Tributyltin Chloride: Past Legacy, Present Use, and Future Responsibility
Looking Back: How Tributyltin Chloride Shaped Industrial Chemistry
Before the world started paying close attention to persistent organic pollutants, tributyltin chloride worked its way into industrial use with impressive speed. Chemists first brought it into the spotlight decades ago, tapping into its knack for controlling growth in the aquatic world. Shipyards valued its antifouling power for keeping barnacles and algae off hulls, which made sea travel more efficient and lowered maintenance costs. The idea seemed almost too good: protect global shipping and fishing from nature’s relentless quest for footholds. Throughout the twentieth century, as production scaled up, tributyltin chloride slotted neatly into paint formulations, wood preservatives, even some plastic manufacturing. Its legacy as a marine biocide rests on a foundation poured during these growth years, setting the stage for both benefit and unintended consequences.
Physical and Chemical Notes: What Makes Tributyltin Chloride Tick
No one in the lab can ignore tributyltin chloride’s distinctive smell and oily appearance. This organotin compound, a colorless to pale yellow liquid, proves soluble in organic solvents like benzene and chloroform. Its melting and boiling points come in handy for separation and storage: the compound doesn’t break down easily, which is both a benefit for industrial life and a sticking point for environmental health. Its molecules blend organic and metallic characteristics — a tin atom at the center, surrounded by three butyl groups and a single chlorine. That chemical structure delivers the toxicity that once seemed so valuable, selectively harming invertebrate larvae in a way that lessened maintenance for ships and treated timber. Its stability presents a practical advantage, only now seen as trouble as tributyltin resists breakdown in soil and water.
The Craft of Making Tributyltin Chloride and Its Transformations
Most commercial processes rely on reacting butyl chloride with metallic tin, using catalysts and specialized reactors. The result: a highly concentrated organotin ready for formulation into antifouling paints or pesticides. Chemists tweak it through reactions that swap out chlorine for other functional groups, or stretch the carbon chains for different properties. Each modification gives the chemist a tool for targeting fungi, bacteria, or marine pests. In the lab, tributyltin chloride also acts as a precursor for other organotin compounds used in organic synthesis — from stabilizers for PVC to intermediates in pharmaceuticals. While modification opens many technical doors, each new product line means another layer of scrutiny for toxicity and persistence.
Technical Details and Labeling: Navigating a Sea of Regulations
Lately, labeling rules have sharpened in response to toxicology findings. Manufacturers and transporters must mark containers for hazardous marine pollutants, with strict limits on workplace exposure. Safety data sheets no longer lurk at the back of filing cabinets — they walk into lab meetings and training sessions, underlining risks in bold print. I’ve seen first-hand how a solid culture of safety hinges on clear, honest labeling, especially in academic and industrial settings where new researchers may not recognize the slim line between scientific curiosity and real-world harm. Not every country enforces the same standards, but the safety bar keeps rising as the public and regulators push for tighter control, especially near water.
Synonyms and Side-Street Naming: The Risk of Confusion
Scientific circles and supply chains often toss around names like TBTCl and tributyltin(IV) chloride. Drop the abbreviation at a shipping dock or a warehouse, and someone will ask: is this the same stuff as tri-n-butyltin chloride or TBTC? Short answer: yes, all these names point back to the same chemical players. The tangle of synonyms underscores a problem that has cropped up in international trade and accident investigations. I once heard about a lab tech in Europe who received the same substance under two labels, and only noticed thanks to a careful cross-check of CAS numbers. Mixing up organotin compounds gets risky — it just takes a simple misunderstanding to start a safety breach.
Staying Safe: Standards in the Real World
Anyone working with tributyltin chloride feels the pinch of regulation and real physical risk. Chronic exposure can hit the immune system, liver, and nervous system, so a mask and gloves don’t seem like overkill. Ventilated fume hoods and detailed waste management rules aren’t just box-ticking exercises; they make sure that what enters the workplace doesn’t leak out into the wider world. Countries like the United States and Japan responded to early toxicity reports with use restrictions, national standards, and sometimes outright bans for antifouling purposes. Still, legacy installations and poor handling leave traces that challenge remediation. Many older shipyards and harbors face expensive clean-up bills, and all that cost underscores the need for better prevention.
