Tributyltin Laurate: Legacy, Chemistry, and Responsible Use
Historical Development
Tributyltin laurate did not enter the chemical industry overnight. Decades back, chemists were already experimenting with organotin compounds, looking for agents that could bring new life to plastics and coatings. By the 1960s and 70s, tributyltin derivatives earned a reputation as performance catalysts and stabilizers, especially as polymers and industrial paints demanded chemicals that offered strength and resilience. In the early days, the incentive came from manufacturing—factories needed compounds that would make their products stretch further, fight off fungal rot, and last longer against weather and use. Scientific advancements soon carried tributyltin laurate into specialty applications where old stabilizers failed to keep up. Over the years, this compound carried both benefits and baggage, with its real-world impact stretching beyond factory walls into environmental debates and new regulations.
Product Overview
Tributyltin laurate stands out as one member of a family of organotin compounds. People in the plastics industry know it for its ability to stabilize polyvinyl chloride (PVC), extending shelf life and making finished products more reliable. Unlike generic additives, this compound provides a specific interaction with chlorine atoms in plastics, cutting down on discoloration, brittleness, and breakdown. Beyond PVC, tributyltin laurate found places in antifouling paints, wood preservatives, and other protective coatings. Manufacturers value it for efficiency—using small amounts can make a significant difference in product stability.
Physical & Chemical Properties
You won’t find tributyltin laurate hiding out as an unremarkable powder. It shows up in commercial shipments as a clear to slightly yellowish liquid, with a faint but unmistakable odor. Chemically, it carries a tin (Sn) atom bonded to three butyl groups and one laurate chain. Its molecular formula is C27H56O2Sn. The laurate part derives from lauric acid, bringing a fatty acid chain to the structure. This setup makes the compound soluble in organic solvents but not in water. Compared to other stabilizers, tributyltin laurate works at lower doses and delivers results quickly during processing. Typical melting and boiling points aren’t the primary concern, as the compound remains stable under regular storage and usage conditions. Density lands at around 1.07–1.10 g/cm³, and it tends to resist decomposition until hitting temperatures above 250 degrees Celsius.
Technical Specifications & Labeling
Chemical suppliers provide tributyltin laurate according to tight technical standards. Purity levels often exceed 95%, with clear documentation of residual tin, impurities, and moisture content. Packaging follows hazardous material guidelines. Drum labels list the UN number (UN 2788), hazard statements for toxicity, and stipulate usage must follow environmental regulations. Anyone handling this product in a warehouse or laboratory knows that its classification as a harmful chemical is not up for debate—sheeting, gloves, goggles, and safe ventilation are not optional. The safety data sheet sits right alongside each delivery.
Preparation Method
Making tributyltin laurate starts with tributyltin oxide and lauric acid, two well-known industrial chemicals. During synthesis, producers combine tributyltin oxide with lauric acid in a solvent like toluene. The mixture heats up, and a reaction swaps the oxygen atoms in tributyltin oxide with the laurate group. Water forms as a byproduct, and removal of water helps drive the reaction to completion. Filtration and distillation clean up the mixture, leaving a viscous, concentrated liquid. Quality control at this stage makes a difference—impurity levels influence final performance in plastics and coatings.
Chemical Reactions & Modifications
The chemistry behind tributyltin laurate attracts researchers interested in organic and organometallic reactions. The tin atom forms a strong bond with the laurate group, but under harsh conditions, these bonds break down. Hydrolysis in acidic or basic media produces tributyltin hydroxide and lauric acid. Exposure to UV light or strong oxidizers can also break apart the molecule—a point of concern for stability in outdoor or high-stress environments. Over the years, chemists tinkered with the structure to tweak properties, swapping the laurate group with others for different performance in specialized applications. As technology changes, so do the targets for modification.
Synonyms & Product Names
Tributyltin laurate goes by a few names in the market. Some suppliers call it lauric acid, tributyltin ester. Others stick to the IUPAC nomenclature—tributyl(laurato)stannane. Commercial brands sometimes shorten the name or embed it within a line of stabilizers. CAS number 3091-25-6 consistently tracks the substance across regulatory filings and international shipments. Chemists might just call it TBTL in technical circles or research notes.
