Tributyltin Methacrylate: Science, Safety, and Sustainability
Roots of Tributyltin Methacrylate
Looking back at the late 20th century, chemists searching for better antifouling coatings for ships stumbled onto the unexpected power of tributyltin compounds. Shipowners faced a sticky problem—literally—when barnacles and algae weighed down hulls and sent fuel bills through the roof. Tributyltin methacrylate wasn’t the first compound in the tin family; it entered the scene after early organotin paints revolutionized marine shipping with efficiency few could ignore. Chemists soon realized that attaching a methacrylate backbone made this compound compatible with acrylic polymers, giving rise to stronger, longer-lasting coatings. A new era began, not so much out of ambition for chemical novelty, but from practical needs on the water and growing concern about wasteful fuel use.
What Makes Tributyltin Methacrylate Tick
Tributyltin methacrylate has a kind of dual personality. The molecule, part way between organometallic and organic chemical, features three butyl groups bonded to tin, with a methacrylate tail stuck on the end. This makes for oil compatibility and straightforward curing in resins—just right for blending into ship paints. Compared to old-school copper-based biocides, tributyltin methacrylate forms a stable bond with polymers, improving the overall resistance of coatings to water and impact. On the technical side, methacrylate monomers let it participate in standard radical polymerization, so manufacturers can control everything from drying time to final hardness.
The Nitty-Gritty: Preparation and Chemical Behavior
Making tributyltin methacrylate starts with tributyltin oxide or chloride and methacryloyl chloride, often catalyzed in a controlled environment. The synthesis isn’t complicated for an experienced hand and yields a thick, colorless liquid. As for its performance, the compound brings more than just antifouling kick; the bulky tin groups disrupt microbial membranes, leading to real-world results on ocean-going hulls. Once embedded in a polymer matrix, its release from the surface slows, extending the life of marine coatings compared to old organotin paints.
Real-World Names and Applications
Ship paint chemists and ocean engineers recognize tributyltin methacrylate by any number of names: TBT-methacrylate, tributyltin ester, or even just “tin-acrylate” in casual shop-floor talk. Sometimes technical papers call it “tributyltin 2-methylpropenoate.” It turns up everywhere from oil rig legs to fishing gear, wherever marine life lurks and corrosion threatens. For decades, shipyards treated it like magic paint, attributing smoother hulls and fuel savings to this single ingredient. Yet, the story does not stop with coatings. Laboratory scientists have tinkered with the monomer to develop antibacterial plastics, anti-microbial hospital surfaces, and more, though these uses trail far behind marine paints.
Inside the Lab: Properties and Reactivity
The physical character of tributyltin methacrylate sets it apart. It holds up against seawater, resists breakdown under sunlight, and flows easily enough for quick mixing. From personal experience in academic labs, its sharp, pungent odor fills the workspace, calling for careful handling. The tin-carbon bonds hold strong under heat, but aggressive oxidizers or acids threaten decomposition. With a methacrylate group on board, the compound jumps into addition polymerizations—making it a favorite for resin creators looking to tweak antifouling power as needed.
Safety Standards and Working Practices
Early users paid little attention to safety—until biologists started seeing snails and oysters vanishing from harbors coated in TBT paints. Scientists uncovered that tributyltin compounds caused imposex in mollusks, blurring the line between pest control and environmental disaster. Shipping regulators responded, leading to stricter protocols and outright bans in many countries. Today’s standards demand gloves, goggles, and local fume extraction at every stage. Disposal rules reflect the compound’s persistence; it isn’t something anyone can just dump down the drain. Shipyards and research labs found ways to substitute alternatives or enclose application procedures, reducing worker exposure and environmental leakage. A focus on education and monitoring remains high on the list wherever organotins appear.
Impact on the Ocean and Human Health
Evidence tying tributyltin methacrylate to marine ecosystem damage keeps stacking up. Studies warn that small concentrations shut down shellfish reproduction, threaten food webs, and weaken predator species. The “miracle” of barnacle-free hulls came at the expense of long-term ocean health. WHO advisories point to possible harm from chronic exposure, though the risk to consumers mostly comes from contaminated seafood. In the early 2000s, international agreements like the IMO’s International Convention on the Control of Harmful Anti-fouling Systems went into effect, forcing the shipping industry to pull back. This shift left a gap for chemists to fill—finding safe, effective replacements without replicating organotin toxicity.
