Tin Tetrachloride Pentahydrate: A Drive through History and Progress
Historical Development
Progress in chemical sciences moves hand in hand with the materials that fuel research. Tin tetrachloride pentahydrate reflects this journey. Its roots trace back to early laboratory work in the 19th century, just as industrial chemistry began to reshape society. Chemists recognized the value in tin compounds, especially for their reactivity with water and other elements. For a long time, tin tetrachloride drew attention mainly in the context of inorganic synthesis and the production of organic tin derivatives. Modern processing and analytical techniques have since unearthed more about its behavior, driving new uses across laboratory and industrial landscapes. Everyday exposure to this compound has broadened as technology touched more industries, with every era adding another layer to its reputation and uses.
Product Overview
Tin tetrachloride pentahydrate stands out in the family of tin chemicals for its unique physical build and responsive properties. Its chemical formula, SnCl4·5H2O, hints at a complex structure with a central tin atom surrounded by four chloride ions and hydrated with five water molecules. Unlike the anhydrous version, the pentahydrate resists aggressive hydrolysis, making it manageable in both storage and application. Technicians know right away the value this version brings when balancing reactivity and stability in their formulations. In coatings, catalysts, and electronics, handling properties matter almost as much as final performance. The pentahydrate offers a workable middle ground between tin’s raw power and the delicacy needed for precision tasks.
Physical & Chemical Properties
Physical characteristics set clear boundaries for how to handle and store tin tetrachloride pentahydrate. It appears as a white, crystalline solid, more prone to losing moisture to air than some expect on first encounter. Solubility becomes truly noticeable: in water, the compound dissolves with ease, but this often unleashes a sharp hydrolytic reaction that releases hydrochloric acid and forms tin dioxide. This reaction is no trivial matter for lab techs and engineers, since it transforms the working environment in seconds if left unchecked. Thermal stability stays moderate; higher temperatures drive off water in stages before the substance decomposes into the less manageable anhydrous chloride. The combination of volatile chlorine chemistry, willingness to react with bases, and affinity for moisture shapes both opportunities and hazards.
Technical Specifications & Labeling
Lab and industrial catalogues describe tin tetrachloride pentahydrate with extraordinary precision. Standard technical specs detail content purity (often upwards of 98%), granule size, pH in solution, solubility, and moisture content. Labeling calls for careful adherence to international regulations, such as GHS and REACH, to inform about dangers like corrosivity, environmental impact, and personal exposure risks. Product data sheets remind us to wear gloves, maintain proper ventilation, and store containers snugly closed in dry spaces away from heat sources. Good catalogues provide QR codes or batch numbers, so entire product lines stay traceable, which matters more than most realize when working with volatile chemicals.
Preparation Method
Production of tin tetrachloride pentahydrate leans heavily on the controlled reaction between tin metal and hydrochloric acid—a straightforward process at first glance. Applying heat, chemists bubble chlorine gas over tin, creating tin tetrachloride vapors. These dissolve in cold water, yielding a solution that crystallizes on cooling to form the pentahydrate. Every step involves close monitoring: excess chlorine or uneven cooling alters the hydrate content or leads to unwanted byproducts. Safety protocols come into sharp focus here, since exposure to concentrated acids and halogens brings risk of burns and respiratory distress. Large-scale producers automate portions of this process, installing sealed chambers, scrubbing systems, and temperature controls for precise outcomes.
Chemical Reactions & Modifications
Tin tetrachloride pentahydrate reacts vigorously with water, bases, and organic compounds. Water triggers immediate hydrolysis, generating hydrochloric acid and a white precipitate of hydrated tin oxide. This property anchors its use in complex syntheses, as chemists harness it as a Lewis acid catalyst. In organic chemistry, exposure to alcohols and amines opens new pathways, leading to alkoxy- or amino-tin derivatives that advance the manufacture of stabilizers, pharmaceuticals, and specialty resins. Chlorine exchange reactions, where the chloride groups swap for organics, build a bridge into tin-organic chemistry. Its ability to shift form fast underlines both its utility and the need for care—many industrial accidents root in underestimating the speed and heat of these reactions.
Synonyms & Product Names
Confusion often arises with chemical names. Tin tetrachloride pentahydrate travels under titles like Stannic chloride pentahydrate, Tin(IV) chloride pentahydrate, and SnCl4·5H2O. Some catalogues stamp additional branding for proprietary blends or tailored product lines. Whether searching English, German, or Chinese sources, it pays to keep alternate names in mind to ensure consistency in sourcing and handling. Product synonyms reflect subtle regional and commercial preferences, and missing one in a database means wasted time and sometimes disrupted research.
