An Editorial Look at Trichloromethyl Chloroformate: Insights and Realities
Historical Development
Trichloromethyl chloroformate did not just arrive on lab benches overnight. Decades back, chemists searching for reactive intermediates in organic synthesis chased after volatile compounds that promised higher yields and more control over reactions. As pressure mounted for more selective synthesis pathways—especially for pharmaceuticals and agrochemicals—chemical manufacturers focused on chloroformate derivatives. In the late 20th century, research on phosgene derivatives brought trichloromethyl chloroformate into sharper focus, as scientists hunted tools that could acylate compounds more efficiently than earlier options. The compound benefited from the rise of industrial research consortia, where sharing knowledge across universities and industry made process optimization possible. In my years consulting with synthetic chemists, time and again, they cited the growing pains of handling phosgene and how safer, more versatile reagents started to fill labs and production floors out of real necessity, not theory. Trichloromethyl chloroformate stands on the shoulders of that long, trial-heavy process.
Product Overview
Anyone who has opened a bottle of trichloromethyl chloroformate knows its sharp, pungent odor means business—this is not a chemical you treat lightly. Known for its use as an acylating and chlorinating agent, it plays a key role in the synthesis of carbamates, isocyanates, and other functionalized molecules. Ask a bench chemist why they reach for this over alternatives, and you’ll likely hear it’s about reactivity and selectivity. Trichloromethyl chloroformate does the job where other reagents stumble, offering efficiency that matters in both research and scale-up environments. In the pharmaceutical sector, it answers the call for bioactive molecule construction with fewer byproducts and cleaner work-ups. Year after year, demand for reliable, high-purity batches keeps suppliers improving their purification and handling protocols, as the end-users—often researchers developing new medicines—cannot risk contamination or inconsistent reactivity. Stories of failed syntheses due to subpar reagents echo throughout chemical development, driving the push for rigor in production.
Physical & Chemical Properties
This compound appears as a colorless to yellowish, highly volatile liquid with a density greater than water. It boasts a boiling point well below many common organic solvents, making containment hardware critical. Ask most organic chemists—they worry about volatility as much as reactivity, since rapid evaporation raises both exposure risk and waste. Highly soluble in nonpolar solvents, trichloromethyl chloroformate does not play well with moisture. Water triggers decomposition, generating phosgene and hydrochloric acid, both of which pose serious health risks and demand careful fume hood work. Its reactivity toward nucleophiles underpins its popularity. While many chemicals demand special handling, few offer such a blend of potency and volatility, shaping operational habits and laboratory design itself.
Technical Specifications & Labeling
Labeling in any lab begins with respect, especially for trichloromethyl chloroformate. Regulatory requirements call for clear hazard symbols, precise storage information, and up-to-date expiry dates. In practice, every container sports more handwritten warnings than most other bottles on the shelf. From my experience, colleagues mark not only the date received, but also the frequency of opening, given the compound’s instability in air and propensity to degrade. Technicians calibrate balances and dosing systems meticulously to counteract the volatility, keeping track of losses and recalibrating procedures. All this isn’t about paranoia—it comes from first-hand knowledge of emergency room visits traced back to mistakes in labeling or careless handling. The cost of cutting corners isn’t abstract; it’s measured in ambulance rides and lost research hours.
Preparation Method
Manufacturing trichloromethyl chloroformate traditionally involves reacting phosgene with methanol derivatives or trichloromethanol. Production scale-up shifted from small-batch glass reactors to tightly sealed industrial vessels, minimizing exposure and improving yield consistency. Engineers and chemists often recount early struggles with runaway reactions and phosgene leaks. As plant safety protocols evolved, controls for temperature, pressure, and reagent purity tightened. Automation brought greater reproducibility and lower occupational risk, but also demanded vigilance—automation does not remove the danger, it just changes its face. Lab-scale preparation remains rare due to the hazards of phosgene, prompting most researchers to purchase from specialists rather than attempt in-house synthesis. It’s a reminder that industrial chemistry is less about brute force, more about systematic risk management.
Chemical Reactions & Modifications
Trichloromethyl chloroformate reacts swiftly with amines, alcohols, and phenols, forming carbamates and carbonates that figure prominently in pharmaceuticals and agrochemicals. Functional group tolerance often sets it apart from related reagents. In real-world usage, researchers value its precision—side reactions stay minimal if you respect the moisture sensitivity and reactive profile. Chlorination reactions extend its utility, letting organic chemists fashion complex intermediates that serve drug discovery and advanced material synthesis. Playing fast and loose with this reagent courts disaster, as traces of water or careless temperature control can flood the system with phosgene, creating crisis instead of progress. Over years of consulting, I’ve seen whole campaigns derailed by improper quenching or a rushed work-up, underscoring the delicate choreography required for consistent results.
