Tribromomethane: More Than a Chemical Footnote
Historical Roots and the Long Trail of Tribromomethane
Tribromomethane carries a legacy that stretches back over a century. Early chemists, drawn to its distinctive properties, dug into its potential both in the lab and out in the world. It was Carl Löwig who first brought tribromomethane to the science community, recognizing its connection to the broader family of haloforms. For scientists of the time, this class of compounds opened doors to understanding halogen chemistry, reactivity patterns, and even the deeper questions that lay behind why certain compounds acted the way they did. The story of tribromomethane reflects the trial-and-error explorations of generations who built today’s chemistry toolbox.
Getting to Know Tribromomethane
Tribromomethane, which you might hear called bromoform, stands out as a colorless to pale yellow liquid with a sweet, heavy odor. The heft of it hits you straight away—a density much closer to mercury than most organic liquids. Anybody who’s handled it in the lab can recall how it pours with a certain weight. Its boiling point sits lower than water but high enough to demand some caution while heating. As it sits on the bench, it barely dissolves in water, but it mixes easily with ethanol, ether, and other organics. These characteristics aren’t just lab trivia; they run the show when it comes to the challenges and possibilities of using tribromomethane in industry and research.
Labeling, Specifications, and the Practical Details
Labeling for tribromomethane draws from years of regulation and practical experience. Every bottle you’ll find includes hazard warnings reflecting its irritant properties, potential for organ toxicity, and environmental risks. Labels usually point out purity standards, often 98% or higher for research-grade material, which buyers seek for more predictable results in synthesis. Technical data sheets cover common contaminants, such as traces of dibromomethane, because small differences in purity can throw off an entire batch or skew research conclusions. That’s something chemists working late in the lab learn after a failed experiment or two.
Preparation: Classic Routes Meet Modern Tweaks
The go-to method for making tribromomethane in a lab draws on the haloform reaction. Add bromine to acetone or ethanol in the presence of a base, and bromoform drops out as a heavy layer at the bottom of the vessel. This reaction has worked reliably for generations. Industrial producers, always pushing for efficiency, refine the process by optimizing temperature, base concentration, and purification steps to boost yield and minimize waste. Even so, bromine isn’t a substance to take lightly, so modern setups include fume hoods and containment to keep both people and the environment safe. While the basic chemistry hasn’t shifted much since the 1800s, safety culture and the drive to reduce emissions give today’s syntheses a much different feel.
How Tribromomethane Behaves In and Out of Solution
Tribromomethane stands as a classic example of a molecule loaded with bromine atoms, each ready to participate in chemical reactions. It doesn’t play well with strong bases or with reducing agents like zinc dust—a reaction that can yank off bromine atoms and produce all sorts of byproducts. Environmental breakdown keeps folks up at night, too, since tribromomethane can degrade to release more dangerous compounds. These reactions keep researchers on their toes, especially when bromoform heads out of the lab and into larger-scale applications or finds its way into nature through industrial discharges or water chlorination.
Names That Travel the Globe
Though science doesn’t play favorites with names, tribromomethane goes by plenty. Bromoform pops up in most English-language texts, while other languages have their own versions. The array of names—tribrommethan, methyl tribromide, acetylene tetrabromide (in error sometimes)—reflects how a single molecule can weave itself through different national and research traditions. For someone digging through research archives, knowing these synonyms matters when trying to piece together a full picture of what’s been tried, discovered, and sometimes forgotten.
Safety and the Human Side of Chemical Handling
Working with tribromomethane demands respect for both its immediate hazards and the longer-term risks. Inhaling its vapors leads to dizziness, headaches, or even unconsciousness if exposure hits high levels—stories you hear from old-timers who remember more relaxed lab days. It also shows toxicity to organs like the liver and kidneys. Lab safety guidelines hammer home the need for gloves, protective eyewear, and strong ventilation—rules meant not just for bureaucratic compliance but to keep people healthy. Spills require prompt cleanup, and any waste heads straight into hazardous disposal, not down the sink. From my own lab days, nothing sparked a quick hustle like hearing a bottle of bromoform hit the benchtop a little too hard.
