technical Specifications & Labeling
Labeling trichloronitromethane comes with warnings that go way beyond those found on household cleaners. Its toxicity, volatility, and environmental persistence require hazard pictograms, robust documentation, and strict documentation throughout professional handling. I remember my first experience unboxing a sealed bottle of this stuff; you don’t forget the glove protocol and the triple checks for vapor-tight seals. Clarity in technical details—from concentration to purity—matters a great deal, not just for compliance but because a misstep in diluting or transferring can end a workday in the hospital. Handling standards exist for good reason: every chemical cabinet I’ve ever used stores this substance in the coldest, most isolated shelf for a reason. Explaining this to new researchers is never an optional step; it’s a necessity.
Preparation Method
Historically, trichloronitromethane emerged from the controlled chlorination of nitromethane, a process that highlights the mass-market methodology of pouring raw chemical feedstock into tightly monitored reactors. In the lab, the reaction feels both simple and perilous: you introduce chlorine gas to nitromethane under precisely managed conditions—temperature, light, and often catalysts—all while ready for sudden, exothermic jolts. I remember double-checking every clamp and vent the first time I saw this prepared in a university lab; years of stories about accidental mishaps replays every time chlorine is involved. Even in modern facilities, automation and remote handling do not erase the need for close supervision. Byproducts—sometimes as toxic as the main product—show why waste management plans must never get overlooked. The urge to streamline this reaction for cost or speed always battles against the practical need to ensure safety at every stage.
Chemical Reactions & Modifications
Trichloronitromethane acts as both a tool and a challenge in synthetic chemistry. Its reactivity provides a jumping-off point for complex chemical elaborations, acting as a precursor for certain pharmaceuticals, dyes, and specialty polymers. The nitro and trichloro groups enable unusual transformations under the right conditions, making it a clever ingredient in advanced syntheses. Yet, too often, such promise gets weighed down by the byproducts—sometimes carcinogenic or persistent in the environment. Scientists have spent decades investigating ways to make its downstream chemistry safer and less wasteful. The relentless push in green chemistry circles aims to tame these reactions, on the hunt for conditions that suppress hazardous offshoots and reduce chlorinated waste streams. Despite these efforts, trichloronitromethane does not make compromises lightly. It brings value, but only if the laboratory or factory accepts the complicated choreography required to keep harm in check.
Synonyms & Product Names
Names in the chemical world often reflect both tradition and market. Trichloronitromethane answers to chloropicrin in agriculture and environmental remediation circles. Other historical names—Nitrochloroform, for instance—reveal a time when early chemists named compounds for their structure or reactivity, not consumer appeal. Each name tells a fragment of the story: the compound’s shifting roles in science, commerce, and, regrettably, conflict. During World War I, the chemical under murky synonyms became an ingredient for chemical weaponry, lending a dark edge to its nomenclature. Names stick for good reason; each reflects a stage of technological optimism or historical reminder of the chemical’s double-edged nature.
Safety & Operational Standards
Of all the chemicals I’ve worked with, trichloronitromethane commands the strictest regime of handling, storage, and emergency preparedness. Direct inhalation triggers coughing, tearing, and, in high doses, pulmonary edema. The memory of skin exposure is hard to forget—irritation follows fast, with gloves quickly swapped out for thicker, acid-resistant varieties. Industrial guidelines spell out full-face protection, air extraction systems, and robust spill procedures. Regulation in North America and Europe zeroes in on workplace exposure—often demanding monitoring and logs for even brief work sessions. Many legacy incidents in broken-down labs or agricultural mishaps haunt the regulatory discussion; nobody wants a repeat of those costly or deadly mistakes. Safety training, for me, became less about checking boxes and more about respecting the reality of risk. Supervising younger technicians proves the stakes are real: not just compliance, but genuine protection.
Application Area
Trichloronitromethane plays a complicated role in pest management, particularly as a fumigant for soil and stored grain. Farmers and pest control specialists appreciate its high efficacy against a vast array of insects, fungi, and nematodes. Yet, the push for “clean” or organic solutions constantly debates the chemical’s future. Concerns over residue, off-gassing, and potential ground water leaching animate local and international restrictions. In remediation work, it knocks out stubborn soil-borne hazards where biodegradation often fails. Researchers have also poked at its use in chemical synthesis for specialty organics, but regulatory heat drives many industries toward alternatives. Through my own experience supporting agricultural technology startups, the knife-edge between potent efficacy and environmental trade-off always sparks heated meetings—balancing crop protection with ground truth on health and regulation.
