Thioglycolic Acid: A Closer Look at its Story, Science, and Promising Future
Historical Development
Thioglycolic acid traces its commercial story to the mid-20th century, right around the time personal care and industrial chemistry started mingling in daily life. Early researchers noticed the unique way it broke down disulfide bonds, and it didn’t take long before big trends like home hair waving and chemical depilation took off, powered by this pungent, colorless liquid. The acid soon captured a spot in the chemical supply world beyond just beauty, thanks to its reactivity and useful sulfur content. While early uses sometimes skirted thorough safety testing, growing regulatory oversight over the decades forced the field to reckon with careful controls and transparency. This tightening has pushed manufacturers and scientists to innovate on purification, synthetic routes, and product safety, shaping thioglycolic acid into a substance that stands as much for regulatory learning as for chemical function.
Product Overview
Thioglycolic acid goes by several names: mercaptoacetic acid, TGA, or just the “perm acid” in salons. In laboratories, this compound changes the game thanks to the balance between its carboxylic acid and thiol groups. It has earned a seat at the table in industries looking for a powerful bond-breaker or a reliable chelating agent. Demand fluctuates between fields like cosmetics, metallurgy, and polymer work, often driven by regulatory shifts or a spike in consumer self-care trends. Different grades get tailored for specific markets, with purity and free-acid content setting boundaries. The acid keeps finding new niches, as innovators discover ways to use its reactiveness in emerging technologies and greener chemical processes.
Physical & Chemical Properties
Thioglycolic acid usually shows up as a clear liquid with a sharp, unpleasant odor that’s hard to forget once you catch a whiff. Its solubility in water makes it easy to handle in both lab and industrial setups. The molecule stands out for its mix of sulfhydryl and carboxyl groups, giving it a split personality that drives unique reactivity. Concentrated solutions corrode metals and damage skin fast, giving a stark reminder about the need for safe handling. It boils at around 100 degrees Celsius under reduced pressure, and high acidity means it can lower pH quickly, which becomes both a key feature in reactions and a challenge in wastewater management. In personal use products, that same reactivity means that the smallest impurities or formulation errors show up in action — the classic unmistakable “perm” smell in hair salons owes its bite to the acid itself and not some mystery additive.
Technical Specifications & Labeling
Labels on thioglycolic acid containers often highlight its concentration, often ranging from 60% to full strength for industrial reactors, and usually cut down below 15% in consumer products. Alongside warnings, labels stress the need for gloves, goggles, and proper waste disposal. Technical datasheets talk up its molecular weight, solubility, and storage tips, but behind those numbers stands a patchwork of global rules chased by every exporter and formulator. Regulations in Europe, North America, and parts of Asia differ on concentration limits and permissible uses, making compliance a maze for global production. For researchers, all these numbers translate to day-to-day habits: rack placement, acid neutralizers nearby, and always keeping fresh air flowing in the workspace.
Preparation Method
Industrially, thioglycolic acid grows from a reaction between sodium chloroacetate and sodium hydrosulfide, followed by acidification. The process relies on sharp temperature and pH controls to yield a clean product, and technical teams invest effort into minimizing side reactions that can lower purity or generate foul-smelling byproducts. Synthetic tweaks have trimmed waste and improved yield over the years, but large-scale plants continue to juggle worker safety, vapor containment, and costs. At the bench, smaller batches mean easier handling, but even home laboratory enthusiasts know not to underestimate the risks; leaks or accidental splashes give a quick lesson in chemical potency. Water dilution becomes essential before most downstream processing, letting companies ship less hazardous forms while keeping the chemistry almost the same.
Chemical Reactions & Modifications
With a hook to both acids and thiols, thioglycolic acid steps into numerous reactions. It breaks peptide bonds, reduces disulfides, grabs metal ions in solution, and acts as a chain transfer agent in polymerizations. Chemists have developed derivatives by tweaking the molecule, swapping out hydrogens and adding functional groups, to give rise to drugs, specialty resins, and even certain agricultural chemicals. Every industrial process that leans on disulfide bond reduction — hair salons, wool dyeing, leather work — borrows from the same basic reactivity but adapts it to different scales and constraints. Its versatility means even small adjustments in reaction conditions, like tweaking the pH or adding stabilizers, can yield new products with distinct characteristics.
