Triallylamine: A Closer Look at Its History, Use, and Role in Modern Chemistry
Tracing the Path: Historical Development of Triallylamine
Organic synthesis has sparked my curiosity for years, simply because the inventions of past generations serve as building blocks for what we create today. Triallylamine, a tertiary amine with three allyl groups connected to a nitrogen atom, did not emerge overnight. Chemists working in the mid-20th century began probing the reactivity of amines with unsaturated hydrocarbons, prompted by the explosive growth of polymer technology and additive chemistry. Key milestones in its history reflect a push to diversify monomer feedstocks for plastics and to develop new building blocks for pharmaceutical and agrochemical products. In academic papers and patents from the late 1930s onwards, researchers started exploring routes to multi-allylated amines, opening up fresh avenues for the material’s broader deployment. Even today, the evolution of triallylamine reflects the drive to harness simple structures for complex transformations in both large-scale industry and high-value specialty applications.
Looking at the Product: What Makes Triallylamine Stand Out
Step into any lab that handles organic synthesis, and it won't take long to run across bottles labeled "triallylamine". It’s a colorless, flammable liquid that emits a fishy, ammonia-like odor, a scent that lingers in the memory long after leaving a workspace. This molecule plays a quiet but crucial role in the world of specialty chemicals. Its triple allyl group structure imparts a strong reactivity profile, particularly in alkylation and cross-linking reactions. The versatility built into its molecular framework makes it attractive for resin technology, as a cross-linking agent, and for use in creating quaternary ammonium compounds. Chemists value triallylamine not just for these reactivity patterns, but also for its ability to serve as a starting point for advanced chemical modifications. Whether developed for old-school linoleum compounds or cutting-edge polymeric gels, its adaptability underpins its longevity as a mainstay in chemical stockrooms.
Physical and Chemical Properties: Simple Structure, Striking Potential
With a boiling point around 156-160°C, triallylamine stands in the middle of the volatility range for low molecular weight amines, offering enough heat stability for most synthetic processes yet still easy to remove under reduced pressure when necessary. Its low density, under 0.8 g/cm³, means it floats readily atop water, but that doesn’t mean it dissolves efficiently in water; organic solvents serve as a better match. The nitrogen atom, surrounded by those three reactive allyl groups, makes the molecule susceptible to oxidation and radical-based reactions. Any experienced chemist handling the liquid quickly realizes its attributes—energy-rich alkenes, nucleophilic nitrogen, and a structure that positively begs to be functionalized. Conducting chemistry with triallylamine demands respect: it’s volatile, it burns readily, and those vapors can irritate the respiratory system or contribute, in the right (or wrong) mixture, to explosive atmospheres.
Technical Specifications and Labeling: Handling the Details
Regulatory standards for triallylamine mirror those for similar tertiary amines and solvents—tight specifications on purity, limits on water content, and close control over color and odor. Material arrives in tightly sealed, corrosion-resistant drums or bottles. Labels specify not just chemical identity and expected hazards, but storage information, flammability, and mandatory personal protective equipment. The chemical’s UN designation flags it as a flammable liquid with potential acute and chronic toxicity, a fact underlined by vivid hazard pictograms. Laboratories and factories alike keep it contained in well-ventilated storage and workspaces, with specialized exhaust and fire suppression systems. For those buying or handling this liquid, technical expectations rarely leave room for error. Out-of-spec product can spike risks, so regular analytical checks—GC or NMR—help confirm the identity and spot impurities before use.
Preparation Methods: Building Triallylamine from Scratch
Synthetic chemists have explored several approaches for generating triallylamine. The most direct one involves reacting allyl chloride with ammonia or other amines under controlled conditions, using a solvent such as ethanol or water. This nucleophilic substitution demands strict control of reagent ratios and temperature; too much ammonia favors primary amines, too little leads to quaternization. Some variations employ allyl alcohol, passing it through catalytic amination in the presence of acid or base. Modern scale-up facilities stick to robust batch or continuous reactors, using careful addition to limit side reactions and formation of unwanted mono- or diallyl byproducts. Distillation under nitrogen atmosphere helps recover the purified material, leaving behind heavier or more highly substituted amines. Each of these steps—reagent addition, heat control, vacuum application—reflects a century of fine-tuned practice and real-world problem-solving.
