Toluene-3,4-Dithiol: Commentary on Chemistry, Safety, and Modern Use
A Journey Through Its Discovery and Rise
Toluene-3,4-dithiol sits in a unique niche among organic compounds, forging its reputation over the span of more than a century. Chemists in the early twentieth century started focusing on aromatic thiols derived from toluene, attracted by their pungent odors and strong reactivity with metals. Synthesizing small thiol molecules like this one wasn’t just an academic exercise; research tied closely to the challenges of refining fuels, developing new rubber additives, and advancing die chemistry. By the late 1900s, improvements in catalytic hydrogenation and directed metalation transformed what used to be tricky bench experiments into reproducible lab routines. Watching the progression from glass flasks and puzzled frowns to confident columns in organic journals, I can see how curiosity has kept this compound relevant.
What Stands Out Physically and Chemically
Many people picture a clear or yellowish oily liquid when asked about small dithiols, and Toluene-3,4-dithiol fits this view, though its scent can clear a room. Two thiol groups, each attached to the third and fourth carbon of the benzene ring, make it far more reactive than plain toluene itself. The molecule’s heavy, sharp aroma comes from these sulfhydryl groups—and the strength of that odor points directly to its high volatility and reactivity. Solubility leans toward organic solvents; water won’t dissolve it, but ethers and alcohols see no such problem. Sometimes, chemists focus only on large-scale hazards, but even small amounts can pose troubles, both in the lab and environment. The sitting point and boiling point put the compound at risk of fume exposure if mishandled, yet these same properties draw in research teams searching for reactions that need precise control.
Technical Details and the Labels That Matter
Regulations have pressed hard on labeling for chemicals like this one. Toluene-3,4-dithiol carries hazard labels for its volatility, toxicity, and flammability. Laboratories receiving this compound need to log its storage in secure flammable cabinets, never skimping on leak checks or proper seals. The Safety Data Sheet paves the way for further handling, but not all labs can afford elaborate ventilation or detection. Technical data from major suppliers covers purity checks—looking for traces of oxidized byproducts, often with gas chromatography and mass spectrometry. It pays to study these certifications before opening a new bottle because hidden impurities will ruin downstream work.
How Chemists Craft This Molecule
Preparation tends to begin with aromatic substitution on methylated benzenes. A common lab method starts from 3,4-dibromotoluene, reacting it with sodium hydrosulfide under carefully controlled temperatures. Alkali-catalyzed sulfurization works, but careful tuning of stoichiometry makes the difference between a clean yield and a tarry mess. I’ve seen new researchers underestimate the smell; wear gloves, work in a hood, and avoid open flames at every stage. Every bench-top mishap becomes a legend retold to each incoming grad student. Large batches may shift to catalytic hydrogenation on specialized supports, but scaling carries its own set of headaches. Industry processes demand rigorous solvent recovery and waste management to avoid introducing thiols into water streams—much stricter than in early decades.
Chemical Versatility and Laboratory Surprises
The double-thiol structure gives toluene-3,4-dithiol a strong grip in coordination chemistry. Metal ion chelation underpins its use in analytical methods and catalysis research, though anyone who’s tried copper(II) precipitation in an undergraduate teaching lab knows the chaos a few extra microliters unleash. As a building block, the molecule enables the creation of disulfide-linked compounds, tailoring surfaces for sensors or polymer-derived membranes. Researchers playing with oxidative reactions can coax either the disulfide dimer or specific metal complexes, depending on which oxidant they add. It’s one of those rare cases where small changes in the chemistry prompt big shifts in application—from sulfonate intermediates to ligands for heavy metal extraction.
Other Names, Same Challenges
The world of chemical synonyms rarely attracts fanfare, but it shapes transactions and safety logs. Toluene-3,4-dithiol often appears as 3,4-dimercaptotoluene or 3-methylbenzene-1,2-dithiol, depending on supplier catalogs or older literature. Mixing up a digit or letter can bring the wrong compound into a reaction, so experienced chemists always double-check CAS numbers and cross-reference safety data. In the field, clarity in language means fewer mistakes, whether you’re ordering bulk supplies or passing a protocol to a colleague on a busy Monday.
