Tribromoacetaldehyde: Looking Back, Looking Forward
Historical Development
Chemists first took note of tribromoacetaldehyde in the late 19th century, tracing its roots to the same period where organic halogen compounds got their big break. The process of introducing halogen atoms, especially bromine, fascinated early industrial and academic labs. Back then, researchers often stumbled into new compounds by accident, but tribromoacetaldehyde emerged through a more focused effort to build on the understanding of aldehyde reactivity and halogen chemistry. Brominated derivatives started showing up in various journals, with tribromoacetaldehyde gaining a spot because of its stability compared to more reactive relatives. Progress over the next decades happened in fits and starts, mainly because its practical uses remained tied to niche industrial processes and some early pharmaceutical explorations.
Product Overview
Tribromoacetaldehyde carries a reputation as a specialty chemical more likely to be found in a research lab than a factory floor. The molecule includes three bromine atoms attached to an acetaldehyde backbone, which gives it a solid spot among halogenated aldehydes. This combination results in high density and the ability to participate in a handful of selective chemical reactions. It's far less common in day-to-day industry use, unlike its chlorinated cousins, but for those who need this compound, it solves some chemical puzzles that others can’t touch.
Physical & Chemical Properties
Handling tribromoacetaldehyde brings a strong, pungent odor along with a crystalline appearance. The density stands out since heavy bromine atoms pull up the molecular weight. Tribromoacetaldehyde tends to decompose rather than melt cleanly, breaking down into smaller fragments and sometimes giving off irritant fumes. Solubility in water isn’t great, but in common organic solvents, the compound dissolves fairly well, making it approachable for use in typical lab glassware. The three bromine atoms draw extra attention because they prime the molecule for further chemical transformations or degradation, showing strong electron-withdrawing effects on the core functional group. The material doesn’t score any points for stability, especially if exposed to heat or light over time.
Technical Specifications & Labeling
Clear labeling remains important, because careless handling leads to serious hazards. Labels go beyond just concentrations and molecular structure, flagging bromine content, warning about volatility, and listing clear directions for secure, dry storage. Tribromoacetaldehyde typically gets bottled in amber glass—no fancy packaging, just a nod to keeping out light and minimizing the risk of decomposition. Most producers keep the listed purity above 97 percent and include warnings about potential impurities that might skew reactions or boost toxicity. Real-world labs check not only the batch label, but also documentation on handling precautions to prevent any nasty surprises.
Preparation Method
Labs produce tribromoacetaldehyde through a direct halogenation of acetaldehyde, using elemental bromine in a controlled environment. Since acetaldehyde reacts briskly, strict temperature control and the gradual addition of bromine help manage the exothermic nature of the process. Side reactions easily crop up, producing mixed brominated byproducts. Trituration and repeated recrystallization deliver the pure material, although this often means accepting some losses. Waste management starts at the bench, since unreacted bromine and organic byproducts demand special attention to keep hazards in check and avoid unnecessary environmental impact.
Chemical Reactions & Modifications
Tribromoacetaldehyde works like a Swiss army knife in halogen chemistry. The three bromine atoms open doors to substitution reactions that aren’t possible with simpler aldehydes. Nucleophilic displacement stands out, putting the molecule in play as a precursor to other organobromine compounds. Reducing agents strip the bromine off, producing mixed halogenated products, sometimes useful by themselves or as intermediates for more complex molecules. Tribromoacetaldehyde falls apart quickly in alkaline solutions, which can either wreck your reaction or deliver the next building block if planned correctly. Some specialized pharmaceutical syntheses pick up this compound where highly selective halogen introduction is essential, and in a few historical cases, the molecule provided a stepping stone to flavor and fragrance chemicals.
Synonyms & Product Names
Some refer to tribromoacetaldehyde as bromal, drawing a parallel with chloral—the much more famous trichloroacetaldehyde. The name “bromal” shows up sporadically in old texts, usually paired with stories about early medicinal chemistry. It also answers to tribromoethanal or 2,2,2-tribromoacetaldehyde in more formal settings. The different names matter when working with decades-old literature, saving researchers from making avoidable errors in reading or sourcing.