Chemical Reactions and Modifications: Unlocking Synthetic Value
What makes triallylamine such a prized intermediate is its remarkable reactivity across a span of chemistries. The nitrogen atom, with its lone pair, readily forms quaternary ammonium salts when exposed to simple alkyl halides, expanding its role in surfactant and phase transfer catalyst manufacture. The three allyl groups open the door to radical and addition reactions, especially for producing resins, polymers, and cross-linked gels. Peroxides or ultraviolet radiation set off rapid polymerization at these unsaturated sites, forging materials that find their way into electronics, dental composites, and water treatment. On the other end, triallylamine undergoes oxidation to form amine oxides or, under harsher conditions, cleavage to yield aldehydes and nitriles. Skilled chemists use it as a scaffold for further functionalization by both traditional and green chemistry approaches, tuning the molecule for each new generation of materials science challenges.
Synonyms and Naming: Sorting Through the Alphabet Soup
Anyone searching chemical catalogs or literature knows the annoyance of running into a single compound under a tangle of names. Triallylamine, with its roots in systematic nomenclature, pops up as N,N-diallylprop-2-en-1-amine, allylamine, 2-propenylamine, or its official CAS registry number. Long stretches in patent documents and regulatory filings lean on these synonyms, while older academic literature sometimes opts for trade names or regional variants. Keeping track of synonyms ensures researchers avoid confusion that can lead to misinterpretation of data or mislabeling in warehouses and laboratories. Digital systems and inventory management software now track synonyms as standard, but veteran chemists still benefit from fluent translation between these naming systems.
Safety and Operational Standards: Managing Hazards with Experience
Working directly with triallylamine brings persistent safety lessons. Flammable vapors require good ventilation and strict fire controls, especially during scale-up or bulk transport. Accidental releases—splashes, spills, vapor leaks—demand rapid attention, since prolonged exposure irritates skin, eyes, and lungs. I’ve watched standard operating procedures grow out of bitter experience: dedicated PPE including goggles, gloves, and chemical splash aprons, strict “no open flames” zoning around storage, and regular emergency drills. Facility design often separates storage of triallylamine from oxidizers, acids, or reactive metals, as cross-contamination leads to runaway reactions. Attention to local and national regulations, not just for air emissions and waste water, but also for employee exposure and transport, sets the tone for quality operations. Regulatory compliance is not just about paperwork; it reflects a real-world commitment to safe chemistry and the health of workers and communities.
Application Areas and Why They Matter
The uses of triallylamine echo across several industries, serving as a foundational molecule for more complex products. Its most well-known application occurs in the production of quaternary ammonium salts, which act as reactive surfactants for cleaning products, hair conditioners, and fabric softeners. From the perspective of industrial chemistry, its triple-allyl structure makes it a go-to monomer for producing ion-exchange resins and specialty polymers with precise mechanical and electrical properties. Electronic component manufacturers count on it as a crosslinker, given its ability to forge tight molecular networks that stand up to stress and electrical charge. It turns up in water treatment, textile finishing, and even pharmaceutical synthesis, where the need for controlled reactivity outweighs most alternatives. In research settings, its flexibility tempers its toxicity concerns, opening the door for discovery of novel materials with tailored properties.
Research and Development: Pushing Boundaries with New Science
With each decade, the eyes of the research community turn to new and greener ways of harnessing triallylamine’s potential. Computational chemists plot electron density maps to predict new reactivity, while synthetic chemists experiment on scaled-down reactors to prototype advanced polymers or functionalized catalysts. The trend to replace hazardous solvents and optimize atom-economical routes has prompted a renaissance in selective amination and in situ activation methods. Working with colleagues in materials science and catalysis, I’ve seen firsthand how triallylamine unlocks new pathways: the push for recyclable or degradable polymers, new water filtration membranes, or custom-tailored adhesives stems from the versatility of this chemical. Interdisciplinary teams, funded by both public and private grants, hunt for applications that balance safety, performance, and environmental responsibility, using triallylamine as a springboard for next-generation chemical solutions.
