Thallous Acetate: Tracing the Path from Discovery to Tomorrow’s Applications
Historical Development
In the closing years of the 19th century, chemists were racing to understand the properties and potential of newly discovered elements. Thallium, a soft post-transition metal, pulled from the shadows by Sir William Crookes and Claude-Auguste Lamy, soon drew scientific imagination toward compounds springing from its reactivity. Among those, thallous acetate emerged as a curiosity in research, not for its modest cost but its striking, selective qualities. Laboratories in the early 1900s saw thallous acetate appear on shelves, added to the growing catalogue of thallium-based salts. By the 1930s, microbiologists found an unexpected use—its ability to single out enterococci in cultures, enabling more refined studies and food safety protocols. Historic patents and early technical literature show a trail of experiments, from circuit work in physics labs to selective culture plates in public health offices.
Product Overview
Thallous acetate has carved out its role in specialized corners of research and industry. It’s never been a household name or a staple outside laboratories, largely due to its significant toxicity. Supplied as a white, odorless crystalline solid, it delivers the power to differentiate bacteria in culture media, to help with radiological tracing, and to act as a reference material in analytical chemistry. Across decades, bottles labeled with “Thallous Acetate” moved from heavy glass containers sealed with wax to today’s clearly labeled vials, equipped with tamper-evident seals and prominent hazard warnings.
Physical & Chemical Properties
With a formula of C2H3O2Tl, thallous acetate forms crystals that dissolve well in water, making it useful for solutions and culture media. Lifting a sample, a faint metallic sheen gives away the presence of thallium. The salt dissolves rapidly, a sign of the weak ionic bonding in its lattice. At room temperature, thallous acetate remains stable under dry air, but it requires careful handling—exposure to excessive moisture can cause mild hydrolysis. This compound melts just under 110°C, handy for lab workers needing to prepare solutions without specialized heaters. Its density and compactness persist as reminders that handling such heavy-metal salts isn’t trivial.
Technical Specifications & Labeling
Lab supply companies ship thallous acetate with rigorous documentation. Labels spell out molecular weight—264.43 g/mol—solubility parameters (214 g/L at 20°C), shelf life, and batch-specific purity, typically over 98%, confirmed by spectroscopic markers. Regulatory information, including GHS pictograms for acute toxicity, stands in bold on containers, sometimes with additional data for REACH or TSCA compliance. Small print warns of hazardous dust and the need for fume hoods or respirator masks during weighing or transfer. Every shipment carries a certificate of analysis, and suppliers maintain batch records to back trace any safety incident.
Preparation Method
Most thallous acetate stems from the neutralization of acetic acid with thallous carbonate or oxide. In bench-scale production, a chemist combines a measured quantity of thallous carbonate with glacial acetic acid under ventilation, stirring steadily. Bubbling stops as carbon dioxide leaves the mix, signaling completion. Following reflux, the hot solution filters off trace insoluble thallium or carbonate residues. Concentration under vacuum and slow cooling invites crystal formation, which technicians collect, dry, and sieve to uniform particle size. Stringent washing with cold ethanol or water removes any ionic contaminants. Finished lots rest in air-tight jars, ready for downstream use.
Chemical Reactions & Modifications
Thallous acetate engages in exchange reactions common to thallium(I) salts—anions swap in and out readily. In microbiology, mixing with sodium or potassium salts forms double salts with unique selective action. Reacting thallous acetate with sulfide sources, such as hydrogen sulfide, instantly forms thallous sulfide—jet-black and insoluble, used for qualitative ion detection. Adding strong oxidants, chemists force thallium’s oxidation to the trivalent state, though this conversion runs inefficiently. Modified versions with isotopically labeled acetate emerge in tracer studies, pushing research into environmental chemistry and metabolic tracking.
Synonyms & Product Names
Thallous acetate travels under several names: acetic acid, thallium(1+) salt, thallium monoacetate, thallium(I) acetate. Chemical catalogs tag it as TlC2H3O2 or C2H3O2Tl, with vendors listing synonyms to avoid dangerous mix-ups with thallium(III) compounds, which carry even greater hazards. European suppliers often use “acétate de thallium” or similar regional variants, but the core warnings and product identifiers cross borders with little difference.