Applications: Where Tributyltin Chloride Made Its Mark — and Where It Still Lingers
Tributyltin chloride’s real-world reach sprawls further than most casual observers realize. Ship coatings top the list, but its footprint shows up in wood treatments, certain industrial footwear, and as a stabilizer in plastics. The marine painting industry leaned heavily on its promise to slash hull cleanings, with entire fleets undergoing routine reapplications. Timber for houses and railroad ties, once pressure-treated with tributyltin, still lines old towns and tracks in some regions. Chemists pushed into the realm of plastics in search of heat stability, finding that organotin agents like this one could keep vinyls from degrading under sunlight. As new findings pile up about environmental toxicity, restrictive laws and greener alternatives have started to push tributyltin out, but its former dominance lingers in soil, sediment, and living organisms.
Toxicity: The Costs of Convenience and Short-Term Gains
Toxicity studies have sounded alarms about tributyltin chloride for decades. Research uncovered its power to disrupt endocrine systems in aquatic animals, causing everything from abnormal growth to outright population collapse. Mussels and snails show the results on their shells, literally, as TBT triggers disorders like imposex, where female animals develop male features. Data from estuaries in Europe, North America, and Asia tie population drops in oysters and dog whelks to contaminated paint chips. Bioaccumulation means tiny exposures climb the food chain, ultimately raising red flags for people who depend on seafood. As environmental monitoring gets more comprehensive, traces of tributyltin show up in unexpected places, especially in sediment that releases the chemical back into water year after year. Toxicologists, ecologists, and policymakers all agree: the old belief in a safe “threshold” concentration keeps eroding, and clean-up isn’t simple.
Research and Development: New Paths, Old Lessons
Recent years have seen a pivot toward alternatives — both in antifouling coatings and in the stabilization of plastics. Academic researchers and corporate labs seek less persistent solutions, some based on biodegradable ingredients, others pushing mechanical coatings that don’t release biocides at all. Environmental chemists dig into remediation strategies, like finding microbes that break down organotin residues or using activated carbon to trap leaching chemicals. Money and talent flow into these efforts because the days of overlooking long-term impact are ending. Lab models are growing smarter, and so are regulatory tests that check not just for acute toxicity but also for subtle, chronic effects.
The Road Forward: Facing the Future with Open Eyes
The story of tributyltin chloride still offers plenty of lessons about trying to solve a problem in one domain without thinking through the costs in another. As technology advances, the temptation to chase a magic bullet never fades, but lived experience and real data push resource managers, scientists, and regulators to stay skeptical. Dealing with the aftermath of TBT calls for more than banning — it takes monitoring, public engagement, reinvestment in clean-up, and transparent research sharing. In the lab and on the dockside, the culture is shifting. Many industries now share environmental reports and remediation plans that once stayed secret. The next wave of antifouling, wood protection, and material stabilization will reflect these hard-won lessons, making room for compounds measured not just by their effectiveness but by their entire lifecycle impact. That broader perspective, rooted in both experiment and experience, leads toward solutions that respect both economic need and environmental health.
What is Tributyltin Chloride used for?
Where Tributyltin Chloride Shows Up
Tributyltin chloride slips quietly into our lives, usually far away from public attention. In the chemical world, it packs a punch both in industry and in everyday products. Its main role stands out in the manufacture of pesticides, plastic stabilizers, and wood preservatives. Boat owners and shipbuilders once used it widely to coat vessel hulls because tributyltin compounds repelled barnacles, worms, and algae. Ship bottoms stayed cleaner, fuel costs sank, and maintenance took a backseat, so it looked like a money-saving breakthrough.
The Science Behind Its Use
What sets tributyltin chloride apart is its strength as an organotin compound. Chemists value it in catalysis, especially for polymer production and in laboratory synthesis. It can make plastics stronger, last longer, and resist degradation. When you see talk about PVC, tributyltin compounds sometimes reinforce its durability. This sounds technical, but it impacts daily items from pipes to credit cards.