Safety & Operational Standards
Even experienced chemists pay attention to safety protocols with tribromoacetaldehyde. The compound irritates skin, eyes, and the respiratory system, with volatility adding to exposure risks. Short-term exposure leads to coughing, watering eyes, and potentially severe inflammation, especially in a poorly ventilated workspace. Most guidelines call for full PPE, including nitrile gloves, goggles, and fume hoods. The need for extra ventilation doesn’t relax just because the chemical only comes out now and then. Any spill requires swift cleanup with neutralizing agents, not paper towels. The possibility of bromine fumes means only those trained in organohalogen handling should even think about working with this substance in bulk.
Application Area
Tribromoacetaldehyde finds itself most often in chemical synthesis, not mass production. Medicinal chemists still dust off its recipe card for making rare drug candidates that need heavy halogenation without too much fuss. A few polymer chemists experiment with it as a reactive modifier, seeking controlled chain termination or functionalization. Its use in mainstream manufacturing lost momentum because of regulatory and safety hurdles, not lack of performance. Environmental and workplace safety boards view heavily brominated compounds as higher risk, which throttled broader adoption. Standard production lines lean into cheaper and less hazardous alternatives—the handful stepping outside the lines do so for compelling, high-value applications where no substitute quite fits.
Research & Development
Tribromoacetaldehyde’s reputation as a research chemical keeps it in the pages of academic journals, where synthesis methods get minor tweaks nearly every year. Chemists constantly seek milder conditions and cleaner byproduct profiles, with green chemistry principles driving a push to minimize waste and toxic exposure. Studies out of university and pharmaceutical labs probe structure-activity relationships tied to bromine placement, opening up possibilities for new biologically active molecules. Analytical chemists lean on advanced chromatography and spectroscopy techniques, working to identify unknown metabolites or breakdown products. Knowledge gained from these studies trickles down into safety guidelines and informs policy shifts on hazardous materials.
Toxicity Research
As with many organohalogens, tribromoacetaldehyde brings toxicity concerns that drive both lab behavior and public policy. Early studies flagged its strong irritant nature, but newer research tracks both acute and chronic impacts on human and animal health. Tribromoacetaldehyde breaks down into reactive species, and that means higher risk for cellular and genetic harm, especially in uncontrolled settings. The focus has shifted toward environmental impact, since wastewater from chemical plants can deliver trace brominated organics straight into rivers and drinking supplies. Regulators ask for detailed ecotoxicity profiles, all while evidence mounts about brominated breakdown products sticking around in soils and contributing to broader chemical exposure networks. Emphasizing containment, waste treatment, and monitoring has forced labs and manufacturers to keep a tighter grip on every gram through its lifecycle.
Future Prospects
Looking at tribromoacetaldehyde’s future, two roads stand out. Chemists continue exploring halogenated intermediates for the next wave of pharmaceuticals, working around the compound’s hazards with better equipment and stricter protocols. Interest in green chemistry leads to sharper questions about its lifecycle, from synthesis through disposal. Pushes for safer alternatives aren’t likely to completely replace tribromoacetaldehyde in its specialized realms, because for some transformations, only this compound delivers the right reactivity and selectivity. Next-generation research leans into building derivatives with lower environmental footprints, tighter supply-chain controls, and faster decomposition under mild conditions. Limiting exposure, investing in detection and containment technology, and sharing best practices across labs help resolve the toughest safety and regulatory challenges. The spotlight shines less on tribromoacetaldehyde than on flashier molecules, but its hard-won niche and storied past guarantee it won’t disappear from the scientific stage any time soon.
What is Tribromoacetaldehyde used for?
Understanding Its Real Uses
Tribromoacetaldehyde doesn’t pop up in daily conversation, but in the research community, this chemical draws some special attention. Labs rely on it mostly for its role as a reagent and intermediate. The interesting thing about tribromoacetaldehyde lies in how three bromine atoms change the reactivity of the whole molecule, letting researchers tackle problems that would stump simpler compounds.
Ethical Considerations in Chemical Handling
Although most folks outside of chemistry classrooms never cross paths with tribromoacetaldehyde, scientists who use it know to handle it with care. The substance can release toxic fumes, especially when in contact with water or moisture. Safety data sheets flag it as hazardous for very good reasons. Experience teaches that gloves, eye protection, and a good fume hood aren’t optional here—they’re mandatory. People in academic or industrial labs need regular training, and it helps to keep emergency eyewash stations close.
