Tributyltin Benzoate: Moving Beyond Chemistry Toward Responsible Use
History and Product Overview
Tributyltin benzoate didn’t arrive out of nowhere. The search for advanced organotin compounds initially took off in the twentieth century, right as industries started demanding smarter solutions for controlling biological growth on surfaces. My first exposure to the name came in a university lab, where we debated its place among fungicides, molluscicides, and stabilizers. Chemists saw value in its ability to keep surfaces and materials clean of pesky organisms and to stabilize plastics against heat and sunlight. The compound soon carved out a niche for itself, especially in sectors where PVC and other polymers played central roles.
What separates tributyltin benzoate from other tin-based formulas is the deliberate combination of three butyl groups with a benzoate moiety. This structure changes both how the substance behaves and how it interacts with the environment and living things. There’s a sharp lesson here: sometimes, the chemistry that solves industry problems can lead to headaches down the road, especially if overlooked in the regulatory rush or by companies eager to profit before toxicity studies finish.
Physical & Chemical Properties
From a hands-on perspective, tributyltin benzoate typically appears as a solid or oily liquid with a faint odor. Its solubility profile steers users to certain applications—soluble in organic solvents but not fond of water. That means once it’s in a material, rain or moisture alone doesn’t strip it away; this sticks out as both a benefit in product performance and a worry for environmental contamination. The tin center not only defines much of its behavior but makes disposal a complicated affair, not just an afterthought at the end of a production line. Higher melting and boiling points, chemical reactivity toward acids and strong bases, and a readiness to degrade under certain conditions all factor into storage, handling, and long-term planning.
Technical Specifications & Labeling
Manufacturers who use tributyltin benzoate keep a close eye on technical details. These include not only purity—typically measured in percentage terms—but also control of impurities and residual solvents. Accurate labeling on packaging serves two purposes: first, letting workers and regulators know what’s inside, and second, giving fair warning about both dangers and required handling procedures. The industry learned long ago that overlooking clear labeling sets the stage for accidents or improper mixing. Over the years, my own work reinforced how essential high standards on these points are, especially in regions with strict oversight or wherever products might move across borders.
Making Tributyltin Benzoate: Preparation and Reactions
The chemistry behind forming tributyltin benzoate is straightforward in concept but unforgiving in practice. Reacting tributyltin oxide with benzoic acid under controlled heat produces the desired ester. This sounds simple on paper, but production becomes expensive and dangerous if shortcuts are taken with temperature or purification steps. As my colleagues in chemical process management often point out, small mistakes tend to magnify in bulk processes, leading to runaways or off-grade side products. After synthesis, the product usually gets filtered, washed, and sometimes distilled to reach the level of purity demanded by industrial clients. Later, tributyltin benzoate may participate in other reactions: it can act as a source of tributyltin cations for anti-fouling coatings, or react with other organic acids to yield fresh esters for experimental use.
Names and Synonyms: A Maze of Definitions
Anyone researching tributyltin benzoate runs right into a wall of names and identifiers. Sometimes it goes by TBT-benzoate, tributyltin(IV) benzoate, or chemical shorthand like TBtBz. In the real world, this makes accurate tracking across different safety sheets and labels more than a paperwork chore—it’s essential for avoiding mix-ups, especially in storage yards or research inventories. Early experiences in lab safety reminded me that a misplaced synonym often means incompatible chemicals end up together, raising the odds of a fire or toxic release.
Application Territory: Beyond Boat Paint
The golden era of tributyltin compounds hit around the 1970s and 1980s, with broad approval for marine paints, PVC stabilization in construction, and even pesticide blends in agriculture. Its antifouling power kept barnacles, algae, and other marine life from coating ship hulls and water installations. Yet, repeated scientific studies—especially those surfacing in the 1990s—showed strong toxicity to aquatic invertebrates, and the damage didn’t stop at the water’s edge. Small traces started showing up in sediment and even in the tissues of marine mammals. The compound’s mobility and durability in the environment made it a global concern, leading the International Maritime Organization and national agencies to start phasing out applications. As a result, many countries set strict limits or outright bans for some uses, especially in marine paints. The struggle to replace tributyltin benzoate with safer, equally effective alternatives hasn’t ended, highlighting how some scientific breakthroughs bring their own set of problems.