Synonyms & Product Names
Anyone shopping for this acid likely encounters an army of aliases. Chemical catalogs list it under mercaptoacetic acid, sometimes as carboxymethyl mercaptan, and occasionally with abbreviations like TGA. Commercial mixtures, especially those tailored for salons, appear as part of product blends labeled “perm lotion” or “depilatory cream,” where regulations force prominent placement of safety warnings. In the research and industrial world, nearly every distributor has a different product code or variant, meaning even seasoned buyers keep an eye out for cross-checking supplier numbers. Industry jargon varies by region, which has tripped up more than a few cross-border projects over the years.
Safety & Operational Standards
Strict safety protocols developed around thioglycolic acid, driven by a track record of corrosive injuries and environmental spill concerns. Handling concentrated acid without full PPE courts disaster, even from the smallest splash. Fume extraction systems aren’t optional, especially in busy labs where heat ramps up volatility or containers might tip. In industrial settings, automated dosing and closed reaction vessels have become the norm after well-documented exposure incidents. Regulatory authorities require detailed reports on spill planning and waste treatment, and many production sites now run real-time air monitoring where the acid gets sprayed or mixed. Training forms the backbone of safety culture, and repeat drills keep the lessons fresh. Wastewater from manufacturing plants passes through neutralization steps before hitting treatment plants, a move demanded by both local ordinances and broader environmental standards. Failure to follow the playbook racks up fines and, more importantly, puts workers and communities at real risk.
Application Area
Thioglycolic acid found early fame in the world of hair perming and straightening, where it broke the strong disulfide bonds in keratin, letting new shapes hold. Today, the personal care sector still draws much of the acid’s production, but other industries have latched on to its unique properties. Many metal processing plants use it as part of cleaning and etching baths, where the acid’s bite removes oxide or scale and preps surfaces for plating or bonding. In oilfields, field workers pump derivatives into wells to clean drill pipes and improve flow, taking advantage of the acid’s ability to dissolve certain deposits. Other industries, like leather processing and photography, exploit its reactivity for controlled modifications and precise work. Each field demands tweaks in formulation and handling, but the acid’s basic chemical punch stays at the core. Agriculture and electronics research continues chasing potential new uses, with specialty polymers and metal ion scavenging as research frontiers.
Research & Development
Recent years brought a rush of R&D targeting safer derivatives and cleaner production routes. Academic labs have tried enzyme pathways and novel catalysts, hoping to cut out traditional byproducts and energy costs. There’s a growing push for bio-based feedstocks that could replace petroleum-derived precursors, which would shrink both the carbon footprint and downstream pollution. Another active area looks for safer, less-volatile substitutes for certain personal care applications, answering consumer calls for milder, less hazardous products. Analytical methods for detecting trace residues in wastewater have become more sensitive, letting environmental agencies keep a sharp eye on industrial emissions and keeping companies on their toes. Collaborative work between academic chemists, process engineers, and regulatory experts happens more than before, leading to smarter controls and, occasionally, breakthrough approaches that ripple out across multiple markets.
Toxicity Research
Studies over the past few decades made it plain that thioglycolic acid poses both acute and chronic hazards. Even at relatively low concentrations, skin exposure leads to chemical burns, and inhalation of vapors irritates membranes and lungs. Animal studies flagged risks from repeated exposure, showing impacts on red blood cell formation and organ function at high doses. Regulatory risk assessments now cap allowable concentrations in over-the-counter products and limit industrial emissions sharply compared to the less-stringent rules of previous eras. Monitoring blood levels in workers who regularly handle thioglycolic acid forms part of some site protocols, backed by research that linked long-term uptake with subtle metabolic shifts. Community health activists, particularly in countries with bustling personal care industries, lobby hard for transparent ingredient lists and routine inspections, aiming to keep both workplaces and the public shielded from accidental or careless exposures.
Future Prospects
Scrutiny and innovation shape the road ahead. The strongest momentum right now comes from efforts to keep the chemical’s function while lowering its footprint and hazard. Improved reactor designs, sharper process controls, and bio-based syntheses all push the needle toward greener chemistry. In the personal care sector, industry newcomers tout perms and depilatories with “next-gen” molecules inspired by thioglycolic acid but tuned for lower volatility and toxicity. Researchers probe for applications in medicine, where thiol chemistry could deliver targeted drug action with fewer side effects, and in electronics, where precision etching and cleaning with minimal waste can spell real cost savings. Every major shift in regulation — whether on emissions, ingredient transparency, or worker protection — pushes the community toward innovation. The next decade will likely see thioglycolic acid’s role continue to evolve, driven as much by new science as by relentless pressure to do more with less risk to people and the planet.