Safety & Operational Standards
Safety forms the backbone of real-world chemistry. Handling tin tetrachloride pentahydrate demands more than gloves and goggles—it calls for a clear plan for spills, leaks, or sudden exposure. The substance attacks skin and eyes, and its vapors irritate lungs and mucous membranes. I once worked beside a technician caught off guard by a cracked bottle, and the sudden, eye-watering sting of hydrochloric acid still sticks with me years later. Modern operational standards require training, emergency showers, tight labeling, and dedicated chemical fume hoods. Regulatory guidelines from OSHA, EU REACH, and local bodies set firm rules for transport, storage, and disposal. Responsible sites install continuous air monitoring, provide clear signage, and review emergency procedures not just once, but regularly.
Application Area
Industries rely on tin tetrachloride pentahydrate in several arenas. Paint and coatings manufacturers tap its reactivity to modify pigments and give more durable finishes. Glassmakers introduce tiny amounts in surface treatments to produce self-cleaning and low-reflective windows. Electronics firms use it in controlled environments to deposit thin layers of tin oxide, which serve as transparent electrodes in touchscreens and solar panels. In synthetic chemistry, laboratories embrace it as a powerful catalyst for assembling complex molecules, particularly where traditional catalysts fall flat. Water treatment researchers explore its interaction with organic pollutants, trying to unlock new strategies for purification. Its multi-faceted nature means every sector demands its own handling and regulatory plan, shaped by years of trial, error, and adjustment to evolving best practices.
Research & Development
The pace of research involving tin tetrachloride pentahydrate moves faster now than ever. Material scientists study its behavior in nanostructured films, looking for breakthroughs in next-generation electronics and flexible displays. Chemists continue refining catalytic cycles involving organotin intermediates, aiming for greener, safer processes that limit byproduct waste. Environmental scientists examine its breakdown pathways and seek scrubbing or neutralization options suitable for both industrial wastewater and lab-scale spills. Collaboration across industries and universities leads to new patents, giving a clearer picture of both chemical transformations and their environmental implications. This wave of knowledge-sharing pushes the compound into new, sometimes unexpected roles, blurring lines between old-fashioned chemistry and high-tech innovation.
Toxicity Research
Toxicologists place tin tetrachloride pentahydrate under close scrutiny due to its potential impacts on human health and the environment. Acute inhalation or skin exposure leads quickly to burns and irritation, while studies suggest repeated or prolonged contact increases risks of chronic lung or kidney problems. Animals exposed at high concentrations show clear changes in blood chemistry and organ function, underlining the need for rigid exposure limits. Environmental agencies classify tin compounds, especially those releasing free chloride or tin ions, as threats to aquatic life in some contexts. Ongoing research explores long-term effects at trace levels, particularly with rising concern over microcontaminants in water. Toxicity findings steer policy, push for safer workspaces, and shape the design of advanced waste treatment systems.
Future Prospects
Future roles for tin tetrachloride pentahydrate depend on maintaining a balance between utility and safety. Researchers see promise in harnessing its catalytic power for green chemistry, aiming to swap older, dirtier reagents for cleaner alternatives. Electronics continues to beckon, with the compound’s ability to build smooth, conductive tin oxide films supporting hopes for more efficient solar panels, flexible devices, and specialty coatings. Tightening regulations push companies to explore closed-loop recovery, reprocessing, and reduced exposure, so safety keeps improving alongside efficiency. Progress in computational chemistry and real-time sensors offers tools to predict reactions and mitigate risks before they cause harm. Looking back, every leap in understanding owes something to careful observation and adaptation. Peering forward, tin tetrachloride pentahydrate will likely remain in the toolbox—paired with stricter controls and deeper research guiding its use.
What is Tin Tetrachloride Pentahydrate used for?
Everyday Uses Hiding in Plain Sight
If you’ve ever handled glass coatings or read up on advanced electronics, you might have crossed paths with tin tetrachloride pentahydrate—even if you didn’t realize it. This compound pops up not only in research journals but also in real-world products that shape how we live. Stores rarely sell it by name on their shelves, but industries rely on its reactivity and specificity for solutions that need a precise outcome.