Shifts in Research, New Directions
After years of blanket bans and phased withdrawals, researchers aimed at designing new antifouling agents inspired by tributyltin’s powerful properties but stripped of its environmental baggage. Some experiments focus on copper-free biocides, others try surfaces that shed fouling organisms without any chemical toxins at all. Polymer chemists experiment with locked-in organotin units, promising activity inside bulk plastics without leaching into water. Most marine paint manufacturers spent millions redesigning product lines. Attention has also shifted to remediation—using bacteria or fungi to strip tin from contaminated sediments, or advanced filtration to clean up shipyards. In my own years watching marine innovation, the field’s energy shifted away from quick fixes to long-haul, systems-based thinking.
Tomorrow’s Prospects: Beyond Organotin
With tributyltin methacrylate’s controversial legacy, its days in mainstream marine applications have dwindled. Researchers hold out hope for safer, biodegradable antifoulants, combining chemistry and biology to outsmart barnacle glue. Some see possible recycling of tin-based polymers as a way to honor past work, cleaning up the mess instead of ignoring it. Companies and environmental watchdogs now treat every new additive with deep skepticism, requiring long-term, open data on what happens as coatings weather and wear away. Innovation continues, just in a different spirit—less about speed to market, more about safeguarding ocean and human health for the long haul.
What is Tributyltin Methacrylate used for?
What Folks Really Use It For
Tributyltin methacrylate might sound like a mouthful, but it’s found a job in places far from a chemistry textbook. This compound shows up most often in the coatings used on ships and underwater structures. Ship owners and marine engineers don't use it because it’s cheap or flashy—they turn to it because marine growth creates real problems. Barnacles and algae don’t care about paint or fancy metals. Once they latch on, they bring drag, slow things down, and force ships to burn more fuel. Fuel bills go up, maintenance goes up, and small issues quickly pile up. Tributyltin methacrylate brings a hard stop to most of that by stopping critters from settling in the first place.
Nobody likes to scrape barnacles off the hull or schedule extra repairs. Before tributyltin methacrylate came along, paint seldom lasted long in the salty brine. Anti-fouling paints made with this compound changed the game in the 1960s and 1970s. Studies showed cargo ships cruised smoother, saving thousands in fuel costs. Container lines and navies around the world quickly took notice. Even small fishing boat owners saw the point: less time cleaning, more time earning.
Not All Sunshine and Smooth Sailing
Every good solution brings its own headaches. Tributyltin compounds are toxic—not just for barnacles, but for almost every living thing in the water. Science didn’t catch up right away. The signs started with damaged oyster beds and strange changes in snails and other shellfish. Slowly, rules changed. By the 2000s, most countries banned tributyltin-based products in marine paints. The United Nations’ International Maritime Organization called for a phase-out, citing both declining biodiversity and poisoned seafood supplies.
It’s tough to balance performance with responsibility. On one hand, tributyltin methacrylate cut waste from scraping hulls and kept goods moving around the world. On the other, untreated leaching left lasting scars on coastal economies and ecosystems. Coastal folks paid a price in lost income and health hazards. These aren’t distant, academic problems—they hit people right at home and at work.
What Does a Better Future Look Like?
I’ve seen shipyard workers relieved when paint jobs lasted longer. They don’t stand in the sun, scraping barnacles, as often. At the same time, I’ve watched oyster farmers worry when their harvests dipped for reasons no one could wrap their heads around, until science traced it back to toxic runoff. Tributyltin methacrylate is a classic case of progress needing a second look.
The good news: chemists now design paints with new biocides and even natural oils. Copper-based hull paints, slick silicon finishes, and herb-based blends step in for the old tributyltin formulas. These still block barnacles but do far less harm to helpful marine life. Regulators and industry need to stay sharp, watching for side-effects. Ship owners and operators should pay attention to what’s in their paint, and support research into new, less toxic methods. We don’t have one magic answer. Each step forward comes from weighing short-term fixes against long-term losses, then picking a path that helps both boat owners and those who depend on clean water.
Is Tributyltin Methacrylate hazardous to health or the environment?
What is Tributyltin Methacrylate?
Tributyltin methacrylate comes from the family of organotin compounds, often used in specialty coatings and polymers. Manufacturers once blended it into paints, mainly for boats, because it helps stop barnacles and other organisms from sticking to hulls. That sounds helpful, but the story doesn’t stop at clean boats.
Looking at Health Concerns
Tributyltin compounds, including methacrylate forms, raise red flags for toxicology experts. Studies point to their potential for causing immune system problems, liver damage, and even harm to reproductive health. After spending some time digging through databases and health advisories, I found that researchers link tributyltin exposure to hormone disruption. This can trigger changes in fish and shellfish, but also shows up in lab mammals. The risks extend to people working with the raw compound who breathe in fumes or touch residue. Headaches, respiratory problems, and nausea have been reported in manufacturing settings. Sometimes skin contact can cause irritation or allergic responses.