What is thioglycolic acid used for?
What’s Behind the Name?
Thioglycolic acid doesn’t sound inviting or familiar, but it shapes the lives of millions without much fanfare. People might not realize how often they interact with it, especially at salons or in the comfort of their own bathrooms. For decades, salons have reached for bottles containing this chemical to help deliver softer, straightened hair. At home, many reach for it again, thanks to its central role in creams marketed for hair removal.
Chemistry at Work in Everyday Beauty
The biggest chunk of thioglycolic acid sinks into beauty treatments. Wavy hair turns straight or curly, leg hairs vanish—all because thioglycolic acid can bust through the tough bonds that hold each hair strand together. In simple terms, it knocks out the keratin structure, giving hairdressers a way to break curls or consumers a pain-free alternative to shaving. My younger cousins often laugh about my post-college “perm disaster,” a moment I can now blame on thioglycolic acid, not just the stylist’s heavy hands. That experience taught me anyone using chemical treatments needs to read the label and ask questions, because these ingredients are no joke.
Why It Isn’t for Everyone
Some people react badly to thioglycolic acid in personal care products. Skin can get irritated or burn if left too long. Cosmetic chemists must balance its strength, so it removes hair without crossing the line into damage. Long after the first creams hit the market in the 1940s, stories still pop up about rashes and reactions. Dermatologists repeat the same advice each year: patch test on a small area before using on larger patches of skin.
Bigger Uses in Industry
Thioglycolic acid’s reach stretches far outside bathrooms and salons. Car parts, water pipes, and oil wells also get their dose. In these fields, thioglycolic acid attacks rust and cleans metal by stripping away built-up deposits. Factories use it to stabilize plastics and keep some types of electronics working smoothly. In water testing, labs turn to it to help measure iron and other trace metals. It’s not a one-trick pony, and that’s part of the reason it continues to draw attention from both regulators and health experts.
Safety Stakes Keep Rising
Questions about safety follow any chemical with such a mix of uses. Thioglycolic acid doesn’t build up in the body when used in small amounts, but accidents can occur if workers or home users don’t pay attention to instructions. Regulatory groups in Europe, the U.S., and Asia have all set strict limits in products. Wastewater loaded with leftover thioglycolic acid harms aquatic life. Schools and workplaces now focus more on safety lessons and protective gear, aiming to limit direct contact and accidental spills.
Looking for Solutions
Product makers keep chasing better answers—ingredients that can remove hair or clean metal without the side effects. Some are swapping in new plant-based formulas, though they don’t always work as fast. Innovation takes dollars and time, but public demand for safer and healthier options keeps industry on its toes. From where I stand, it makes sense to stay cautious, ask about ingredients, and support businesses investing in transparency and safety. For now, thioglycolic acid keeps solving problems quietly, in products that rarely earn a second thought during a morning routine or at a factory bench.
Is thioglycolic acid safe for use on skin or hair?
Looking at the Science and the Real-Life Risks
Walk into any drugstore and you can find hair removal creams and perm lotions promising quick, smooth results. Behind these products, there’s often a chemical called thioglycolic acid. Chemists use it to break apart the bonds in hair, so that old-school waving and removing hair becomes easy. The convenience comes with questions, though. Is it worth putting this stuff on your skin or hair? Let’s pick it apart and see what people should know before slathering on a lotion with this compound.
What Doctors and Scientists Say
Dermatologists have kept an eye on thioglycolic acid for decades. It works fast because it slices through disulfide bonds in hair’s structure, basically loosening hair at the root. This speed draws in customers, but it’s also where the trouble starts.
I’ve known folks who came away from a hair removal cream with red, irritated patches for a week. Skin absorbs thioglycolic acid on contact, and if someone leaves it on longer than the box says — or uses the cream on broken or sensitive skin — things get rough. Medical literature backs this up, showing that allergic reactions, rashes, and burns happen more often than most realize. The FDA sets strict limits on concentration, usually below 15% for home use, to prevent deep tissue injury. Still, salon professionals sometimes reach for formulas twice as strong, trusting training that not everyone shares.
Who Faces the Greatest Risks?
From personal experience, not all hair or skin types react the same. Folks with sensitive skin, conditions like eczema, or a history of allergies tend to see more drama with thioglycolic acid. Kids and teenagers, too, face more risk because their skin’s thinner.
