Thallous Carbonate: Beyond the Lab Bench
Historical Development
Thallous carbonate entered the world of chemistry more than a century ago, surfacing in laboratories during an era obsessed with cataloging new metals. Early researchers had their hands full exploring its unique structure, especially after discovering thallium in the 1860s. Curiosity over the dense, heavy characteristics of thallium salts stoked plenty of interest. The industrial age saw growing demand for purification techniques, driving chemists to develop more efficient ways to isolate thallous carbonate. Over the years, updates in manufacturing and an improved grasp of thallium’s toxic effects shifted how labs and plants handled the compound, making safety and precision bigger priorities than ever before.
Product Overview
Thallous carbonate, with the formula Tl2CO3, turns up as a white or slightly off-white powder. Factories that handle specialty chemicals often produce it for specific scientific and industrial roles. Its main draw comes from its distinct chemical behavior, especially for folks working with radioactive tracers or advanced ceramics. Some labs keep it stocked in small, tightly sealed containers, always with an eye on its serious hazards. Its physical form makes handling fairly straightforward but not without risk.
Physical & Chemical Properties
This compound usually appears as a fine, crystalline solid with notable density owing to its heavy thallium atoms. It dissolves readily in water, unlike many other carbonates, giving it an edge in certain chemical syntheses. The structure supports stable ionic bonds between the carbonate and thallium ions. In my own time handling thallous carbonate, the powder proved surprisingly persistent; it clings to gloves and stubbornly resists cleanup if spilled. Its high molar mass, almost 468 grams per mole, makes even small quantities deceptively heavy, a trait that can surprise the unwary. The compound maintains stability at normal temperatures but reacts noticeably to strong acids, releasing carbon dioxide gas and turning into thallous salts.
Technical Specifications & Labeling
In regulated markets, thallous carbonate arrives labeled with explicit hazard warnings. Labels often include its CAS number (6533-73-9), physical state, potential effects, and critical safety data. Storage instructions specify locked, ventilated areas far from acids or materials that could trigger accidental release of thallium ions. The sheer hazard level explains the vigilance over labeling—regulatory agencies set tight limits on allowable thallium use and waste to protect workers and communities.
Preparation Method
Industrial preparation of thallous carbonate has evolved, but the main route features the reaction between thallium(I) sulfate and sodium carbonate. In a personal experience during graduate work, I watched technicians dissolve thallium(I) sulfate in water, add sodium carbonate, and carefully stir the mix. White precipitate signaled formation of the desired product. After filtration and washing to remove byproducts, the compound would dry in air-tight trays, often under inert atmosphere to minimize air contamination. This process underscores the importance of well-trained hands and constant monitoring to prevent even trace exposure.
Chemical Reactions & Modifications
Chemists value this carbonate for its readiness to react. Mix it with a strong acid—say, hydrochloric or nitric—and it gives up carbon dioxide rapidly, forming thallous chloride or nitrate. It will also swap ions with various metals, supporting research on ionic conductivity and specialty ceramics. I’ve used thallous carbonate to introduce thallium ions into glasses and crystals, where its unique electron structure can fine-tune optical or electrical properties. Its reactivity, both as a base and a supplier of thallium ions, fits well into many niche but powerful applications.
Synonyms & Product Names
Across different catalogs and research notes, chemists know this compound by several names: thallium(I) carbonate, dicarbonothallium, and Tl2CO3 among them. Some older texts use “thallous carbonate” as a catch-all for thallium(I) salts. Suppliers from different regions may list it under slight variants, but the essential character always comes through in the carbon-thallium chemistry.
Safety & Operational Standards
Handling thallous carbonate means treating every grain with caution. Thallium stands among the most dangerous heavy metals, notorious for its easy absorption through skin, lungs, or digestive tract. Protocols demand full personal protective equipment: gloves, goggles, and masks, plus rigorous decontamination routines. Lab training drills stress the irreversible harm from accidental exposure. Regulatory agencies insist on strict tracking even for short-term lab use, and disposal rules force users to neutralize and secure thallium waste far away from regular sewage systems. These procedures shape organizational culture around chemical safety, setting a higher bar than many other materials.