Toxicity Research: Navigating a Double-Edged Sword
The toxicological profile of triallylamine marks a turning point in how industry treats chemical safety. Unlike some smaller amines, it presents moderate acute toxicity, especially if inhaled or absorbed through skin. Signs of overexposure include headaches, respiratory issues, and skin burns. Long-term data points to potential risks of liver and kidney damage with chronic exposure. Animal studies and cell culture assays shape allowable exposure limits and inform the choice of safer alternatives. My own approach—leaning on thorough risk assessment and mitigation—favors strict controls: handling inside well-ventilated hoods, rapid containment of spills, and regular training on emergency protocols. Ongoing research in industrial hygiene and toxicology aims to clarify the mechanisms of harm and support the design of safer variants with similar chemical utility. Risk, in this case, stands paired with reward, demanding vigilance and vigilance in each operation.
Looking Forward: The Future of Triallylamine
Looking ahead, triallylamine stands at a crossroads in advanced materials science, green chemistry, and safer manufacturing. The growing need for specialty polymers, high-performance resins, and tailored ion-exchange technologies continues to drive demand. At the same time, rising awareness about workplace safety and sustainable practices pressures both manufacturers and users to innovate. Advances in catalytic efficiency, bio-based production, and closed-loop process design could reshape how triallylamine is sourced and deployed in industry. I see university and industry labs test new synthetic pathways that generate less waste, use renewable feedstocks, and lower the burden on workers and the planet. Stronger data on exposure risks and environmental fate will guide regulatory evolution, ensuring the chemical’s benefits do not outpace safe practices. The story of triallylamine charts the ongoing dance between innovation, stewardship, and practical chemistry—reminding everyone that progress relies on learning not just what a molecule can do, but how to use it wisely.
What is Triallylamine used for?
What Triallylamine Brings to the Table
Triallylamine goes by a mouthful of a name, but its influence runs deep behind the scenes of chemistry and industry. Chemists have relied on this clear, flammable liquid for decades to make things that matter—materials, coatings, medicines, and more. Its structure, three allyl groups connected to a nitrogen atom, gives it some useful qualities that are tough to match.
Behind Everyday Items
People don’t run to the store asking for triallylamine, but it helps bring more familiar products to life. Take polymers, for instance. By adding triallylamine, manufacturers can connect molecules in a precise way, leading to gels, adhesives, and plastics that hold up under heat and stress. This trick shows up in paints, floor coatings, dental materials, and even rubber.
I remember touring a lab where folks were tinkering with new water purification materials. They used triallylamine to build a network of molecules strong enough to trap contaminants, but easy to clean between cycles. Instead of tossing out filters, the team just rinsed them, saving loads of plastic every year.
Essential in Pharmaceuticals
Medicine makers use triallylamine when shaping raw chemical ingredients into the building blocks of some drugs. Its knack for forming stable bonds, and reacting only when asked, lets chemists stitch together active molecules with fewer steps and less mess. Every shortcut taken in the lab can translate to cheaper, safer, and more accessible medicines down the line.
If you look at antihistamines, antiviral drugs, or heart medications, chances run high that somewhere in the dozens of reactions on the factory floor, triallylamine played a supporting role. These leaps don’t make headlines, but researchers recognize the difference it makes during scale-up, especially when trying to keep impurities down for patient safety.
Challenges in Handling and Health
Using triallylamine takes some care. It evaporates easily and packs a punch if inhaled or spilled on skin. Factory workers and scientists gear up with goggles and gloves, with fume hoods running strong, especially since the fumes can irritate the eyes, nose, and lungs. In crowded warehouses or labs, poor air flow could mean headaches or worse. Factory rules call for careful storage, clear labeling, and regular toxicity monitoring, since a slipup can turn a routine day into a health scare.