The Research Value
Researchers use tribromoacetaldehyde in organic synthesis. It’s a heavy-hitter when converting simple molecules into building blocks for more complicated drugs or pesticides. Some chemists take advantage of its unique structure to add specific functional groups to larger molecules, or to protect sensitive chemical groups during a series of reactions. The focus isn’t on producing massive quantities. Instead, the value comes from helping scientists build or break bonds in ways that lead to promising products or new discoveries.
Medical and Commercial Applications?
At one time, tribromoacetaldehyde (sometimes called bromal hydrate) saw some use as a sedative. Today, newer and safer drugs do a much better job, so medical workers consider it outdated and risky. Few, if any, medicines now depend on it directly. In commercial manufacturing, this chemical rarely finds a place, and when it does, it usually supports research or quality testing, not everyday mass production. Many countries strictly regulate its use because of safety and environmental reasons.
The Bigger Picture: Chemical Innovation and Responsibility
Responsible scientists follow strict disposal protocols with tribromoacetaldehyde. Dumping leftovers down the drain, or even storing it haphazardly, leads to trouble for both people and the planet. Misuse brings legal penalties, and the long-term environmental effects can sneak up quickly if people skip over safety rules. This reality underlines why chemistry isn’t just about mixing things together; it’s about understanding consequences and taking steps to limit harm.
Better Solutions for Safer Labs
Years ago, my own work in a shared lab taught me how much planning goes into handling substances like tribromoacetaldehyde. Nowadays, with regulations tightening and environmental agencies keeping a closer watch, the focus has shifted toward using the smallest possible amounts—only as much as lab work calls for, not a drop more. Researchers constantly look for safer alternatives by studying molecules that do the same job with fewer risks. Green chemistry efforts encourage everyone to rethink processes so that labs don’t keep leaning on hazardous chemicals unless there’s no other way.
Looking Forward
Progress in the sciences depends on balancing creativity with caution. Tribromoacetaldehyde remains a tool for chemists who know exactly what they’re doing and why. It reminds us to trust expertise, prioritize safety, and never hesitate to ask tough questions about the chemicals we use, and where possible, to aim for safer, smarter choices.
What are the safety precautions when handling Tribromoacetaldehyde?
Knowing the Risks Beyond the Label
Tribromoacetaldehyde sits in many research labs, usually in brown glass bottles with intimidating warning labels. I remember handling it in a chemical prep room, where just opening the cap kicked off a sharp, almost medicinal smell. That first whiff was a clear signal to respect what I was working with. This chemical vaporizes quickly, and those vapors hit the nose and lungs hard. It's not just about uncomfortable fumes—tribromoacetaldehyde can irritate eyes and mucous membranes and, if it gets on skin, it doesn’t wash off easily. Some reports link its ingredients, including bromine compounds, to serious health problems after repeated exposure.
Personal Protection Makes the Difference
Putting on the right gear isn’t just a box to check. Safety goggles shield your eyes, but you’ll want a sturdy chemical splash face shield for anything beyond a tiny sample. Gloves matter too. Too many people grab latex gloves out of habit, but brominated organics slip right through. You want thicker nitrile or neoprene gloves here. I’ve seen folks lose patches of skin because they thought nitrile disposables were enough. A lab coat means less chance of a spill soaking clothes and skin. Lab shoes—best with no mesh—keep drops off your feet. Never trust regular street clothes to do the job here.
Ventilation Changes Everything
Even a little vapor fills a room fast. Tribromoacetaldehyde belongs under a well-maintained fume hood. More than once, I saw colleagues try to shortcut and open containers outside proper ventilation, which left everyone coughing and the air thick for hours. Running a hood isn’t enough if it’s blocked or overloaded with equipment. Check that airflow gauge every session. Fresh air makes all the difference after a spill or accident.
Spill Response—Don’t Wait
Cleanup procedures need repeating until they’re second nature. Small spills—less than a few milliliters—call for absorbing with neutralizing agents designed for halogenated chemicals, not common paper towels or sand. Get all residues off the surface and inside a chemical waste container labeled right. A large spill can’t be tackled alone. Lab personnel should clear out, call the appropriate emergency team, and keep ventilation running to push vapors out of the workspace. Ingesting or inhaling tribromoacetaldehyde in significant amounts puts workers at risk of long-term respiratory or nerve problems according to the latest safety bulletins.