Synonyms & Product Names
Comprehending trichloromethyl chloroformate’s aliases matters more than many realize. It moves through the literature under names like diphosgene, carbonochloridic acid, or trichloromethyl chloroformate. Synonyms cascade from different naming conventions between manufacturers and regulatory agencies. Applied researchers swapping notes often hit walls of confusion from ambiguous nomenclature, reinforcing the importance of double-checking batch numbers, CAS registry details, and specific applications. For chemists transitioning between companies or countries, learning the full suite of names for such critical reagents means faster project onboarding and fewer costly mix-ups. Real experience teaches that administrative oversight in chemical identification can scuttle projects quicker than any failed experiment.
Safety & Operational Standards
Safety culture around trichloromethyl chloroformate runs deep. Every veteran in the lab has a story about an evacuation or near miss. Strict use of fume hoods, splash guards, fixed leak monitors, and personal protective gear keeps risks at bay. Emergency drills aren’t just paperwork—teams treat potential exposure as an ever-present hazard. Training new staff now means hands-on sessions focused on spill protocols, waste disposal, and first-aid for chemical burns or inhalation. In one multinational chemical plant, safety posters hang at every entrance, not out of habit, but as living reminders of hard-won lessons from accidents past. There’s no substitute for institutional memory—a culture that expects vigilance proves its value every time a close call becomes nothing more than a learning moment rather than a headline.
Application Area
Demand for trichloromethyl chloroformate springs from its ability to introduce protective groups efficiently, which remains a linchpin in multi-step organic synthesis. Process chemists in pharma talk about the time savings it brings in modifying amines and alcohols, and how cleaner conversions feed directly into regulatory submissions for investigational new drugs. Agrochemical developers turn to it when building crop protection agents, as the downstream chemistry depends on intermediates made possible by its reactivity. Material scientists, searching for novel polymers or coatings, lean on the carbamate and carbonate transformations enabled by this compound. Among synthetic chemists, reputation travels by word-of-mouth—tools that produce consistent, predictable results become unspoken standards. Trichloromethyl chloroformate sits in that rare category where risk matches reward, shaping not only synthetic routes but the evolution of product pipelines themselves.
Research & Development
Active research on trichloromethyl chloroformate revolves around making its usage safer, cleaner, and more amenable to automation. Academic labs team up with industry partners to devise sealed-system syntheses, minimizing human interaction without sacrificing product quality. Green chemistry initiatives focus on reducing phosgene use, exploring alternative reagents or buffered processes that contain or neutralize byproducts more efficiently. In R&D meetings, technical leads measure progress not just by yield improvements, but by reductions in hazardous waste and exposure incidents. Watching safety statistics trend down proves more satisfying than simply reporting an extra 5 percent yield. Graduate students and postdocs often drive this innovation, eager for methods that let them focus on discovery rather than hazard mitigation. It’s this blend of scientific curiosity and practical necessity that keeps developments rolling forward.
Toxicity Research
Toxicology teams document acute respiratory and dermal risks associated with trichloromethyl chloroformate. Repeated exposure brings a real chance of serious harm, as the compound breaks down to phosgene and acid on contact with biological tissues. Researchers publish case studies based on accidental releases or improper storage, highlighting the need for redundant safety controls. Medical teams associated with chemical facilities receive specialized training on rapid decontamination and supportive care. Regulatory agencies require regular reviews of permissible exposure limits, pushing companies to update engineering controls and monitoring equipment. Lessons string together from each documented incident, producing a living safety protocol that adapts as understanding deepens—a testament to how direct experience shapes best practice faster than regulation alone.
Future Prospects
As environmental and occupational safety expectations climb, chemists keep searching for reagents that can outperform trichloromethyl chloroformate without its drawbacks. Alternative acylating agents attract funding, but few match this compound’s balance of reactivity and selectivity. Process intensification—moving away from batch to continuous flow—looks promising, especially for limiting human exposure. Digital monitoring and predictive safety systems aim to cut accidents further, making once-exotic automation accessible even to midsized labs. Demand for greener, less toxic chemistry drives collaboration between synthetic chemists and engineers, with a shared goal: tools that power discovery yet bring risk down to a whisper. Watching the next generation enter the field, armed with better data and sharper risk awareness, leaves no doubt: chemical practice keeps changing, shaped by both necessity and experience. The coming years may see trichloromethyl chloroformate recede somewhat in favor of safer alternatives, but the lessons learned from its long, eventful run will guide safer, smarter chemistry for years to come.