Environmental Risks Are No Joke
Anyone who has spent time around rivers, harbors, or beaches may notice dead zones where nothing seems to grow. Scientific studies point to tributyltin as a culprit behind some of these toxic stretches. Shellfish and fish populations dropped fast in harbors with heavy use of tributyltin-based paints. It doesn’t break down quickly, so tiny concentrations build up in water and sediment. Over time, creatures absorb these chemicals. Oysters, for example, end up with stunted shell growth and even become sterile in heavily contaminated areas.
Impact on Human Health
Workers exposed to tributyltin chloride may see more than just sticky hands. Researchers have tied it to skin and eye irritation, and sometimes to disruption of the human hormone system. Lab tests suggest it can harm the immune system. Now governments, especially in Europe and the US, place tough rules on its use. Bans rolled out for ships under a certain size. Bigger vessels switching over took longer, but international agreements helped phase out old-style paints.
Alternatives and Solutions
Some companies now turn to less toxic copper-based or silicon-based coatings. These options protect ship hulls and cut down on marine life buildup, without creating such hazardous byproducts. Strict controls mark the purchase, transport, and disposal of leftover tributyltin chloride stocks. Scientists push for regular monitoring of local water quality and more public reporting. As a consumer or someone who works around these chemicals, reading product labels and supporting brands that ditch persistent pollutants can help steer the market in a safer direction.
Looking Forward
Industry continues to search for new answers. Biodegradable coatings and green chemistry hold promise for the future. Stronger environmental standards and better awareness can help keep harmful compounds like tributyltin chloride from spreading. Talking with friends or colleagues about safer alternatives and demanding more transparency from manufacturers keeps this conversation alive—not just for cleaner water, but for healthier communities all around.
Is Tributyltin Chloride hazardous to health?
Tributyltin Chloride: More Than Just a Chemical
Most people haven’t heard of tributyltin chloride until it shows up in the news or spills into waterways. Industry uses it for a handful of things—mostly as a pesticide in paints to stop barnacles from sticking to ships. The worry really starts with how easily tributyltin gets into the environment and sticks around. Once it's in the water, it doesn’t take long to find its way up the food chain.
Hard Evidence on Health Risks
There’s a real reason for concern here. Studies have linked tributyltin chloride to a range of health effects. In aquatic life, it messes with the way snails and fish reproduce. Male snails can turn into females, and fish can have trouble growing up properly. If this stuff can bend biology in the open water, it’s a wake-up call to think about what it does to people.
A big red flag shows up in research where tributyltin gets into the human body. It doesn’t just pass through. It gathers in fat and the liver. This chemical can disrupt the way hormones work—think thyroid, reproductive systems, and even how our bodies store fat. Some testing links exposure to weakened immune response, issues with metabolism, and changes in brain development, especially for younger kids.
Real-World Exposure
You won’t find tributyltin on the label of anything at the store, but it touches our lives more than we like to admit. In one case I looked into, lab workers cleaning up an old marina site found out too late that their gloves and masks barely slowed the chemical. Their blood tests flagged higher levels weeks after the spill. That story sticks with me because it shows how easily this stuff slips through the cracks—literally and legally.
People worry about eating seafood from contaminated waters—and for good reason. Tributyltin moves from plants to tiny sea creatures, then up to fish, which end up on dinner plates. Testing by food safety agencies in Asia and Europe found tributyltin in farmed shrimp, mussels, and clams. Long-term exposure brings more risk for people with liver or kidney problems.
Taking Action Reduces Risk
Tackling tributyltin chloride means facing tough choices in industry and regulation. Governments worldwide have phased out its use in new marine paints and pesticides, but legacy pollution remains in harbors and coastal areas. As someone who’s watched community groups push city officials for better cleanup, I see the power in keeping pressure on lawmakers and agencies. It takes clear testing rules, real penalties for polluters, and funding for cleanup crews.