Beyond the Lab: Where Tribromomethane Ends Up
This chemical finds its way into a surprising patchwork of applications, even as newer materials chip away at old markets. Analytical chemists value bromoform for its high density in mineral separation. Some water treatment plants keep a wary eye on it, since it’s a byproduct formed when chlorine does its job on natural bromide in water. Manufacturers used to make fire-retardants and specialty solvents from it, but growing health and environmental concerns have steadily squeezed out older uses. Today’s applications show up in niche research projects or experimental pharmaceuticals far more than in mass-market consumer goods.
Chasing the Future Through Research
Tribromomethane continues to play a role in basic scientific research, especially when exploring mechanisms of halogenation and dehalogenation. Studies examining how organisms metabolize such compounds teach us about the interactions between halogenated organics and living systems—a puzzle relevant to both toxicology and potential therapies. Work published in environmental science journals highlights tribromomethane as both a canary in the coal mine and a problem to solve: reducing unwanted byproducts in water supplies asks us to understand every step from formation to breakdown in intricate environmental pathways. As new detection tools arrive, the ability to trace minuscule amounts sharpens the debate over safety, risk, and how tightly regulations should clamp down.
Facing Up to Toxicity and the Real-World Challenge
Researchers have laid out the health risks linked to tribromomethane, with animal studies confirming the kinds of organ toxicity and carcinogenic potential suggested by lab data. Regulatory agencies across Europe and North America now limit both occupational exposure and environmental release. Making those limits stick demands solid workplace training, robust tracking of chemical flows from factory to landfill, and investment in better filtration and containment. Those I’ve met in industry and academia agree that responsibility doesn’t end with compliance but stretches into waste reduction and persistent efforts to substitute safer alternatives where practical.
Looking Ahead: New Questions and Better Solutions
The future of tribromomethane centers not on expanding its market but on managing legacy issues, sharpening detection methods, and deepening our understanding of its chemistry. As industries around the globe shift toward greener chemistry, tribromomethane stands as a key case study—a compound with genuine value yet unmistakable hazards. Solutions lie in blending classic industrial hygiene with modern detection technology and pushing academic research into more sustainable alternatives. The story of tribromomethane isn’t just a matter of chemical properties; it’s a challenge for anyone who believes in balancing scientific usefulness with the demands of health and the environment. The best work ahead will come from teams willing to look past the status quo and imagine safer, smarter chemistry for future generations.
What is tribromomethane used for?
What Exactly Is Tribromomethane?
Tribromomethane, often known as bromoform, looks like a colorless to pale yellow liquid with a sweetish smell. Chemical plants and laboratories keep it under lock and key because it’s pretty potent stuff. It carries the formula CHBr3, which puts it in the same family of chemicals as chloroform, only with three bromine atoms attached.
How Tribromomethane Lands in Laboratories
Laboratories rely on tribromomethane for a handful of specialized jobs. For one, it provides a super-dense liquid for separating minerals. Geologists and mineralogists drop rocks or soil into tribromomethane to sort minerals based on how fast they sink or float. This trick lets scientists isolate grains that carry valuable information, like traces of ancient life or buried ore deposits. I remember a fellow student in university finding a zircon grain, only visible after using a dense liquid. That sample helped date a volcanic eruption half a billion years ago. It’s a reminder that sometimes, the heavy chemicals bring light to lost chapters of Earth’s story.
Role in Chemistry and Research
Chemists sometimes turn to tribromomethane as a reagent. It helps whip up new molecules or tease apart complex mixtures. Drug researchers use it to test reactions or study the breakdown of pollutants in water, pushing science forward. But there’s also a flip side: because the substance evaporates easily and could affect health, science labs follow strict rules and keep exposure to a minimum. This calls for good ventilation, gloves, and a sharp eye for spills.