Safety & Operational Standards
Tributyltin compounds don’t get a free pass. Industry veterans know their toxicity profile from years of experience and increasingly strict oversight. Safety regulations require robust personal protective equipment and detailed standard operating procedures. Chemical plants and research labs must install exhaust ventilation and monitor airborne concentrations. Waste collection and disposal systems must eliminate any chance of runoff or accidental release. Safety standards extend through the whole chain—production, transport, storage, and end-use. Ignoring these measures risks both individual health and environmental consequences.
Application Area
Most tributyltin laurate heads into PVC manufacturing plants, blended in to preserve strength and color during both processing and long-term use. It is particularly useful for wire insulation, injection-molded products, and some specialty films. Besides plastics, antifouling paints for ships and marine structures took advantage of tributyltin laurate because of its biocidal effect—it disrupts the growth of algae, barnacles, and other marine life. Restrictions in recent years pushed coatings producers to consider alternatives, but historic applications still echo in legacy infrastructure and shipping.
Research & Development
Scientific attention continues to track tributyltin laurate, with teams studying both its performance in plastics and its environmental behavior. Adjusting the base chemistry for lower toxicity and improved degradation remains a top challenge. Environmental chemistry labs monitor breakdown products and study their movements in soils and water. University polymer labs sometimes rebalance formulations to reduce dependency on organotin compounds. Ongoing research looks at less hazardous tin stabilizers, biobased alternatives, and advanced recycling methods to keep plastics durable while cutting down risks.
Toxicity Research
Toxicologists studied tributyltin laurate over several decades, prompted by incidents of pollution near manufacturing sites and reports of marine disruption. This substance can cross biological membranes, and animal studies show its ability to harm aquatic life at very low concentrations. Organs like the liver and immune systems are especially vulnerable. Regulators in Europe and other regions responded with severe limits and outright bans in ship paints and agricultural coatings. The scientific record reflects ongoing concern—testing for worker exposure, environmental contamination, and ecosystem impacts does not let up. Safety recommendations focus on well-ventilated working spaces, sealed processes, and specialized disposal.
Future Prospects
The world can’t turn the clock back on tributyltin laurate, but change is gathering pace. Many industries now look at biobased stabilizers or metal-free alternatives. Research into "greener" organotin compounds continues, but long-term prospects depend on finding additives that can match performance with a fraction of the health risks. Some PVC manufacturers pivoted to calcium-zinc additives or new blends that keep plastics tough and colorfast with fewer environmental tradeoffs. Shipping and coatings industries press for non-toxic antifoulants that protect marine structures without jeopardizing aquatic ecosystems. Each solution means tradeoffs—in cost, performance, and compatibility. Stakeholders want regulators, chemists, and industry leaders to keep working together, putting health and sustainability on the same level as durability and profit.
What is Tributyltin Laurate used for?
Getting to Know Tributyltin Laurate
Tributyltin laurate often pops up in conversations about chemicals in industry and environmental safety. It’s a compound with some big roles, mostly tied to its ability to stop things from growing where they shouldn't. The main reason manufacturers reach for it comes down to its antifungal and antibacterial punch. This property made it a favorite for coatings, especially in places where moisture, bacteria, and fungi could spell trouble.
Common Uses—Paints, Plastics, and Beyond
Take a look at the hull of an old ship or the pipes snaking under farmland. Tributyltin laurate has been around to keep barnacles and unwanted organisms from sticking to ships, and to keep algae and fungus in check in water systems. Boat paints, some plastics, and even some industrial textiles got dosed with the stuff for its strong biocidal effect.
The story doesn’t stop at boating. Manufacturers use this chemical in making certain PVC products. It works as a stabilizer during the production of vinyl, helping to extend the life of products like cables, flooring, and roofing. If you grew up with vinyl tiles underfoot, tributyltin laurate might have helped them shrug off mold and mildew for years.