On a personal front, people can push for tighter food safety standards and support seafood that’s tested for contamination. Sharing data, supporting local waterway monitoring, and joining in beach cleanups may seem small, but every bit helps. Knowledge and strong advocacy go a long way toward making sure tributyltin doesn’t slip back into the shadows.
How should Tributyltin Chloride be stored and handled?
Getting Real About the Risks
Few chemicals draw a stronger reaction among lab workers and environmental specialists than tributyltin chloride. This isn’t a compound you want to treat casually. Tributyltin compounds, used for decades in marine paints and industrial processes, left a trail of environmental scars behind. Tributyltin chloride stands out for its toxicity, potential to disrupt hormones, and the long-term headaches it causes if handled carelessly. Having spent time in both research and industrial settings, I’ve seen too many close calls not to respect these risks.
The Right Room and Storage Method
Any facility working with tributyltin chloride needs a dedicated, well-ventilated chemical storage room. You’re not just looking for a spot on a shelf. Give this chemical a cool, dry, and dark place, tucked away from incompatible substances like acids, oxidizers, and bases. Label everything clearly; don’t let sloppy handwriting decide someone else’s fate. Storing it in tightly sealed glass containers can help slow down hydrolysis, but it’s not magic. Routine checks to make sure seals haven’t broken or vapor hasn’t escaped save a lot of trouble later.
Personal Protection: Not an Afterthought
Goggles, gloves, and full lab coats are a must. Regular nitrile gloves break down too quickly, so opt for butyl or laminated options for any extended work. I’ll never forget the time a drop soaked through my gloves — it only took seconds before my skin tingled, and a quick dash to the eyewash station became necessary. Never underestimate how quickly tributyltin chloride can irritate your skin, eyes, and respiratory tract. Use a fume hood every time you open a container. If the ventilation system’s acting up, lock that container and walk away. No chemical process is worth risking your health over a shortcut.
Handling Spills and Accidents
One spill turns a quiet lab into chaos. Those without training scramble, and those who practice spill response calmly get to work. Tributyltin chloride can’t touch water or basic sorbents. Specialized absorbents, gloves, and face shields should always be close by. Everyone in the area should know the drill: evacuate if there’s even a hint of trouble, activate the emergency response plan, and air out the room. Once I saw a junior researcher panic and try to mop up a spill with paper towels — things went downhill fast. After that, our group reviewed chemical safety training and put up bigger, bolder instructions.
Disposal Shortcuts Lead to Long-Term Problems
You can’t pour tributyltin chloride down the drain and hope for the best. Disposing of this compound demands licensed chemical waste handling. I’ve watched labs pay huge fines and face shutdowns after improper disposal, and that doesn’t even scratch the surface of damage to local water supplies. Keep a detailed log of what goes out with every hazardous waste pickup. Responsible companies schedule routine training and work with certified waste managers. Trust in established disposal channels — backyard chemistry causes problems for years to come.
Culture of Accountability
It’s tempting to believe that one person’s habits don’t matter much in a bigger operation. That belief leads to minor mistakes turning into major emergencies. Building a culture where people look out for each other, double-check labels, and aren’t afraid to call out unsafe storage conditions makes everyone safer. Open conversation, transparent incident reporting, and regular reviews of procedures help stop disasters before they start. Respect for tributyltin chloride’s hazards doesn’t show fear; it shows wisdom — and it keeps everyone alive, healthy, and able to get home at the end of the day.
What are the safety precautions for working with Tributyltin Chloride?
The Real Hazards
Tributyltin chloride doesn’t play around. Exposure, even in small amounts, can trigger skin and eye irritation, as well as more severe effects if vapors or fine particles go airborne. Over the years, plenty of folks working in labs or industry have learned how it can chew through gloves, damage lungs, and even linger on surfaces in places with poor ventilation.
People sometimes underestimate just how much care organotin compounds demand. I remember an old colleague who thought a dust mask would suffice. Not even close. A few hours later, a trip to occupational health made it crystal clear that respirators and proper gloves aren’t optional for jobs like this.