Behind the Science
Tin tetrachloride pentahydrate doesn’t just show up for kicks; it actually solves tough problems most of us don’t see. Glass manufacturers count on it for specialty coatings, especially those that block ultraviolet light. You’ll find it in lab settings where making pure tin oxide matters, whether for sensors, transparent electronics, or solar cell substrates. The stuff reacts quickly in water, so those tasked with working with it often wear gloves and goggles, keeping safety at the top of the mind.
A laboratory tech once told me about their first trial synthesizing transparent conductive films. The compound’s sensitivity to moisture forced them to adapt. Instead of working in a rushed environment, they had to slow down, check equipment, and pay close attention to the ambient humidity. In short, these kinds of chemicals don’t allow shortcuts if quality matters.
Hidden Value in Industrial Processes
Industries don’t just pick chemicals at random, and tin tetrachloride pentahydrate offers a few perks. It dissolves in a way that supports controlled hydrolysis, setting the stage for producing ultra-pure tin oxide. High-purity materials matter not only for tech gadgets but also for medical and analytical devices. Devices measuring trace heavy metals often need stable electrodes or glassware made under strict conditions—something this compound supports.
Environmental and Health Responsibilities
The flip side to robust chemicals is risk. Tin compounds can be hazardous in the wrong hands. For anyone using tin tetrachloride pentahydrate, storage routines and waste disposal can’t become afterthoughts. I learned pretty early in my science career that a forgotten bottle or sloppy labeling spells trouble, not just for workers but for water systems downstream of a disposal drain. Regulatory bodies like the EPA and OSHA keep companies on their toes, making sure protocols stay fresh and training stays up to date. High school labs rarely get their hands on this chemical; seasoned professionals keep a steady guard.
Possible Paths Toward Safer and Greener Practices
While tin tetrachloride pentahydrate plays a vital role, we can’t ignore calls for safer substitutes. Some research groups look at alternatives for glass coating, aiming for less corrosive or more biodegradable options. Others refine recycling methods, trying to recover tin and chlorine components for reuse. One promising angle highlights better waste capture systems, where companies collect vapors and neutralize acidic byproducts before they ever reach the environment.
If I had to sum it up: this is one of those chemicals that pushes us to ask hard questions about performance versus responsibility. Getting the benefit without the baggage means paying attention from start to finish—whether in the factory or in the research lab.
What is the chemical formula of Tin Tetrachloride Pentahydrate?
Unpacking the Chemical Formula
In the world of chemistry, accuracy matters, especially with formulas that can either clear up confusion or add to it. Tin tetrachloride pentahydrate, with the chemical formula SnCl4·5H2O, brings together a core of tin tetrachloride with five bound water molecules. This combination stands out. It’s not just about molecular weights or stoichiometry. The water molecules alter how the substance behaves.
Why the Pentahydrate Form Counts
Chemists and folks working with chemicals see a big difference between anhydrous and hydrate forms. That water really changes how tin tetrachloride can react, store, and even transport. In my days working in a chemical supplies warehouse, the pentahydrate version always showed up with more stable packaging than the dry form. We didn’t see the same corrosion or fume headaches with the hydrate. That’s the effect of those five locked-in H2O molecules.
You could pour it without getting overwhelmed by the fumes. That practicality matters on a busy floor. Companies that produce specialty coatings, for instance, put a premium on safety and ease of handling—which makes pentahydrate versions attractive. They don’t gamble with pure anhydrous material when the hydrate works.
Legacy and Modern Uses
People have leaned on tin compounds for centuries, stretching from the tin cans in old pantries to high-tech applications like glass coating and the electronics industry. Tin tetrachloride acts as a catalyst, a starting point for making other tin chemicals, and occasionally, as a disinfectant. Hydration doesn’t just dilute the substance. It can impact reaction rates, solubility, and shipping safety, with fewer headaches due to lower volatility.
Safety and Handling
Handling snappy chemicals brings calls for better safety. One exposure incident I watched stick with a coworker for years. Tin tetrachloride itself produces clouds of hydrochloric acid if spilled in humid air. The pentahydrate form tempers this effect. You’ll find fewer ugly surprises if a container leaks, although gloves and eye protection still remain the rule. According to safety sheets, pentahydrate forms are less aggressive, but there’s no room for shortcuts.