Concerns on Environmental and Health Fronts
There’s a catch here, and it’s a big one. Research since the 1980s highlights the dangers this chemical brings to waterways and wildlife. Scientists from institutes like NOAA and groups like the World Health Organization flagged tributyltin compounds as particularly toxic to marine life. Tiny doses can turn the tide for entire animal communities, causing reproductive problems in fish and shellfish, and they stick around in the environment much longer than most folks think.
Human health enters the mix, too. Prolonged exposure links with problems ranging from skin and eye irritation to more serious issues involving the nervous system and hormone function. Seeing these risks, many countries banned or heavily restricted its use, especially in marine paints. The European Union and the United States led the way, pushing industries to seek safer alternatives.
Chasing Alternatives and Solutions
So where does that leave industries? Shifting away from tributyltin laurate takes effort and investment, but the tide is slowly turning. Safer antifouling agents, made from copper or silicon-based materials, started replacing tributyltin in ship coatings. Vinyl producers looked at tin-free stabilizers, and modern biocides now aim to focus their action where needed, breaking down faster in the environment.
Spotting green-certified labels or products from manufacturers committed to responsible chemical use now matters more than ever. It helps to ask questions and demand transparency. Every time a builder chooses materials free from persistent pollutants, it means fewer toxins find their way into waterways or our homes.
Battling pollution starts with knowing what ends up in products all around us. Chemicals like tributyltin laurate show how old solutions can linger beyond their welcome. Keeping up with the science, pushing for tougher safety reviews, and supporting better options drive real change in industries and help protect communities.
Is Tributyltin Laurate hazardous to health or the environment?
What Tributyltin Laurate Actually Means in Daily Life
Tributyltin laurate pops up in more places than most people expect. Boat paints, wood treatments, and plastics sometimes rely on this chemical to fight off fungus and bacteria. The promise of keeping things clean and mold-free sounds good from the outside. Walking around a boatyard, the whiff of chemical protectants hangs in the air, and folks rarely ask questions about what’s floating in that scent. Years ago, I didn’t blink twice at the slick, bright finish they left behind. Over time, I started reading more labels—and the story got complicated.
Why Scientists and Regulators Raise Red Flags
It’s not hype. Research from groups like the European Chemicals Agency points out tributyltin compounds don’t just wash away. They stick around in water and soil and don't break down easily. The chemical builds up in fish, mussels, and other marine life. An old neighbor who worked as a fisherman told me about deformed shells on oysters they pulled up near painted hulls. Turns out, tributyltin-based paints got blamed for stunted growth and weird shell shapes throughout coastal communities.
By the late ‘90s, international watchdogs started clamping down. The United Nations flagged high toxicity for aquatic life. The pesticide breaks through skin, disrupts hormones, and damages immune systems. These aren’t just headline-chasing statements. Studies in environmental toxicology journals provide data showing links between these chemicals and birth defects in snails, as well as thyroid and liver disruption in mammals.
Impacts on People—Not Just Sea Creatures
Calling tributyltin laurate an “environmental hazard” only tells half the story. Indoor environments matter, too. Wood preserved with tributyltin winds up in homes and sheds. While sanding down planks salvaged from a marina project, I noticed nose irritation and raw skin after a few hours. At the time, no one on the job site wore gloves or filtration masks. Only later did I discover workplace safety guidelines recommend skin protection and good ventilation for even small-scale exposure.
The main concern goes deeper: tributyltin laurate messes with hormone systems. According to findings in Environmental Health Perspectives, just small doses can disrupt the reproductive system in humans and animals. These compounds can cross into the food chain, especially in seafood and even in livestock living on contaminated land.
Real Changes to Limit Harm
More countries are banning or restricting tributyltin laurate. The International Maritime Organization stopped ships from using paints made with it. Many states require safer wood preservatives and label marine coatings more clearly. Some hardware stores stopped offering products containing tributyltin after local activism.
Transparency counts. Folks deserve to know if a deck stain or hull paint contains harmful chemicals. Labels need plain language about health risks, not just technical terms buried in the fine print. Authorities must enforce these rules, not just at the docks, but in hardware aisles and online stores.
Safer options exist on the market, from copper-based paints to natural wood sealants. Alternatives take patience; switching requires time for research, adjustment on worksites, and sometimes a bit more money up front. Still, the long-term health and environmental gains make it worth the trouble, especially after watching the slow but real recovery of shellfish beds once restrictions kicked in.