What are the main uses of Trichloromethyl Chloroformate?
The Backbone of Chemical Synthesis
Any chemist who’s worked in a lab knows that progress often happens one clever reaction at a time. Trichloromethyl chloroformate, sometimes called diphosgene, shows up in places where pinpoint control over a process matters. Organic synthesis tasks rely on it to help form carbonates, carbamates, and ureas. These aren’t just textbook structures—pharmaceuticals and crop protection products start out as building blocks like these. Years ago, I watched a senior scientist demystify the hazards and rewards of using this chemical. The respect was clear, and so was the sense of opportunity: if you got the handling right, you could make compounds you’d struggle to synthesize any other way.
Role in Pharmaceutical and Agrochemical Research
Drug discovery jobs don’t hand out shortcuts. Researchers draw on trichloromethyl chloroformate because it helps insert a carbamoyl group exactly where it’s needed. Many modern pharmaceuticals—including some life-saving antibiotics and cancer drugs—take shape through reactions triggered by this reagent. Give it to the hands of a skilled chemist, and you’ll see it unlock challenging molecular architectures. Take crop protection: trichloromethyl chloroformate has shaped the backbone chemistry for insecticides and herbicides that support global food supply. This impact becomes real not just in patents and publications, but in every harvested crop that resists pests longer thanks to reliable active ingredients.
Polymers and Dyes: Shaping Modern Materials
Folks working in plastics and advanced coatings depend on reactions that require a balance of reactivity and selectivity. Trichloromethyl chloroformate steps in to help create specialty polymers. Some flexible foams and high-performance coatings trace their origins to these carbonylation reactions. The world of dyes also leans on this chemistry. Textile and display industries require colorants that last longer, fade less, and offer sharp clarity. The underlying molecular changes, invisible to the naked eye, come about thanks to the transformations made possible with trichloromethyl chloroformate.
Safety and Environmental Responsibilities
This chemical doesn’t forgive carelessness. As someone who’s worked in academic and industry settings, respect for hazardous reagents isn’t just about following rules. It’s about protecting health and the community. Trichloromethyl chloroformate can release phosgene gas. No operator wants that risk, so labs and plants invest in solid ventilation and proper personal protective gear. Industry groups promote regular training and risk assessments. The movement toward greener chemistry presses onward—researchers look for alternatives that reduce risks while preserving the reaction options that make innovation possible.
Improving Access for Researchers
Not every lab around the world finds it easy to handle or purchase trichloromethyl chloroformate. Transportation, storage, and regulation set real limits on who gets to use this reagent. Smaller teams or those in developing regions sometimes lose out on research opportunities. Shared facilities, better distribution networks, and consistent regulatory guidance could help. Policymakers and industry leaders would do well to keep channels open for education, storage, and disposal—supporting the next breakthroughs while guarding against risk for people and our planet.
What safety precautions should be taken when handling Trichloromethyl Chloroformate?
The Real Risks Behind the Name
Trichloromethyl Chloroformate sounds complicated, and honestly, it lives up to the name. Anyone working with it in labs or production lines already knows the thing can turn nasty fast. Most folks haven’t heard about this chemical, but in the right industry, it’s got a reputation. Breathing it in or spilling it on your skin can lead to some serious damage. The chemical releases phosgene gas, which historians remember from World War I as a chemical weapon. Makes you wonder what we’re doing with it these days, but the truth is, it’s still useful for making medicines and specialty chemicals. The danger doesn’t simply lurk; it lands hard when you get too casual.
Basic Gear Isn't Enough
Lab coats and nitrile gloves can stop most things, but not Trichloromethyl Chloroformate. I’ve watched someone faint before because their mask slipped and the air filtration wasn’t tight enough. Splash goggles barely cut it. The stuff produces vapor that finds its way into the tiniest gaps, and it stings like a wasp if it hits your eyes or skin. Full-face respirators and rubber gloves shield much better. The right gear needs to mean chemical-resistant suits and respirators with cartridges graded for acid gases or organic vapors. Routine checks for rips or leaks matter as much as wearing the gear in the first place.