Application Area
Thallous carbonate never turns up in consumer products, but it makes itself useful in research, medical diagnostics, and electronics. Laboratories use it to synthesize other thallium compounds for radioactive tracer work, especially before safer alternatives came along. In more advanced settings, scientists dope certain glasses and ceramic materials with thallium to improve electrical or optical responses. Some detectors and imaging devices have relied on thallium-based components for improved sensitivity. Geochemists sometimes rely on it as a laboratory standard, appreciating its reliable reaction profile in analytical work.
Research & Development
Research into thallous carbonate continues to unfold new insights, especially for materials science. A few years back, interest grew in how thallium ions modify the electrical conductivity of oxide ceramics. Teams working on superconductors and special sensors sometimes use thallous carbonate to create new alloys or test ionic movement in solid structures. I’ve seen collaborations between universities and industry spark new patents, particularly in the race for more sensitive diagnostic devices. At the same time, creative modifications to reduce toxicity or improve handling are gaining attention, with green chemistry efforts rethinking paths toward safer thallium handling.
Toxicity Research
Toxicologists take thallous carbonate seriously. Human exposure to thallium, even at parts-per-million, leads to unpredictable poisoning affecting nerves, skin, and powerful control centers in the brain. Historical data from industrial workers and accidental poisonings have sharpened risk awareness. I keep a vivid memory of safety lectures outlining how thallium can hide in body tissues before triggering painful symptoms weeks later. Modern studies focus on early detection, treatment protocols, and ways to shield workers from accidental contact. Ongoing animal research aims for deeper understanding of uptake and long-term effects, hoping to improve both medical and environmental management.
Future Prospects
Growing pressure for safer, greener chemicals might eventually shrink thallous carbonate’s application space, but some high-stakes research areas still lean on its rare combination of reactivity and ionic heft. Advances in electronic devices keep experts looking for novel ways to harness thallium’s unique properties while cutting risk. Alternative compounds won’t always match its performance, so future years will likely see chemists striving for substitutes or sealed-use formats that never expose thallium to handlers or the environment. Responsible stewardship, smarter containment, and relentless innovation will shape the future of thallous carbonate, balancing discovery with safety every step along the way.
What is Thallous Carbonate used for?
The Reality Behind Thallous Carbonate
Thallous carbonate is not a substance you find at the local hardware store or even most science supply shops. Over the years, it’s managed to gain a reputation as both an intriguing chemical tool and a dangerous material. Chemistry classrooms rarely mention it, but it pops up in advanced research and a few specialized industries. Its main uses come down to its unique combination of thallium and carbonate and the chemical behaviors these bring.
Why Researchers Seek Thallous Carbonate
In the lab, thallous carbonate comes in handy for producing other thallium compounds. Chemists prefer it as a source of thallium ions because it dissolves well in acids, giving up thallium for use in more complicated reactions. These reactions bring forward materials that end up in everything from semiconductors to specialty glass. Thallium-based products, including thallous carbonate, have played a role in developing infrared detectors and crystal growth. A good example: to make thallium bromide or iodide crystals for infrared optics, scientists often start with thallous carbonate. The process involves pretty precise handling and careful storage because thallium, in almost any chemical form, ranks high on the list of toxic substances.
Old Medicine and the Shadows of Risk
Years ago, thallous carbonate made waves in the medical world. Doctors tried it in treatments for skin diseases, and some even used it as a depilatory. That changed quickly after people started getting sick, sometimes fatally so. Thallium accumulates in the body and causes nerve damage, hair loss, and even death at high enough exposure. Modern medicine has banished thallous carbonate. Anyone tempted to see it as a historical curiosity only needs to look at old case reports to grasp why safety rules today stay strict and why researchers handle it under supervision.