What are the storage and handling requirements for Tributyltin Laurate?
Why This Chemical Demands Care
Tributyltin laurate shows up in a long list of applications—antifouling paints, some industrial sealants, and stabilizers for plastics. It’s powerful, but it doesn’t just quietly help with your project. The compound brings serious risks for human health and the environment, which makes how you store and handle it a big deal.
Physical and Chemical Traits
In the lab, tributyltin laurate presents itself as a colorless to pale yellow liquid. There’s a relatively low volatility, but its toxicity profile stands out. It can absorb through the skin and can’t be trusted in environments with casual safety measures. A bit of mishandling can mean big trouble.
Storage Practices That Matter
I’ve seen chemicals stored all sorts of ways—old sheds, corner cabinets, and high-tech lockers. For tributyltin laurate, none of the casual routes cut it. Keep it locked away in a cool, dry, and well-ventilated spot. Direct sunlight and temperature swings can affect stability. Use corrosion-resistant shelves: stainless steel or chemical-grade plastic. Every drum or bottle should have a tight seal, labeled not just with a name, but with hazard warnings that anyone can figure out. Store away from acids and oxidizers, because even small leaks or splashes can start nasty reactions. An accident here isn’t just a mess—it’s an immediate containment crisis.
Personal Experiences with Handling
I’ve worked with people who thought gloves alone would do the job with organotins. It’s never that simple. Tributyltin laurate needs full personal protective equipment: nitrile gloves, splash-proof goggles, long sleeves, and, if splashing is possible, a face shield and chemical apron. Chemical fume hoods make a dramatic difference. Spraying, heating, or mixing out in the open sends vapors and fine droplets right into the work area, which puts skin, lungs, and eyes on the frontline. Even low exposure leaves lasting impacts. People have reported headaches, skin burns, and chronic irritation in barely-ventilated shops.
Disposal and Disaster Preparation
Tributyltin compounds don’t break down quickly in the environment. Environmental Protection Agency guidelines put them near the top for regulation. For small spills, cover the area with absorbent material designed for organics, scoop it up with non-sparking tools, and store cleanup waste in a labeled hazardous waste drum. Don’t think ordinary drains or trash bins will work—municipal systems can’t handle this stuff. Every storage site should keep spill kits nearby and conduct yearly safety drills. Even in small shops, folks benefit from drills where they actually use the spill kits. Safe disposal runs through licensed hazardous waste handlers, and paperwork backs up every container that leaves the site.
Safer Workplaces and Health Impacts
The science on organotin toxicity remains solid: repetitive exposure links to immune suppression, developmental issues, and hormone disruption. The industry pushes for safer substitutes, but for groups stuck with tributyltin laurate, training is a game-changer. Regular refreshers can keep new team members and veterans out of harm’s way. Real stories and hands-on training trump binders of written procedures. Clear communication builds a sort of culture where people don’t put “just a little” chemical in an unsafe place because they ran out of room elsewhere.
Looking Ahead
If you work with tributyltin laurate, set the tone by demanding secure storage, consistent labeling, and sharp attention to personal safety. These steps cut risks way down and make workplaces healthier for everyone involved. Setups that skip these details set up workers—and their communities—for tough lessons later on. Respect for the dangers gives everyone a better chance of staying healthy at the job.
What are the physical and chemical properties of Tributyltin Laurate?
Tributyltin Laurate: More Than Just a Compound
Tributyltin laurate grabs attention for good reason. Its applications in industrial chemistry stretch across fields like plastics, antifouling paints, and even biocidal formulations. Anyone who works hands-on with plastic production, or tries to keep marine equipment free from barnacles and algae, has come across it either directly or through product labels. On the surface, it gives off an oily vibe, gleaming with a faint yellow color. This is not something to splash around or dismiss. It carries a distinct odor, one that reminds careful users of its presence and potency, especially in confined spaces.