Making Connections: Water Treatment and Tribromomethane
Human hands don’t only bring tribromomethane into the world. It emerges by accident too. Municipal water systems that disinfect with chlorine can generate small amounts of tribromomethane when bromide, often naturally present in water, reacts with chlorine. Studies link long-term exposure to high concentrations of this substance to a higher risk of certain cancers and problems for the liver and kidneys. Regulations now cap how much ends up in tap water. This pushes water plants to improve filtration and source water that’s lower in bromide. Public health watchdogs routinely test and publish water quality, an effort that came after decades of learning the hard way about chemical byproducts in drinking water.
Concerns in the Ocean and Beyond
Nature also produces tribromomethane. Seaweed and algae pump it out as a byproduct of their metabolism. Marine scientists noticed that this chemical plays a part in moving bromine from the sea to the atmosphere. Up there, it can nudge ozone chemistry. While the amounts are tiny compared to industrial chemicals, even small traces matter when talking about the ozone layer’s health. This raises questions about how much industry should add to the load and how careful we ought to be with what leaks into rivers and oceans.
What Can Be Done?
Some see tribromomethane as just a tool for science and industry. Others see a warning: if we release too much of it, risks follow us home. Public health agencies watch water sources and push for better treatment. Scientists hunt for safer alternatives and invent new ways to test minerals that don’t call for dense toxic liquids. Sometimes it means changing a cleaning routine at a water plant, or switching to disinfectants that spark fewer chemical byproducts.
What matters most is clear communication. People deserve to know what lands in their water or air. Inventors and engineers can play a part by designing safer methods, while watchdogs sharpen the rules. Only then does knowledge about chemicals like tribromomethane serve the public good — not just the next research paper.
Is tribromomethane hazardous to human health?
Understanding the Risks
Tribromomethane, often called bromoform, pops up in swimming pools, disinfected drinking water, and as a byproduct in some industrial processes. If you’ve ever walked into a pool facility and caught that whiff of “pool smell,” you’ve been exposed to a family of substances that can include tribromomethane. This chemical doesn’t belong on any public health wish list, even in small amounts.
Decades of water safety research show that tribromomethane forms during water disinfection with chlorine or bromine. It’s not deliberately added, but comes out of the chemical interactions between source water and disinfectants. Studies published by environmental agencies, including the US Environmental Protection Agency (EPA), warn about links to problems ranging from skin and eye irritation to increased risk of cancer after long-term exposure. According to the International Agency for Research on Cancer, tribromomethane lands in the “possibly carcinogenic to humans” category. No one relishes drinking or inhaling something with that label.
Why Worry Matters
My background in environmental science underscores just how quickly small exposures add up. While a single glass of water may not send you to the hospital, the slow buildup of these compounds can turn into something much worse over time. In the past, I worked on water quality projects in rural communities where water treatment was basic at best. Even low levels of tribromomethane, when consumed over years, raised red flags for community health. We noticed headache complaints and skin rashes, symptoms often brushed aside but worth paying attention to when linked to chemical exposure.
Research from both the Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO) points to long-term effects, like liver and kidney damage. The punchline? Even tiny amounts, over many years, can chip away at good health. In pregnant women, high exposure has been associated with low birth weight or problems during pregnancy, which makes it particularly concerning in areas where drinking water disinfection cannot be easily changed.
Reducing the Risk
Regular water testing stands as one obvious way for communities to learn what's flowing through the taps. Municipal water systems in the United States must report levels of disinfection byproducts under the Safe Drinking Water Act. Anyone concerned can ask their local provider for these results. Installing home filters that use activated carbon helps remove many organic compounds, including some amount of tribromomethane, according to both EPA studies and consumer reports.
On a larger scale, cities and towns can tweak their water disinfection practices by using better source water, limiting the amount of organic material before adding disinfectant, or switching to alternative methods like UV treatment or ozone, which can cut down on byproduct formation. These options need careful oversight but have real promise in lowering tribromomethane without sacrificing safety.
Why It Matters Beyond the Headlines
Most people trust the water that pours from their faucet or the pool they swim in. As someone who’s dug into the details of water safety, I see the gap between official standards and what keeps communities truly safe. Staying informed about substances like tribromomethane puts the power back in the hands of families and neighborhoods, and pushes regulators to keep health front and center.