Research & Development: Seeking the Middle Ground
Modern research keeps circling back to a core concern: how to maintain the industrial benefits of tributyltin benzoate while managing or replacing its toxic legacy. Material scientists have tried tweaking the molecular structure, aiming for more controllable degradation profiles or improved compatibility with specific polymers. Others work on alternative stabilizers, leaning on calcium-zinc or organic substitutes, though these technologies demand trade-offs in both price and performance. Growing up around a coastal community, I noticed firsthand how stakeholders—including regulators, environmentalists, and manufacturers—rarely moved at the same pace. Progress means not only inventing new molecules but also updating regulations, funding long-term toxicity monitoring, and training workforces in safe use and handling of emerging chemicals.
Toxicity: Science and Public Policy Collide
Toxicity research paints a bracing picture. Animal studies link tributyltin benzoate exposure to disrupted endocrine systems, shell deformations in mollusks, and even developmental challenges in higher organisms. I can recall early news reports sparking public anger in port cities as local fish populations declined and shellfish beds showed unusual growth patterns. Lab work has confirmed lower-dose risks, particularly for aquatic life, with bioaccumulation proving more persistent than experts first predicted. Despite regulatory pressure, legacy contamination persists in many coastal sediments, raising difficult questions around environmental cleanup and the health of food webs. Debates in scientific journals stress the need for ongoing research, not only on direct effects but also on complex mixtures including heavy metals and other organic pollutants. The takeaway from these years of data is clear: effective stewardship of tributyltin benzoate must involve robust testing, independent monitoring, and a healthy dose of skepticism toward easy answers provided by manufacturers or lobbyists.
Looking Ahead: Responsible Transitions
Shifting beyond tributyltin benzoate challenges both industry and regulators. Some advocate for fully green chemistry substitutions, which would remove organotins—a category with a well-documented impact on public health and the environment—from the industrial toolbox. Industry groups often warn that performance or price will suffer, but technological progress suggests otherwise. Researchers now focus on encapsulation, controlled release coatings, and biodegradable alternatives—piloting new compounds and delivery systems in both the laboratory and in field applications. My experience counseling startups suggests that the future belongs to companies valuing openness, third-party testing, and transparent risk communication. Working across the chemical and environmental sectors, I’ve seen how hard it is to balance the demand for high-performance materials with the public’s right to a clean, safe environment. The ongoing dialogue between users, regulators, and the public may not always run smooth, yet it’s the surest route toward meaningful progress and a lasting legacy beyond the next product cycle.
What is Tributyltin Benzoate used for?
Used in Plastics, But the Impact Runs Deeper
Tributyltin benzoate doesn’t land in the daily news, but its fingerprints sit in a lot of products most people use all the time. This compound usually ends up inside PVC pipes and cables. Manufacturers lean on it not to add color or shine, but for stability. Polyvinyl chloride, the technical name for PVC, has a tendency to break down over time—sunlight, heat, and even normal aging chip away at the material. Without the right stabilizer, plastic pipes go brittle, wires crack, and flexible hoses stiffen much faster than they should. Tributyltin benzoate protects these materials, holding the structure together for the long haul.
Keeping Fungus and Microbes at Bay
The usefulness of tributyltin benzoate stretches beyond physical strength. It acts as a biocide, which blocks the growth of fungus and certain microbes inside and outside plastic products. In damp climates, mold can creep into the smallest crannies of cables and hoses, causing unseen damage until something fails. Flexible electrical wire covers, garden hoses, and pool liners all benefit from this action. Living in a region with frequent rain and hot summers, I’ve seen first-hand how cable sheaths without additives quickly develop mold streaks and strange smells. This compound has pulled its weight where humidity rules.