The Environmental Angle
Disposal of thallous carbonate demands special attention. It doesn’t break down easily in the environment, and waterways spiked with even a little thallium can become hazardous to fish and mammals. Old factories and abandoned labs where thallium compounds were used often require expensive cleanup. There’s a necessary balance between scientific curiosity and environmental safety. Judging from news stories about polluted groundwater, the need for monitoring and regulation is clear. Avoiding problems in the first place beats trying to clean up the mess later.
Making Thallous Carbonate Safer
Lab supervisors and workplace safety officers have a direct influence on how these risks get controlled. Special containers, spill management plans, and air monitoring stop leaks and exposure. Whenever authorities flag a chemical for strict controls, it’s because history shows what goes wrong if safety slips. Investing in safe workstations, proper storage, and constant training turns the lab from a danger zone into a place for real progress without the health risks.
Looking Forward
Future use of thallous carbonate depends on ongoing work to find safer alternatives for thallium in electronics and science. Advances in material science open doors to replacement materials that sidestep the toxicity. Industries willing to invest in greener chemistry see long-term savings by cutting both medical risks and cleanup costs. Still, where thallous carbonate remains essential, up-to-date protocols and full transparency protect both the people in the lab and the environment outside its doors.
Is Thallous Carbonate hazardous or toxic?
An Insider's Look at Thallous Carbonate Risks
Thallous carbonate rarely pops up outside of specialist settings, but anyone who works around chemicals or studies toxicology has heard the warnings. This compound contains thallium, a heavy metal infamous for its toxicity. Thallium made headlines decades ago after being misused in criminal cases. These dangers linger in labs and facilities that handle thallous salts.
I’ve worked with enough industrial teams to know — workers approach any thallium compound with serious caution. Skin exposure or inhalation opens the door to accidental poisoning. Thallous carbonate isn't used as commonly as other thallium compounds, though its toxic effects follow the same dangerous playbook.
What Makes Thallous Carbonate So Risky?
Chemists don’t have to handle much thallium to risk getting sick. The carbonate form dissolves in water and acids, making it more available to the body after accidental ingestion or contact. Swallowing even a small amount can spark a cascade of health problems: nausea, nerve pain, and sometimes permanent damage to organs. Chronic exposure builds up in bones and tissues, keeping the risk alive long after contact stops.
Research proves its hazard is no myth. The U.S. Environmental Protection Agency classifies thallium compounds as hazardous. Chronic low-level exposure is tied to hair loss, weakness, and changes in mental status. High doses can be fatal. Industry standards like OSHA and NIOSH set strict exposure limits, signaling just how dangerous this material gets, even in trace amounts.
Who Faces the Greatest Danger?
Laboratory technicians, chemists, and workers in specialty manufacturing sometimes face unavoidable contact. In places where environmental controls lag, bystanders or neighbors to poorly contained facilities can run into trouble. Thallium doesn’t just vanish; it can seep into soil or water, making chemical hygiene and waste treatment a big deal.
Years ago, visiting a plant on the East Coast, I saw employees insisting on double gloves, lab coats, and full respirators when handling thallous salts. One veteran manager showed me logs tracking every gram of waste out of sheer necessity. The memory stays fresh whenever I read about accidental thallium poisonings.
What Can We Do to Reduce Risk?
Strong training stands as the first protective layer. I’ve watched safety officers drill new hires with relentless focus — not just on the danger, but on immediate steps to take after a spill or splash. Cleanup teams need direct access to spill kits and running water. Proper labeling goes far beyond red stickers; clear identification and sturdy storage containers keep misplaced samples from slipping into the wrong hands.
Engineering solutions help as well. Local exhaust hoods above work areas, airtight containers, and routine air monitoring play a crucial role. Good policies mean chemical stocks don’t pile up in forgotten corners.
Communities and environmental programs demand transparency. Local agencies need clear communication from industries using thallium compounds. Testing neighborhood water and soil after industrial releases shields neighbors and wildlife from lasting harm.
A Persistent Problem, but Not an Unmanageable One
Serious chemicals don’t look forgiving in the lab notebook or under the microscope. Thallous carbonate gives no breaks. Treating it as hazardous at all points — storage, handling, and disposal — isn't just habit; it’s the only way to keep people, and the world beyond the lab door, safe.