Research & Development
For many years, trichloronitromethane drove laboratory innovation. Its use in method development, analytical calibration, and as a reagent in synthetic routes fueled graduate research and funded university projects. Over time, the chemical’s growing regulatory burden prompted a shift more toward novel decontamination pathways, alternative crop protection technologies, and surface chemistry applications. The appetite for innovation persists, but newer inquiries focus on safer analogues, breakdown methods, and encapsulation strategies to limit exposure. At the heart of this research stands a conviction: any practical innovation with such compounds needs to prove itself both on a technical level and in meaningful risk reduction. Finding that intersection defines the next chapter of chemical progress.
Toxicity Research
Every encounter with trichloronitromethane comes with a backdrop of toxicology: respiratory distress, acute irritation, and possible chronic health effects mark decades of animal and, regrettably, human data. Inhalation studies in rodents confirm what farming communities long suspected—lung injury and systemic effects can follow exposures above recommended thresholds. Regulators and health agencies pulled together incident databases, case reports, and epidemiological reviews, building a mountain of evidence that steers the tight limitations around its commercial and research use. Community advocacy and environmental monitoring shaped these policies. At the same time, scientists continue parsing the breakdown products and bioaccumulation risks, pressing for better detection, repair, and antidotes. Health workers and first responders keep current on new research not out of curiosity, but as a matter of personal and public safety.
Future Prospects
Looking ahead, the story of trichloronitromethane represents both a challenge and an opportunity. Industry finds itself under pressure to provide cleaner, safer pest control while maintaining yield and cost guarantees for a hungry world. Regulatory agencies eye phased bans in key markets, shifting the commercial frame toward alternatives and even biotechnological pest management. Chemists still focus on remediation techniques and greener substitutes with the same efficacy but a fraction of the risk profile. My experience consulting for tech developers shows a familiar pattern: the strongest innovations happen when teams accept that legacy chemicals cannot be “de-risked” through paperwork alone. Ongoing dialogue among regulators, workers, manufacturers, and environmental scientists provides hope for new practices—a future where potent molecules are governed not just by capability, but by accountability.
What is Trichloronitromethane used for?
Where Trichloronitromethane Shows Up
Trichloronitromethane, also known as chloropicrin, shows up in places where most folks don’t expect it. Someone who has ever tended crops or worked with old wood can cross paths with it. In agriculture, it acts as a soil fumigant. Ask any farmer who's dealt with stubborn fungi or nematodes: crops grow stronger where troublemakers in the soil don’t get free rein. Chloropicrin takes down these unseen pests before a single seed sees daylight, protecting harvests and helping keep food on the table.
Wood preservation also leans on trichloronitromethane. Moisture, warmth, and neglect turn wood into a playground for rot, fungus, and insects. Using chemicals like this one lets railroad ties, utility poles, and even old log cabins stand firm much longer than they otherwise could. While it can’t work alone, in combination with other preservatives, it keeps costly repairs at bay.
Hard Lessons Learned
Not many people realize this compound once defended lives in a different way. Anyone who has read about the First World War might recognize it as a chemical weapon—used to drive soldiers from their trenches with its irritating properties. The world learned quickly about its dangers. Since then, rules have tightened everywhere. In the United States, for example, the EPA controls how and where it gets used. Those lessons stick: safe handling and proper training sit at the center of every modern use.
Health and Safety
Contact with trichloronitromethane brings strong reactions. Eyes tear up, skin stings, lungs burn. Early in my days working near treated fields, I saw workers drop everything after a small leak started. Respirators, gloves, and strict guidelines protect those on the frontline. Residents near treated soil often worry about drift and water contamination, and those concerns hold weight. Studies have tracked how it can move with wind or water, affecting nearby homes or wildlife. Responsible application—choosing the right day, checking the wind, sealing soil—matters for everyone living nearby.
