Trichloroacetonitrile: A Closer Look at Its Legacy and Future
Historical Development
Trichloroacetonitrile has traveled an interesting road since its origins in the late nineteenth century. Chemists searching for versatile building blocks discovered that nitriles with halogen substitutions could open up a new toolkit for synthesis. While other halogenated compounds like chloroform or carbon tetrachloride got most of the headlines, trichloroacetonitrile found its place among chemists who needed a little more reactivity from a nitrile group, coupled with the ability to manipulate chlorines. Laboratories have long valued it for the sharp reactivity it brings, and it’s become an enduring presence in the chemical catalogs. To most non-specialists, the name barely registers; for those in synthetic chemistry or specialized manufacturing, its fingerprint runs through decades of published work and patents.
Product Overview
With a prismatic cyanide smell and a dense, liquid texture, trichloroacetonitrile isn’t the friendliest substance to meet, but it plays a large role behind the scenes in research and industry. In many cases, this material enables transformations and pathways for other, more familiar drugs, polymers, and agricultural tools. Its value lies less in its glamour and more in its workhorse reliability and high reactivity. I learned early on that this is the kind of chemical you respect: clear purpose, straightforward to use for those who know what to expect, and definitely not meant for experimentation without deep familiarity.
Physical and Chemical Properties
From a practical standpoint, trichloroacetonitrile boasts a fairly high boiling point, a compact molecular structure, and substantial density for its molecular weight. The sharp, acrid odor lingers in the lab air, a warning that this compound is best worked under proper ventilation. Its solubility profile focuses on organic solvents, which suits its use in organic synthesis well. The real draw, though, comes from its trio of chlorines attached to an already-active nitrile group. Where other nitriles sit quietly, this one sparks curiosity amongst chemists—reactivity that can slice through carbon frameworks or attached to functional groups as needed.
Technical Specifications & Labeling
Bottles of trichloroacetonitrile in the lab share common themes: prominent hazard markings, amber glass to block light, and labeling that never underplays the dangers. Every batch comes with purity data, and small manufacturers work hard to keep contamination low. It’s the sort of product you can’t casually forget on a bench; a minor spill can create an overpowering stench, and its status as a lachrymator means even a little on exposed skin or in the air can force a hasty retreat for fresh air. Handling guidelines stress immediate containment, not just for legal liability, but for personal safety born out of experience.
Preparation Method
Let’s not sugarcoat it: trichloroacetonitrile is usually made industrially from trichloroacetaldehyde (chloral) and ammonia or related nitrogen sources. This reaction steers through a tightly controlled process, where the stoichiometry and temperatures call for precision and patience. For years, synthetic textbooks presented variations, mostly tweaking yields and purification steps. The best setups channel waste away from the lungs and eyes of anyone in the vicinity and harvest the liquid product under cool, dry conditions. Fume hoods aren’t optional. That old proverb about knowing your materials rings especially true with this stuff, as even skilled chemists keep an antitoxin protocol handy.
Chemical Reactions & Modifications
Trichloroacetonitrile stands out because it’s not just a passive solvent or inert building block. Instead, it offers chemists multiple points of attack. The chlorines fall off—or swap partners—with strong nucleophiles, opening doors to unique intermediates not easily reached another way. When a nitrile needs to be converted, the three chlorines promote powerful electron withdrawal, making the carbon even more susceptible to further transformation. Its usefulness in cyclization reactions, in the formation of heterocycles, and in selective reductions makes it a staple in advanced synthesis. I recall a day in graduate school when a single miscalculation with trichloroacetonitrile delayed an entire week’s schedule—it demands respect in every drop.
Synonyms & Product Names
The chemical world offers plenty of aliases for trichloroacetonitrile: 2,2,2-Trichloroacetonitrile, Cyanotrichloromethane, and Trichloroethanenitrile, to name a few. In catalogs, the CAS number usually settles confusion, but around the lab, the shorter name is common parlance. Any practitioner soon learns to double-check the label; those three chlorines can make for nasty surprises if mistaken for another nitrile or misread in haste. There’s an object lesson in humility every time someone orders the wrong isomer or reaches for this compound when a less aggressive nitrile is needed.