Environmental Impact Stands Out
Organotin chemicals became infamous for environmental persistence. Once tributyltin gets into water, it doesn’t just break down and disappear. It clings to sediment and collects up the food chain. Some of my friends in environmental science describe seeing shellfish deformities along harbors and coastal zones known for heavy boat traffic—places where paints with tributyltin saw regular use. Fish populations dropped, and mussel beds in some areas never bounced back fully after bans came into force. The World Health Organization and European Union have labeled tributyltin as a substance of very high concern because of these effects. Even low levels cause marine snails to develop male characteristics regardless of sex. This strange change, called imposex, leads to collapsing snail populations because breeding fails.
Why Prevention Matters
Chemical hazards don’t respect borders. Anyone who grew up near rivers or the coast might remember advisories warning against eating shellfish after chemical spills. I remember seeing warnings posted along the shore during the late 1990s. Local fishers worried about the effect on their catch and the future of their jobs. Public health agencies and environmental groups worked to monitor contamination and push for regulation. These experiences showed me that prevention is cheaper and safer than cleaning up later. Bans phased out tributyltin in some products, but legacy uses linger in old coatings and sediments. Kids, wildlife, and communities still face risks from past contamination washing out during storms or dredging projects.
Practical Steps Toward Safer Alternatives
Solving problems like this comes down to shared responsibility between industries, regulators, and consumers. Technology exists for safer marine paints. Some use silicone or copper-based alternatives, which, while not perfect, avoid the cascading effects of organotin pollution. Workers need strong safety protocols, including modern ventilation systems, personal protective equipment, and real training—not just paperwork. Governments can strengthen monitoring and enforce penalties for illegal use or dumping.
People deserve to know what goes into products around their homes and waterways. Full transparency, clear labeling, and smarter purchasing choices drive the shift toward safety. In my view, communities thrive when local leaders support green chemistry programs and invest in cleaning up polluted harbors. Tributyltin methacrylate serves as a cautionary tale—respect for science and real-world experience must guide decisions on industrial chemicals. Progress starts with facing risks honestly and acting together to protect health and nature.
What are the main applications of Tributyltin Methacrylate?
Understanding Its Impact in Marine Coatings
Any conversation in the shipbuilding world or the yachting circles eventually runs up against the challenge of barnacle and algae buildup. Tributyltin methacrylate (TBTMA) stepped onto the scene primarily to fight this ongoing headache. I’ve watched maintenance costs spiral for ship operators as hulls get slow, fuel bills climb, and downtime mounts. Paints with TBTMA changed the landscape back in the 1970s by keeping hulls smoother for longer. These coatings stopped marine life from sticking, slashing energy use and keeping ships in service, not docks.
During my stint working with a marina on the Gulf Coast, captains swore by the anti-fouling performance. The coatings kept hulls free of drag for entire seasons, and the difference often meant fewer scrapes and sand-downs midyear. Ship owners felt the savings, and I saw environmental groups raise concerns—it wasn’t just about performance; these compounds leached into the water and built up in food webs. With such a stark tradeoff, regulatory bodies responded, eventually limiting their widespread use.
Specialty Polymers and Industrial Resins
TBT methacrylate doesn’t stop at hulls and propellers. Manufacturers use it to tweak polymer structures, adding a distinctive edge to certain plastics and resins. In labs focused on specialty coatings, this compound adds stain resistance and weatherability that doesn’t just fade. Product engineers want surfaces that shrug off dirt or withstand tough environments, particularly in industries like architecture or heavy equipment. This knack for chemical durability translates to products lasting through harsher seasons or more abrasive treatment.
In my circle, researchers once pursued TBTMA as an option for water-resistant coatings on critical bridge parts, where rust and wear would otherwise bite hard. Results varied; performance often impressed, but the environmental persistence returned as a sticking point over time.
Pressure from Regulations and the Search for Safer Alternatives
Governments and watchdogs pushed back once evidence stacked up around toxicity. I remember heated town hall meetings near marinas, where fishers and scientists handed out brochures showing the ecosystem effects: snail populations tumbling, shellfish harvests shrinking. Eventually, most rich countries placed strict limits or outright bans on TBTMA in marine paints. Watchdogs pointed to long-lasting residues—the compound doesn’t drop out of a food chain quickly.
Innovation thrives when up against a wall, and the coatings industry hustled to fill gaps. Silicones, copper-free antifoulants, and biocide-release systems grabbed market share. Green chemistry is a constant work in progress; replacements aim for fouling control without polluting stretches of shoreline for years after application.