Supply Chain and Purity Issues
Labs and factories chase purity numbers, but hydrate forms can complicate calculations. Five water molecules add to the mass. Recipes change if you swap out the anhydrous for the pentahydrate. Distributors label drums accordingly, but mix-ups still happen. I’ve fielded frustrated calls from lab techs who expected 100% SnCl4 and got a surprise—yield calculations flew out the window. Training and clear labeling save a lot of headaches and wasted materials.
Looking Forward
Industries lean on the pentahydrate for its blend of safety and function. Demand for careful labeling and worker safety keeps growing. Manufacturers keep tightening up purity specs while balancing costs related to extra water weight. Solutions to persistent mix-ups come down to keeping a steady line of communication between producers, shippers, and end-users. People need clear chemical names and correct formulas, and it's worth pushing for robust education at every step.
How should Tin Tetrachloride Pentahydrate be stored?
Real Hazards Require Real Precaution
Tin tetrachloride pentahydrate may sound like a chemical only chemists need to worry about, but plenty of professionals across academic labs, industry, and education rely on it. Once you’ve worked around chemicals that can bite back, following basic storage rules starts to feel like an act of respect—for both the material and those who handle it. Storing tin tetrachloride pentahydrate takes attention to detail, genuine caution, and a commitment to building a safe working environment. A single oversight, like leaving the wrong cap off or ignoring a leaky bottle, could put workers at risk or set off a larger incident.
Moisture and Tin Tetrachloride Pentahydrate Don’t Mix
This compound absorbs water from the air, which makes it tricky to store. If you let moisture in, the tin tetrachloride pentahydrate can decompose. The fumes that result—mainly hydrochloric acid—damage not just property, but also your lungs and eyes. Most labs keep it in tightly sealed, corrosion-resistant bottles. Glass or high-quality plastic containers work well. I learned quickly in my time working in a college lab that sealing chemicals is more than good practice—it saves your nose and skin from awful burns and keeps the air in your workspace clear.
A dry, cool location helps maintain stability. Fluctuating temperatures push more water vapor into the air, which spells trouble for sensitive chemicals. In reality, few shop-bought cabinets are enough. Ventilated and climate-controlled storerooms show up in well-funded labs for exactly this reason. If budget goes tight, at least tuck the bottle away from sources of humidity—sinks, radiators, or exterior doors. Label shelves clearly and keep the storage area clean to avoid cross-contamination.
The Problem with Ignoring Risks
Tin tetrachloride pentahydrate doesn’t just attack skin and eyes. Any accidental spillage releases hydrochloric acid vapors, which can eat through metal shelving and equipment. That costs real money and halts experiments. Worse, vapors can drift toward unsuspecting colleagues. Old incidents still come up in workplace safety meetings—one employee staggers from a store room clutching their face, and everyone gets a sobering reminder to double-check lids. Strong containers don’t just satisfy bureaucrats—they keep the workplace running and prevent painful accidents.
Practical Steps for Everyday Storage
- Use original packaging or a tightly sealed replacement. Never transfer to unlabelled containers.
- Store in a dry, cool space—preferably a locked chemical cabinet away from heat and humidity.
- Keep away from incompatible materials: bases, water sources, and oxidizers pose serious risks if mixed.
- Line the storage area with chemical-resistant trays. These catch any spills before they become disasters.
- Make safety equipment easy to access. Goggles, gloves, and a fume hood should be steps away, not a walk down the hall.
- Label storage shelves clearly. This speeds up inspections and helps new staff avoid rookie mistakes.
Training Makes All the Difference
No set of rules replaces training. Staff who handle tin tetrachloride pentahydrate should practice dealing with emergencies—like spill kits and eyewash stations—with real products. Talk through chemical hazards and what can really happen if water or other reagents spill nearby. The human memory holds onto stories of accidents as much as procedures in a binder.
Building Trust Through Responsibility
Handling dangerous materials builds a culture of care. Storage isn’t some formality—it’s the shield between routine work and disaster. Keeping tin tetrachloride pentahydrate in a properly prepared space fits neatly into the bigger picture: protecting people, safeguarding research, and avoiding the kinds of mistakes that lead to regret.
Is Tin Tetrachloride Pentahydrate hazardous or toxic?
The Real Dangers Behind Tin Tetrachloride Pentahydrate
People rarely stumble across tin tetrachloride pentahydrate at hardware stores or in the kitchen cupboard. It’s a specialty compound, mostly popping up in labs and certain factories. Still, most chemicals that show up behind locked cabinet doors didn’t get there by being easy to handle. This one’s got a reputation, and it’s worth looking at why.