Safety & Operational Standards
Personal experience offers one clear message: do not ignore the risks with trichloroacetonitrile. The chemical loves to irritate mucous membranes, causing burning eyes, skin rashes, and breathing issues. That’s not scaremongering, just honest trailblazing from scientists who’ve spilled it on the bench or inhaled a waft of fumes on a bad lab day. Practices like wearing gloves, goggles, and working under a certified fume hood aren’t optional—they’re minimum preparation. Waste goes into sealed containers well away from heat sources or organic debris. Over years in the lab environment, I learned to spot newcomers who took the warnings lightly; they rarely made that mistake twice.
Application Area
Trichloroacetonitrile rarely sees direct consumer use. Its destiny lies deep in the trenches of organic chemistry, pharmaceuticals, and select agricultural chemistry. It helps synthesize α-chloro carboxylic acids, pharmaceuticals, dyes, and specialty intermediates. In research labs, it enables transformations that might seem out of reach otherwise, bridging molecular gaps for medicinal chemistry or pesticide research. While better-known chemicals attract attention, this one quietly greases the wheels, letting new reactions and molecules appear where old methods failed. I’ve seen it mentioned in late-night research marathons and patent sprints—the kind of tool that sits silent in the background, quietly shifting the possibilities.
Research & Development
Innovation with trichloroacetonitrile hasn’t slowed. Scientists probe ways to harness its reactivity with less environmental fallout. Environmental chemists pursue greener syntheses or work to tame the toxic impact of manufacturing, while medicinal chemists use it for synthesizing intricate active molecules. Its electron-poor carbon center lets it feature in cutting-edge routes, from heterocycle assembly to polymer science. R&D labs now put greater emphasis on lifecycle safety, emission controls, and alternative reagents, but trichloroacetonitrile’s powerful chemistry keeps it in the conversation. Young chemists sometimes eye it warily, aware of the risk-reward balance, but the results often justify its continued use in the right hands.
Toxicity Research
Exposure to trichloroacetonitrile can turn a productive day into a lesson in chemical safety. Studies confirm that the compound has pronounced acute toxicity via inhalation, skin contact, and ingestion. It targets the central nervous system and lungs at high doses, and repeated contact brings chronic health issues. Over time, regulations and workplace practices grew more strict for a reason—too many stories of accidental exposure and long-term effects taught hard lessons. Major health agencies watch developments closely, and toxicologists push for safer handling and disposal. Experience has taught me the importance of rigorous protocols—even seasoned professionals have stories of accidental exposure that caused hours or days of discomfort.
Future Prospects
Chemists face a balancing act with trichloroacetonitrile. The need to build new molecules faster and cleaner has some pushing for safer and greener alternatives, while others refine ways to contain emissions, recycle waste, or reformulate processes for minimal human exposure. There’s real potential in new applications, especially as computational chemistry opens windows into mechanisms few had considered. Industry continues to navigate compliance and liability, pushing for closed systems and automated production lines, while research teams work to make sure that risky materials like trichloroacetonitrile offer maximum benefit for minimum cost—both in safety and budget. One thing’s certain: experience and caution will always remain more valuable than any fancy new technology, especially with a substance this powerful and unforgiving.
What is Trichloroacetonitrile used for?
Trichloroacetonitrile in Chemical Synthesis
Trichloroacetonitrile steps into countless chemical labs as a fierce nitrile compound. I've seen precise organic syntheses lean on this molecule for its reactive nature, which pops up in pharmaceutical factories and academic research alike. The moment you want to whip up certain amides, carboxylic acids, or heterocyclic skeletons, this compound turns useful. One common way scientists use it: converting alcohol groups to nitriles, an essential move in drug development. In real research, getting from one functional group to another defines the entire outcome, so trichloroacetonitrile becomes a bit like an overlooked utility player on a baseball team—steady and reliable, even if few fans buy its jersey.
Role in Pharmaceutical Manufacturing
Pharmaceuticals don’t just happen. Every effective medicine you’ve seen on the market, from old antibiotics to new cancer drugs, depends on a supply of clean intermediate chemicals. Trichloroacetonitrile helps create those intermediates, especially where standard reactions struggle. When pharmaceutical chemists want to build complex rings—the backbone for plenty of modern medicines—they reach for reactive compounds like trichloroacetonitrile. The yield matters; nobody wants waste or lengthy purification. It helps keep those margins thinner, keeps labs safer, and helps get novel drugs to the shelves a little faster.