On the production side, triallylamine creates waste that can’t get dumped down the drain. Responsible companies collect, neutralize, or recycle as much as possible. The stakes rise with the volumes handled in chemical plants, so regulators keep a sharp eye on disposal. It’s a balancing act—using the best tools in chemistry, but never cutting corners when people or the planet could get hurt.
Smarter Approaches and the Road Ahead
Chemists keep finding ways to work safer and cleaner. Some new labs install real-time air sensors that catch leaks as soon as they start. Others cycle used triallylamine back through special filters, squeezing more life out of each batch. Regulatory agencies like the EPA and OSHA lay down tough standards that push companies to use chemicals with discipline, not just profit in mind.
Anyone relying on triallylamine—whether to make high-grade coatings or critical medicines—faces the same bottom line: stay sharp, stay safe, and don’t forget that every molecule matters. What happens in a beaker or reactor can ripple into real-world impact. As industries shift toward greener and more efficient processes, the lessons learned from using triallylamine help chart a better course for everyone.
What is the chemical formula of Triallylamine?
Understanding Triallylamine
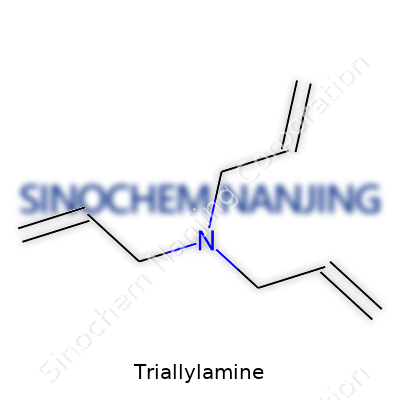
Triallylamine pops up in conversations among chemists and manufacturers as a specialty amine, known for its unique structure and reactivity. Its chemical formula, C9H15N, speaks to its makeup: nine carbon atoms, fifteen hydrogens, and a single nitrogen atom. In basic terms, the molecule features a nitrogen atom bonded to three allyl groups. Each of these allyl groups has a backbone of three carbons, organized in a way that brings out a useful double bond for later chemical reactions.
Why Structure Matters in Chemical Synthesis
In everyday practice, the layout of atoms decides how any chemical behaves and what role it can play. Triallylamine pulls ahead thanks to those three unsaturated allyl groups, making it more versatile compared to typical alkylamines. People tinkering with new polymers or resins often add Triallylamine to their process. For instance, it helps introduce crosslinks—those essential bridges that hold synthetic materials together. The double bonds don’t just sit there; they offer reactive handles for linking to other molecules during polymerization.
Personal Experience with Triallylamine in the Lab
I remember the first time I encountered Triallylamine as a grad student. We needed a compound with enough flexibility for both organic synthesis and further functionalization. Most amines performed well in simple substitution reactions, but few provided the same ease of adding on custom side chains through those allyl groups. This flexibility meant fewer steps in the syntheses, which cut down on both cost and time. In industrial labs, every shortcut counted. Debugging a process, we tried switching from a regular tertiary amine to Triallylamine, hoping for better control over product structure. The improvement turned some heads; polymer properties checked out, and downstream steps simplified.
Environmental and Safety Considerations
Handling Triallylamine isn’t risk-free. As with other amines, it carries an odor, irritates the eyes and skin, and demands careful storage. Chemical workers always use tight-fitting goggles and gloves and check that ventilation is working right. Long-term studies show that exposure can affect the nervous system, reinforcing the call for robust safety measures. From an environmental perspective, the potential for volatile organic compound (VOC) emissions is real. Responsible companies invest in closed reactors and scrubber systems that capture stray fumes. Cutting down runaway emissions protects workers and the surrounding air.