What are the storage requirements for Thallous Carbonate?
The Risks Live Beyond the Label
Thallous carbonate doesn’t show up in everyday news, but most folks would be better off never seeing it at all. This chemical packs some serious risks – heavy toxicity, environmental hazard, and its potential for misuse. Labs and industries may use it for testing or specific manufacturing processes, yet the moment it comes through the door, every gram demands respect.
Knowing the Hazards Inside Out
Thallium’s record for causing health problems starts at the smallest exposures. It’s a poison with a history—linked to nerve and organ damage as well as broader environmental contamination. I read case reports from the CDC about accidental exposures that ended in hospitalizations, even death. Once thallium salts get into the body, they stick, causing decades of problems. Airborne powder can settle unnoticed too, adding exposure routes. For these reasons, strict storage rules matter.
Dry, Sealed, and Solitary: Keeping Risks at Bay
Too many chemical storage mishaps happen because someone ignored the basics. Thallous carbonate thrives on dryness: if it touches moisture, it starts to react, so a tightly sealed container becomes non-negotiable. I remember a visit to a research facility where all their dangerous compounds, especially any with thallium, sat behind double-locked cabinets. Desiccants aren’t fancy, they’re a must. Silica gel packs or comparable drying agents cut down humidity and keep the risk of reactions low.
Temperature and Light – Not Just Fine Print
Sunlight and temperature swings spark trouble with a lot of chemicals, not just thallous carbonate. Direct sunlight can break down certain chemical bonds, possibly forming thallium oxide, which nobody wants. The storage spot should stay cool and shaded. I’ve seen plenty of storage rooms decked out with blackout shades and temperature alarms, and there’s good reason for the hassle. Consistency keeps everyone safer, especially with thallium compounds.
No Roommates Allowed
Stacking incompatible chemicals together has spelled disaster for more than one facility. Thallous carbonate does not belong next to acids or oxidizers. Just think about the risks: accidental mixing could release toxic gases or trigger fires. Proper labeling stands as a frontline defense, not just for regulatory peace of mind, but so everyone—maintenance, lab workers, or emergency responders—knows exactly what they’re dealing with, even during a crisis.
Accountability in Practice
Safe storage relies on human diligence. Every container needs some sort of sign-out log or tracking system. At one lab, we kept a simple notebook—every withdrawal, every return, signed off. This wasn’t busywork. It turned out to be invaluable during audits and, more importantly, if an incident occurred. Faster tracking can mean faster medical attention, and that alone could save lives.
Training: More than a One-Off Lecture
Staff turnover in labs isn’t rare, so relying on orientation day to cover thallium safety doesn’t cut it. Ongoing training works best. I’ve led refreshers where participants practiced emergency procedures, not just heard about them. Honest assessment: the more hands-on, the better the recall under stress.
Bottom Line: Responsibility Above All
Thallous carbonate may gather dust on a shelf for months, but every day it sits there is another chance for something to go wrong. Cutting corners just isn’t worth it. In handling hazardous chemicals, listeners sometimes roll their eyes at the layers of locks and logs—until that one moment when safety planning proves itself. The right storage steps make the difference between a safe lab and tomorrow’s headline tragedy.
What precautions should be taken when handling Thallous Carbonate?
Why This Compound Demands Respect
Thallous carbonate is not your everyday lab chemical. It’s a white powder that packs a dangerous punch, combining the toxicity of thallium with the tendency of carbonates to disperse in air. This stuff can easily sneak into the body through inhalation, ingestion, or even skin contact. Thallium’s toxicity has been documented for more than a century, leading to nerve damage, hair loss, and, in severe cases, organ failure. No amount of expertise justifies dropping your guard around it.
Don’t Trust Gloves Alone
I remember leaning on the comfort of disposable gloves in the past, thinking they’d shield me from any risk in the lab. Thallous carbonate slices through this kind of complacency. Over time, lab reality drives home the need for real protection: thick, chemical-resistant gloves, eye protection, and a fitted lab coat. Even with gloves, frequent changes matter, because thallium compounds can seep through or linger on surfaces unseen.