Learning from the Journey of TBTMA
Experience with TBTMA teaches a hard lesson: high performance usually comes with high responsibility. I’ve seen product teams lean into environmental testing protocols and adopt life-cycle analysis, not to check regulatory boxes, but to understand where the next “miracle chemical” might backfire. Stakeholders—from manufacturers to end users—deserve transparency about what goes into paints, plastics, or coatings. Clear labels and thorough research help the broader community make informed choices.
While TBTMA still has a niche in some tightly controlled settings, its story underscores the need for continuous improvement, not only in chemistry but also in stewardship. Lessons learned fuel the drive for safer innovation and better environmental safeguards in every new generation of materials.
How should Tributyltin Methacrylate be stored and handled?
Why the Way We Store Chemicals Matters
Anyone who’s stepped into a lab even once learns quickly—storage isn’t just about shelf space. Tributyltin Methacrylate is a mouthful, and there’s nothing casual about it. It’s one of those chemicals you don’t want leaking, spilling, or left in a sunlit window. Scientists around the world have flagged its toxicity, particularly its impact on aquatic organisms. Accidents with this stuff can have ripple effects running all the way from the lab sink to riverbanks far beyond.
Straightforward Storage Rules
Basic rules aren’t just academic—they’re the kind of knowledge you keep in your pocket because you know what a headache clean-up becomes. Tributyltin Methacrylate calls for storage in tightly closed containers. Once air creeps in, there’s a higher chance the chemical degrades and dumps unwanted fumes. The label must always face outward. Half the safety comes from knowing what’s inside each bottle, especially when you’re pulling a late night or swapping shifts.
Heat is another silent enemy. Keep this compound away from furnaces, heaters, or even that window with afternoon sun blasting through. Chemical stability hangs on reasonable temperatures, so a cool, shaded cabinet is the best place. Even labs working in sweltering areas can make use of AC units. If space comes at a premium, prioritize the risky stuff—like Tributyltin Methacrylate—for your coolest shelf.
Flammable liquids belong far away. This isn’t just regulation. One spark from a volatile neighbor, and you’re looking at a chain reaction, not just a bigger mess. Even in a pinch, don’t stack incompatible bottles together just because the shelf is empty.
Handling: Less Is More
I learned the hard way that limits on handling protect everyone. Pouring or transferring should happen with gloves, goggles, and a working fume hood overhead. Don’t lean on hasty workarounds—nothing ruins a day like splashing skin with something described as “hazardous upon contact.” I once saw a colleague try to wipe up a drip with a standard paper towel—bad idea. Dedicated chemical-resistant materials and a spill kit close by make all the difference.
After each use, seal the bottle right away. Don’t walk off for a coffee break with it open—exposure to air, dust, or curious hands spells trouble. Wipe down threads to keep the cap from gluing itself shut or letting residue escape.
Labeling, Training, and Emergency Prep
Every bottle needs clear, visible labels with handling and hazard details. I’ve seen new staff skip this step since it feels redundant when everyone “already knows.” It only takes one new technician or a distracted day for a mistake to happen. Updating Safety Data Sheets in visible spots and running refreshers every season builds the kind of safety culture that catches small problems before they snowball.
Labs thrive by staying organized and watchful. Setting up emergency eye wash stations, restocking gloves, and doing monthly spill response drills might sound dull, but nothing feels as reassuring in a real emergency. The real lesson? Don’t trust luck—routine habits save time, money, and sometimes, people’s health.
What are the safety precautions when working with Tributyltin Methacrylate?
Why Safety Matters Here
Anyone spending time around paints, plastics, or ship coatings has bumped into tributyltin methacrylate. It helps fight marine growth, which keeps ships running smooth and fuel costs down. Trouble comes with its power. Tributyltin compounds can cause serious harm, both to people and to the environment. Hands-on experience in industrial labs taught me how easily small slips with chemicals like this turn into big regrets. Awareness and solid habits keep damage at bay.
Handling with Proper Protection
Gloves, goggles, and lab coats represent the basics. Splashy stories about minor skin contact leading to rashes or blisters pop up too often in workrooms. But there’s more—nitrile gloves beat out latex here, as tributyltin chews through the wrong materials. Safety goggles need a snug seal; this compound stings eyes badly. Lab coats protect your usual clothes, making sure you’re not tracking contamination home. Respirators step up whenever you handle powder or strong fumes because the vapor can do real harm to lungs and nervous systems. Poor air quality in tight workspaces builds up exposure fast. That lesson sticks if you’ve ever worked overnight in a cramped shop and ended the morning light-headed.