Physical Properties That Matter
Tributyltin laurate shows up as a liquid at room temperature. Its melting point sits below the normal room temperature, so nobody handles it as a solid in day-to-day workflows. Pouring it feels like handling a light oil. That makes it easier for manufacturers to mix it into PVC or paint bases without too much prep. Its density sits around 1.05 to 1.10 grams per cubic centimeter. Having spent time around chemical containers, I know this leads to a bottle that feels hefty for its size, giving some clue about its robust molecular makeup.
Solubility tells a different story. Drop it in water and nothing exciting happens—it refuses to dissolve much at all. That explains why it’s so tough on marine life but doesn’t wash away with the tides. Still, put it in something organic, like toluene or ethanol, and it mixes in smoothly. It clings to nonpolar surfaces and resists breakdown, which is both a perk and a curse. Shipping and storage always demand real care because any spills cling to surfaces and stick around.
Chemical Properties: Reactivity and Stability
Looking deeper, the heart of tributyltin laurate comes from its structure: a tin atom bonded to butyl groups and a laurate tail. That tin-carbon backbone makes it fiercely stable. With experience in the lab, it’s clear why industry values this trait. Once in a product, it won’t give up its place easily, keeping its antimicrobial punch for years. But it reacts to light and strong acids or bases, so nobody leaves it unprotected in sunlight or in open chemical baths. I’ve seen what heat or unexpected contamination can do—breakdown products are a headache, sometimes even more toxic than the original compound.
Burning tributyltin laurate in a fire releases tin oxides and organic fragments, so every warehouse with this compound keeps large warning labels and foam extinguishers nearby. A chemical spill on a workbench calls for full protective equipment. It can irritate skin on contact, and breathing in its vapors over time harms health. Literature and safety data confirm organotin compounds like this tend to accumulate in living organisms, causing trouble in aquatic food chains.
Safety, Regulation, and Responsible Use
Several global organizations have kept a close eye on tributyltin laurate. The International Maritime Organization and local governments have banned or restricted its use in paints and coatings to curb its environmental footprint. Studies from the 1970s and 1980s revealed that even low concentrations disrupt endocrine systems in marine snails and fish. Efficient alternatives get more attention these days, with scientists and engineers focusing on less persistent and less toxic compounds.
Despite strong regulation, tributyltin laurate keeps popping up where industrial performance outweighs ecological risk. I’ve seen factories implement closed systems, recycling used solvents, and adopting real-time leak detection sensors. These steps cut down on accidental exposure and limit the amount that escapes into waterways. Still, safe storage, strict handling protocols, and ongoing worker training carry the most weight in protecting people and ecosystems.
What are the safety precautions when working with Tributyltin Laurate?
Understanding the Risks in Real Life
Tributyltin laurate turns up in specialty chemistry labs and certain manufacturing shops. Its toxic reputation isn’t for show. The compound can cause real harm if it gets in your system. The science outlines it as a heavy-duty skin and eye irritant, and toxic if inhaled or swallowed. Workers who don’t respect it may face nerve problems, headaches, or worse. It can get under your skin—literally, because it absorbs right through.
Growing up around people who took on rough jobs at plants, I saw how easy it was to take shortcuts. Maybe gloves seemed too bulky or goggles fogged up in summer. But stories of colleagues out sick for weeks after chemical exposure got people paying attention. Hospitalizations for chemical burns or breathing issues aren’t rare when safety slips.
The Real Gear—Not Just Recommendations
Sturdy personal protective equipment always beats a warning label. Rubber or nitrile gloves, chemical goggles, and a face shield protect you from splashes or spills. Long sleeves and full-length pants keep it from brushing your skin. Make sure the gloves fit right—loose cuffs invite mistakes.
A good respirator means more than fresh air. Most open spaces don’t filter out vapor or fine droplets. Work with it in a fume hood or a well-ventilated spot. Dust masks from the hardware store won’t cut it; you want a cartridge rated for organic chemicals. Nose and mouth protection can’t be skipped for even a quick pour.
Simple Habits That Prevent Pain
Keep work areas tidy and wash hands often. It isn’t enough to just wipe your fingers on a rag. Running water, basic soap, and regular breaks keep chemicals from building up on skin. Never use solvents like acetone to clean up—those can drive the compound deeper into your body.