Supporting Responsible Chemistry Through Knowledge
Knowing the formula of Triallylamine isn’t just a rote fact for tests or trivia. It arms researchers and manufacturers with clues about how to use, modify, and dispose of the compound responsibly. Safety data should always follow the molecule wherever it goes. Providing up-to-date safety sheets and making sure all team members respect PPE protocols keeps accidents rare. Companies committed to Responsible Care policies see less turnover and better morale.
Looking Ahead: Improving Chemistry for All
Safer, greener production methods keep gaining traction, and Triallylamine producers take stock of alternatives too. Recyclable solvents, improved capture technologies, and stricter monitoring make a difference in reducing exposure for both humans and wildlife. Researchers, equipped with a solid sense of chemical structure and real-world challenges, make smarter choices that ripple out from the lab to the wider world.
Is Triallylamine hazardous or toxic?
The Basics
Triallylamine crops up in chemical supply catalogs with its trio of double-bonded allyl groups, ready for action in polymer and resin manufacturing. At the lab bench or factory floor, it looks like a clear, colorless liquid with a fishy scent that’s hard to ignore. Most folks outside of industrial chemistry likely won’t bump into it, but if it shows up in your environment, questions about safety are reasonable.
Contact and Exposure Risks
Handling any chemical often introduces risk, and triallylamine isn’t a charming exception. It evaporates easily at room temperature, so vapors can sneak into the air without much warning. Short exposures through the skin or eyes cause burning, redness, even blistering. A splash is genuinely painful. Inhaling its fumes, gassed off from poorly sealed bottles or spills, irritates the nose, throat, and lungs. Headaches, nausea, and dizziness follow exposure, no exaggeration needed.
The trouble with inhaling triallylamine doesn’t stop at the surface. Prolonged or heavy exposure damages internal organs such as the liver and the kidneys. Animal studies show long-term inhalation leads to respiratory problems and other serious effects. Exposure limits exist for a reason—OSHA and NIOSH peg the recommended airborne concentration at a fraction of a part per million. This is no CYA regulation; the expertise behind it grows from years of research and a parade of industrial accidents, big and small.
Fire crews and plant workers can vouch for triallylamine’s flammability. Its vapors ignite with minimal encouragement, lighting up at room temperature. Mixed with air and a stray spark, it does more than burn. It forms explosive mixtures, a detail that’s left scars on buildings and memories alike. Stories about warehouse fires always mention how crews worked around containers of flammable amines like this one, using foam, careful planning, and clear headcounts.
Toxicity and Chronic Impact
At a molecular level, triallylamine doesn’t play nice with human biology. It reacts with cell membranes and proteins, causing direct damage. Chronic low-level exposure in research animals links to both cancer risk and reproductive problems, though clear human data stays elusive. That said, enough structural similarity with better-studied toxic amines stirs caution among toxicologists. The “probably carcinogenic” cloud hovers, and old-timers who worked unprotected in poorly ventilated plants talk about unexplained illnesses among colleagues.
Real-Life Handling, Safer Practices
For professional environments, experience shows that dust masks and goggles won’t cut it. Full-face respirators and chemical-proof gloves, along with tight-fitting lab coats or coveralls, offer real protection. Chemical hoods or dedicated extraction units pull the vapors away from breathing zones. Emergency eye-wash stations save the day when spills go sideways. Good engineering beats luck—better ventilation, regular air monitoring, and training keep incidents to a minimum.
Two decades in labs taught me the value of strict inventory and regular labeling checks. I’ve seen old triallylamine bottles crystalize around caps, a recipe for leaks. Insisting on routine inspections and never leaving open containers unattended became second nature. Proper disposal happens through incineration by licensed hazardous waste handlers, never the regular trash or sinks.
Looking Forward
Firms that substitute safer compounds in place of triallylamine make a difference, both for workers and the environment. Green chemistry programs encourage switching out these kinds of amines as soon as viable options come up. Safety culture grows not from checklists, but shared stories—the folks who speak up about close calls or reveal shortcuts keep everyone sharper.