Ventilation: Not Just an Extra Step
It shocks me how some places treat ventilation as an afterthought. Fume hoods don’t just clear the air; they save lives. Trichloromethyl Chloroformate evaporates quickly, and if you’re not keeping the air moving and filtered, people can end up breathing the sickly-sweet scent well before realizing the damage done to lungs. Anyone who sweats underneath their PPE knows opening a window is never enough. Certified chemical fume extraction is worth every penny spent.
Storage and Spill Smarts
Tragedy usually starts with storing the chemical on the wrong shelf or without checking for leaks. Exposure to water, sunlight, or heat can turn a small bottle into a hazardous mess. Even a bit of moisture can set off a reaction that fills the whole area with fumes. More than once, I’ve seen emergency showers and eyewash stations blockaded by storage boxes or carts, which never ends well if there’s a spill. Keeping containers closed, checking seals, and putting the chemical away from incompatible substances really makes a difference.
Training and Emergency Plans
No engineer or scientist, no matter how smart, should decide to “wing it” with this stuff. Proper training on each step—from opening a bottle to cleaning up a drip—keeps mistakes from turning deadly. If something spills or someone gets exposed, everyone needs to know the drill: evacuation, first aid, and who to call. Regular drills and visible emergency contacts stick with people a lot better than thick manuals. Nobody plans on accidents, but solid training and practice keep response quick and clear.
Final Thoughts on Collective Responsibility
Trichloromethyl Chloroformate isn’t an everyday problem for most people, but where it’s used, safety isn’t optional. Wearing the right gear, sticking to good habits, and insisting on strong ventilation and training help everyone get home safe. If leadership ignores one of these steps, it’s worth reminding them that lives rest on these decisions. No shortcut is worth the risk.
What is the chemical formula and molecular weight of Trichloromethyl Chloroformate?
Understanding the Basics
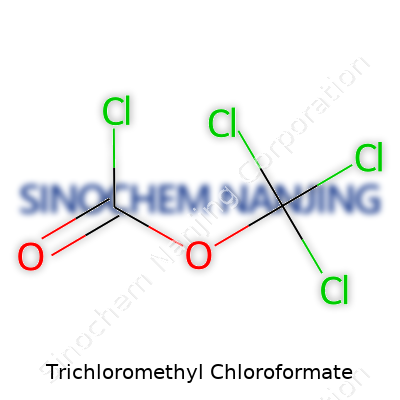
Most people don’t run into trichloromethyl chloroformate every day, but it pops up in the labs, chemical plants, and regulatory conversations that keep our modern world going. Its chemical formula, C2Cl4O2, brings together two carbon atoms, four chlorine atoms, and two oxygen atoms. Molecular weight lands at 197.84 g/mol. These aren’t just numbers for textbooks—they carry weight in safety management, research, and even environmental policy.
Where the Risks Hide
Personal experience teaches that understanding the chemistry isn’t just about memorizing formulas. Roughly two years ago, I watched a seasoned chemist pause before opening a bottle labeled C2Cl4O2. One sniff of escaping vapor signaled trouble. Getting a reminder that this compound breaks into phosgene—one of the nastiest toxins from World War I—changed the way many in that lab handled reagents. Health risks from trichloromethyl chloroformate go beyond everyday lab mishaps. Just a small spill releases phosgene and hydrochloric acid, both hazardous to breathe. The Environmental Protection Agency has flagged this stuff for extra care, not just for workers but for communities downwind of manufacturing sites.
Regulatory Landscape and Compliance
Regulators never treat chemicals like C2Cl4O2 lightly. The Occupational Safety and Health Administration asks for detailed documentation and strict containment practices. Looking at the material safety data sheets, you notice a pattern—minimal exposure, swift containment, and readiness for spills. Laws keep workers and the environment safer, but following these rules also costs time and money. Some facilities switched to less toxic alternatives, but replacing C2Cl4O2 isn’t always possible when it's the only reagent that gets a reaction right.
Industrial Uses and Challenges
Trichloromethyl chloroformate finds its way into making medicines, dyes, pesticides, and specialized plastics. Chemists appreciate how it delivers the right chemical groups to other molecules, speeding up synthesis. The work process never gets routine. Handling comes with extra layers of gloves, special storage cabinets, and reliable ventilation. Emergency showers and respirators stand within reach, not just as a formality but as part of daily workflow.
Seeking Better Solutions
Reducing risk starts with knowledge. Chemists who understand molecular weight and structure take fewer risks and communicate hazards clearly. Digital tracking systems now spot inconsistencies fast—if chemical quantities don’t add up, the system flags it. Training staff to spot early warning signs or using real-time gas sensors helps catch leaks before they turn into emergencies. In some universities and companies, teams work on greener alternatives, developing reagents that deliver the same chemical punch but break down into safer byproducts. Investment in innovation here could pay back later, cutting down on medical bills, cleanup efforts, and legal headaches.