According to the American Academy of Dermatology, even people with “tough” skin can end up with sores or blisters if they ignore the instructions. Warm showers before use, too much cream, or using it on the face spells trouble.
Long-Term Effects
Most people focus on the rash or sting that shows up right after using a depilatory. Scientists say repeat exposure over months could thin the skin, sometimes cause pigment changes, and even make some people sensitive to other ingredients down the line. Cases of long-term issues are rare compared to the millions of uses, but scars and color patches can last for years.
Better Choices and Improvements
A patch test saves a lot of misery. Rubbing a small dab of product on the inside of an elbow twenty-four hours before going full-on offers the best early warning. Creams with aloe or less acid-heavy ingredients now crowd store shelves for a reason. Brands that list full ingredients and concentrations offer a layer of trust that random knock-offs can’t match.
Salons and at-home users both benefit from gloves, timers, and cool water rinses. There’s no harm in swapping thioglycolic acid-based creams for sugar waxing, epilators, or simple shaving, especially where the risk seems high.
Why It Matters
People value smooth skin and easy hair removal, but they shouldn’t trade safety for speed. Reading labels, checking concentrations, and listening to your body matters every time. Manufacturers and stylists both play a role, sharing better education and transparency. Chemical shortcuts can save time, but the side effects sometimes linger far longer than the stubble.
How does thioglycolic acid work in hair removal products?
What Really Happens With Chemical Hair Removal
Pulling up a tube of depilatory cream, I remember high school days, that tight smell, and hoping for smooth skin before pool parties. Like many people, I gave up waxing and shaving for those hair dissolvers. At the center of the action sits thioglycolic acid. People rarely know much about it, but it runs the show whenever hair melts off after a few minutes with a cream or gel.
Thioglycolic acid packs a punch. It’s a small molecule with a sulfur group, and that sulfur does more than clear a room; it breaks the bonds that keep hair sturdy. Every strand of hair uses keratin as its backbone, which relies on disulfide bridges for strength. Thioglycolic acid steps in and cuts those bridges. With those bonds gone, hair fibers collapse and let go from the root. This goes deeper than a shave. Razors just slice at the surface, leaving the tip behind. Chemical removers break hair at a molecular level—suddenly that “melted” texture makes sense.
Not All Smoothness Is Created Equal
It’s no secret: chemical hair removal is quick, painless (usually), and cheap. A study from the International Journal of Cosmetic Science showed thioglycolic acid could break down hair within five to ten minutes. For coarse growth, it sometimes takes a bit longer, but most products hit a sweet spot between skin safety and effectiveness. Overuse or ignoring the instructions leads to burns. Speaking from experience, that red patch sticks around longer than smooth skin ever does. Product labels warn about this, and the rules matter here.
Many folks turn to these creams for accessibility. Not everyone can shell out cash every month for waxing or splurge on laser treatments. Thioglycolic acid makes it possible for someone to get smooth legs, arms, or even facial hair gone at home. For people with mobility challenges or sensitive skin, this option gives some independence. Dermatologists say allergic reactions remain relatively rare, but folks with eczema or damaged skin need to be careful.
Getting to the Root of Safety and Sustainability
The challenges with thioglycolic acid don’t end with personal burns or rashes. Most products use a mix of stabilizers, fragrances, and bases to buffer the harshness of the acid. Some of these additions cause irritation or trigger allergies. The smell often sticks around because sulfur-based compounds never go unnoticed. Companies try covering it up, but anyone who’s ever used a depilatory remembers that whiff. A 2021 review published in “Dermatitis” pointed out that a chunk of reported reactions came from those extra ingredients, not always from thioglycolic acid itself.
Waste matters, too. The leftovers from hair removal creams usually end up in the trash, and some of those chemicals head down the drain. These products can contribute to water pollution. Environmental agencies in different countries keep an eye on personal care chemicals, and stricter regulations have started taking shape. Companies push more biodegradable formulas now, which feels like a step in the right direction.
What Could Make It Better?
Looking at the big picture, chemical hair removal sticks around because it works fast and doesn’t break the bank. If manufacturers keep improving buffers and swap out harsh additives for gentler ones, the user experience will improve. Public info campaigns and clear labeling can protect people with allergies or vulnerable skin. On the green front, R&D teams in the personal care industry test plant-based or less abrasive ways to break keratin bonds. Each new product comes with its pros and cons, but no easy fix yet matches the easy, at-home feel that thioglycolic acid offers. As long as people want a shortcut to smooth skin, the chemistry challenge continues.