The facts about triallylamine remain clear: treat it with respect, invest in protection, and don’t take unnecessary chances. Health and safety rules get written in the wake of avoidable harm. Nobody regrets doing too much to keep people whole.
How should Triallylamine be stored?
Recognizing Triallylamine’s Hazards
Triallylamine doesn’t pop up in most people’s lives, but folks working with chemistry or industrial processes know it deserves respect. This chemical’s amine smell can fill a room quickly. Contact with the eyes, skin, or lungs causes real problems. Left open, it evaporates readily and reacts with oxygen, which ramps up risk. Keeping it safe means giving real attention to where and how it’s stored.
A Spot That Gives Peace of Mind
Years ago, I visited a plant where men in faded coveralls made sure every amine was inside a separate fireproof cabinet. Their logic was simple: less oxygen, less heat, zero flames nearby. This was no fancy protocol, just old-fashioned caution. Modern guidelines echo these points. Triallylamine calls for cool, dry storage, away from sunlight or any ignition source. Even fluorescent lights that hum overhead can heat up a storage shelf unnoticed. I’ve seen cases where one bottle’s poor placement led to a chemical odor leeching across several floors—no fun for anyone.
Choosing Containers That Hold Up
Not every container takes kindly to Triallylamine. Glass with airtight seals or steel drums lined with compatible plastics stand up best. Polyethylene offers chemical resistance, but any cap should screw on tight enough to block fumes from sneaking out. Labels must not fade, peel, or smudge. Last year, a college student grabbed what looked like a bottle of acetone—label worn, lid cracked open. One whiff told him he’d made a mistake. Sharp labels and sturdy seals matter as much as the container itself.
Keep It Separate, Keep It Safe
Acids and strong oxidizers do not mix well with Triallylamine. Storing them on different shelves or, even better, on opposite sides of the room helps prevent accidents that happen faster than you’d expect. I once heard about an older facility that lumped chemicals together in a back closet. Someone spilled an acid once, and ended up with a cloud of gas that sent staff running for the exits. Physical distance between incompatible materials and clearly taped labels aren’t just nice add-ons—these moves save headaches, or worse.
Regular Checks Beat Surprises
Modern safety means little without a pair of sharp eyes. Each month, check for leaks, faded labeling, or swelling on containers. Spills may look minor, but the chemical scent lingers and spreads. My own workplace tasks someone weekly to inspect every shelf and log what needs replacing or repackaging. This routine, simple as it is, caught a slow leak behind a box last spring—fixing it early dodged a much bigger problem. Problems left alone do not improve on their own.
Clear Communication Fuels Safety
No one should have to guess what’s on a shelf. New staff or visitors must know what’s stored, where, and why. Signs should read clear even in fading light. Simple briefings save time and prevent panic if anything goes wrong. Small habits—doors closed, lids tightened, spill kits nearby—have a decent track record for keeping folks safe. Relying on everyday caution and double-checks beats learning lessons the hard way.
What are the safety precautions when handling Triallylamine?
Understanding Triallylamine’s Risks
Triallylamine isn’t a household chemical and hasn’t ever shown up in my garage, but I’ve seen it used in specialty labs, mostly in polymer jobs. This colorless liquid looks innocent, but as soon as you uncap it, that tells a different story. You catch the sharp, fishy smell, and after working with it, nobody forgets to respect it after the first time it stings their eyes or nose. The safety data is plain: it’s flammable, corrosive, and gives off nasty vapors. Knowing that, cutting corners with Triallylamine feels like a quick way to end up at the emergency room.
Donning the Right Protective Gear
Goggles and gloves are the starting point, not the fallback. Standard nitrile gloves work if nobody pokes holes in them. Splashing a drop on bare skin may not burn at first, but give it a little time. Full sleeves and a lab coat make sense. The skin around the wrists tends to get overlooked, and I’ve seen the red patch it leaves.