Navigating Safety and Good Practice
Safe handling of thiols calls for more than just gloves and goggles, especially for those with less training. Most stories about strong-smelling leaks in academic labs come down to rushed technique or fatigue. Proper ventilation keeps fumes out of the working zone, but waste disposal remains the biggest environmental hurdle. Disposal of residual dithiol—either by oxidation to disulfides or through incineration—demands oversight and regular monitoring for leaks. Chronic inhalation in unventilated spaces affects the central nervous system and respiratory tract, with allergic reactions posing further risk for sensitive staff. Current guidelines in major laboratories recommend closed-system transfer, double-sealing vials, and routine air checks in high-use areas, but older university labs may still lag on upgrades.
Where Science and Industry Put This Molecule to Work
Much as it might surprise outsiders, Toluene-3,4-dithiol keeps busy in a string of specialized roles. In analytical chemistry, it builds strong complexes with transition metals, helping isolate or detect trace metals in environmental samples. Chemical manufacturing firms add it to formulations for new synthetic routes, benefiting from its active hydrogen atoms. Surface scientists rely on it for gold-thiol self-assembly, laying out the backbone for biosensors or nanoelectronic switches. In rubber vulcanization, it acts as a curative, boosting product resilience—and anyone who has followed rubber chemistry knows how tough regulations on tire additives have become. A handful of research programs look at dithiols as starting points in medicinal chemistry, especially when exploring metal-based therapeutic agents. These projects speak not only to the chemistry but also to the collaboration between academics and industry, sometimes leading to patent disputes and, rarely, new commercial blockbusters.
What Researchers Keep Discovering
Research on toluene-3,4-dithiol doesn’t stand still. The molecule remains central to basic studies probing electron transfer, redox chemistry, and ligand design. Lately, green chemistry has begun to nudge even the purest synthetic labs toward less toxic solvents and lower residue outputs; a target that fits with using dithiols in recyclable catalytic systems. In advanced materials science, linking thiols to graphene or other two-dimensional sheets offers a rich area for physical chemists looking to create sensors with high selectivity. Mass spectrometry teams in pharmaceutical development appreciate its usefulness as a derivatization agent, both in detecting trace metals and in labeling peptides.
Toxicity and Working Realities
Trapping the full risk of Toluene-3,4-dithiol inside lab protocols sometimes feels incomplete. Acute toxicity by inhalation is well-documented, with symptoms stretching from headaches to lung irritation. Skin contact can prompt redness or allergic reactions, making regular use of nitrile gloves a no-brainer. Chronic exposure, even at low levels, links to sensory neuropathy and respiratory disease in animal studies. Regulations in the EU and North America push for regular air quality monitoring and exposure limits—steps that may save careers and lives. In my experience, letting complacency settle in can backfire quickly; safe storage and real-time monitoring far beat the consequences of a single slip-up.
Looking Ahead to New Possibilities
Future prospects for Toluene-3,4-dithiol tie tightly to the shifts in green chemistry, materials synthesis, and pharmaceutical research. As industry standards crank down on environmental discharge, chemists will keep refining reaction conditions to lower waste and develop safer derivatives. Materials science teams staying at the edge expect to draw on the unique reactivity of dithiols for new types of low-cost, sensitive sensors. Collaborative projects between academia and industry grow stronger as analytical techniques unlock new uses for what looks, at first glance, like another smelly, hazardous chemical. Handling the compound with respect—for both its benefits and its dangers—ensures new generations of chemists carry these lessons forward, shaping safer labs and cleaner technologies in the years ahead.
What are the main applications of Toluene-3,4-Dithiol?
A Closer Look at Toluene-3,4-Dithiol
Toluene-3,4-dithiol often shows up in lab supply catalogs, but most people outside certain scientific circles don’t cross paths with it. This isn’t the sort of compound you find on hardware shelves or in craft store aisles. What drives its demand hides deep in the world of specialty chemistry, with a surprising reach into everyday products and critical industrial processes.
Role in Chemical Synthesis
Chemists value Toluene-3,4-dithiol for its reactive two-thiol structure. In my own university days, watching peers in organic chemistry labs, I saw how versatile these dithiols can be. They serve as key building blocks for synthesizing biologically active molecules—sometimes the backbones of enzyme inhibitors or anti-cancer drug candidates. Not long ago, several published studies explored dithiols as crucial reagents for modeling how proteins form and maintain their three-dimensional structures. This helps researchers attempting to unravel mysteries inside our cells or build better pharmaceuticals.