Personal Protection: No Excuses
Before opening a bottle of tributyltin chloride, check your gear. Splash goggles block droplets, and a full-face shield offers another layer during transfer or mixing. I’ve seen nitrile gloves dissolve under some tough chemicals, but tributyltin chloride needs those thicker, chemical-rated gloves, along with a long-sleeved lab coat. Lab coats should never leave the work area—throw them right in the dedicated hamper for proper laundering. Safe storage of street clothes matters, too; it keeps outside contamination at bay. Boots or closed-toe shoes are a must.
Engineering Controls Beat Complacency
Working in a chemical fume hood gives an obvious advantage. I trust benches with solid airflow, confirmed by those little flutter strips or digital monitors. Don’t assume last week’s safe fume hood still performs the same today. Testing is critical before starting. If a hood shows weak airflow, flag it for repair fast. Process changes can’t skip vented enclosures. Even a single use outside containment invites trouble for everyone in the area.
Handling and Storage: Ditch Bad Habits
Storing tributyltin compounds away from open space and heat sources can’t just be a guideline—it’s basic discipline. Fire-resistant cabinets with labels keep things organized and reduce the risk of accidental mixing or knock-overs. It pays off sticking to the rule of never pipetting by mouth or handling these chemicals anywhere except dedicated work zones. Spills can turn small problems into evacuation drills if people cut corners.
Training Isn’t Just a Form
Annual safety training shouldn’t be a paperwork exercise. Working through real incident stories drives home what can go wrong. In my experience, hands-on drills—imagining where the chemical could splash, how to rinse eyes, how to grab the spill kit—build the muscle memory that posters on the wall can’t match. Supervisors need to walk the walk. If leadership skips steps or rushes procedures, everyone else will pick up those habits, and accidents follow.
Emergency Response: Know the Drill
Eyewash stations and safety showers need clear access at all times. My old lab kept these areas spotless, because you never know when seconds count. Spill kits should sit within arm’s reach of work benches and storage spaces, fully stocked with absorbent pads and neutralizers. Keep the emergency contact chart in plain sight, never locked up in a file cabinet. Quick thinking and easy access turn a close call into a story, not an injury report.
Better Solutions Going Forward
Some workplaces now use substitutes—safer reagents with less chronic toxicity. Still, many research goals rely on tributyltin chloride, and shortcuts just aren’t worth the price. Sticking to strong safety culture, regular training, and solid personal habits makes the difference between a safe day and a disaster. Ask questions. Make safety meetings part of the real workflow, not an afterthought. Every one of us is responsible for making dangerous materials less dangerous, every single day.
What is the chemical formula and appearance of Tributyltin Chloride?
A Closer Look at Tributyltin Chloride
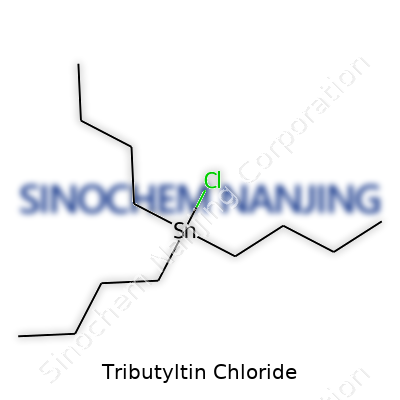
Whenever someone starts digging into organotin compounds, tributyltin chloride pops up pretty quickly on the radar. Its chemical formula is C12H27ClSn. This tells you it’s a molecule built from twelve carbons, twenty-seven hydrogens, a chlorine atom, and a single tin atom holding it all together. In real life, it doesn’t look flashy. The compound usually shows up as a colorless to pale yellow oily liquid. Open a bottle of it in the lab, and the sharp, biting odor jumps out. The smell isn’t the friendliest—most experienced chemists keep it well sealed and prefer working in a fume hood.