Are there any side effects or risks associated with thioglycolic acid?
What Thioglycolic Acid Really Does
Thioglycolic acid pops up anywhere from beauty salons to industrial workplaces. You’ll see it labeled in hair removal creams, hair perms, and lab supply catalogs. The stuff packs enough punch to break down tough protein bonds. Quite effective for its jobs, but that doesn’t mean it comes risk-free.
Everyday Exposure: At Home and in Salons
If you’ve ever used depilatory creams, you may have felt a burning or tingling feeling. Redness and skin irritation show up often, especially for folks with sensitive skin. The acid works by literally breaking the structure of hair, but it doesn’t always stop there—it can break through the outer layer of your skin, too. That stinging is your body’s early warning system. Washing the product off right away usually solves the problem, but things can get ugly fast if you leave it on longer than the recommended time.
Some people come away with allergies after a few exposures. Symptoms range from mild rashes to severe swelling. In rare but real cases, breathing in thioglycolic acid during application may set off asthma-like symptoms, especially in salon workers who handle the chemical every day. Monitoring for strong odors and adequate room ventilation makes a difference but isn’t always enough for everyone.
Serious Health Risks and Long-Term Concerns
Thioglycolic acid can do more damage at high concentrations. Industrial-grade products often contain levels that burn or corrode skin on contact. Even diluted versions can cause chemical burns or blisters if misused. Eyes need special protection, as splashes have led to vision problems or even blindness in accident reports.
Repeated exposure calls for caution. Occupational studies linked regular workplace contact to skin conditions like dermatitis and respiratory issues. The National Institute for Occupational Safety and Health classifies thioglycolic acid as a hazardous substance, and the U.S. Environmental Protection Agency keeps it on its watchlist because of potential health and environmental effects.
Why the Risks Matter and What to Do
Products that deliver fast results usually get people’s attention, but the drawbacks deserve equal notice. Over-the-counter versions slip through with warnings on the back of the bottle, but real safety depends on following instructions. Dermatologists regularly remind their patients to patch test before putting anything new on a large area of skin.
Some sectors have called for stricter labeling and better public education. Salons and factories that use thioglycolic acid benefit from improved ventilation, gloves, and eye protection. Proper disposal helps keep this chemical out of waterways, which means less harm for fish and aquatic plants. Schools and training programs that focus on safe chemical use also support long-term wellbeing for workers in cosmetology and manufacturing.
The Case for Respect and Precaution
Treating chemicals with respect comes from understanding what can go wrong. Thioglycolic acid gets the job done in many products, but respect for the risks should follow every use. Focusing on safety—at home, in salons, or on factory floors—protects both users and the environment. With knowledge, people steer clear of trouble and still get the results they want.
Can thioglycolic acid be used on sensitive skin?
Trying Out Hair Removal Creams With Sensitive Skin
Shopping for a hair removal product can feel like gambling if your skin stings easily. I remember my first teens; I grabbed a drugstore depilatory and thought I’d found a shortcut to smooth legs. Minutes later, my calves turned red, spots burned, and my bathroom smelled like rotten eggs. I only learned later that thioglycolic acid drives these fast-working creams, breaking down hair’s proteins at the root. The same action cuts through keratin, but sometimes it doesn’t stop at the hair — it can attack the skin too.
Thioglycolic Acid Works — Sometimes Too Well
Waving away hair in minutes sounds good on a commercial, but sensitive skin can pay a price. Thioglycolic acid gets its power from breaking sulfur bonds in the hair, turning once-tough strands into jelly you wipe away. Sensitive skin, by its nature, can’t put up much of a fight against chemicals. Redness, itching, stinging, and even blisters can follow if the formula’s too strong or left on too long. It’s not just unlucky first-timers — dermatologists get plenty of folks who react even using “for sensitive skin” versions.
Listening to Science on Safety
Clinical studies back up these complaints: chemical burns and allergic reactions happen, and the risk gets higher with repeated use. Reports from the American Academy of Dermatology make it clear: thioglycolic acid’s power comes with a flip side. Hair removal creams work, but they can disrupt the skin’s natural barrier. That allows germs in and moisture to leak out, which sets up peeling, inflammation, and sometimes infections. If your skin already acts temperamental with soaps or fabric, the chances of trouble only go up.