Materials Science and Polymers
A lot of polymer science comes down to finding the right links. Toluene-3,4-dithiol steps in as a cross-linking agent. Take vulcanized rubber, for example. With its two sulfur-rich arms, this compound provides strong bridges between polymer chains, changing softness and stretchiness into reliable toughness. That sort of molecular splicing spells the difference between a brittle material and one that’s built to last. If you’ve ever admired a durable industrial gasket or wondered why some medical tubing outperforms others, chemical tweaks like this play a bigger role than most people realize.
Flavor and Aroma Chemistry
The story takes a turn toward the senses. Sulfur compounds have an outsized impact on flavors and smells, for better or worse. Toluene-3,4-dithiol doesn’t end up in your food, but its relatives do, especially in the wine world. Enologists monitor dithiols closely; the tiniest traces help unlock layered scents in Sauvignon Blanc or Cabernet. Analytical chemists often use this compound as a calibration reference or precursor when developing detection methods for these “aroma impact molecules,” ensuring the results are reliable. Mistakes in identification can cost wineries years of brand-building effort, so accuracy matters.
Environmental and Analytical Testing
Beyond food and drink, environmental scientists and industrial hygienists keep Toluene-3,4-dithiol on hand for testing. Certain heavy metals, especially mercury, react strongly with its sulfhydryl groups, producing measurable changes. This clever chemistry underpins several detection kits and lab assays. Back in an internship, I saw field researchers pull out glass vials with solutions of this type before testing soil or water for contamination near old manufacturing sites. Reliable detection depends on precise reactions, not guesswork, so compounds like this offer assurance during high-stakes investigations.
Toxicity and Safe Handling
Like any potent chemical, handling Toluene-3,4-dithiol takes more than just gloves. Exposure risks run high because sulfur compounds can trigger headaches or worse if mishandled. Strict safety training forms the backbone of every workplace using it. From the proper fume hoods to secure storage, companies willing to invest in high-quality equipment and ongoing training end up with fewer incidents and better long-term results. Regular medical monitoring and personal protective equipment can turn what could be a hazardous shop into a model of safety.
Finding Balance: Progress and Precaution
Toluene-3,4-dithiol underlines how small-volume specialty chemicals still matter on big stages. Whether helping develop medicines, toughen up everyday materials, sharpen analytical tools, or keep labs honest about contamination, its applications ripple far beyond the bottle. The right mix of scientific know-how, rigorous safety, and open communication ensures that its potential brings more benefits than risks.
What is the chemical formula and molecular weight of Toluene-3,4-Dithiol?
The Formula and Structure Tell a Story
Toluene-3,4-dithiol packs a punch in the world of aromatic chemicals. Its chemical formula is C7H8S2. This molecule brings together a classic toluene backbone with two thiol groups hanging off the benzene ring at the 3 and 4 positions. These sulfur groups place it in a category of compounds that grab the attention of chemists looking for reactivity and functionality. The molecular weight measures out at about 156.27 grams per mole. These numbers aren’t just technicalities tossed around in labs—they influence every aspect, from how researchers handle the chemical to how industries put it to work.
Applications that Reach Well Beyond the Lab
Sulfur isn’t rare in industrial chemistry, but having two thiol groups tethered to a toluene ring gives this compound a distinctive edge. In my years of tinkering with aromatic sulfur compounds, the stench always marked thiols as powerful players in the lab. One whiff, and you never forget the lesson: potent functionality often comes with memorable drawbacks. This trait makes Toluene-3,4-dithiol ideal for organic synthesis as both a reactant and a building block. It shows up in the synthesis of specialty polymers, as a cross-linking agent, or as a starting point when developing chelating agents for heavy metal capture. None of these uses demand a huge visible spotlight, yet they power daily life in silent, crucial ways—think fuel purification, advanced electronics, and medicine.
Why Purity, Handling, and Knowledge Matter
With sulfur in the structure, reactivity jumps up noticeably. I've watched mishandled thiols create headaches for lab safety, causing unwanted reactions, stink, and contamination. This compound is no exception—both its chemical potency and strong odor require airtight containers, thoughtful ventilation, and strict procedures. Anyone who’s worked with aromatic thiols knows the importance of vigilance. Even small leaks can lead to big problems, ranging from equipment corrosion to occupational health risks. Open discussions about handling, healthy respect for personal protective equipment, and real investment in ventilation prevent bigger issues down the line. Large-scale use, naturally, multiplies these concerns.