Safety & Operational Standards
Anyone opening a jar of thallous acetate understands the stakes—thallium salts pose serious, even life-threatening risks. The compound’s fine crystalline texture allows for airborne dust that can slip unnoticed into airways or across skin. Gloves and fume hoods serve as default gear, while storage goes into locked cabinets marked for toxic substances. Spill response relies on dedicated vacuums with HEPA filters and disposable wipes for scrubbing residue. Disposal routes demand specialist waste contractors—thallium persists in the environment and accumulates in living tissue, so landfill or drain disposal remains off the table. Research labs must track every milligram, and audits run frequent to ensure no accidental exposures.
Application Area
Microbiology led the way in practical uses of thallous acetate. Enterococcus, a persistent bacterium in food and water, gets highlighted on thallous acetate plates, letting food safety teams identify outbreaks with speed. Water quality labs pour plates for fecal contamination checks, public health authorities responding to early signs of contamination. Beyond that, radiochemistry uses thallous acetate as a reference or intermediate for synthesizing radioactive tracers. In biotechnology, researchers sometimes look for rare properties or take advantage of thallium’s behavior in ionic channels for basic neuroscience studies. Despite its power, its danger keeps it locked out of the classroom and away from broad industrial application.
Research & Development
Academic papers continue to track new tweaks to thallous acetate’s utility. Media developers constantly look for better, safer growth promoters in bacteriological testing, but thallous acetate holds ground for its precision and sensitivity. Environmental scientists assess its fate in soil and water, working to understand pathways that might drive unintended exposure. Innovations linger at the interface of chemistry and life sciences, using thallous acetate in small-scale labeling experiments. Meanwhile, environmental and regulatory researchers try to map out replacement protocols that preserve selectivity without the same toxic risks.
Toxicity Research
Medical literature tells a cautionary tale about thallium salts. Thallous acetate’s toxicity mechanisms resemble other thallium(I) compounds—it slips easily into biological systems, mimicking potassium and disrupting cellular function. Acute poisoning brings rapid gastrointestinal distress, nerve pain, and even hair loss, while chronic low-level exposures build up quietly in organs. Research teams study chelation therapies and quicker diagnostics, hoping to shrink danger for the few cases where accidental exposure occurs. Regulatory threshold values have been ratcheted down repeatedly, and guidance documents stretch to several pages, underscoring how little room for error research communities can afford.
Future Prospects
Thallous acetate stands at a crossroads. Its capability as a microbiological agent and research material is undisputed, yet rising environmental and health concerns spur a search for alternatives. Regulatory crackdowns in North America and Europe push academic and industrial users toward less hazardous materials whenever the precision edge can be sacrificed. Research grant priorities increasingly reward methods with green chemistry profiles. At the same time, frontier applications—nanoscale tracing, rare organism studies—keep it relevant in tightly controlled settings. With proper stewardship, thallous acetate may remain a tool for breakthrough work, but its days as a go-to solution for standard tests look numbered. Scientists continue to debate how to balance its powerful selectivity with the moral mandate to protect researchers and the environment from persistent, invisible threats.
What is Thallous Acetate used for?
Looking at Thallous Acetate in the Lab
Labs often stock plenty of chemicals, but thallous acetate tends to stand out in microbiology. Through years of working alongside microbiologists, I’ve seen how it becomes crucial for isolating certain bacteria, especially the ones causing problems in food safety or health care. To get a little more hands-on, this compound works best in selective media — it helps spot specific germs by making the environment less friendly to others.
Take Thallous Acetate Citrate (TAC) Agar. Researchers turn to it when they need to investigate potential Salmonella outbreaks. Salmonella loves crowded places, and it often hides among other bacteria when food goes bad. Adding thallous acetate to culture media slows the growth of less dangerous bacteria, so Salmonella has nowhere to hide. This single chemical can turn a routine test into a potentially life-saving diagnosis by leading health professionals straight to the target organism.
