Thallium Sulfate: A Straight Talk on Its History, Science, and Hard Realities
Historical Development
Thallium sulfate emerged out of late 19th and early 20th-century chemical breakthroughs. Back then, chemists were still mapping the curtain between curiosity and catastrophe. Having spent plenty of my time poring over murky historical journals, I see old records that show thallium, once called “the poisoner's poison,” filtered into lab routines with little caution. This compound quickly raised eyebrows in the scientific community because of its deadly properties along with its surprising effectiveness as an agent in certain agricultural products and rat poisons. By mid-20th century, manufacturers churned out thallium sulfate for everything from pest control to glass making, before toxicity studies forced a change of course. The substance’s popularity tanked, as public health authorities got better at flagging real dangers, and people realized this wasn’t just another common salt.
Product Overview
Thallium sulfate looks like a colorless or white crystalline powder. Folks in the chemical trades recognize its fine, powdery touch, which dissolves well in water. Used without much fuss in earlier decades, it sometimes fooled handlers into letting their guard down. Products like this—simple in appearance, deadly underneath—remind me of stories where experienced professionals missed the hidden risks, simply because it didn’t look dangerous. In education and industry, the realization dawned late: sometimes the most benign-looking substances pack the worst punch.
Physical & Chemical Properties
Thallium sulfate comes with some tricky properties. Its water solubility stands out; only a modest amount of stirring leaves no trace behind, at least to the naked eye. The compound absorbs moisture from the air if left exposed, which can lead to clumping. If you’ve ever handled it, you know it doesn’t smell or signal its presence by color, making accidental exposure a lurking worry. Chemically, it stays stable under normal temperatures and pressures, but heat, acids, and light can lead to breakdowns or generate dangerous byproducts. These physical traits, combined with its readiness to slip easily into the bloodstream, make it a stealthy hazard wherever it turns up.
Technical Specifications & Labeling
Anybody with experience unpacking chemicals from a supplier will tell you the thallium sulfate label comes festooned with warning symbols. The convention is bold and blunt: toxic, handle with gloves and in a ventilated space. This isn’t just regulatory red tape. One whiff or skin contact can set off symptoms, so you’ll see careful language about quantity, purity levels, and batch data. That’s born from decades of mistakes and injuries, not just legal caution. Having sorted through storerooms full of reagents, I’ve grown to trust high-quality, thorough labeling. In the case of thallium sulfate, those warnings aren’t just for the shelf—they’re for your life.
Preparation Method
Thallium sulfate doesn’t just occur in nature; making it usually means combining thallium metal or oxides with sulfuric acid. The process itself seems simple on paper: reacting the constituents in water, letting the sulfate crystallize upon cooling, then filtering and drying. In reality, each step carries risk. Even those used to handling toxic substances talk about the peculiar nervousness that comes with preparing this compound. From firsthand lab work, equipment choice and ventilation take on real urgency, since even tiny spills or airborne dust act faster than most people expect. Chemical manufacturers shifted their methods over time, tweaking processes to reduce exposure to staff and the environment, as regulatory agencies clamped down hard.
Chemical Reactions & Modifications
Inside the beaker, thallium sulfate shows a willingness to react and create new compounds under the right push. Add strong acids, and thallium ions might migrate into other chemicals, producing soluble thallium salts. Chemists once experimented with these reactions to generate pigments, specialty glasses, and even in medical imaging, though those uses faded due to the health costs. In real-life settings, the compound’s reactivity isn’t just a technical detail—it demands a kind of respect and constant alertness. Having seen accidents from overlooked side reactions, I give thallium sulfate a wide berth during experiments that push it past its comfort zone.
Synonyms & Product Names
Old textbooks and stockroom logs sometimes call thallium sulfate by other names: thallous sulfate or simply TL2SO4. Some commercial catalogs still list those vintage labels, a relic of the days when naming conventions were stuck in the wild west era of chemistry. Navigating synonyms means tracing through historical documents, safety archives, and technical papers. This confusion around names caused real messes in the past—leading to a push for clarity in modern chemical databases—so nobody reaches for the wrong bottle and puts themselves or others at risk.
Safety & Operational Standards
Thallium sulfate’s reputation for toxicity shaped every operational rule around it. Only trained hands should manage this stuff, and never without full personal protective gear. Chemical hygiene plans recommend fume hoods, double gloves, and emergency decontamination stations nearby. Stories circulate among lab veterans about near-misses, late-diagnosed skin contact, and frantic rushes to medical help. Companies and universities shifted their protocols after every incident, sometimes only after hard lessons. Regular exposure limits show how seriously regulators take the dangers, and disposal protocols run pages long. Anyone collecting hazardous waste for a living knows the anxiety that comes with every thallium sulfate container, and for good reason.
Application Area
Thallium sulfate’s use across the decades tells a cautionary tale. Early on, agricultural sectors used it as a rodenticide until countless poisonings forced a ban for most outdoor uses. Some glassmakers prized thallium in specialty glass, owing to its ability to alter refractive indexes, and semiconductors sometimes saw small-scale laboratory work. Medical researchers, too, dabbled in it for diagnostic imaging, but the health risks choked off that avenue. In my view, the story here isn’t just about what this compound could do, but about the grudging acceptance that its harms outpaced its benefits. Nearly every modern application is tempered with heavy oversight, endless risk assessments, and alternatives always standing by.