How should tribromomethane be stored safely?
Why Care About Tribromomethane Storage?
Anyone working with tribromomethane faces a job that’s a little more serious than keeping the janitor’s closet in order. Known also as bromoform, this chemical’s sweet odor hides something far more concerning. Exposure can irritate the eyes and skin, and inhaling too much can lead to headaches, dizziness, even central nervous system problems. Breathing in vapors for long stretches or spilling some on skin isn’t just uncomfortable — it’s dangerous. I recall a research lab mishap where sloppy storage sent fumes drifting halfway down a hallway. Nobody enjoyed that clean-up. A few key moves can prevent that kind of headache.
Relying on Real Science, Not Luck
The chemical structure of tribromomethane pulls moisture from the air, so it doesn’t just evaporate and vanish. It finds its way into lungs and waterways if given a chance. In hot conditions, its volatility ramps up. Heat or sunlight sets off chemical reactions — nobody enjoys an accidental lab fire or toxic exposure.
Facts matter. The Centers for Disease Control (CDC) points out bromoform’s possible cancer risks and its tendency to disrupt water supplies when poorly managed. Not every facility faces the same threats, but the science lines up across every workplace that chooses to store this stuff.
Building a Storage Space that Actually Works
Many try to cut corners, sticking bottles on random shelves or next to flammable cleaning supplies. That’s a recipe for a news headline nobody wants written. Sturdy, sealed glass containers with tight-fitting lids stop leaks. Keep that bottle somewhere cool and dry. A locked chemical cabinet, away from sunlight and sparks, makes spills less likely.
Ventilation transforms a risky room into a safer one. Good air exchange limits vapor buildup, reducing the odds of breathing in something harsh. I once stepped into a closet without a fan while hunting for pH strips. Bromoform’s sharp odor hung heavy for hours. Never ignore that nose — annoyance signals bigger dangers lurking.
Addressing the Human Factor
Safe storage isn’t just about strong glass and labels. Everyone sharing a lab or warehouse needs real training. Slapping a warning sign on a cabinet doesn’t teach anyone what to do during a spill or breakage. Staff should run through spill drills and know exactly who to call in an emergency. It helps to keep up-to-date safety data sheets nearby — not buried in a drawer, but out where people actually see them.
Regular checks matter. I’ve seen leaking stoppers and fading hazard tapes on too many old shelves. Inspect those bottles, replace cracked lids, and check for sticky residue. Leaks never announce themselves politely.
Thinking Ahead
Labs and factories aren’t the only ones who need to store tribromomethane. Smaller workplaces pop up, and online retailers move dangerous goods to curious hobbyists as well. Carefully monitored inventories and strong local rules for chemical storage set the bar higher for everyone. Learning from environmental groups and regulatory agencies stops accidents before they happen. Nothing beats a proactive mindset in a chemical storeroom.
All these steps pay off not just in health and clean air, but in peace of mind. There’s satisfaction in knowing you’ve taken every real-world precaution — because ignoring risks leads to stories that nobody wants to tell again.
What are the physical and chemical properties of tribromomethane?
Physical Profile of Tribromomethane
Tribromomethane shows up as a colorless to faintly yellow liquid with a sweet odor that anyone who’s worked in a chemistry lab might recognize. Its molecular formula is CHBr3, making it a heavier relative of both chloroform and methane. With a boiling point hovering near 150°C, it doesn’t evaporate quickly at room temperature, so it tends to linger as a liquid under most conditions. Out of curiosity, holding a vial of tribromomethane, you can feel the density—this liquid is much heavier than water, almost two and a half times more dense, sinking straight to the bottom of a beaker.
The high density comes from the three bromine atoms packed tightly around the central carbon. At just under 9.5 g per 10 mL, tribromomethane outpaces many common laboratory liquids. Cold weather won’t freeze it solid until temperatures drop well past the reach of your home freezer, sticking around as a liquid down to minus 56°C. Compared to its cousin bromoform, tribromomethane has similar solubility in cold water: not much at all—less than 3 grams per liter—but it mixes well in alcohol, ether, or chloroform. If you pour it out, you’ll notice it doesn’t really mix evenly with water and tends to layer out instead.