Concerns About Human and Environmental Safety
Being useful doesn’t mean being harmless. Tributyltin compounds raise eyebrows in the science and public health worlds. Research traces these chemicals in aquatic environments, where they collect in sediments and, more worryingly, living organisms. Oysters, mussels, and other sea creatures concentrate tributyltin. Studies published by the Environmental Protection Agency and the World Health Organization link these compounds to developmental issues in aquatic life, like “imposex” in snails, where females grow male organs—a sign the chemical messes with hormones. Eating seafood from polluted areas can mean the substances make their way up the food chain.
Communities and regulators have started to notice. In places like the European Union and United States, rules restrict how tributyltin compounds enter the water system or products that might touch food. Consumer groups want clear answers. I remember conversations with local parents worried about what leaches from plastic baby toys and teething rings—questions that stick with you long after the products themselves are gone.
Searching for Better Solutions
Safer alternatives attract investment, but switching isn’t simple. Many stabilizer chemicals don’t match tributyltin’s performance for both durability and antifungal properties. Some companies move toward calcium-zinc or organic-based stabilizers, which create less pollution and lower toxicity, though these substitutes still struggle to match longevity and strength in tough environments. Tracking results from switching to these newer compounds in large-scale applications—municipal water pipes, for instance—matters. It takes long-term studies, plus honest reporting from both industry and watchdog groups, to weigh the real costs.
Companies and regulators face a real balancing act. Cutting corners means bigger repair costs or even health risks years down the line. At the same time, moving slowly risks contaminating the environment, with real consequences for public health and wildlife. Transparent data, conversations between scientists, industry, and communities, and adopting tested safer chemicals where possible can keep both products and people safer.
Is Tributyltin Benzoate safe for humans and the environment?
Understanding the Issues Around Tributyltin Benzoate
Tributyltin benzoate has popped up in recent conversations about chemical safety. It's a compound that sees use in agriculture and industry, particularly as a biocide and a stabilizer. Many companies lean on this chemical to keep products mold-free or to protect materials from degrading. Before digging deeper, it’s crucial to get a sense of why health and environmental experts show real wariness toward tributyltin compounds.
What Science Says About Human Risk
If someone works around tributyltin benzoate, exposure might happen through skin contact, breathing vapors, or even by accident during food handling. Concerns come from the broader class of tributyltin compounds, which scientists recognized as endocrine disruptors. That means they can interfere with hormones in living creatures, rattling metabolism, growth, and even reproduction. High-profile studies show these effects in both animal and lab settings.
Some researchers point to nerve toxicity, liver damage, and immune problems linked to prolonged or heavy exposure. Factory workers and farmers face more risk than everyday folks, but since tributyltin residues contaminate seafood—especially shellfish—the public isn't off the hook. The main issue here lies not only in what's already known, but also in what hasn't been properly studied. Human long-term safety data still looks patchy. European regulators, reading the warning signs, already moved to restrict tributyltin uses across many industries.
Environmental Impacts and Ecosystem Health
Tributyltin's bad reputation grew in part from its disaster on marine life. In the past, boat paints used this compound to keep barnacles off hulls. Fish and shellfish populations near marinas started to crash, with female snails growing male sex organs, causing reproductive chaos. Even small amounts stuck around in sediments for years.
The substance doesn’t just vanish after use. It can accumulate in soils and waterways, spreading up the food chain and weakening whole ecosystems. Predators eat contaminated prey, and the toxic effects pass up to birds and mammals. Some rivers and harbors still wrestle with these leftovers. On land, tributyltin runoff harms insects, aquatic organisms, and even soil bacteria that help plants grow.
Why the Debate Matters—and What To Do
It’s easy to think chemistry happens behind closed lab doors far from regular life. What gets sprayed on crops or painted on ships ends up in our environment, sometimes cycling back into the food we eat. Tributyltin benzoate offers one sharp example of how useful products can carry major downsides. Farmers and manufacturers have pressure to produce at scale while minimizing risk, both for workers and for communities downstream.