Storing and Transporting Safely
I’ve seen accidents start long before the chemical gets used. Tribromoacetaldehyde needs cool, dry, locked storage, far from acids, bases, or flame sources. A shelf on a shared rack won’t cut it. Double containment in sealed vessels prevents leaks. During transport—even across a hallway—lift the bottle in a secondary tray just in case. Never carry it in pockets or bags. Sometimes, small steps prevent big headaches later.
The Big Picture—Training Saves Lives
Safety training isn’t just paperwork. Regular refreshers, hands-on drills, and open conversations about near-misses save careers and, possibly, much more. I still remember a fire department captain saying, “It’s not about if someone will make a mistake. It’s about when.” Folks working with tribromoacetaldehyde need fully stocked eyewash stations, spill kits, and emergency contacts posted clearly. Keeping everyone honest with scheduled safety audits means the lab stays not only productive but also safe.
Small Steps, Fewer Regrets
If there’s one lesson I’ve taken from time with halogenated organics, it’s that nothing replaces caution and respect for chemistry. Trust the gear, use the fume hood, never cut corners on cleanup, stay up to date with training, and speak up when things stray from safety. The small routines—gloves checked for pinholes, hoods inspected, bottles triple-checked—save skin, lungs, and sometimes lives.
What is the chemical formula of Tribromoacetaldehyde?
Breaking Down the Formula
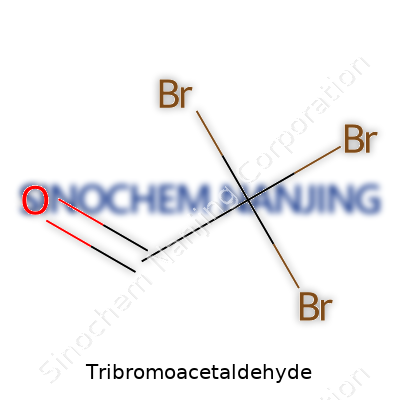
Tribromoacetaldehyde carries the formula C2HBr3O. Chemically, this compound builds on the acetaldehyde backbone. Swap out three hydrogens on the methyl group for bromine atoms and the structure changes in a way that’s easy enough to picture if you’ve sat in a sophomore organic chemistry class. Some folks remember it as bromal, a close cousin of chloral—the stuff once used in old sedatives.
The backbone here matters. Acetaldehyde is CH3CHO. Toss in three bromines on that methyl side and the result: CBr3CHO. Understanding this formula helps more than just chemists. In labs, clarity on molecular makeup keeps you from mixing up compounds with similar names, which could end with an expensive glassware mess or a late-night cleanup shift.
Why Tribromoacetaldehyde Matters
Tribromoacetaldehyde isn’t something popping up in daily headlines, but its structure and properties matter in research and chemical manufacturing. This compound’s heavy load of bromines ramp up both reactivity and hazard potential—safety goggles have never looked so good. In organic synthesis, compounds like this often belong to the toolkit used in building new molecules. That can mean pharmaceuticals, dyes, and when transformed, it leads to derivatives with special properties.
Brominated compounds get attention because of more than just lab uses. Certain byproducts formed during water disinfection also carry multiple bromine atoms, raising questions about health and the best ways to keep water safe from both germs and unwanted chemical residues. Tribromoacetaldehyde, with its structure, helps researchers compare behaviors and outcomes of various halogenated compounds.
Health and Environmental Concerns
Chloral hydrate once slipped into hospital settings for sleep. Tribromoacetaldehyde, close in structure, never reached the same popularity, but its related chemistry signals potential health concerns if handled carelessly. Bromine atoms increase density and can heighten toxicity compared to non-halogenated relatives. Release into the environment calls for watching water sources and understanding reaction pathways, since old industrial practices relied on brominated organic chemistry far before environmental oversight tightened.
Workers exposed to brominated aldehydes should run proper ventilation and wear protective gear. Researchers track breakdown products because some related compounds linger in the environment, bind tightly to soil, and sometimes get into water streams through industrial runoff. Clean-up isn’t cheap or simple. Municipal water plants now invest in monitoring and removing such contaminants to ensure what comes out of a tap won’t wind up a toxic case in tomorrow’s news.
Getting Facts Straight With Chemical Literacy
It’s easy to mix up similar compound names, especially under pressure. A clear grasp of chemical formulas protects not just the reputation of a lab or a business, but also public health. Today’s students face hundreds of formulas, and memorizing them isn’t enough. Mislabel a sample and a chain reaction starts—from paperwork snags to real health hazards. Resources like PubChem and the Merck Index serve as important stops for cross-checking details, not just guessing based on naming conventions.