Trichloroacetonitrile and Pesticide Production
If you’ve ever wondered about the contents of a pesticide label, chances are there's a web of chemistry behind it. Trichloroacetonitrile gets used by many agrochemical plants as a raw component in manufacturing certain pesticides and herbicides. Modern food systems often depend on these to prevent crop loss. A compound like trichloroacetonitrile makes the difference between a bug problem ruining acres of crops and a stable food supply. Its use in intermediates for pesticides keeps input prices manageable, which translates to steadier grocery bills at the store.
Safety and Environmental Concerns
You hear 'trichloro-' and the mind drifts to safety issues. That’s fair. My time shadowing chemists in the field taught me the lesson: treat every potent chemical with respect. Trichloroacetonitrile requires personal protective gear, proper ventilation, and strict storage rules. Accidental releases or improper handling can lead to health hazards—not just for workers but for nearby communities and the water supply. The world is more alert than ever to chemical exposure risks. European safety standards and US regulations classify trichloroacetonitrile as hazardous, enforcing guidelines for its transport and disposal.
Reducing Risks and Shaping Better Policies
Public health depends on balancing practical needs with careful safeguards. Advocating for tighter oversight or more comprehensive waste management aligns with EPA and global safety targets. I’ve met researchers pushing for green chemistry approaches, replacing harsh chemicals with safer agents. In the lab, it often means spending a little extra to ensure long-term wellbeing. Companies and universities investing in decontamination systems and monitoring protect their staff—and the surrounding neighborhoods. By pushing for alternatives, training everyone involved, and backing regulatory science, society can handle compounds like trichloroacetonitrile responsibly.
Looking Forward
The role of trichloroacetonitrile links back to industries that touch nearly everyone—medicine, farming, and chemical innovation. Access to reliable, high-quality chemicals shapes research, manufacturing, and daily living. The best outcomes come from experienced professionals following the science, listening to regulators, and putting safety at the front. That’s how progress avoids the kind of shortcuts that end up on front-page headlines for all the wrong reasons.
What are the safety precautions when handling Trichloroacetonitrile?
The Risks That Come With Trichloroacetonitrile
Trichloroacetonitrile means business. Sitting in a bottle with its sharp chemical scent, it causes fierce irritation to eyes, skin, and lungs. Even a short sniff can set off coughing or burning in your throat. Spilling this liquid can spell trouble if you skip proper protection—especially for people who spend years in a lab or plant environment, not just a few minutes for a demo.
Why Serious PPE Is Non-Negotiable
Drawing from my own time hunched over a lab bench—cramming for finals or cranking out samples for research—I saw right away that forgetfulness gets punished. Splashing a drop of trichloroacetonitrile on bare skin means racing for the eye-wash or safety shower. At my old workplace, anyone who handled this chemical wore splash goggles, gloves made of nitrile or butyl rubber, and a solid lab coat. Thicker gloves work better. Latex or vinyl tends to break down, spreading risk instead of blocking it. I took precautions seriously, more than once finding myself grateful for that extra barrier.
Air Quality Makes a Difference
One whiff of trichloroacetonitrile, and you understand why ventilation comes up so often in safety manuals. Open benches or crowded workshops can turn a minor mistake into a big problem. Fume hoods are more than just fancy enclosures—they pull vapors away before you breathe them in. I remember a batch that got mixed a bit too aggressively, sending vapors up in an instant. The hood caught it, and I thanked the old controls for never letting me work with the sash too high.
Storage Isn't Just About Neatness
Chemical spills in storage rooms stick in your memory. I once reorganized a cluttered shelf and bumped into an old, poorly labeled bottle of trichloroacetonitrile. Luckily, our team kept incompatible substances apart—anything with moisture or bases was banished to another part of the room. Leaky bottles always got swapped out fast; you don’t want this stuff eating through a cap or mixing with water by accident. Temperature control matters too. Trichloroacetonitrile sits best in a cool, dry spot, inside a fire-proof safety cabinet. Labeling has to be clear, not faded or hidden behind other bottles.