Environmental and Practical Challenges
Farming communities, environmentalists, and scientists share strong opinions about soil fumigants like trichloronitromethane. Some argue we should stop using it, but the reality on farms makes things complicated. Removing it without solid backup plans leads to pest problems and crop losses. Nobody wants food prices soaring or pests moving unchecked. At the same time, nobody wants long-term health or environmental harm.
Safer alternatives and smarter use make a difference. More farms now rotate crops or use cover crops—old-fashioned moves that break the cycle of pests and diseases. Scientists keep chasing new pesticides with shorter persistence and lower toxicity. Regulations work best when authorities listen to both growers and neighbors and adjust rules as they learn.
Balancing the Risks and Benefits
What started as an aggressive defense tool now sits in a balancing act: enough benefit to keep food growing, not enough risk to endanger health or soil. My experience tells me most folks who use trichloronitromethane don’t take its risks lightly—they’ve seen what can go wrong. Farming, construction, and utility industries rely on it not from carelessness, but because sometimes nothing else works as well. Good science, smart rules, and honesty with the public keep its risks in check.
Is Trichloronitromethane hazardous to health?
Understanding Trichloronitromethane
People rarely talk about trichloronitromethane outside of labs and technical circles. Some call it chloropicrin or PS, but the facts around this chemical should matter to anyone working near agriculture, soil fumigation, or certain industrial plants. My own encounters with this substance came during a high school internship focused on agricultural chemistry; the memory of sharp, eye-stinging fumes still sticks around. The first lesson I learned: trichloronitromethane shouldn’t be handled lightly.
Health Hazards of Trichloronitromethane
The dangers aren’t stories you hear just to worry you. Trichloronitromethane acts as a strong irritant. When a small amount gets into the air, it can quickly irritate the eyes, throat, and lungs. Symptoms kick in almost instantly—tearing, coughing, chest tightness. Many know it as a chemical once used in World War I as a tear gas. If something finds a way into military arsenals, expecting risks isn’t paranoia; it’s common sense.
Long-term exposure can hit even harder. Chronic effects may show up as bronchitis or asthma-like symptoms, even with occasional exposure in agricultural spraying or improper handling. Anyone spending time around greenhouses and farms might have heard stories about people sent home with headaches, dizziness, or worse after exposure.
The International Agency for Research on Cancer classified trichloronitromethane as a Group 3 agent. Research still debates clear links to cancer, but this doesn’t let the chemical off the hook. Evidence already points to genetic and cellular damage in some lab experiments. My own look at toxicology journals revealed documented DNA disruption in lab animals, which should stay in the back of your mind. Even without a smoking gun for cancer, repeated exposure never leaves anyone better off.
Where You’ll Find Trichloronitromethane
Fumigants and pesticides mark the main use for this compound. Farmers use it to sterilize soil, prepping for the next crop. Some water treatment processes used to lead to small amounts of trichloronitromethane showing up as a by-product—luckily, tighter regulations have clamped down on that side of the problem.
Even so, accidents still happen. Poorly ventilated greenhouses, leaky containers, and improper application have all caused injuries. It doesn’t take a major spill to poison a workspace; sometimes a missed label or a cut corner is all it takes. Seeing a coworker rushed outside, coughing and red-eyed, made clear the importance of proper protective gear. Most of us who’ve handled hazardous chemicals remember not the routine, but the close calls.
Steps for Safer Handling and Better Outcomes
Strict safety standards offer solid protection against trichloronitromethane. Goggles, gloves, and sealed respirators do more than look official—they stop burns and breathing trouble. Anyone working with crop fumigants or chemical products should receive detailed training, not just a quick talk.
Every workplace should keep clear labels and up-to-date safety sheets. Spills or leaks call for instant cleanup, not a wait-and-see approach. Modern sensors and ventilation systems help a lot, but even the oldest warehouse benefits from a good open window and a crew that knows their stuff.
Alternatives exist for many uses. Some places now rely on less toxic soil treatments, thanks to changing regulations and a push for better health practices across the board. Strict enforcement forks the path between daily risk and safer routine. Over time, the right systems and habits keep acute mishaps out of the news and off medical records.