Final Thoughts from Experience
A world built on industrial chemistry relies on trust in the people holding the beakers and on the numbers in the formula. With C2Cl4O2, knowing and respecting its chemical identity shapes every step, from the first order to safe disposal. Facts don’t tell the full story—it takes vigilance, teamwork, and, at times, a healthy fear of the unexpected lurking behind a simple label.
How should Trichloromethyl Chloroformate be stored?
The Chemistry and the Challenge
Trichloromethyl chloroformate carries a reputation for being tough to handle. It’s a clear liquid, but don’t let that fool you—this compound reacts quickly with moisture, generates corrosive gases, and can sting the lungs just from vapors. Anybody who’s ever opened a bottle of it in the lab knows not to underestimate its kick. Somebody in the chemical supply chain has to face the tough reality: mishandling even a few liters of this stuff can lead to serious burns or a lockdown-sized emergency.
Why Proper Storage Decisions Save More than Materials
This chemical reminds me of tales from older colleagues, who’d recall leaky stoppers leading to sleepless nights and tough phone calls. Those lessons stick. Sticking a bottle of trichloromethyl chloroformate on a basic chemical shelf invites disaster. Heat, sun, or a humid afternoon in the storeroom—each can trigger reactions before anyone gets a warning. That’s not a risk worth testing.
Fire departments and chemical safety boards both agree: use steel cabinets rated for flammable and corrosive chemicals. People who work with these substances trust locking acid cabinets—not cheap plastics or woods—because they carry self-closing doors and ventilation, sometimes with automatic suppression systems. That way, a cabinet can slow down an accident and give humans a chance to react before injuries happen.
What Experience Teaches about Containers and Placement
Glass seems like a good call—trichloromethyl chloroformate eats through some plastics, and metal containers mean corrosion. Always go for bottles with tight, corrosion-resistant caps and clear labeling—no scratched-out abbreviations or old stickers. In one incident a few years back, faded labels fooled a junior tech, and she treated a bottle like ethanol. She stopped when she spotted the sharp chemical scent. Sometimes, vigilance saves the day, not just rules.
Keep these containers far from any water source or chemical that could set off a runaway reaction. That often means a shelf at eye level, deep inside a ventilated cabinet, never stacked above head height where a slip could turn dangerous. Folks in the safety office double-check for separation from amines, bases, and any products with active hydrogens—even a single shelf gap keeps incompatible chemicals from crossing over.
Why the Extra Steps Can’t Be Skipped
It’s tempting to get casual after a few routine months, but shortcuts with chemicals like trichloromethyl chloroformate cost too much. In a busy teaching lab, a leaky cap once triggered a slow pressure buildup. I remember the sound—a soft hiss when the cabinet door opened. One lab tech’s quick call to the supervisor got everyone moving in time, and no one got hurt. That episode cemented a culture of double-checking locks and storing only what’s needed, not a drop more. Lower volumes mean less chance for trouble if human error enters.
Responsible storage doesn’t end with the chemical itself. Safety goggles, face shields, gloves, and acid-resistant aprons stay close by. A standardized log for inventory keeps everyone alert, and regular inspections spot corrosion or leaks before the worst happens. Thoughtful workflow and a culture of respect for potent chemicals support everyone’s health. These experiences build habits that keep the whole team safe and the lab running without interruption.
What are the potential health hazards of Trichloromethyl Chloroformate exposure?
Why Trichloromethyl Chloroformate Matters
Working around chemicals in a lab or an industrial setting brings its own risks, but some substances deserve extra caution. Trichloromethyl chloroformate often pops up as a reagent in making pharmaceuticals and agrochemicals. Its role sounds technical, but its real effect becomes dangerously clear in the wrong setting. Years of dealing with hazardous materials showed me how things can turn quickly from safe to threatening. This chemical stands out because it reacts strongly when mixed with water or takes in moisture from the air, producing toxic gases almost instantly.
Health Risks That Can't Be Ignored
A single whiff or accidental touch can start a chain of trouble. The fumes from trichloromethyl chloroformate irritate the eyes and throat, sometimes causing breathing difficulties. People unlucky enough to get it on their skin often talk about painful burning and blistering. This isn’t rare speculation—health agencies recognize its dangers. According to the U.S. National Institute for Occupational Safety and Health (NIOSH), inhalation can cause coughing, chest tightness, and, in heavy doses, fluid buildup in the lungs.