Uses—and Why It Matters
Tributyltin chloride isn’t just another chemical with an intimidating name. For years, industries have turned to it for work as a stabilizer and catalyst in various reactions. Shipbuilders leaned on compounds like this one for anti-fouling paints. These paints make life difficult for barnacles, algae, and other hitchhikers on ship hulls. The main reason it became so popular was efficiency. In the short term, tributyltin compounds outpaced earlier coatings in stopping growth and keeping ships faster in the water.
Here’s the rub: tributyltin chemicals do the job almost too well. People who’ve worked around harbors or tracked marine science will remember cases where the chemical leached into the water, building up in living organisms. Researchers found mutations in oysters and issues in fish. One French study in the 1980s linked the presence of imposex (where female marine snails grow male organs) directly with tributyltin pollution. Just a small leak could hurt an entire local ecosystem.
Safety and Regulation
Tributyltin chloride feels pretty slick between your fingers, but this is not a chemical to handle without proper gear. Exposure risks include skin irritation, eye damage, and respiratory issues if inhaled. From firsthand lab experience, a drop on the skin usually gets washed off right away, no exceptions. Those sharp smells in the air serve as a warning. Not long ago, it was more common to find it in shipyards or plastics factories, but new regulations have changed the landscape. The International Maritime Organization banned organotin compounds in anti-fouling paints in 2008. Some countries cracked down even sooner.
Getting off tributyltin chloride isn’t easy; industry always wants cheap, long-lasting solutions. Some manufacturers still use it as a reagent. Strict storage rules and disposal procedures help keep the worst problems in check. Polyethylene containers and chemical-resistant gloves are the norm. Experienced workers read safety data sheets as closely as instructions for a new tool because it pays to treat this liquid with respect.
Searching for Alternatives
Engineers, chemists, and environmental scientists are all trying to switch the world away from tributyltin chloride. New generations of anti-fouling coatings use copper or even silicone. These alternatives don't build up in marine life the way tin compounds do, though nothing comes with zero tradeoffs. In the lab, green chemistry approaches are helping labs reduce organotin use, run cleaner reactions, and generate less waste. Cleaner ships, safer water, and healthier workers matter more today than ever before. Every move away from toxic legacy substances means stronger ecosystems and better human health in the long run.
| Names | |
| Preferred IUPAC name | tributyl(chloro)stannane |
| Other names |
Chlorotributyltin Tributylchlorostannane Tributylstannyl chloride TBTC Tributyltin(IV) chloride |
| Pronunciation | /traɪˈbjuːtɪl.tɪn ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 1461-22-9 |
| Beilstein Reference | 1710942 |
| ChEBI | CHEBI:34760 |
| ChEMBL | CHEMBL1223051 |
| ChemSpider | 54625 |
| DrugBank | DB13732 |
| ECHA InfoCard | 100.031.083 |
| EC Number | 202-932-7 |
| Gmelin Reference | 8197 |
| KEGG | C18287 |
| MeSH | D014265 |
| PubChem CID | 6437 |
| RTECS number | WH6780000 |
| UNII | 4O2C70VV7O |
| UN number | UN2788 |
| Properties | |
| Chemical formula | C12H27ClSn |
| Molar mass | 325.97 g/mol |
| Appearance | Colorless liquid |
| Odor | unpleasant |
| Density | 1.198 g/cm3 |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.05 mm Hg (20 °C) |
| Acidity (pKa) | 12.2 |
| Basicity (pKb) | 4.0 |
| Magnetic susceptibility (χ) | -87.0e-6 cm³/mol |
| Refractive index (nD) | 1.488 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 354.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -248.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6352.8 kJ/mol |
| Pharmacology | |
| ATC code | V09CX03 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes skin burns and eye damage. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H314, H410 |
| Precautionary statements | H260, H302, H314, H317, H330, H410 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 96 °C |
| Autoignition temperature | 120°C |
| Lethal dose or concentration | LD50 oral rat 47 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 2,311 µg/kg |
| NIOSH | WH6950000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 250 mg/m3 |
| Related compounds | |
| Related compounds |
Tributyltin oxide Tributyltin hydride Tributyltin acetate Tributyltin fluoride Trimethyltin chloride Triphenyltin chloride |