Someone dealing with trichloronitromethane owes it to themselves and coworkers to treat the chemical as more than an ordinary headache. Health, after all, rarely offers second chances once things go wrong.
What are the safety precautions for handling Trichloronitromethane?
Understanding the Risks
Trichloronitromethane, better known as chloropicrin, brings a nasty gut punch to anyone not respecting its bite. Most folks bump into chloropicrin because it's used in agriculture or pest control, and it once served as a chemical weapon. It makes eyes water, skin itch, and breathing tough. If you’re working with it, you really feel the need for serious precautions. I’ve seen colleagues lock up for hours after only a splash on the sleeve. Nothing about it invites carelessness. The stuff’s volatile, and its fumes fill a room faster than you’d expect, making simple mistakes a big deal.
PPE Isn’t Optional
Suiting up comes first. Good gloves—nitrile or other chemical-resistant material—take the sting out of accidents. Chloropicrin chews through thin latex, so sticking with the right gloves counts for a lot. Eye protection needs to seal up tight, not just cheap lab goggles but full-face gear that blocks vapors and splashes. Respirators, not dust masks, keep the lungs clear. Cartridge filters made for organic vapors or a positive pressure air supply work best. I always check the fit; if you can smell anything, you’re not sealed up right. Boots and aprons help ward off spills, as this chemical seeps through regular clothes in no time flat.
Work Space Speaks Volumes
With trichloronitromethane, location matters. Outdoor spaces give you an out if a spill happens, but that isn’t always possible. In labs or sheds, ventilation ramps up the safety. Fume hoods pull air away from your face, and air exchange lets you breathe easier. Keep work surfaces clean and uncluttered. It’s easy to avoid a spill when you don’t have to reach around old coffee cups or stray tools. My own habit is to keep only what I plan to use within arm’s length. Dump chemicals, soiled rags, or empty containers right after use—never leave them lying around because someone else could stumble into trouble.
Handling and Storage Matter More Than Folks Think
Before opening any container, check the label twice and surround yourself with what you need—a spill kit, emergency eyewash, plenty of soap and water. Store trichloronitromethane in tight, labeled bottles away from heat, sunlight, or food. This chemical breaks down with heat, and the gases build up fast. Never stack it with strong bases, reducing agents, or anything flammable. I learned early on that even a tiny leak turns into a call for the hazmat team if you’re sloppy about storage. Fire extinguishers, emergency showers, and exits should remain within easy reach, because seconds matter in a mix-up.
Emergency Know-How
Training doesn’t stop after a first lesson. Regular drills make sure that if something goes wrong, muscle memory takes over. If exposed, don’t tough it out. Rinse the eyes or skin for at least fifteen minutes and swap contaminated clothes right away. Seek medical help even if you feel better—chloropicrin can cause delayed effects. For spills, ventilate the area, evacuate if needed, and wear full gear before cleanup. Never try to save on protective gear or rush through decontamination.
Solutions and Good Habits
Most of the trouble with trichloronitromethane happens when workers skip steps or cut corners. Regular training, open conversations, and watching out for each other go further than rules printed on a wall. Trust in experience, but never in routine—complacency opens the door for accidents. Industries must invest in safety, upgrading equipment and personal protective gear, because medical bills and recovery time always cost more than prevention. Real safety grows from respect for the risks and a commitment to look out for everyone on the team.
How should Trichloronitromethane be stored?
Understanding Why Safe Storage Matters
Trichloronitromethane—some folks know it as chloropicrin—packs a punch when it comes to chemical reactivity. Anybody who’s worked in a lab or an agricultural setting knows this compound for its sharp smell and its use as a pesticide and soil fumigant. Personally, after handling compounds that react to temperature, sparks, or just plain air, I've learned respect comes first, not last.
Respecting What You're Dealing With
This stuff doesn’t just irritate the eyes or lungs; mishandling creates explosive results, sometimes literally. Stories float around scientific circles of accidents caused by ignoring simple rules. That's why folks who store trichloronitromethane keep it locked up under strict controls.