Many countries, catching on to the wake of pollution and health risk, started setting stricter guidelines—or even full bans. There’s a push for alternatives that break down quickly or pose less hazard. Some companies already switched to safer biocides and stabilizers, showing that progress is possible when public health takes priority over short-term savings. More open science, regular monitoring, and new rules could keep risky substances out of the food chain and away from sensitive habitats. Chemical safety isn’t abstract—it's personal and collective, woven into the air we breathe and the water we rely on.
What are the storage and handling precautions for Tributyltin Benzoate?
Understanding the Risks
Tributyltin benzoate falls into a group of chemicals known for toxicity in both workplace and environmental settings. This compound, often used as a stabilizer in plastics and as a biocide, can cause serious health issues if handled without care. Anyone working with it, from warehouse operators to lab workers, faces risks like skin irritation, eye damage, and longer-term organ harm. Accidents or lapses in attention can turn a normal day into a medical emergency or an environmental cleanup operation.
Essential Storage Standards
Storing tributyltin benzoate safely means more than putting it on a shelf. Chemical stability often depends on controlling temperature, light, and humidity. Heat or sunlight can accelerate decomposition, increasing the chance of hazardous fumes or leaks. I've seen shipping areas ruined by accidental exposure of volatile chemicals to heat sources, triggering unnecessary emergencies that better storage could have prevented.
Use tightly-sealed, clearly-labeled containers. Glass works better than many plastics—tributyltin compounds can soften or even degrade some types. Place each container away from acids, strong bases, or oxidizers, since mixing can spark dangerous reactions. Never decant into food containers, which can confuse less experienced staff or new hires. Always keep the original label showing clear hazard symbols and the chemical’s name.
Ventilation helps prevent accumulation of vapors. Air flow systems need regular checks and prompt repairs after even minor failures. Proper air exchange lessens the impact of small leaks that might otherwise go unnoticed until someone falls sick.
Safe Handling Habits
Working with tributyltin benzoate requires more than gloves and safety goggles. Chemical splash goggles, face shields, and gloves rated for organotins help, but they’re only as good as their condition and fit. Tired gloves have failed in my own hands after weeks of careless reuse, so regular replacement is key.
Wear protective clothing with no exposed skin. Wash hands and any equipment that touches the chemical before leaving the work area. Never eat, drink, or touch your face until all safety gear has come off and you’ve cleaned up. Accidents often happen after hours or during clean-up, not during organized production runs when everyone pays close attention.
Anyone handling the chemical must know about emergency showers and eyewash stations, and know how to use them under stress. Training for spills and exposures can't stay theoretical—if you’ve never done a drill, you’ll freeze when it matters most. Spills must get contained right away, and people without training should leave the area rather than trying to help. Keeping spill kits accessible and stocked makes all the difference.
Keeping People and the Environment Safe
Tributyltin compounds are toxic to aquatic life. One small spill down a drain can pollute far beyond the workplace. Collect waste in well-sealed containers and hire certified hazardous waste handlers for disposal. Attempting to clean with water or household cleaners only spreads the mess and increases the risks for everyone.
Being proactive—checking labels, reading safety data sheets, paying attention to expiration dates—protects both health and the environment. Chemical safety isn't just about following rules; it’s about keeping colleagues healthy and building a workplace people trust.
How should Tributyltin Benzoate be disposed of after use?
Why Proper Disposal Isn’t Optional
Tributyltin benzoate (TBTB) isn’t a household name, but people using specialty paints and plastics often run into it. Its chemical makeup makes it a powerful biocide. The EPA identifies TBT compounds as toxic for the aquatic environment. Even small amounts can threaten ecosystems. Throwing leftover TBTB into regular trash or down the drain sends toxins directly into water sources, where they linger for years. Communities living near water bodies pay the price, with fishing bans and loss of safe drinking water. I’ve seen reports from places where improper handling led to fish kills and beach closures. Those aren’t things to brush off.