The drive for accurate information goes beyond preventing mistakes. It means safer experiments, more stable supply chains, and cleaner communities. Chemical literacy, for students and professionals, reduces risks and builds trust in the research powering everything from medicines to clean water. One wrong digit in a formula like tribromoacetaldehyde shifts the meaning, and safety, of a project. That’s why getting the formula C2HBr3O right isn’t just an academic exercise—it’s a line between trust and trouble in chemistry today.
How should Tribromoacetaldehyde be stored?
Storing Trouble or Staying Safe?
Work in chemical labs long enough, you hear stories about people taking shortcuts. They say, “We always keep that bottle right there, it’s never moved.” Then someone bumps the shelf, or the temperature spikes, and things go wrong. Tribromoacetaldehyde offers a lesson in why the rules matter. The stuff isn’t famous like mercury or cyanide, but it packs a punch—handling it carelessly leaves nobody safe.
Why Tribromoacetaldehyde Demands Respect
This compound holds three bromine atoms bonded to an aldehyde. What does that mean in normal terms? Watch out for the fumes. Tribromoacetaldehyde tends to give off hazardous vapors that irritate your eyes and lungs. People in labs with old ventilation systems know exactly what a bad fume can do. Add in moisture from the air, and the substance can break down faster than you’d expect, sometimes creating even nastier byproducts.
Stories from academic labs and small manufacturers all point to the same realities. Small leaks can slip by unnoticed until the sharp, acrid odor spreads. When clean-up happens, it’s usually under pressure. Emergency showers and constant scrubbing leave everyone on edge, especially if someone skipped their gloves or eye protection. Cleaning up the mess takes patience and knowledge. People learn quick—once is enough.
Realities of Best Storage Practice
I’ve seen what goes on behind the scenes—labels peeling off because bottles get handled too much. Some insist on keeping everything under their benches for easy reach, but that’s plain risky. A good setup depends on three things: a dry location, a cool temperature, and tight seals. Heat speeds up decomposition. Humidity encourages the release of toxic fumes. A forgotten cap means the workspace fills with something nobody wants to breathe.
Glass seems like the obvious choice for storing aggressive chemicals, and it usually works—unless you pick a bottle with a leaky stopper. So many spills have started from cracked glass or plastic that went brittle. Secondary containment trays catch leaks before they hit the floor. These trays should not just be placeholders; they save people hours of stressful decontamination every year. Good ventilation is not only a luxury but a necessity. Fume hoods see regular use, especially when people open containers or move material. If your storage shelf sits across the room from a working hood, you’re gambling with everyone’s health.
Training Matters as Much as Equipment
Even the best storage system runs on trust—people doing their jobs thoroughly. Experienced staff show newcomers where things go and why the rules stick. Everyone who handles tribromoacetaldehyde learns this stuff isn’t forgiving. Marrying good training with routine audits gives warning signs before danger escalates. If a bottle feels sticky or smells off, it doesn’t sit around while someone sends an email about it. You take action, log the problem, and start the clean-up.
Solutions That Work
Investment in chemical inventory management pays back every year. Electronic tracking cuts down on lost containers or expired stock, and helps organize regular checks for leaks or container breakdown. Some facilities add extra humidity control or temperature alarms—a small up-front cost compared to the consequences of a spill. Insisting on proper labeling, clear hazard tapes, and accessible safety data sheets means nobody claims ignorance. It’s the simple steps—solid storage, airtight record-keeping, and a clear safety culture—that turn potential disasters into non-events.
Is Tribromoacetaldehyde hazardous to health or the environment?
What Are We Up Against?
Tribromoacetaldehyde doesn’t turn up in everyday life. Most people haven’t heard of it. Think of it as a specialty chemical, mostly handled in labs or factories. But just because it flies under the radar doesn’t mean it can’t cause trouble. This compound brings together three bromine atoms and an aldehyde group, which already hints at a molecule you ought to treat with caution.
Health Hazards: A Real Concern
Breathing in tribromoacetaldehyde or getting it on your skin isn’t something anyone should take lightly. Brominated compounds have a mixed track record in human health. Over the years, I’ve watched chemical safety evolve, especially as more evidence ties halogenated substances to both acute and chronic problems. Exposure can irritate eyes, skin, and lungs, much like an accidental splash of bleach or formaldehyde. That’s only the tip of the iceberg. Long-term or repeated exposure ramps up the risk, hitting the liver and kidneys especially hard.