What To Do If Something Goes Wrong
No one loves drills or reading safety posters, but practice makes response automatic. I once watched a new technician hesitate when he splashed some on his glove. Training kicked in—off went the gloves, straight to the wash station, and a check for any chemical exposure. Our supervisor stressed calling for help, even if you “feel fine.” Inhalation means fresh air and a visit to a doctor. Spills, even small ones, get covered with inert material—something like sand or a spill pillow—and scooped with care. Never mop up with bare hands or rags.
Trust Science, Not Guesswork
The rules for handling trichloroacetonitrile are strict because people paid the price before. Poisonings, burns, and even fires make up the history of chemical safety. Every regulation and checklist gets built from real accidents. New employees get hands-on safety training, and even old hands at the job update their knowledge. Reliable suppliers provide detailed Safety Data Sheets. Rely on those instructions, never rumors or shortcuts, and double-check everything before every step.
Culture and Accountability Lead to Fewer Mistakes
A solid safety culture does more than avoid fines. It keeps teammates healthy enough to return home after every shift. In labs and factories where supervisors and techs talk through the potential hazards, mistakes drop fast. Clear signage, accessible personal protective equipment, and ongoing conversations—these help keep trichloroacetonitrile, and anything like it, in check. Safety grows from habits, not just rules.
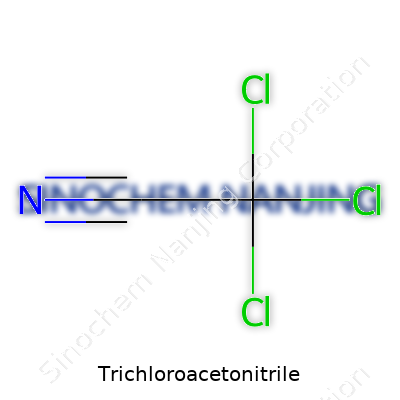
What is the chemical formula of Trichloroacetonitrile?
Knowledge From Chemistry Labs and Real-Life Application
Ask anyone who’s spent a few semesters in a chemistry lab—few names stick in your brain like Trichloroacetonitrile. This mouthful of a compound boils down on paper to the chemical formula C2Cl3N. Those five letters and numbers may not mean much to most people, but in research or industrial settings, every character counts. The arrangement of atoms, the number of bonds, and the placement of those chlorines dictate what sort of chemical magic or trouble this substance brings into the room.
Breaking Down the Formula
With two carbon atoms, three chlorine atoms, and one nitrogen atom, Trichloroacetonitrile does more than just sit quietly in inventory. The structure places the chlorines all around one carbon, with the nitrile group (that’s the -CN hook) on the neighboring carbon. Chemical structures like this set off alarm bells in the safety training courses I remember—chlorinated compounds, especially small ones, often sneak through gloves and evaporate in no time.
Understanding Use and Significance
This compound’s most well-known role pops up in the world of organic synthesis. You can find references to it as a reagent, a building block, and sometimes as an intermediate in drug or pesticide making. Researchers like it because it tends to react reliably, and those three chlorines attached in a row open up all sorts of possibilities. The fact remains, though, that any chemist working with it treats the bottle with no small amount of respect. Trichloroacetonitrile releases fumes you don’t forget, and toxicology data backs up the caution you see in every safety manual.
Risk Connected to the Formula
The chemical formula doesn’t just say what’s inside; it predicts how a molecule behaves. Those three chlorine atoms ramp up both chemical reactivity and potential human health risk. Exposure through inhalation or skin contact leads to real problems. The past decades show enough research to warrant strict handling. Regulatory agencies list Trichloroacetonitrile, and guidelines spell out ventilation, gloves, eye protection, and waste disposal. In my time working on smaller scale syntheses, yearly refresher training on handling chlorinated reagents carried the same weight as lab fire drills. Nobody wants to be caught off guard.
Toward Safer and Smarter Use
The chemistry community spends a lot of time on new ways to achieve the same outcomes with less hazardous compounds. But as of now, Trichloroacetonitrile still earns its spot on research and industrial shelves. People working with the compound should demand the latest safety data and shouldn’t hesitate to speak up about handling procedures. Relying solely on tradition in chemical processes ignores what scientists continue to learn about long-term health and environmental effects.