Chemical Behavior—What to Expect
Tribromomethane reacts in all the ways those familiar with organic bromides might expect. Chemists sometimes use it for haloform reactions, thanks to its ability to deliver the trihalomethyl group reliably. Since it’s fairly unreactive under standard conditions, it stores stably in brown bottles away from the light, so long as nobody leaves the cap loose. When heated with strong bases, the molecule gives off carbon monoxide and forms salts—a trait that landed it a place in classic textbook demonstrations.
Working with tribromomethane, gloves become non-negotiable. Exposure risks aren’t limited to skin irritation; this compound can be toxic if inhaled or absorbed. Studies dating back decades link it to effects on the nervous system, and because it’s denser than air, accidental spills tend to settle low, making ventilation a priority in enclosed spaces. Tribromomethane also resists breaking down in the environment, where it can accumulate, leading to stricter handling rules in labs and industrial sites.
Human Health and Environmental Impact
Long-term safety stands out as a real concern. Tribromomethane can contaminate water supplies, mostly through disinfection byproducts when chlorine reacts with organic matter. Multiple research groups—including national health authorities—report that even low levels become worrisome with regular exposure. It’s proven to act as a central nervous system depressant, and animal studies link prolonged use to liver and kidney changes. The risk is less about a single accident and more about slow accumulation through improper waste disposal.
In my experience in school labs, teachers drove home its hazards by letting us read data sheets and real case reports, not just aiming for compliance, but helping us grasp how everyday chemicals can have long-standing effects.
Tackling the Risks
Solutions aren’t found in flat bans but in better oversight, handling, and education. Ventilated fume hoods, sealed storage bottles, and real-time monitoring cut the likelihood of accidental exposure. Waste collection needs to follow hazardous protocol, making sure tribromomethane doesn’t slip into general drains. Facilities benefit from adopting stricter labeling and training, reinforcing a culture of safety from the first day students enter a laboratory.
On a broader level, regular review and updating of safety guidelines, plus investment in water treatment upgrades, protect both employees and the public. For home chemists and professionals alike, understanding tribromomethane’s chemical quirks isn’t just academic; it’s a matter of public health and responsibility.
Is tribromomethane soluble in water?
Looking at Solubility Through Lived Experience
Tribromomethane, sometimes called bromoform, brings up memories for anyone who has handled it in a lab. Transparent, with a sweetish smell hovering somewhere between medicinal and sharp, this chemical tells its own story well before it hits the water. The debate around whether tribromomethane dissolves in water gets settled pretty quickly—watching a drop try to blend in says it all. It’s not eager to mix, and that has real-world implications that stretch from lab notes to water quality reports.
Why Limited Solubility Matters Outside the Textbook
Working in environmental labs paints a vivid picture of what low solubility means. In practical terms, tribromomethane’s water solubility hovers at roughly 3 grams per liter at room temperature. Compare that to classic solvents or salts, and you see a chemical that wants to keep to itself. This plays into how it behaves as a water contaminant, affecting water supplies—even if it doesn’t flood the system. The compounds formed in water treatment plants when chlorine meets the natural stuff in the water can include tribromomethane. And even at low concentrations, the story doesn’t end there.
Tracing Health and Safety Concerns
The risk side often gets overlooked. Bromoform crops up when disinfecting water, a process that keeps communities safe from bacterial diseases. But its presence raises flags for other health concerns. The US Environmental Protection Agency (EPA) and the World Health Organization keep limits on how much bromoform can hang around in drinking water. The main reason? Chronic exposure, even at low levels, brings cancer risks—the science shows enough to prompt action. For professionals, that means regular monitoring. For the rest of us, trust in public water systems depends on those checks and balances.