Safety Always Matters
Safety stays front and center any time thallous acetate comes out. This isn’t a chemical for casual use. Even just a tiny bit, if mishandled, can pose significant health risks. According to the CDC, exposure can hurt the nervous system and kidneys if it’s inhaled or swallowed. I can’t recall a time when a reputable lab handled it without proper gloves, eye protection, and a well-maintained fume hood. The need for such strict precautions builds trust with lab workers and the public. If everybody in the lab follows procedures, patients and researchers avoid dangerous mistakes.
Focusing on Public Health
I’ve seen public health labs lean on thallous acetate, mostly because bacteria like Proteus and Pseudomonas can get rambunctious and crowd out the ones causing illness. Add this chemical to the mix, and less hardy microbes fade into the background. This approach helped public health experts pinpoint sources of waterborne outbreaks. Without thallous acetate, tracking a typhoid outbreak can drag on, risking more harm to communities.
Thinking about the Downsides
No discussion is real without weighing trade-offs. Thallous acetate lands on hazardous chemicals lists in the U.S. and EU for good reason. Its benefits in the lab don’t outweigh the risks if it ever winds up in the wrong hands or isn’t disposed of carefully. I’ve watched labs take extra steps with neutralizing and packing solid waste, and no one treats this as a side job. Proper labeling, safety drills, and regular audits prove vital, not just for compliance but to keep people safe.
Where to Go from Here
Some researchers keep looking for safer alternatives that deliver the same results. That innovation matters, especially when dealing with routine food safety testing or research in countries that might not have robust waste handling processes. With better investment and collaboration, safer additives or digital testing methods could replace hazardous compounds. Until then, the people using thallous acetate have to keep safety, public health, and environmental impact in constant view. That’s the only way its benefits can outweigh its risks for the long haul.
Is Thallous Acetate toxic or hazardous?
Thallium and Human Health: A Risk Not Worth Ignoring
Walking into a lab, most people probably don't think about the powders on the shelves. Thallous acetate raises different concerns. This chemical, with its smooth-sounding name, packs a serious punch. Just looking at the facts, it takes little exposure to start causing real harm. The thallium in thallous acetate can disrupt the nervous system, trigger nausea, and make hair fall out in clumps. Those symptoms point to nerve and kidney damage—problems that don’t clear up with a few days of rest and hydration.
How Toxicity Unfolds: My Take From Lab Experience
Having worked in an academic chemistry lab, I remember the red-tape wrapped around thallium compounds. It wasn’t just for show. The acetate salt of thallium enters the body quickly, whether through skin or air. A person doesn't need to swallow it. Breathing dust or letting it linger on hands is enough to bring on trouble. Early signs like tingling fingers or aching muscles can creep up slowly. Often, the real problems stay hidden until they've done their damage—a trait that sets thallious acetate apart from more forgiving chemicals.
Chemical Hazards Extend Beyond the Bench
It’s tempting to think hazards stay contained in metal cabinets. The reality: thallous acetate leaves the lab with the people who handle it. Traces under fingernails, on phones, or in washing machines at home put families at risk. Symptoms show up late, so by the time someone notices something off, thallium has already wound itself deep into the body’s system. Unlike other heavy metals, thallium resists the body’s efforts to break it down or push it out.
Looking at the Numbers: No Safety in Small Amounts
A few milligrams—less than a pinch—can send someone to the hospital. The World Health Organization calls thallium one of the most dangerous toxic substances. Even environmental exposure at low levels can harm wildlife or seep into water. Countries have set tight limits for how much thallium can appear in drinking water, food, and soil.
What Makes Thallous Acetate so Deadly?
Some chemicals warn you. They burn your nose, sting your skin, or have a strong smell. Thallous acetate moves quietly. It has no odor. It doesn’t sting. That means someone can use it without realizing they’ve picked up a dangerous dose. This silent toxicity plus the long-lasting effects in the body make labs treat it with a level of caution reserved for few other chemicals.
Solutions: Safer Practices Aren’t Optional
Finding substitutes is often the smartest choice. For example, microbiology labs once counted on thallous acetate to isolate certain bacteria, but safer media have replaced it in many protocols. For those who still handle it, strict rules really do save lives. Personal protective gear, designated lab spaces, and decontamination steps afterward all matter. Waste from anything containing thallous acetate must go straight to hazardous disposal. No rinsing down the drain, no sweeping up into a common trash can. Sharing hard facts and real stories about thallium exposure in schools and labs keeps people from brushing off the danger.