As demand for safer workplaces and greener chemistry grows, teams need accessible education and investment in improved gear—closed-system reactors, modern fume hoods, better analytical tools—to manage the risks that come with compounds like this. The formula C2Cl3N may look simple printed on a label, but it signals a lot more work than casual handling suggests.
How should Trichloroacetonitrile be stored?
Respecting the Substance
Trichloroacetonitrile doesn’t get much attention outside of chemical circles, but it deserves respect from anyone handling or storing it. In labs and industrial spaces, trichloroacetonitrile serves as an important intermediate, and I’ve seen firsthand how small oversights in its storage can turn into health emergencies.
Health Hazards Demand Caution
Exposure to trichloroacetonitrile can cause severe irritation in the eyes, skin, and respiratory system. If the chemical leaks or breaks down, it can produce toxic gases like hydrogen chloride and cyanide. These aren’t just scary words on a safety sheet—lab workers have ended up in the hospital for skipping protective gear. Storing this chemical properly isn’t about following a checklist; it’s about looking out for people’s health.
Choosing the Right Container
Sturdy, sealed containers aren’t negotiable. Glass or high-quality, chemical-resistant plastics work well. Metal containers tend to react with halogenated organic compounds, so they should stay out of the picture. A loose lid or cracked seal risks both leaks and contamination, so regular inspections feel like common sense, not just protocol.
Cool, Dry, and Shelter from Light
Heat turns trichloroacetonitrile into a different beast. In warm rooms or direct sunlight, it decomposes faster, leading to dangerous fumes and degraded material. A cool, dry storage area, away from heat-generating machines and exposure to sunlight, increases safety for everyone nearby. High humidity causes condensation issues and, indirectly, corrosion on metal fixtures. Placing containers away from sinks, pipes, and damp floors keeps moisture out of the equation.
Avoiding Incompatible Chemicals
Acids and bases cause reactions with trichloroacetonitrile that nobody wants in a storage room. Combining this chemical with strong oxidizers, alkaline materials, or reducing agents creates violent or toxic outcomes. Arranging shelves so incompatible chemicals never share a spill tray reduces that risk. In chemical storage, physical distance matters as much as labeling.
Labeling, Ventilation, and Security
Labels should state the full chemical name and hazard class in bold, readable print. If workers have to hunt for hazard information, someone will eventually get hurt. Good ventilation prevents the buildup of fumes from slow leaks or accidental drips. Proper vent hoods suck out hazardous air, making accidental exposure less likely. Storage rooms should stay locked. Only trained staff need access, so accidents and theft stay at a minimum.
Regular Training: The Not-So-Secret Ingredient
Safe storage goes beyond room temperature and containers. People make the biggest difference. Teams need regular refreshers and updates on storage guidelines, spill handling, and emergency response. Having clear instructions nearby—posted right on the storage room wall—saves precious time during a crisis. Training isn’t about passing safety audits; it’s about sending everyone home healthy.
Disposal Counts as Storage
Unused trichloroacetonitrile and contaminated gear call for immediate, careful disposal. Outdated bottles shouldn’t linger on shelves as a forgotten hazard. Arranging for pickup by certified hazardous waste handlers closes the loop. Watching a careless disposal turn into a workplace crisis taught me to never put off that step.
Wrapping Up With Safety in Mind
Protecting people and the environment from trichloroacetonitrile takes commitment to good practices, solid training, and regular checks. Each small precaution adds up to a safer workplace and one less reason for an emergency room visit.
What are the health hazards associated with Trichloroacetonitrile?
Understanding the Chemical’s Risks
Trichloroacetonitrile builds its reputation as a specialty chemical used in making pharmaceuticals and agricultural chemicals. Folk in labs and factories might run into it during production or research. This stuff mostly shows up as a colorless liquid, but don’t let the plain look fool you. Behind the scenes, it’s packing some punch in the hazard department.