Don’t Let Spills Spread
Chemical spills always turn into a scramble if you don’t plan ahead. Keep spill kits handy—absorbent pads, neutralizers, and safe disposal bags help contain messes in seconds. It’s no exaggeration to say a student in our old lab once cleaned up a couple ounces without proper gear and missed class for a week. Good ventilation matters, too. Fume hoods and exhaust fans eat up vapors before they hang around. Make it a habit to triple-check work surfaces and tools for residue before and after each use.
Waste and Environmental Care
Throwing out tributyltin like household trash isn’t just illegal—it’s dangerous. This chemical lingers in water and soil for years, hitting shellfish and other marine life hardest. Workplaces I’ve seen take waste seriously, collecting all leftovers and contaminated gear in properly labeled barrels. Certified waste companies pick up the barrels to process with special incinerators. Quick rinses don’t cut it; those end up in the pipes and flow straight to rivers. Training everyone on these steps helps more than any rulebook alone. Knowing where the waste goes and why it’s dangerous keeps shortcuts in check.
Health Checks and Training
Companies that see safety as real, not just red tape, run training every few months. People learn to spot red flags for early poisoning—loss of balance, skin tingling, headaches—and know who to call. Doctors may do regular blood checks to catch problems before they grow. Honest reporting of small accidents gets everyone thinking about prevention, not blame. Some of the best places I’ve worked bake these habits into the culture, not just the manuals. This brings trust, and trust keeps people open about mistakes and near misses.
Building a Culture That Cares
Tributyltin methacrylate does useful work but only in skilled hands. Knowing the risks first-hand, I don’t take shortcuts. Personal experience proves strong habits, real teamwork, and a respect for the chemical keep people safe day after day. Staying alert, talking openly, and updating safety steps as we learn more gives everyone the best chance at health.
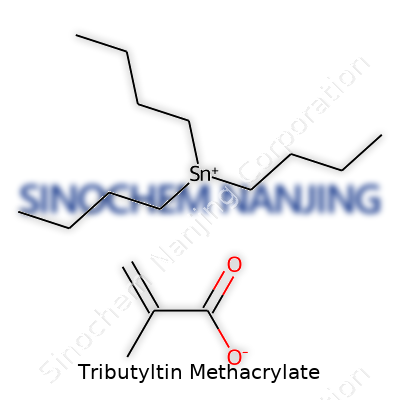
| Names | |
| Preferred IUPAC name | tributylstannyl 2-methylprop-2-enoate |
| Other names |
Methacrylic acid, tributylstannyl ester Methacrylic acid tributyl tin ester TBMA Tributylstanninyl methacrylate |
| Pronunciation | /traɪˈbjuːtɪl.tɪn ˌmɛθ.əˈkrɪl.eɪt/ |
| Identifiers | |
| CAS Number | 2155-70-6 |
| Beilstein Reference | 1722966 |
| ChEBI | CHEBI:34579 |
| ChEMBL | CHEMBL444213 |
| ChemSpider | 158965 |
| DrugBank | DB14005 |
| ECHA InfoCard | 01c0c0b0-7eb3-4c8c-9383-09e8174b3a15 |
| EC Number | 215-175-9 |
| Gmelin Reference | 78725 |
| KEGG | C18166 |
| MeSH | D031506 |
| PubChem CID | 156726 |
| RTECS number | WN0175000 |
| UNII | UB857H60ZA |
| UN number | UN2527 |
| Properties | |
| Chemical formula | C19H36O2Sn |
| Molar mass | 419.19 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Mild characteristic |
| Density | 1.19 g/cm3 |
| Solubility in water | Insoluble |
| log P | 2.78 |
| Vapor pressure | 2.4 x 10^-3 mmHg (20 °C) |
| Acidity (pKa) | 13.3 |
| Basicity (pKb) | 13.96 |
| Magnetic susceptibility (χ) | -7.38 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.474 |
| Viscosity | 1.38 mPa·s (25 °C) |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 619.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −554.91 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H226, H301, H314, H410 |
| Precautionary statements | P261, P264, P273, P280, P302+P352, P305+P351+P338, P312, P321, P330, P391, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-2-2-W |
| Flash point | Flash point: 114°C |
| Autoignition temperature | 250 °C |
| Lethal dose or concentration | LD50 (oral, rat): 105 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 250 mg/kg |
| NIOSH | GJ9130000 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Tributyltin chloride Tributyltin oxide Tributyltin acetate Tributyltin fluoride Tributyltin benzoate |