Solutions: Balancing Utility and Safety
Before starting any project, I check safety data sheets, talk with chemists who’ve logged hours with the compound, and look for up-to-date best practices. For toluene-3,4-dithiol, standard protocols mean using fume hoods, donning gloves, and working in small batches. Waste management often gets too little attention, so clear labeling and proper disposal rank high on the jobs list. Proper training for anyone new to the lab helps avoid costly mistakes and keeps injuries at zero. Manufacturers can help by delivering high-purity material in sealed, warning-labeled containers that keep the product stable longer and avoid leaks in shipping.
Bringing Facts to the Forefront
The point isn’t to make chemical work harder. Instead, knowledge, practical training, and sensible investment in basic safety gear keep this compound useful and out of headline-making accidents. It’s never about reinventing the wheel—just keeping everyday science honest, transparent, and responsible. Toluene-3,4-dithiol exemplifies how one string of atoms can fill an important spot in high-stakes, real-world chemistry.
How should Toluene-3,4-Dithiol be stored and handled safely?
Understanding the Chemical’s Risks
Toluene-3,4-dithiol belongs to the family of dithiols — organosulfur compounds that can irritate skin, eyes, and lungs just by being there. Open a bottle in a regular room, and the odor alone lets everyone know. Exposure over time, even to small amounts, often leads to headaches, dizziness, and even worse, possible allergic reactions. These facts are not scare tactics; they reflect real experiences in the lab. There’s enough real-world evidence warning us not to get casual with storage or handling.
Personal Experience Drawing the Line
Back in the chemistry lab, I remember being surprised how quickly fumes got out of hand. Even those who felt comfortable with lab work realized how important it was to double-check their protective gear. Chemical splash goggles weren’t optional. Gloves—no shortcuts allowed. Nitrile usually held up better than latex, which quickly broke down when exposed to sulfur-based liquids.
Ventilation Can’t Be Ignored
No fume hood meant trouble. I learned the hard way after one colleague opened a sealed vial without proper airflow. The sharp, rotten smell filled the workspace fast. Colleagues reacted, covering mouths and getting outside. For sensitive compounds like this, simple lab ventilation doesn't cut it. A dedicated chemical fume hood with direct exhaust is essential for safe use, not just a nice-to-have.
Proper Containers Matter More Than Labels
Toluene-3,4-dithiol eats through thin plastic over time and seeps through mineral-oil seals. Glass bottles with airtight, chemical-resistant caps kept leaks under control. Labels faded, but nobody ignored the hazard warning symbols and the physical safety data sheets next to storage shelves. Double-containment using secondary trays or bins helped prevent dangerous spills reaching work surfaces or drains.
Spill Response Starts with Training
A chemical spill tests everyone’s readiness. Training saved time and nerves. If you see a puddle or breakage, there’s no running for paper towels. Absorbent pads made for hazardous chemicals keep the mess from spreading, and every team I worked with made eye wash stations a short walk from every bench. Emergency showers close to the workspace made a difference, especially when panicked hands fumbled with gloves or didn’t remember to button lab coats.
Secure Storage Sends a Message
Locked chemical cabinets protected against unauthorized access. Restricted keycards or logbooks reminded staff that this compound isn’t handled lightly. Away from acids and bases, toluene-3,4-dithiol sat on shelves built for stability — no sagging, no uneven stacking. Storage areas faced periodic checks; any sign of leakage meant the whole tray came out for a deep clean or repacking. That diligence made the difference between an orderly workplace and a dangerous one.
Solutions from Building a Culture of Safety
Promoting strong safety culture starts with open, practical discussions, not just checklists. Repeated drills prepared teams to respond to accidents before panic set in. Regular review of chemical inventories helped prevent expired stock from causing surprises. Encouraging every team member to speak up if safety slips didn’t only protect them — it protected guests, contractors, and everyone passing nearby.
Using Authority and Clear Evidence
The facts are straightforward: handling organosulfur chemicals safely reduces injury, keeps the workspace functional, and shows respect for everyone present. Nobody who knows the effects wants shortcuts. Companies and research institutions now follow strict protocols based on decades of lessons, not because regulators force them to, but because real people learned those lessons the hard way.
What are the common hazards and safety precautions for Toluene-3,4-Dithiol?