Containers should get labeled right away. Never trust a memory or a sticky note. Double-check lids and caps after every use. Take a minute to set up spill trays, and keep emergency eyewash and a shower nearby. Quick action in the first minute after a splash matters more than anything.
Stay Ahead of Emergencies
Training pays off. At my last job, new hires spent hours walking through worst-case scenarios with real gear—not just slideshows. Having a routine, practicing an eyewash or spill drill, and memorizing the placement of exit doors makes the difference when things turn bad in a hurry. Know the emergency numbers by heart or taped near the phone.
Waste disposal can’t be a back-burner task. Used rags, gloves, and containers shouldn’t land in a regular trash can. Talk to supervisors or chemical management teams about the legal steps in your area. Environmental damage can ruin lives outside the lab; keeping toxins out of the water or landfill is everyone’s job.
Keep Watching Each Other’s Backs
Nobody gets it right every day. But calling out risky moves, loaning an extra set of gloves, or reminding someone to secure a cap can stop an injury. Many of the worst workplace accidents happen on easy jobs, not the big projects. Working with something as unforgiving as tributyltin laurate, attitude matters as much as knowledge. Trust your senses, respect small warnings, and walk away from distractions. In the end, everyone goes home safer that way.
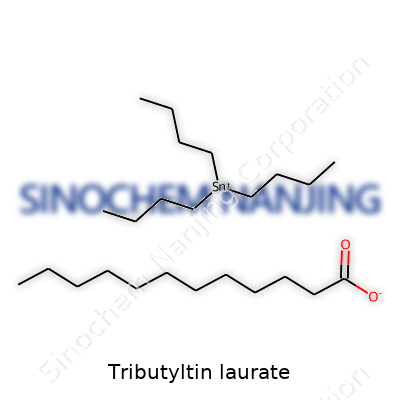
| Names | |
| Preferred IUPAC name | tributylstannyl dodecanoate |
| Other names |
Lauric acid tributylstannyl ester Tributyltin dodecanoate TBTL |
| Pronunciation | /traɪˈbjuːtɪl.tɪn ˈlɔːr.eɪt/ |
| Identifiers | |
| CAS Number | 3091-25-6 |
| Beilstein Reference | 1461213 |
| ChEBI | CHEBI:34916 |
| ChEMBL | CHEMBL3332289 |
| ChemSpider | 3249200 |
| DrugBank | DB16653 |
| ECHA InfoCard | 03c9d7f8-0dea-47a7-82b8-347a0c64b372 |
| EC Number | 247-759-0 |
| Gmelin Reference | 81815 |
| KEGG | C19201 |
| MeSH | D014266 |
| PubChem CID | 25262799 |
| RTECS number | WH6780000 |
| UNII | LUO8I60NAM |
| UN number | UN2922 |
| Properties | |
| Chemical formula | C39H80O2Sn |
| Molar mass | 661.55 g/mol |
| Appearance | Colorless liquid |
| Odor | characteristic |
| Density | 1.05 g/cm3 |
| Solubility in water | Insoluble |
| log P | 4.92 |
| Vapor pressure | <0.0001 hPa (20 °C) |
| Basicity (pKb) | 6.2 |
| Magnetic susceptibility (χ) | -74.0e-6 cm³/mol |
| Refractive index (nD) | 1.488 |
| Viscosity | 30 mPa.s (25°C) |
| Dipole moment | 2.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 887.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | V09CX03 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330, H314, H317, H410 |
| Precautionary statements | Precautionary statements of Tributyltin Laurate: "P201, P202, P260, P264, P270, P271, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P311, P321, P330, P362+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2-W |
| Flash point | 110°C |
| Autoignition temperature | 250°C |
| Lethal dose or concentration | LD50 oral rat 224 mg/kg |
| LD50 (median dose) | LD50 (median dose): 118 mg/kg (oral, rat) |
| NIOSH | TYF |
| PEL (Permissible) | PEL: 0.1 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
Tributyltin oxide Tributyltin chloride Tributyltin acetate Tributyltin fluoride Tributyltin nitrate |