Personal Experience on Practical Solutions
Cleanup starts with understanding the chemistry. In every lab module focusing on volatile organics, the first move always involved checking containers, ensuring waste streams channeled the chemical to proper disposal. Given tribromomethane’s low affinity for water, activated carbon comes up as a reliable fix; it snatches the molecule away thanks to how carbon interacts with organic chemicals. Water plants often turn to granular activated carbon filters or advanced oxidation to resolve these concerns before water hits anyone’s home.
Learning from the Field
Spending time with environmental engineers drove home the value of source control. Reducing the organic matter that finds its way into treatment plants limits byproduct formation like bromoform in the first place. That could mean better watershed protection, smarter land use or improved pre-treatment methods. Cleaning up after the fact works, but cutting off the supply? That’s more efficient and less expensive over the long term.
The Importance of Awareness and Ongoing Research
The foundation of trust in water systems lies in transparency. People want to know that what’s in their glass won’t make them sick. Public agencies publish results and push for stricter regulations as research continues. Better monitoring tools and new cleanup technologies reach municipal plants faster today than ever before. What began as a question of simple chemistry ends up shaping public health policy and guiding investments in infrastructure. The story of tribromomethane reminds us that every detail in water chemistry deserves our attention—and benefits from solutions born both in the lab and in common sense experience.
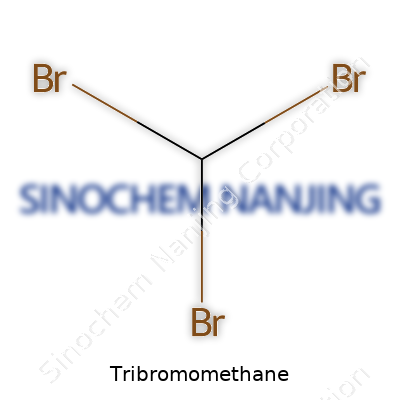
| Names | |
| Preferred IUPAC name | Bromoform |
| Other names |
Bromoform Methenyl tribromide Tribrommethane Bromatum Tribromoform |
| Pronunciation | /traɪˌbroʊmoʊˈmɛθeɪn/ |
| Identifiers | |
| CAS Number | 75-25-2 |
| 3D model (JSmol) | `JSmol` string for Tribromomethane (bromoform) is: ``` BrC(Br)Br ``` |
| Beilstein Reference | 1209220 |
| ChEBI | CHEBI:31301 |
| ChEMBL | CHEMBL1377 |
| ChemSpider | 5937 |
| DrugBank | DB03255 |
| ECHA InfoCard | 03bc480e-0000-4ecc-844c-fbbf2a96f1bf |
| EC Number | 200-854-6 |
| Gmelin Reference | 822 |
| KEGG | C06407 |
| MeSH | D001969 |
| PubChem CID | 5946 |
| RTECS number | PA4900000 |
| UNII | 7M8SOE8830 |
| UN number | UN2515 |
| Properties | |
| Chemical formula | CHBr3 |
| Molar mass | 252.73 g/mol |
| Appearance | Colorless to yellow crystalline solid |
| Odor | sweetish |
| Density | 2.89 g/cm³ |
| Solubility in water | 8.5 g/L (20 °C) |
| log P | 1.97 |
| Vapor pressure | 5.86 kPa (at 25 °C) |
| Acidity (pKa) | 0.8 |
| Basicity (pKb) | 15.63 |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.597 |
| Viscosity | 0.967 cP (20 °C) |
| Dipole moment | 1.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -24.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −56.9 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AX05 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332, H351 |
| Precautionary statements | P210, P261, P273, P280, P301+P310, P304+P340, P305+P351+P338, P330, P501 |
| Flash point | Flash point: 71°C |
| Autoignition temperature | 400°C |
| Explosive limits | Explosive limits: 11-15% (in air) |
| Lethal dose or concentration | LD50 oral rat 1,600 mg/kg |
| LD50 (median dose) | 5.7 g/kg (rat, oral) |
| NIOSH | PB6125000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Tribromomethane: "50 ppm (skin) |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Bromoform Chloroform Dibromomethane Diiodomethane Fluoroform Iodoform Methane |