The Risks Lurking with Toluene-3,4-Dithiol
Toluene-3,4-dithiol often lands in specialty labs and chemical manufacturing work, and the reality is—it’s no innocent bystander. Even at first glance, the pungent, sulfur-heavy chemical smell is a warning. Eyes and lungs don’t appreciate it. Touching the skin or catching a whiff without protection can leave you with strong irritation or a nasty headache. My first brush with it left my hands tingling despite gloves, and over time, I learned this is not the kind of irritant to ignore.
Inhaling fumes sends vapor straight into your body. Short-term exposure may bring on nausea and dizziness, and over long periods, it can affect the nervous system. At high enough levels, the body struggles: difficulty breathing, chest pain, and an odd taste linger. Eyes burn and water. Skin may blister. Nothing subtle here. Animal studies have shown clear toxic effects on organs when exposure runs unchecked.
The Fire and Spill Problem
Lab stories about toluene-3,4-dithiol spills make the rounds because the chemical’s low flash point means fire becomes a real danger. Any tiny spark or hot surface could ignite vapors. One drop on an exposed surface doubles as a slip and a fast way to a chemical burn. Most of us in labs learn quickly to check all bottle seals and keep sources of ignition far from storage cabinets. A mess made with this chemical does not wipe away like spilled water.
Spills pick up a strong, rotten-egg stench that clings to everything. If it gets into water systems, there’s a risk for wildlife and local drinking supplies. Ordinary cleaning won't help. Most places restrict drainage after a spill, and disposal requires a trained hand. I watched a careless drain dump cause a full shutdown for hours just to deal with a tiny puddle.
Smart, Everyday Precautions in Handling
The first rule has always been hard barriers: goggles, nitrile gloves, and lab coats. Not just for show. One slip-up with a cracked glove, and your skin pays for weeks. Fume hoods run constantly where this stuff is in use, and the lab air gets monitored for vapor build-up. A good practice is never working alone—someone needs to spot problems fast and call for help.
Every container carries clear hazard labels and gets double-checked before use. Spill kits designed for solvents and sulfur compounds stay close, stocked with absorbent pads, neutralizing agents, and heavy bags for disposal. Leftover or waste material never gets tossed in the regular trash; trained personnel pick it up, record the amount, and send it on for special handling.
Reducing Trouble Down the Line
Good engineering controls save more headaches than any warning sign can. It means using chemical-resistant benches and making sure any vent system works as it should. Investing in detector alarms for leaks helps. Teams stick to strict protocols and pay close attention to symptoms of exposure, no matter how minor. Training never stops.
Companies that depend on toluene-3,4-dithiol stay ahead by regular reviews, updating safety data sheets, and building a responsive culture that rewards vigilance. Only well-maintained equipment and clear emergency plans see routine success. You start seeing less “it won’t happen here” and more active discussions about hazards and response plans.
Keeping Focus on Safety
Dealing with toluene-3,4-dithiol means making respect for the chemical part of the daily routine. Good habits guard against hidden harm. Smart storage, careful handling, and transparent reporting never take a back seat. In the end, it’s personal mindfulness mixed with strong team habits that keep the lab out of trouble and everyone healthy.
Where can I purchase high-purity Toluene-3,4-Dithiol?
Access Challenges and Why Purity Matters
Tracking down a chemical like toluene-3,4-dithiol poses a unique challenge for researchers and industry folks alike. I remember searching for rare compounds during a research stint—frustration came more from logistics than from the science itself. Provenance and purity shape everything; you can’t trust results or build a process if what’s in the bottle only “sort of” matches what the label claims.
Chemicals with functional groups like thiols are notorious for sensitivity and odor issues—one whiff, and the whole lab knows someone’s been at work. Getting high-purity material stops experiments from going sideways due to leftover contaminants. Authenticity checks, certificates of analysis, HPLC and NMR data: these matter. Oddly enough, they’re what many sales reps may avoid talking about in fancy brochures. Look for the data, not the promise.
Where Suppliers Stand
Major chemical distributors keep toluene-3,4-dithiol on their books, but not on their open-facing storefronts. Instead, it’s almost always “procure upon request,” and often there’s a screening step. Sigma-Aldrich, TCI, and Alfa Aesar have broad catalogs. Custom requests go through specialists with compliance restrictions, especially for anything smelling like precursor material for more regulated compounds.
Those who work in academic settings know the game: a purchase order never moves alone. Expect multiple emails, sometimes old-fashioned phone calls, and requests for your project’s details. The endless paperwork might seem overkill, but the end goal matches Google’s E-E-A-T guidance—trust. You didn’t just walk down to the store. Someone’s vetting that you know what you’re doing.