Let’s be real: keeping things safe isn’t about following boring textbook rules—it’s about making sure no one gets hurt and property doesn’t go up in smoke. The Centers for Disease Control and nationally recognized safety bodies recommend cool, dry storage, far from sunlight, heat sources, or anything flammable. You don’t want a runaway reaction; you want peace of mind.
Choosing the Right Space and Container
For anyone storing trichloronitromethane, a well-ventilated chemical storeroom away from work areas beats leaving it in a closet or on a shelf near the office microwave. Metal cabinets with flame-proof finishes have become my go-to for chemicals like this. Anything glass or steel usually works for containers. Seals matter—leaky lids let vapors escape, and there’s nothing worse than realizing you’ve created a hazard just by screwing a lid on too loose.
Humidity, Heat, and Separation
Humidity wears down labels and causes corrosion inside metal containers. Too much heat ramps up trichloronitromethane’s volatility, turning a manageable chemical into a disaster looking for an excuse. Premium labs often check temperature and humidity daily. From experience, I believe simple dehumidifiers and fans make all the difference.
People often forget the threat of mixing. Trichloronitromethane and strong alkalis or even some metals don't mix well. A few years back, a neighbor at a shared lab moved some bottles around without thinking, putting this compound next to bases—it nearly caused a mess. Keeping incompatible materials at a distance takes little effort and can prevent a headline-making incident.
Small Precautions, Big Outcomes
Even in routine handling, small actions matter. Someone wearing protective goggles and gloves stands a better chance if something spills or leaks. Have an eyewash station close by and make sure labels carry clear warnings. Do regular checks on storage areas, not just for leaks, but also for faded warnings.
A simple rule I always stick with: never store more than you’re going to use in a reasonable time. Inventories piling up raises the risk, especially if folks retire or staff changes hands and labels get lost. Practical steps—like registering what you have and planning disposal—support a culture of safety.
Room for Improvement
Training beats every high-tech gadget. Clear communication among staff and regular reminders keep everyone sharp. There’s also growing interest in automated alert systems for volatile compounds, sending notices if temperature or humidity drift into danger zones. It doesn't take much to make a difference.
Talk to anyone with scars from old chemical mishaps and they'll say: smart storage saves. By focusing on airtight containers, temperature control, and keeping tabs on compatibility, workplaces keep risk low and everyone goes home safe.
What is the chemical formula and structure of Trichloronitromethane?
What Makes Trichloronitromethane Unique
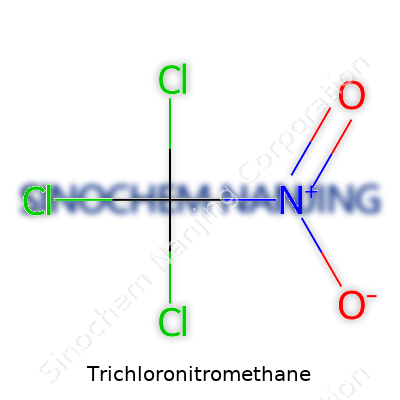
Trichloronitromethane, known to many in the chemistry field as chloropicrin, draws attention for its chemical backbone and historical impact on both agriculture and industry. Its formula, CCl₃NO₂, describes a single carbon branching out to three chlorine atoms and one nitro group. The structure can be written as Cl3CNO2. This straightforward formula leads to a shape where the carbon atom anchors the bulk of the molecule, letting both the chlorine atoms and the nitro group dictate much of its behavior in chemical reactions.
Real-Life Encounters: Beyond the Textbook
Years of experience in handling soil fumigants and pest control chemicals has led me into direct encounters with trichloronitromethane's properties. The pungent smell and eye-watering strength hit hard – not just for humans, but for most living organisms. This isn't just a byproduct of its chemical setup; it's evidence of how chemistry blends with ecology and public safety. In places where tomatoes, peppers, and strawberries grow on large acreage, the compound’s use keeps fields healthy by targeting nematodes, weeds, and fungal diseases. This effect is possible because the arrangement of chlorine atoms and the nitro group makes the molecule volatile and reactive with organic material in soil.