Fumes from Triallylamine don’t play around, either. Even folks with strong noses notice the irritating odor quickly. Proper fume hoods stay on, with the sash down low enough to shield your face. No fume hood, no handling. That’s something I learned the hard way—headaches and watery eyes last hours after exposure.
Aligning with Storage Best Practices
Storing Triallylamine is straightforward: keep it cool, dry, and away from fire sources. This stuff catches fire more easily than some solvents—even static discharge could spark it off. Flammable cabinets, grounded and kept locked, turn into must-haves. Combining it with oxidizers isn’t just a rule—chemists who’ve seen weird color changes in the bottle know that mixing can trigger reactions that ruin more than a workday.
Containers, too, tell their story—tight-sealing glass or steel works. Polyethylene may break down over time, so nobody trusts leftover dropper bottles.
Emergency Actions that Save Skin
Spills aren’t common, but they happen to even careful hands. Having absorbent pads, vinegar (for neutralization), and a chemical waste bin at arm’s reach isn’t just protocol. Once, a small spill on a benchtop soaked through a student’s lab notebook in twenty seconds. Fast action with absorbents kept things from spreading. Keeping an eyewash and shower station working means nobody has to fumble if Triallylamine gets on skin or in the eyes. Flushing for at least 15 minutes isn’t a suggestion—it literally keeps tissue from burning.
Training and Attitude Make the Difference
Being told to respect Triallylamine comes from experience more than manuals. I’ve watched seasoned researchers walk new folks through procedures, double-checking PPE and storage every single time. Accident logs show that complacency usually triggers incidents, not lack of supplies. Building a culture where nobody shrugs off safety drills means people go home healthy.
The same habits that keep labs safe apply in the rare specialty shops where Triallylamine pops up. Reporting up-to-date safety procedures, sharing first-hand stories, and pressing for updated fume hoods and equipment are solutions that actually work. It’s not about paranoia, just a daily commitment to walking out as safe as you walked in.
| Names | |
| Preferred IUPAC name | N,N-Diallylprop-2-en-1-amine |
| Other names |
TAA Tris(2-propenyl)amine Triallylamin N,N-bis(2-propen-1-yl)prop-2-en-1-amine |
| Pronunciation | /traɪ.əˈlɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 102-70-5 |
| Beilstein Reference | 1718737 |
| ChEBI | CHEBI:34831 |
| ChEMBL | CHEMBL74678 |
| ChemSpider | 7950 |
| DrugBank | DB13925 |
| ECHA InfoCard | 17be779a-50e8-4486-b22e-7c2d10586a4f |
| EC Number | 203-672-5 |
| Gmelin Reference | 8413 |
| KEGG | C06535 |
| MeSH | D013034 |
| PubChem CID | 7903 |
| RTECS number | UC0175000 |
| UNII | 7H4S4H34QF |
| UN number | UN2618 |
| Properties | |
| Chemical formula | C9H15N |
| Molar mass | 127.21 g/mol |
| Appearance | Colorless liquid |
| Odor | fishy |
| Density | 0.789 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 1.63 |
| Vapor pressure | 4 mmHg (20°C) |
| Acidity (pKa) | 8.5 |
| Basicity (pKb) | 4.38 |
| Magnetic susceptibility (χ) | -8.11 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.437 |
| Viscosity | 0.6 mPa·s (25 °C) |
| Dipole moment | 1.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 148.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -22.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4157 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS05 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H301, H311, H314, H331, H373 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P311, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2-AIR |
| Flash point | -18 °C (closed cup) |
| Autoignition temperature | 215 °C |
| Explosive limits | Lower 1.1%, Upper 4.7% |
| Lethal dose or concentration | LD50 oral rat 226 mg/kg |
| LD50 (median dose) | LD50 (median dose): 250 mg/kg (oral, rat) |
| NIOSH | SN1680000 |
| PEL (Permissible) | 5 ppm |
| REL (Recommended) | 100 mg/m3 |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Monoallylamine Diallylamine Triethylamine Trimethylamine Triisopropylamine |