Breathing and Skin Trouble
People who’ve worked with harsh chemicals know that a whiff or a drop where it shouldn’t go can mean a pretty tough day. Trichloroacetonitrile falls in this league. Breathing in the vapor hits the nose and throat, leading to coughing and choking. Eyes start to burn. Folks with longer exposure risk headaches and, in bad cases, passing out. Liquid contact with skin strips away oils, giving way to redness, blisters, and pain. Even a splash across regular work clothing can seep through over time.
Damage Goes Deeper
Laboratory animal studies flag bigger concerns. The chemical can break down in the body to form cyanide, one of the most dangerous toxins out there. Cyanide chokes off a body's ability to use oxygen, and that can lead to organ damage or death if things get serious. Folks in chemistry or pesticide plants tell stories of colleagues needing immediate first aid or hospital runs after accidental splashes or leaks.
Lingering Dangers
A room that’s held this chemical needs a solid clean-up. Any left-over vapor in the air can stick around, settle into furniture or gear, and come back to get someone later. Even though most people don’t handle trichloroacetonitrile every day, improper storage or poor ventilation in workplaces has led to unexpected fumes and lingering health problems among workers.
Worker Protection and Oversight
Health agencies pay close attention to chemicals like this. In the United States, the Occupational Safety and Health Administration (OSHA) gets involved, setting exposure limits where possible. The National Institute for Occupational Safety and Health (NIOSH) lists it as a substance requiring strong controls — gloves, face shields, full protective clothing, and fume hoods. Even so, stories still circulate about accidents from slips in procedure or old equipment. In 2013, a spill at a production facility sent several workers to the emergency room. Quick action limited damage, but that event serves as a warning.
Better Safety Means Better Health
From my own days working in a plant, strict routine checks and fresh air systems did more to protect folks than all the safety posters money could buy. Training makes a difference. Nobody wants to watch a friend or co-worker doubled over from chemical fumes. Simple steps matter — double-check seals, don't skip protective gear, keep emergency showers and eyewash stations ready.
Switching to safer alternatives, where possible, also carries weight. Some countries favor chemicals with a lower risk profile. If trichloroacetonitrile can’t be dodged, strong rules and constant vigilance must stay in place. A company that values its people never cuts corners on safety. Everyday routines in chemical labs or plants save more lives than fancy new gear.
| Names | |
| Preferred IUPAC name | Trichloromethanenitrile |
| Other names |
Cyanotrichloromethane Trichloromethyl cyanide |
| Pronunciation | /traɪˌklɔːroʊˌæsɪtoʊˈnaɪtraɪl/ |
| Identifiers | |
| CAS Number | 545-06-2 |
| 3D model (JSmol) | `data="C#NCl(CCl)Cl"` |
| Beilstein Reference | 1209223 |
| ChEBI | CHEBI:35911 |
| ChEMBL | CHEMBL186477 |
| ChemSpider | 6788 |
| DrugBank | DB14035 |
| ECHA InfoCard | 03-2119944712-50-0000 |
| EC Number | 203-686-6 |
| Gmelin Reference | 923 |
| KEGG | C19268 |
| MeSH | D014248 |
| PubChem CID | 6574 |
| RTECS number | GC5950000 |
| UNII | 58781A2UWS |
| UN number | UN3430 |
| CompTox Dashboard (EPA) | DTXSID2020726 |
| Properties | |
| Chemical formula | C2Cl3N |
| Molar mass | 149.39 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Pungent |
| Density | 1.336 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 1.90 |
| Vapor pressure | 24.9 mmHg (20 °C) |
| Acidity (pKa) | 11.4 |
| Basicity (pKb) | pKb = 14.56 |
| Magnetic susceptibility (χ) | -53.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 0.682 cP (20°C) |
| Dipole moment | 1.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 220.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -7.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -184.1 kJ/mol |
| Pharmacology | |
| ATC code | V6CQA |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P280, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 37 °C |
| Autoignition temperature | 515 °C |
| Explosive limits | Lower: 10.5% Upper: 44% |
| Lethal dose or concentration | LD50 oral rat 160 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 160 mg/kg |
| NIOSH | SN2980000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 ppm (0.41 mg/m³) |
| IDLH (Immediate danger) | IDHL: 10 ppm |
| Related compounds | |
| Related compounds |
Acetonitrile Chloroacetonitrile Dichloroacetonitrile Bromoacetonitrile |