Smarter Choices for Sensitive Skin
It’s tough finding a perfect answer. Avoiding thioglycolic acid altogether feels tempting, but alternatives don’t always guarantee safety or ease. Waxing rips at the hair and skin itself, and shaving brings the risk of nicks or razor burn. Laser hair removal, though permanent, can cost hundreds and sometimes brings its own reactions. Some people lean toward sugaring or physical trimmers. Each option carries its own learning curve and drawbacks.
Dermatologists suggest patch testing, but brands often downplay this step or bury it in tiny print. Realistically, many skip the fuss and apply creams directly — then deal with problems later. Medical experts stress the need to watch for burning or tingling right away and rinse off at the first sign. Thick creams designed for sensitive skin can reduce the risk a bit, usually using less acid or buffering with emollients. Still, even “gentle” formulas can trigger problems if left on for extra minutes or used on broken skin.
What Works For Me and Others Like Me
Over years of trial (and plenty of error), I stick with shaving or trimming, especially on my legs. If hair removal creams ever tempt me again, I only use them on small spots, always patch first, and slather on a simple moisturizer afterward to calm the skin. Finding an unscented, nourishing lotion helps, since fragrance is another trigger. Consulting a dermatologist sounds dramatic at first, but after seeing friends land with chemical burns, I see it as smart rather than excessive caution.
Those with sensitive skin shouldn’t feel embarrassed about being picky. There’s no shame in staying careful. Chemical shortcuts look easy, but your skin’s comfort and health will always be more important than speed.
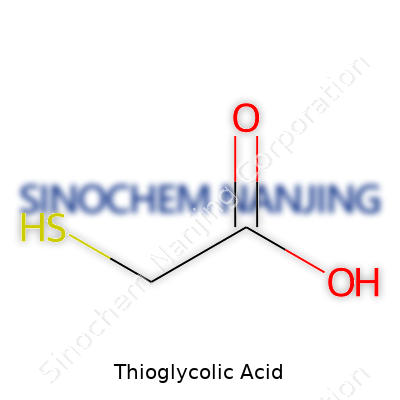
| Names | |
| Preferred IUPAC name | 2-sulfanylacetic acid |
| Other names |
Mercaptoacetic acid Thiovanic acid Acetothioglycolic acid HSCH2COOH TGA Acetic acid, mercapto- Monothioglycolic acid |
| Pronunciation | /ˌθaɪ.oʊ.ɡlaɪˈkɒl.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 68-11-1 |
| Beilstein Reference | 1207176 |
| ChEBI | CHEBI:132990 |
| ChEMBL | CHEMBL1438 |
| ChemSpider | 5471 |
| DrugBank | DB09428 |
| ECHA InfoCard | 03c9d7e0-8b18-4047-bf49-cac139d5a3a2 |
| EC Number | 200-677-4 |
| Gmelin Reference | Gmelln 7872 |
| KEGG | C00788 |
| MeSH | D017529 |
| PubChem CID | 6277 |
| RTECS number | XN7175000 |
| UNII | 4E1Q1O784S |
| UN number | UN1949 |
| CompTox Dashboard (EPA) | DTXSID4020818 |
| Properties | |
| Chemical formula | C2H4O2S |
| Molar mass | 92.12 g/mol |
| Appearance | Colorless to pale yellow transparent liquid |
| Odor | disagreeable |
| Density | 1.33 g/cm³ |
| Solubility in water | Miscible |
| log P | -0.38 |
| Vapor pressure | 0.35 mmHg (25°C) |
| Acidity (pKa) | 3.55 |
| Basicity (pKb) | 10.6 |
| Magnetic susceptibility (χ) | -37.0e-6 cm³/mol |
| Refractive index (nD) | 1.507 |
| Viscosity | 5-15 cP (25°C) |
| Dipole moment | 3.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 132.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -388.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -554.0 kJ/mol |
| Pharmacology | |
| ATC code | D11AX52 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Harmful if inhaled. |
| Precautionary statements | P210, P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P330, P363, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-Acid |
| Flash point | 79 °C (174 °F; 352 K) (Closed cup) |
| Autoignition temperature | 215 °C |
| Lethal dose or concentration | LD50 (oral, rat): 252 mg/kg |
| LD50 (median dose) | LD50 (median dose): 848 mg/kg (oral, rat) |
| NIOSH | WWG35000NN |
| PEL (Permissible) | 0.2 ppm |
| REL (Recommended) | 20.0% |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Cysteine Mercaptoethanol Thioacetic acid Dithiothreitol |