Regulations: Not Just Red Tape
Laws around TBTB disposal didn’t appear for no reason. Several studies in the 1980s and 1990s wrote the ugly story — malformed sea life, collapsing oyster populations. International conventions, like the Stockholm Convention, aim to eliminate persistent organic pollutants such as TBT derivatives. In the U.S., the Resource Conservation and Recovery Act (RCRA) weighs in. Under RCRA, TBTB counts as hazardous waste. Factories and users can’t ignore labeling or storage rules. Labeling informs waste haulers what they’re dealing with; without it, mistakes can put employees at risk.
What Works: Steps That Keep People and Water Safe
It’s always tempting to look for a shortcut. Yet with TBTB, cutting corners brings real harm. Users finish with a product, collect waste in closed, labeled containers that block leaks. Storing that container away from sunlight, water sources, and foodstuffs prevents accidental exposure. Waste management companies specializing in hazardous chemicals take it from there. They use incineration at high temperatures in approved facilities, which breaks down the compound into safer substances. Landfill is not safe for untreated TBTB. Landfill leachate can eventually reach groundwater. There’s no room for improvisation: listed hazardous waste always travels with detailed documentation, including manifests that follow the shipment from site to incinerator.
Worker and Community Health: A Real-world Look
Chemical workers shouldn’t shoulder all the risk. Protective gear and training work best together. Broken containers or accidental spills call for an immediate response from trained teams, never regular maintenance staff. Mistakes happen—I’ve read case studies where improper spill cleanups led to respiratory illness in employees and nearby residents. Outreach and training sessions—simple talks, not just paperwork—can cut through confusion.
What Should Change
Wider education can help. Companies must share disposal procedures not only with employees but also with contractors and waste handlers. Regulators can offer clearer, industry-tailored guides, using everyday language and local case studies instead of legalese. Investment in alternative compounds may ease disposal concerns for the next generation. Until that day, everyone involved has a responsibility to treat TBTB waste with the seriousness it deserves. I support businesses that go the extra mile, double-checking manifests, funding waste audits, and even inviting third-party inspections.
Summary of Solutions: Protecting Water and People
Disposing of TBTB isn’t a quick toss-and-go. Label waste, store it securely, hand it off only to companies licensed for chemical incineration, and track each step. Accidents should meet prompt, trained response—never improvisation. Widespread, plain-language training, plus strict compliance, provides the best shot at keeping waterways and communities clean.
What is the chemical formula and structure of Tributyltin Benzoate?
Getting to Know Tributyltin Benzoate
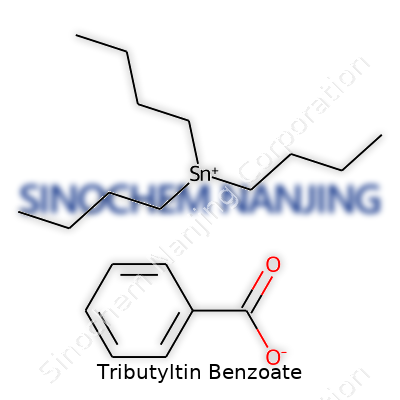
Tributyltin benzoate stands as an organotin compound, catching attention mainly for its role in industry and its environmental impact. The chemical formula looks like this: C4H9)3SnO2CPh, which breaks down to (C4H9)3SnOCOC6H5 in plain language. Imagine a core of tin bonded to three butyl groups and a benzoate moiety. The structure draws from both its organometallic side and its aromatic carboxylate side, which shapes not just its reactivity but its fate in the environment.
Why Learn About the Structure?