Airborne Danger Isn’t Just Theory
Experience teaches that powders spread more than we expect. With thallous carbonate, dusting a benchtop can turn into a real risk of inhalation. Lab protocol recommends working inside a certified fume hood. Dry compounds generate tiny particles you can’t see or smell, but the body isn’t fooled. Respiratory masks rated for toxic dusts—like NIOSH-rated P100 respirators—provide another line of defense.
Contaminated Clothing Spreads Hazards
I once watched a colleague absent-mindedly wipe his hands on his coat before lunch. Food and drinks in contagion areas guarantee trouble. With thallous carbonate, outside-lab clothing shouldn’t ever cross into work zones. Immediate handwashing and showering after handling this chemical turn into more than just routine—they’re acts of self-preservation.
Thallium’s Subtle Harm
Symptoms of thallium poisoning creep up gradually: fatigue, stomach pain, tingling in fingers. Search old medical reports and you’ll find cases where exposure escaped detection for years. Because these signs don’t scream chemical exposure at first, regular medical surveillance for those handling thallium compounds isn’t bureaucratic overkill—it’s essential.
Safe Storage: Think Beyond the Label
It might seem enough to keep thallous carbonate in a labeled jar in the usual chemical cabinet. In reality, storage needs more consideration. Separate it from acids, because acids free up toxic thallium ions. Keep containers tightly shut, and regularly check for residue or leaks. A dedicated, locked space prevents unauthorized access, and inventory tracking ensures no one accidentally misplaces or mishandles it.
Why It All Matters
No matter how experienced someone feels in the lab, thallous carbonate’s history forces respect. Even experts have faced exposure by skipping one glove change or ignoring a sneaky spill. The best way out isn’t fear, but careful routine. Ongoing training, clear protocols, spill kits ready at hand, and open conversation about near-misses send a message: safety isn’t an afterthought, it’s a daily discipline.
Finding Better Alternatives
Plenty of scientific advances replace toxic thallium salts in routine work. Double-check if a less hazardous substitute could do the same job, because sometimes it’s smarter to eliminate a hazard than engineer around it. This approach spares both the handler and the environment downstream, since thallium lingers and accumulates outside lab walls.
Responsible Waste Handling
Thallous carbonate waste requires strict disposal. No drain can deal with thallium safely; licensed hazardous waste handlers must take over. Every exposure incident so far started with a shortcut—throwing away a contaminated wipe in the regular bin, for example. Treat every speck you generate like a biohazard.
Everyone Counts
Safety routines only work if every person in the chain follows them. From chemist to cleaner, knowing thallous carbonate’s risks and sticking to best practices keeps everyone healthy. Every laboratory worker deserves that peace of mind.
What is the chemical formula and appearance of Thallous Carbonate?
Looking at Thallous Carbonate
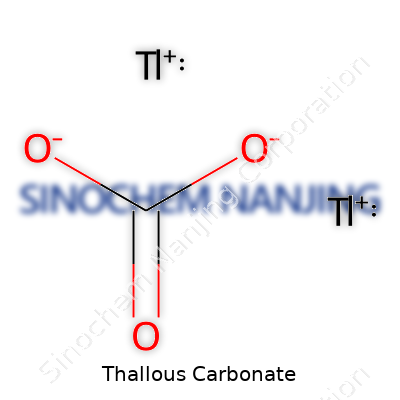
Most folks don’t bump into thallous carbonate on a regular Tuesday. Schools spend time drilling in chemical formulas early on, but the ones making headlines usually show up outside the classroom. Thallous carbonate, also known as thallium(I) carbonate, carries the formula Tl2CO3. This formula signals two thallium atoms for each carbonate group. Arranging the elements this way says plenty about how it behaves and how careful users need to be.