Digging into the Structure: How Arrangement Shapes Behavior
The carbon center in trichloronitromethane stands out due to its partial positive charge, caused by the three chlorines. Each chlorine, a strong electron-withdrawing group, pulls density away from the carbon, increasing its reactivity. The nitro group, on the other hand, lends its own share of electronegativity and oxygen content. This means the molecule doesn’t just evaporate easily; it also acts as a strong electrophile, making it effective in both pest control and as a lachrymatory (tear gas) compound in warfare. In daily life, these features translate to sharp toxicity – dangerous, but controllable when handled responsibly.
Historical Footprint and Safety Concerns
Over the decades, trichloronitromethane’s reputation carries both progress and caution. Its role as a chemical war agent in World War I pulled it from the lab into the battlefield. Later, farmers and scientists found value in its ability to sterilize soil. The same molecular traits that break down organic threats in dirt also threaten respiratory health and groundwater safety. Companies working with this compound need clear processes and strong ventilation to keep workers and nearby communities safe. I’ve seen training sessions stretch on for hours just to ensure no step gets skipped.
Weighing Risks and Rethinking Use
Scientific review today challenges us to look past single-molecule solutions. Research shows trichloronitromethane can linger in soil and water, and traces sometimes appear in produce. Public health groups and the Environmental Protection Agency have set strict limits, pushing the pursuit of alternatives and containment strategies. Crop rotation, biofumigation, and targeted drip systems present less-harmful ways to manage pests, all driven by understanding the detailed chemistry and behavior of such compounds.
The Road Forward: Making Chemistry Work for Us
Learning about trichloronitromethane tells a wider story. Knowledge of its formula and structure gives us tools to develop safety protocols, improve agriculture, and protect lives. Monitoring, investment in safer substitutes, and education at every level create paths where chemical expertise directly supports healthier land, safer food, and stronger communities. Science doesn’t stay trapped in a formula—it shapes decisions in the real world, every single day.
| Names | |
| Preferred IUPAC name | Trichloronitromethane |
| Other names |
Chloropicrin Nitrochloroform |
| Pronunciation | /traɪˌklɔːroʊˌnaɪtroʊˈmiːθeɪn/ |
| Identifiers | |
| CAS Number | 76-06-2 |
| 3D model (JSmol) | `JSmol.loadInline("data/mol/CCl3NO2.mol")` |
| Beilstein Reference | 1365051 |
| ChEBI | CHEBI:39241 |
| ChEMBL | CHEMBL156926 |
| ChemSpider | 14816 |
| DrugBank | DB04467 |
| ECHA InfoCard | 100.003.245 |
| EC Number | 203-641-2 |
| Gmelin Reference | 6076 |
| KEGG | C19190 |
| MeSH | D014245 |
| PubChem CID | 6414 |
| RTECS number | YX9625000 |
| UNII | 3HSC41V67Q |
| UN number | 4472 |
| CompTox Dashboard (EPA) | DTXSID0026604 |
| Properties | |
| Chemical formula | CCl3NO2 |
| Molar mass | 197.38 g/mol |
| Appearance | Colorless to pale yellow oily liquid |
| Odor | pungent |
| Density | 1.657 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.43 |
| Vapor pressure | 2.4 mmHg (@ 20 °C) |
| Acidity (pKa) | 0.86 |
| Basicity (pKb) | 8.86 |
| Magnetic susceptibility (χ) | -58.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.478 |
| Viscosity | 2.31 mPa·s (20 °C) |
| Dipole moment | 2.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 308.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -40.1 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -136.8 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AX05 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes severe skin burns and eye damage. May cause respiratory irritation. Toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H370 |
| Precautionary statements | P261, P273, P280, P301+P312, P305+P351+P338, P337+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2 |
| Flash point | 45 °C (113 °F; 318 K) |
| Autoignition temperature | 410 °C (770 °F; 683 K) |
| Explosive limits | Explosive limits: 6-14% |
| Lethal dose or concentration | LD50 (oral, rat): 160 mg/kg |
| LD50 (median dose) | LD50 (median dose): 160 mg/kg (rat, oral) |
| NIOSH | PB87165426 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Trichloronitromethane: 0.1 ppm (0.65 mg/m³) |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH: 25 ppm |
| Related compounds | |
| Related compounds |
Tribromonitromethane Iodoform Chloropicrin Fluoroform Bromoform Dibromochloromethane |