Handling Tin Tetrachloride Pentahydrate is No Joke
Every bottle, container, or drum labeled with tin tetrachloride pentahydrate brings an edge of risk. Splash some on your skin and burning or blistering can follow. Your eyes get in contact – expect searing pain, maybe blurred vision, possibly worse. Its fumes don’t go easy on respiratory systems either; just a whiff can set off coughing, choking, and sometimes more severe lung irritation. Those who’ve worked chemical cleanups learn respect quickly after one mistake.
The main player in its hazards is the reaction with water. Mixed with even small amounts of moisture, tin tetrachloride pentahydrate releases hydrochloric acid, and that stuff is corrosive. A few drops on a countertop spell trouble if not wiped up quickly. Inhalation of the fumes can send workers rushing for fresh air and maybe medical attention. CDC and NIOSH both list it under substances to watch closely in workplaces. These are not agencies known for being alarmist.
The Facts Don’t Paint a Pretty Picture
Some folks might think a little chemical here or there gets diluted and forgotten by the body. Tin tetrachloride pentahydrate does get absorbed. At high exposure, animal studies have linked it to respiratory problems, gastric irritation, and sometimes organ damage. No one wants to find out firsthand what chronic exposure might do. The International Chemical Safety Cards (ICSC) and European Chemicals Agency classify it as hazardous to health.
For the environment, it’s not exactly a friend. Leaks or spills near water sources can lower pH rapidly, threatening aquatic life. Any release to the outside gets flagged, with cleanup teams wearing proper gear. In cities with aging pipes or limited wastewater controls, careless disposal multiplies the risk.
Taking Steps to Limit Harm
Solving the safety issue isn’t about sounding alarms for every beaker of chemicals. Smart habits and clear rules make a running difference. Proper gloves and tight-fitting goggles stop most mishaps before they go from story to hospital trip. Ventilated spaces, chemical fume hoods, and regular spill drills earn their keep fast in any workplace using tin tetrachloride pentahydrate. Emergency showers and eyewash stations should be nearby; no one expects an accident until it happens.
Supervisors and safety trainers ought to keep information handy, easy to read, and up to date. Material Safety Data Sheet (MSDS) copies belong out in the open, not buried in a dusty drawer. A good policy reaches home too: label everything clearly so nobody mistakes one jar for another during a hectic day.
Most lab workers have stories that remind them of the cost of shortcuts. The key is learning from each minor incident so nobody faces a major one. Respecting tin tetrachloride pentahydrate means understanding it and giving it the precautions it deserves—not letting fear set rules, but knowledge. Anyone working with chemicals owes it to themselves and their crew to keep hazards in check.
What precautions should be taken while handling Tin Tetrachloride Pentahydrate?
Why This Chemical Demands Respect
Tin Tetrachloride Pentahydrate shows up in research labs and industry plants more often than most people realize. It serves as a key player in producing organotin compounds, catalysts, coatings, and sometimes glass etching. The real concern is how harshly this compound reacts with moisture and skin. My time in a university lab taught me not just to read safety data, but to watch seasoned techs treat chemicals like this as if they were snakes—unpredictable and dangerous if you get careless.
A Glimpse into Real Risks
Spilling a few drops on the tile releases fumes that burn your nose and sting your eyes. The hydrate acts much like the anhydrous form. Tin Tetrachloride Pentahydrate reacts with water to form hydrochloric acid, which can damage skin and eyes in moments. Even a quick brush with the liquid leaves a white, irritated patch. Statistics reported by chemical safety agencies show burns and respiratory irritation among the top health complaints after exposure. This is no bench toy, and a casual attitude often turns into regret.
Essential Precautions Everyone Should Follow
Wear protective gear: Gloves, goggles, and even a lab coat stop most accidents from turning into medical crises. Nitrile gloves and splash-proof goggles did more than follow a checklist in my experience—they protected my hands and vision day after day. Nitric acid-resistant aprons take this up a notch in industrial settings.
Use fume hoods: Ventilation isn’t optional. The smell hits hard even from a distance. Good hoods suck up vapors fast, stopping that burning in the throat and eyes. Your regular office fans don’t cut it; only dedicated chemical hoods get the job done for real safety.
Handle with dry tools and containers: Water turns neat crystals or solution into an acid-spewing mess. Dry glass vessels and plastics prevent surprise reactions. Some colleagues kept silica packets around just to be sure everything stayed bone-dry between uses.