I’ve seen what happens when someone underestimates a chemical’s danger. Rushed procedures or poor protective gear almost always lead to burnt skin, watery eyes, or far worse. For sensitive groups—like those with asthma or other lung conditions—the odds of a severe reaction jump even higher. Workers in poorly ventilated factories or labs risk long-term problems like chronic bronchitis or, in extreme cases, damage to the cells inside the lungs.
Why Safe Handling Changes Everything
Placing labels and warning signs isn’t just red tape. Good training, solid personal protective equipment, and clear safety protocols change outcomes. In my early days in a university lab, we drilled response scenarios for chemical spills over and over. At the time, it felt like overkill. Later, those routines made all the difference when an intern knocked over a flask and fumes started spreading. Quick action kept everyone safe, but only because everyone recognized the signs and moved fast.
The Environmental Protection Agency and OSHA both require strict handling and storage procedures for chlorinated chemicals. Fume hoods, proper gloves, and safety goggles do more than check boxes—they block out most of the danger. A good culture of safety means everyone respects the risks and never shortcuts the process.
Lowering Risks in the Real World
Reducing exposure takes more than gear. It starts with solid education. New staff or students need to see real examples and hear stories, not just read manuals. Regular checks on safety equipment and storage tanks prevent leaks and mishaps. If management skips these steps, accidents sneak in. Labs and factories should have emergency eyewash stations and showers installed near working zones, since quick rinsing can stop a minor accident from turning catastrophic.
Companies that take the time to regularly update safety plans and invest in fresh equipment see fewer injuries. Medical surveillance programs pick up early warning signs, helping workers get treatment before health problems settle in for good. Honest reporting—without fear of punishment—means issues get addressed fast rather than hidden until it’s too late.
Keeping Perspective
Trichloromethyl chloroformate isn’t the only chemical with serious health hazards, but its particular risks remind us that one slipup can change a career or a life. Staying informed, practicing respect for the substance, and refusing to neglect basic safety steps—all of these shape how things end up for workers and the communities around them.
| Names | |
| Preferred IUPAC name | Trichloromethyl carbonochloridate |
| Other names |
Chlorodicarbonic acid trichloromethyl ester Carbonochloridic acid, trichloromethyl ester Trichloromethyl chloroformate Trichloromethyl carbonochloridate Carbonochloridic acid trichloromethyl ester |
| Pronunciation | /traɪˌklɔːroʊˈmɛθɪl klɔːrˈɒr.mə.feɪt/ |
| Identifiers | |
| CAS Number | [721-56-2] |
| Beilstein Reference | Beilstein 97035 |
| ChEBI | CHEBI:38960 |
| ChEMBL | CHEMBL226675 |
| ChemSpider | 12313 |
| DrugBank | DB08655 |
| ECHA InfoCard | 03a2e278-5fa7-409d-8309-0ddbda6dfa84 |
| EC Number | 205-710-6 |
| Gmelin Reference | Gmelin Reference: **90649** |
| KEGG | C19572 |
| MeSH | D014258 |
| PubChem CID | 6574 |
| RTECS number | GF5950000 |
| UNII | J3R8O0785C |
| UN number | UN2580 |
| CompTox Dashboard (EPA) | DTXSID6020263 |
| Properties | |
| Chemical formula | C2Cl4O2 |
| Molar mass | 183.38 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent odor |
| Density | 1.57 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 1.85 |
| Vapor pressure | 13 mmHg (20°C) |
| Acidity (pKa) | 1.3 |
| Basicity (pKb) | Trichloromethyl Chloroformate does not have a reported pKb value. |
| Magnetic susceptibility (χ) | -49.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4680 |
| Viscosity | 1.798 mPa·s (20 °C) |
| Dipole moment | 1.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.6 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -214.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -471.7 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. H317: May cause an allergic skin reaction. H330: Fatal if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P210, P260, P280, P301+P330+P331, P303+P361+P353, P305+P351+P338, P310, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Autoignition temperature | 250 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 90 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 80 mg/kg |
| NIOSH | IT6475000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Trichloromethyl Chloroformate is 0.05 ppm (0.4 mg/m³) |
| REL (Recommended) | 0.05 ppm |
| IDLH (Immediate danger) | 2 ppm |
| Related compounds | |
| Related compounds |
Dichloromethyl chloroformate Chloromethyl chloroformate Methyl chloroformate Ethyl chloroformate Trichloromethanol Phosgene |