Legal and Safety Pitfalls
Regulations around materials with strong thiols usually zero in not out of paranoia, but experience. In the 1990s, a simple flask could cause real headaches if someone spilled too much or sent it down the wrong drain. Handling and storing stuff like toluene-3,4-dithiol needs attention: think ventilated enclosures, tight packaging, access logs, and up-to-date MSDSs. If you haven’t spent an afternoon reviewing chemical hygiene plans and compatibility tables, now would be the time.
In my time, labs sometimes skipped steps and paid the price—corroded benches, emergency calls, and lost credibility. High-purity supplies cut risks on both data and health, but you still must keep the paperwork ready and the fume hoods clear.
What to Check Before Ordering
Digging into supplier details avoids trouble. Legitimacy goes beyond a polished website. Look for verifiable addresses, global certifications, and customer service that isn’t just a black hole for your inquiry. Request analytical data before payment. Peer groups, forums, and university resource offices sometimes have leads. Resist quick-fix options from grey-market suppliers; tempting as they appear, traceability gaps turn into real safety problems.
Best Practices and Alternatives
Sometimes custom synthesis houses with a solid track record step in, offering tailored batches and real-time progress updates. Costs go up, but reliability and safety usually follow. If supply dries up, skilled chemists know side routes and sometimes adapt their work without cutting corners.
High-purity toluene-3,4-dithiol isn’t just another bottle on a shelf. It shows how chemical supply, regulation, and research goals intertwine—built on more work, not less. For every serious project there’s a parallel need for skepticism, patience, and honest supplier relationships. If you can balance all three, you end up with something better than “off-the-shelf.” You get results you can actually trust.
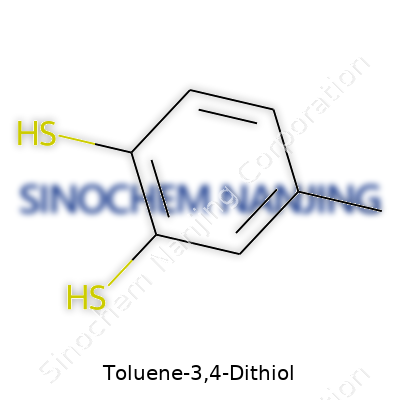
| Names | |
| Preferred IUPAC name | 3,4-Benzenedithiol |
| Other names |
3,4-Dimercaptotoluene 3,4-Dithiolotoluene 3,4-Toluenedithiol Toluene-3,4-dimercapto |
| Pronunciation | /ˈtəʊljuːˌiːn ˌθaɪˈaɪ.tɒl/ |
| Identifiers | |
| CAS Number | 496-73-1 |
| 3D model (JSmol) | `/assets/model-3d/JSmol/109-46-6.jmol` |
| Beilstein Reference | 1208527 |
| ChEBI | CHEBI:87211 |
| ChEMBL | CHEMBL523789 |
| ChemSpider | 251504 |
| DrugBank | DB14096 |
| ECHA InfoCard | 100.027.707 |
| EC Number | 205-030-9 |
| Gmelin Reference | 108137 |
| KEGG | C02526 |
| MeSH | D017929 |
| PubChem CID | 14703 |
| RTECS number | XZ3850000 |
| UNII | XIY1G1A3BO |
| UN number | 2810 |
| Properties | |
| Chemical formula | C7H8S2 |
| Molar mass | 156.26 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | unpleasant |
| Density | 1.27 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 1.95 |
| Vapor pressure | 0.36 mmHg (25°C) |
| Acidity (pKa) | 40.9 |
| Basicity (pKb) | 10.01 |
| Magnetic susceptibility (χ) | -51.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.626 |
| Viscosity | 0.913 cP (20°C) |
| Dipole moment | 2.11 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 331.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -7.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2456 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06, GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H373, H411 |
| Precautionary statements | P210, P233, P260, P264, P270, P271, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P337+P313, P362, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 266 °F (130 °C) |
| Autoignition temperature | Autoignition temperature: 535°C (995°F) |
| Explosive limits | Explosive limits: 1.2–7% |
| Lethal dose or concentration | Lethal dose or concentration: LD50 oral (rat): 400 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Toluene-3,4-Dithiol: 1600 mg/kg (rat, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1-10 mg |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Toluene Thiophenol Benzene-1,2-dithiol Toluene-2,5-dithiol 4-Methylcatechol |