Physical Appearance: More than Meets the Eye
The solid comes as a white crystalline powder. It looks clean, almost harmless at first sight, like the salts sitting near many ovens or hidden in laboratory drawers. That similarity makes thallous carbonate’s true nature even riskier. The powder dissolves well in water, making it easy to mix up solutions or for it to migrate if not handled with caution. From my own years in labs, any fine white powder demands a double-check: label, source, and material safety, every time.
Hazards Tied to Its Properties
Thallium doesn’t play around. Medical texts and accident reports point to thallium salts as some of the most toxic substances humans can encounter. Thallous carbonate doesn’t buck that trend. Exposure can happen by inhalation, swallowing, or even through the skin. Once it enters a person’s system, thallium moves along nerves and muscles, bringing a batch of symptoms — from stomach pain and skin issues to more severe, life-threatening problems. In labs or plants, seasoned staff double-glove and use fume hoods for even short handling.
Tracing Its Use Cases
This compound doesn’t show up often in consumer products but earns its place in specialized industries. Mining, glass manufacturing, and academic research sometimes call for the properties of thallous carbonate — either for chemical analysis or as a reagent in syntheses. There’s history too: thallium compounds saw use in rat poison decades ago, until the world recognized how easily things could go wrong.
E-E-A-T Approach: What Keeps Us Safe?
Sticking close to current guidance from the Centers for Disease Control and Prevention (CDC) and Materials Safety Data Sheets, safety remains non-negotiable around thallous carbonate. Facilities keep strict controls on access and storage. Proper labeling and real-time employee training become essential. Spot checks and emergency spill procedures keep everyone sharp. I’ve seen how even one missed safety step can spiral into a hospital trip — facts don’t change because someone missed a sign.
Better Practices and Safer Futures
Alternatives exist for some of thallous carbonate’s roles, and swapping in less toxic compounds stands as a smart move whenever possible. Research in green chemistry keeps pushing for safer reagents and solvents. On-site, managers can invest in modern ventilation systems and improved protective gear. Transparency also matters: every worker needs to know what’s on their bench and how to react if something goes wrong. That open-door policy cuts down confusion and mistakes.
Thallous carbonate’s formula Tl2CO3 looks simple on paper. In real life, the risks and safe handling show why science isn’t just about formulas. It’s about people, responsibility, and the discipline to keep things safe for the long haul.
| Names | |
| Preferred IUPAC name | carbonic acid; thallium(1+) salt (2:2:1) |
| Other names |
Thallium(I) carbonate |
| Pronunciation | /ˈθæləʊs ˌkɑː.bəˈneɪt/ |
| Identifiers | |
| CAS Number | 6533-73-9 |
| Beilstein Reference | 359872 |
| ChEBI | CHEBI:131378 |
| ChEMBL | CHEMBL1201647 |
| ChemSpider | 157368 |
| DrugBank | DB14543 |
| ECHA InfoCard | Thallous Carbonate (ECHA InfoCard: 100.029.721) |
| EC Number | 208-934-2 |
| Gmelin Reference | GmELIN 2044 |
| KEGG | C18769 |
| MeSH | D013802 |
| PubChem CID | 159410 |
| RTECS number | XG0700000 |
| UNII | 7Y76T13A7H |
| UN number | UN1705 |
| Properties | |
| Chemical formula | Tl2CO3 |
| Molar mass | 468.82 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 7.11 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | -0.77 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 8.72 |
| Magnetic susceptibility (χ) | −37.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.733 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 147.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -816.6 kJ/mol |
| Pharmacology | |
| ATC code | V09FX01 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or absorbed through skin. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P301+P310, P302+P352, P304+P340, P308+P311, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Lethal dose or concentration | LD50 oral rat 21 mg/kg |
| LD50 (median dose) | TDLo=40 mg/kg (oral-rat) |
| NIOSH | TH2022500 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | REL (Recommended): 0.1 mg/m³ |
| IDLH (Immediate danger) | 15 mg/m3 |
| Related compounds | |
| Related compounds |
Thallium(I) sulfate Thallium(I) nitrate Thallium(I) oxide |