I remember a story from the lab: a spill involving a brominated solvent. The cleanup team wore full personal protective gear for a reason. These chemicals can enter the body on contact, and nobody wants to wrestle with the aftershocks, from persistent coughs to chemical burns. For tribromoacetaldehyde, its similarity to other toxic brominated aldehydes raises red flags across regulatory boards. Some aldehydes act as carcinogens. It’s not a leap to see how this one could follow suit, especially if handled with bare hands or stored in unventilated rooms.
Threats to the Environment
Dumping tribromoacetaldehyde into drains or waterways runs the risk of hurting aquatic life. Bromine atoms don’t break down easily in the environment. Once released, these chemicals can travel far, getting into streams and turning up in drinking water. Marine animals and plants face bigger risks. Substances like this can mess with their reproductive and neurological systems. A study led by the US Environmental Protection Agency found persistent brominated compounds piling up in fish tissue. This builds up through the food web, threatening not just wildlife, but also humans who rely on fish for food.
Staying Safe: What Works
To guard against these dangers, nothing beats proper handling. Anyone who works with tribromoacetaldehyde should have material safety data sheets handy. Fume hoods, gloves, and goggles are as essential as an umbrella in a downpour. Training workers to recognize symptoms and respond to spills protects lives. Regular risk assessments, along with emergency protocols, reduce accidental exposure.
Disposal is a major piece of the puzzle. Industrial users must follow hazardous waste regulations to the letter. Incineration or specialized chemical treatment keeps this compound out of rivers and soil. Some countries require companies to track and report any emissions or leaks. These laws make an actual difference—transparency and accountability check reckless shortcuts and keep both neighbors and nature safe.
Pushing for Better Solutions
Safer alternatives should take priority wherever possible. Drop-in replacements don’t always exist, but research into greener chemistry brings results. I’ve seen industries swap out toxic chemicals for ones with less baggage once the risks pile up. Until those alternatives hit the shelves, heavy regulations and tight controls need to continue.
Government agencies and independent groups need to keep gathering good data. Gaps in toxicology reports spell trouble. Continued research, coupled with strong oversight, puts science and common sense ahead of profit. No chemical acts in a vacuum—each one shapes real lives and real places.
| Names | |
| Preferred IUPAC name | 2,2,2-Tribromoethanal |
| Other names |
Bromal Bromal hydrate Acetaldehyde tribromide |
| Pronunciation | /traɪˌbroʊmoʊəˈsɛtəldaɪd/ |
| Identifiers | |
| CAS Number | 115-20-8 |
| 3D model (JSmol) | `3D model (JSmol) string` for **Tribromoacetaldehyde**: ``` C(C(Br)(Br)Br)=O ``` |
| Beilstein Reference | 1104192 |
| ChEBI | CHEBI:47777 |
| ChEMBL | CHEMBL49044 |
| ChemSpider | 57372 |
| DrugBank | DB14015 |
| ECHA InfoCard | 100.012.060 |
| EC Number | 208-119-8 |
| Gmelin Reference | Gmelin 8331 |
| KEGG | C19322 |
| MeSH | D014265 |
| PubChem CID | 643048 |
| RTECS number | AG8925000 |
| UNII | I6C6565918 |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C2Br3HO |
| Molar mass | 376.729 g/mol |
| Appearance | White to yellow crystals or crystalline powder |
| Odor | pungent |
| Density | 2.64 g/cm3 |
| Solubility in water | Soluble |
| log P | 0.2 |
| Vapor pressure | 0.11 mmHg (20 °C) |
| Acidity (pKa) | 0.86 |
| Basicity (pKb) | 0.35 |
| Magnetic susceptibility (χ) | -54.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.688 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -145.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -801.8 kJ/mol |
| Pharmacology | |
| ATC code | N05CM06 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Warning |
| Hazard statements | H302, H314 |
| Precautionary statements | P260, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | Closed cup: 110°C |
| Autoignition temperature | 185 °C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 695 mg/kg |
| NIOSH | JN8225000 |
| PEL (Permissible) | `PEL: Not established` |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Chloral Iodoform Bromoform Acetaldehyde Chloroacetaldehyde |