Final Thoughts: Staying Vigilant Protects Everyone
Thallous acetate isn’t a relic from riskier days. It’s in use today, and its hazards are real. Trust in protocols and constant awareness keeps people and communities safe. If health counts for anything in science, then taking thallous acetate seriously comes first every single day.
How should Thallous Acetate be stored?
Thallous Acetate: Not Just Another Chemical
Thallous acetate looks pretty unassuming on the shelf. A white powder, often kept in microbiology labs and used mostly as a selective agent for isolating certain bacteria. Underneath that plain appearance hides real risk. This compound contains thallium, a heavily toxic element, and safety slips can come with very high stakes. I’ve worked in environments where the importance of proper storage got drilled into everyone, because the risk wasn’t just a theoretical possibility. The threat to human health is real, proven, and immediate.
Toxicity Makes the Rules
European and North American health agencies set strict controls for thallium compounds. Inhalation, ingestion, and even skin contact can quickly lead to poisoning. You won’t always notice right away either. Effects start off quietly: tiredness, headache, maybe some neuralgia creeping into the hands and feet. By the time most realize, real harm turns up. Watching a former colleague battle thallium toxicity changed the way I treat every container.
Location: Separate, Secure, and Away from Food or Drugs
Every lab should keep thallous acetate in a locked chemical storage cabinet. This cabinet belongs far from places where anyone eats, drinks, or works with medicines and reagents for food testing. Mixing up containers during a busy shift feels all too easy unless systems offer total separation. Holding the key isn’t just about liability—it’s about keeping out accidents and, worst case, deliberate misuse.
Container Choice and Labeling
I never cut corners on container quality. Glass or high-grade plastic keeps the substance from reacting, and screw-top lids stop air or humidity from creeping inside. Clear, disease-warning labels matter every bit as much as the lock. Words like “DANGER: TOXIC” or standardized hazard pictograms on the outside wake up anyone who handles the bottle. Handwritten tape invites confusion and mistakes when tiredness kicks in at the tail end of a shift.
Environmental Controls
Temperature and humidity aren’t trivial, either. Thallous acetate should avoid direct sunlight, heat, and moisture. Even a modest leak in a humid room brings out caking or contamination, leading to inhalation risk as dust, or chemical reactions no one wants. Our storage room uses independent ventilation and refrigeration for sensitive materials—expensive, but one mishap can cost far more.
Checks, Inspections, and Never Working Alone
Every good routine includes regular checks. Inventory tracking flags if the container ever gets lighter than it should, or if seals come loose. Any time thallous acetate comes out, it stays logged—no exceptions. New people receive full training before they even touch the cabinet. Lone work gets banned in our system; no one handles the most dangerous items without a partner in the room. Too many cases prove that most incidents happen when shortcuts get taken or someone winds up alone in a crunch.
Reducing Risks for the Future
Alternatives to thallous acetate don’t work for every test, but reevaluating protocols allows labs to reduce the chances for exposure. Some institutions now require written justification before ordering, hoping to phase out non-essential uses altogether. I applaud these efforts – less thallium on the shelves means less risk hanging over everyone’s heads. Until replacements become widespread, following these basic storage practices keeps harm at bay.
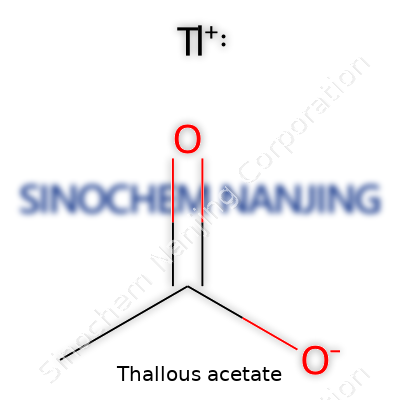
What is the chemical formula and molecular weight of Thallous Acetate?