Keep spill kits nearby: Accidents happen even to experts. Spilled tin tetrachloride pentahydrate shouldn’t wait for a supervisor to clear it. Labs and plants stock calcium carbonate or other neutralizers right at the bench for fast action. Paper towels don’t solve this sort of mess—you need neutralizer and a waste container labeled for hazardous material pickup.
Label and store correctly: Clear labeling with hazard symbols and date breaks confusion, especially during late-night work or shift changes. I’ve seen coworkers nearly mix up bottles because a faded label hid behind a stack of flasks. Leak-proof, sturdy bottles stand up to the known corrosiveness and keep vapors locked down.
Learning from Those Who Came Before
Stories circle in labs about a friend or professor splashed by a “small” spill. There’s usually a moment of panic, a dash to the eyewash station, and then a call to campus health. It doesn’t take a big spill or years of exposure for these risks to show their hand. The real lesson relies on learned habits and steady attention to each step, rather than trusting luck or shortcuts.
Solutions That Actually Work
Training newcomers with hands-on demonstrations rather than dull PowerPoint reveals the seriousness of this work. Refresher workshops each year keep skills sharp. Industry reforms in the last decade brought better ventilation systems and required more thorough labeling. These changes support safer workplaces—not just because regulators ask, but because people have seen what’s at stake.
Tin Tetrachloride Pentahydrate: Handle with Care
Chemicals like these make modern manufacturing and science possible, but they demand respect. Facing the dangers with the right equipment, procedures, and mindset keeps everyone healthy and productive. The safest labs rely on strong protocols, practical training, and a healthy layer of caution learned from experience.
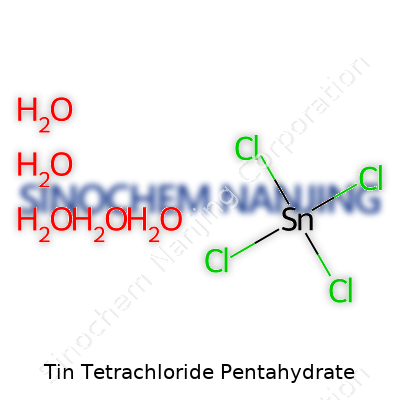
| Names | |
| Preferred IUPAC name | pentaaquatetrachloridostannum(IV) |
| Other names |
Stannic chloride pentahydrate Tin(IV) chloride pentahydrate |
| Pronunciation | /ˈtɪn tɛtrəˌklɔːraɪd ˌpɛntaɪˈdreɪt/ |
| Identifiers | |
| CAS Number | 10026-06-9 |
| Beilstein Reference | 3926152 |
| ChEBI | CHEBI:75956 |
| ChEMBL | CHEMBL1201820 |
| ChemSpider | 20792950 |
| DrugBank | DB14578 |
| ECHA InfoCard | 100.028.834 |
| EC Number | 231-588-9 |
| Gmelin Reference | 7787 |
| KEGG | C18699 |
| MeSH | D013975 |
| PubChem CID | 159410 |
| RTECS number | XU7176000 |
| UNII | 289U35YC92 |
| UN number | UN3266 |
| Properties | |
| Chemical formula | SnCl4·5H2O |
| Molar mass | 338.60 g/mol |
| Appearance | Colorless crystalline solid |
| Odor | Pungent |
| Density | 2.36 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.1 |
| Vapor pressure | 14 mmHg (20 °C) |
| Acidity (pKa) | -1.5 |
| Basicity (pKb) | -4.8 |
| Magnetic susceptibility (χ) | \-63.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.619 |
| Viscosity | 1.87 cP (20 °C) |
| Dipole moment | 4.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 242.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −900.9 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Corrosive, causes burns to skin and eyes, harmful if swallowed, inhalation may cause respiratory irritation |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H335 |
| Precautionary statements | P260, P264, P271, P280, P301+P330+P331, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 2 1 0 |
| Lethal dose or concentration | LD50 oral rat: 750 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1000 mg/kg |
| NIOSH | WW5250000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Tin Tetrachloride Pentahydrate: 2 mg/m³ (as tin) |
| REL (Recommended) | Store at +2°C to +8°C |
| Related compounds | |
| Related compounds |
Hydrated Metal Halides Tin(II) Chloride Tin(IV) Chloride Tin(IV) Bromide Lead(IV) Chloride |