Understanding structure isn’t just for test-taking. From direct lab experience and reading incident reports, the architecture tells a story. Tributyltin holds its toxic reputation because tin binds to organic groups so efficiently. The three butyl groups sticking out from the central tin atom create a molecule that resists breakdown in water. The benzoate region, linked through an oxygen atom, brings aromatic stability and changes how the molecule interacts with biological systems—essential in antifouling coatings. These coatings strip life from ship hulls to keep them slick, but the breakdown products flow into harbors and bays.
Environmental and Health Considerations
Facts point to the persistence of tributyltins. In the late 20th century, researchers spotted odd deformities in marine snails and fish living near boatyards. Personal conversations with marine biologists confirmed that tributyltins disrupted hormone systems, leading to reproductive failures. The structure, with its hydrophobic arms and aromatic ring, slips past cell membranes and binds tightly in places tin never went before the age of chemistry. Tributyltin benzoate lingers in sediments and climbs food chains, endangering top predators and, eventually, people through seafood.
Responsible Chemistry and Alternative Paths
Chemists have tinkered with formulas to try to reduce toxicity while keeping barnacles off ships. Experience in research labs proved that breaking up the tributyltin group weakens its grip on life’s machinery, but replacing these powerful agents takes compromise. Copper-based paints work, but bring their own risks and runoff problems. Silicone coatings physically peel organisms away, though cost slows adoption.
Water testing labs and coastal watchdog organizations track organotin residues, but cleaning up means more than simply writing new rules. Pollution doesn’t pause because of good intentions, and tempers flare when industry faces off against public health advocates. It helps to remember that molecular structure shapes policy as much as test tubes do.
Future Lessons from a Single Molecule
The formula and structure of tributyltin benzoate wrap up chemical elegance and environmental challenge in one package. Lab work from decades past didn’t always predict how tough these molecules would prove in the wild. It takes voices from science, industry, fishing crews, and coastal families to weigh risks and imagine futures where chemistry helps rather than harms. Tributyltin benzoate stands as proof that a single chemical’s structure can ripple far past the lab bench, shaping stories in the water and in our lives.
| Names | |
| Preferred IUPAC name | tributylstannyl benzoate |
| Other names |
Benzoic acid, tributyltin ester Tributyltin benzoate TBT-benzoate |
| Pronunciation | /traɪˈbjuːtɪl.tɪn ˈbɛn.zəˌweɪt/ |
| Identifiers | |
| CAS Number | 4098-16-2 |
| 3D model (JSmol) | `/data/structures/all/3d/Tributyltin_Benzoate.jmol` |
| Beilstein Reference | 1772962 |
| ChEBI | CHEBI:34782 |
| ChEMBL | CHEMBL2106417 |
| ChemSpider | 2229343 |
| DrugBank | DB13338 |
| ECHA InfoCard | 100.222.137 |
| EC Number | 225-650-6 |
| Gmelin Reference | Gmelin Reference: "Gmelin 417068 |
| KEGG | C19235 |
| MeSH | D014249 |
| PubChem CID | 156119 |
| RTECS number | WN5250000 |
| UNII | V3K77G34GE |
| UN number | UN2588 |
| Properties | |
| Chemical formula | C25H36O2Sn |
| Molar mass | 491.18 g/mol |
| Appearance | Colorless transparent liquid. |
| Odor | Odorless |
| Density | 1.198 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 3.89 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 10.35 |
| Basicity (pKb) | 4.20 |
| Magnetic susceptibility (χ) | -61.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.541 |
| Viscosity | Viscosity: 8 mPa·s (20°C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 104.7 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | V09CX02 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H314, H410 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P260, P262, P273, P280, P301+P310, P302+P352, P305+P351+P338, P308+P313, P370+P378, P391, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-2-2-W |
| Flash point | 113°C |
| Autoignition temperature | 250°C |
| Lethal dose or concentration | LD50 oral rat 219 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 176 mg/kg |
| NIOSH | B0282 |
| REL (Recommended) | REL (Recommended): 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Tributyltin chloride Tributyltin oxide Tributyltin acetate Tributyltin laurate Triphenyltin hydroxide |