Chemical Identity and Key Properties
Thallous acetate steps forward with the chemical formula TlC₂H₃O₂, built from one thallium ion (Tl⁺) and the acetate ion (C₂H₃O₂⁻). Its molecular weight rounds out to about 264.42 g/mol. This isn’t just another salt from a chemistry shelf; it proves crucial in specialized environments, most notably microbiology labs where accuracy and sensitivity matter.
Why Thallous Acetate Draws Attention
Researchers don’t pick thallous acetate as a casual additive. It brings a selective edge to microbiology media, especially for isolating bacteria like Streptococcus or enterococci. Here, thallium ions act as a gatekeeper, holding back many unwanted competitors and letting specific bacteria thrive for easier identification or study. This selectivity streamlines work, cutting down on time wasted chasing false positives.
Significance Stretches Beyond the Lab
Handling thallous acetate isn’t everyone’s idea of routine chemical work. Thallium compounds have a track record of toxicity, which demands respect and knowledge during use. Over the years, society learned hard lessons from thallium’s past in rat poison and window cleaning solutions. Stories of environmental exposure, accidental poisonings, and industrial mishaps left a mark that shaped today’s careful handling protocols.
Safety and Environmental Responsibility
Studies confirm that thallium’s toxic effects can build up quietly in living organisms, especially at the nerve endings and kidneys. In the lab, proper gloves, face protection, and fume hoods keep the risks at bay. Outside the lab, clear regulations and waste management procedures block thallous acetate from drifting into public waterways or the food chain.
The United States Environmental Protection Agency (EPA) designates thallium as a priority pollutant for a reason. In my experience supporting chemical handling teams, every training session emphasized protocols: lock up stocks, document every gram, track waste, keep emergency protocols ready. These standards keep people and the environment out of harm’s way—and frankly, they’re not too much to ask when dealing with a heavy hitter like thallium.
Looking Ahead: Solutions and Alternatives
Many ask: do we need to keep using thallous acetate, or can science shift to safer options? Advances in molecular diagnostics, like PCR and rapid antigen testing, have started to ease reliance on toxic compounds in labs. Still, thallous acetate holds a niche where certain rapid or highly selective tests demand its unique characteristics.
Switching to less hazardous agents remains a work in progress. Some groups explore potassium tellurite or sodium azide as alternatives for bacterial selection, but trade-offs in sensitivity and specificity keep thallous acetate in rotation for select cases. Change moves slowly in scientific settings, especially when public health decisions ride on proven accuracy.
Final Thoughts
Knowing thallous acetate’s formula—TlC₂H₃O₂—and molecular weight—264.42 g/mol—shouldn’t turn into trivia. It’s a reminder of chemistry’s reach and responsibility. People who work with thallous acetate view it not as a simple reagent, but as a substance demanding respect, skill, and awareness of its double-edged impact on society and the natural world.
What are the safety precautions when handling Thallous Acetate?
Thallous Acetate and Real Risks
Thallous acetate isn’t a common chemical you find on a grocery shelf. The main thing that sets it apart from everyday substances is its toxicity. Thallium compounds have a reputation for serious health risks. Even small doses, over time or by accident, can lead to big trouble: neurological problems, damage to internal organs, hair loss, and sometimes fatal poisoning. Lab workers and students who’ve spent time around thallium learn early how important respect for safety is with this stuff.
Why Gloves and Goggles Matter
Direct contact with thallous acetate must be avoided. Anyone I know who works with this chemical always wears nitrile gloves, lab coats, and sturdy goggles. Skin absorbs thallium quickly, so washing your hands isn’t enough if you spill any on yourself. It makes a difference to double-check protective gear for holes or tears before every use. Chemical splash goggles add vital coverage, especially since thallous acetate can irritate eyes very quickly.
Fume Hoods are Essential
Respiratory exposure counts as the most dangerous route for thallous acetate accidents. Working in a well-functioning fume hood cuts that risk. You can’t always smell or see thallium dust in the air, but a tight workspace without strong airflow can lead to inhalation. Colleagues who skipped using a hood ended up with headaches or low-grade symptoms that lingered. OSHA lays out requirements because even a tiny inhaled dose can cause problems.
Labeling and Storage: Not a Task to Skip
Effective labeling isn’t bureaucratic busywork. Unmarked bottles and poorly written labels cause accidents. Storing thallous acetate in sturdy, clearly labeled containers with hazard warnings prevents mix-ups. Workers in my labs never use food containers or random glassware for toxic chemicals. Locked chemical storage cabinets, usually designed for poisons, keep unwanted hands out. Full inventory checks once a month prevent forgotten or deteriorating supplies from piling up.
Cleaning Up Matters as Much as Preparation
Accidents with thallous acetate don’t usually involve dramatic explosions, but spills can be easy to underestimate. Small spills demand immediate cleanup with thallium-appropriate absorbent materials and proper disposal, following hazardous waste procedures. After every session, benches get wiped down with cleaning agents suited for thallium compounds. Used gloves, towels, and wipes always go straight into labeled toxic waste bins, not the regular trash.
Training and Medical Readiness
Every lab worker dealing with thallium gets special training before touching the stuff. Training doesn’t stop at learning chemical hazards. People also memorize emergency procedures and know who to call for help. Medical support stands ready, and so does thallium exposure antidote information. Quick access to showers and eyewash stations cuts the time between accident and response. In my experience, regular drills and real-life practice saved precious minutes during emergencies.
Solutions to Keep People Safer
Reducing the use of thallous acetate altogether where possible has had positive effects. Some researchers now turn to less toxic alternatives after careful review. Institutes I’ve worked with started using centralized supply systems rather than storing multiple stocks across different rooms, limiting how much thallium anyone has on hand. Safety officers check procedures, knowing that rules written on paper only matter if people follow them daily.
The Takeaway for Anyone Handling Thallous Acetate
No shortcut exists for safety with thallous acetate. From first unsealing a bottle to the final waste pickup, each step matters to protect health. Culture makes a difference: when people value safety over speed or convenience, chemical work stays secure. Those lessons go home with you, too—and benefit every workplace in the long run.
| Names | |
| Preferred IUPAC name | thallium(I) acetate |
| Other names |
Acetic acid, thallium(1+) salt Thallium(I) acetate Thallium acetate |
| Pronunciation | /ˈθæ ləs əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 563-68-8 |
| 3D model (JSmol) | `thallium(1+) acetate[Th+].CC(=O)[O-]` |
| Beilstein Reference | 3940767 |
| ChEBI | CHEBI:132950 |
| ChEMBL | CHEMBL1201656 |
| ChemSpider | 21420 |
| DrugBank | DB11212 |
| ECHA InfoCard | 100.029.131 |
| EC Number | 205-488-0 |
| Gmelin Reference | 7774 |
| KEGG | C18664 |
| MeSH | D013792 |
| PubChem CID | 24859 |
| RTECS number | AB5950000 |
| UNII | NJ3V6P0Y6B |
| UN number | UN1707 |
| CompTox Dashboard (EPA) | CompTox Dashboard (EPA) of product 'Thallous Acetate' is **DTXSID4020490** |
| Properties | |
| Chemical formula | TlC2H3O2 |
| Molar mass | 265.42 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 3.56 g/cm3 |
| Solubility in water | Soluble |
| log P | -1.285 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.7 |
| Basicity (pKb) | 13.4 |
| Magnetic susceptibility (χ) | -42.0e-6 cm³/mol |
| Refractive index (nD) | 1.744 |
| Viscosity | Viscous liquid |
| Dipole moment | Zero Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -723.9 kJ/mol |
| Pharmacology | |
| ATC code | V09FX01 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; causes damage to organs; suspected of causing cancer. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P201, P202, P210, P260, P264, P270, P273, P280, P284, P301+P310, P302+P352, P304+P340, P308+P311, P314, P320, P330, P361+P364, P370+P378, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Autoignition temperature | 370 °C (698 °F; 643 K) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 47 mg/kg |
| LD50 (median dose) | 250 mg/kg (rat, oral) |
| NIOSH | WH2625000 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | REL (Recommended Exposure Limit) for Thallous Acetate is "0.1 mg/m3 (as Tl), as a TWA". |
| IDLH (Immediate danger) | 40 mg/m3 |
| Related compounds | |
| Related compounds |
Lead(II) acetate Potassium acetate Sodium acetate |