Research & Development
Even as regulators clamp down, researchers haven’t forgotten thallium sulfate altogether. Labs sometimes explore its electrochemical properties, hunting for safer ways to exploit its unique ion behavior. Advances in analytical chemistry keep looking for new detection systems, hoping to catch trace contamination quickly. I’ve seen research into analogs—compounds with similar properties but reduced toxicity—pick up speed as people seek that delicate balance between utility and safety. Thallium’s impact on environmental studies has also grown, as scientists measure long-term soil and water contamination and devise new remediation technologies. The spirit in the research community: study the beast so we know where it lurks and how to deal with it safely.
Toxicity Research
No talk about thallium sulfate skips over toxicity. Human history offers a grim roll call: accidental and intentional poisonings, slow-developing symptoms, and difficult diagnosis. Animal studies repeatedly confirm what human cases made clear—a few milligrams per kilogram can cause hair loss, nerve damage, kidney and liver problems, and death. Chronic exposure points to cancer risks and birth defects. Medical journals detail antidotes like Prussian blue, but those only buy time and don’t guarantee a full save. I’ve read health reports that underscore just how easily this compound sneaks past common safety nets—the body absorbs thallium fast, and it’s slow to come out. This hard data underscores why so many countries banned it from general sale, and why laboratories remain some of its last legal strongholds.
Future Prospects
Looking ahead, the market for thallium sulfate shrinks every year, and with reason. Public health weighs heavier on the scales than unique chemical properties. Innovations in materials science and clean chemistry now focus on alternatives that dodge thallium’s legacy of harm. Some teams view thallium sulfate as a benchmark in environmental toxicology—using it to calibrate their detection systems and safety procedures, not for mass deployment. In my experience, even the boldest research proposals involving thallium sulfate rarely get far unless they draw a convincing roadmap for containment and risk management. Realistically, future prospects mean more environmental monitoring, improved cleanup methods, and further restrictions, not a revival. The biggest lesson: never underestimate the cost of convenience—thallium sulfate reminds us that sometimes the price is simply too high.
What is Thallium Sulfate used for?
Rare Chemical, Risky Business
Thallium sulfate isn’t something most people run into at the hardware store or pharmacy. This substance has a nasty reputation and for good reason. In the early twentieth century, it earned a spot as a key ingredient in rat poison. People used it across continents, thinking it offered an easy fix for rodent infestations. The results turned out disastrous. The same poison that killed rats seeped into crops, farm animals, and people, triggering tragedies everywhere it got loose.
History Shapes Regulation
Reports from the World Health Organization detail countless poisonings from thallium compounds in the mid-1900s. Farmers, children, even entire families suffered after exposure. This isn’t just about bad luck. Thallium sulfate’s toxicity made it hazardous for almost any living thing. As time passed, countries banned its use for pest control. Regulators learned the hard way, sometimes only after workers lost their health or communities lost loved ones.
Limited Industrial Value
Some chemists and researchers still use thallium sulfate in tightly controlled environments, mostly in laboratory research. Analytical chemists run tests with it, especially in studies involving precious metals or semi-conductors. In some labs, researchers expose plants to thallium sulfate to understand toxic effects and study how contamination spreads in food chains. Its high solubility and reactivity make it useful for these experiments, but strict safety protocols surround every vial.
No Place in Daily Life
Hospitals avoid thallium sulfate. Even though thallium itself was once considered for medical imaging, the dangers buried those ambitions. The risks just outweigh any benefits. In medicine, safer radioactive compounds replaced thallium salts.
Why It Matters
The story of thallium sulfate has shaped how public health officials approach chemical regulation. Lessons from mass poisonings in Mexico, Australia, and China forced regulators to prioritize health over convenience. Facts matter here: thallium disrupts cell function, cripples the nervous system, and builds up enough in the body to kill slowly.
Today, you won't find thallium sulfate on shelves. It’s a controlled substance in much of the world. Laws bar its sale to anyone who doesn’t have a solid reason to handle it. Even scrapyards and recyclers keep an eye out for this chemical, knowing one careless move can poison their whole workplace.
Safer Alternatives, Smarter Choices
The thallium sulfate story reminds us not every scientific breakthrough belongs in stores or public use. Better pest control options have come along—methods that don’t pollute water, poison families, or ruin crops. Integrated pest management, physical traps, even rat birth control pills exist now, all tested for safety first.
I’ve toured farms and spoken with growers worried about keeping crops safe. They know shortcuts don’t lead anywhere good. The best growers rely on updated research, keep a close eye on toxic substances, and push for stricter enforcement. Community health can’t take a back seat to quick fixes. That lesson, learned from the damage caused by thallium sulfate, deserves repeating. Public safety relies on remembering what happened when dangerous tools landed in the wrong hands.
Is Thallium Sulfate toxic or hazardous?
Why Thallium Sulfate Catches Attention
Most folks haven’t heard much about thallium sulfate, but in labs and some old pest-control tales, this stuff pops up. Thallium earned its bad reputation through the damage it caused before people understood how it creeps through the body and lingers in the environment. If you ever get handed a vial marked with its name, it’s time to remember a bit of chemistry and a dose of caution.
How Dangerous Can It Get?
Thallium itself carries a heavy weight on the toxicity chart. Mix it with sulfuric acid and you get thallium sulfate. The trouble really starts with how quietly it slips into the body. Touch, taste, or even breathe dust containing thallium sulfate—each way finds a shortcut to your organs. Once inside, this stuff mimics potassium. Cells can’t tell the difference, so thallium sweeps throughout the body, nestling in the brain, liver, and kidneys. That mix-up leads to nerve damage, gut pain, and heart issues. Even a few milligrams are enough to make a person seriously ill.
Real-World Experience Shows Its Risks
In the past, thallium sulfate saw use in rat poison. It worked so well that people pushed for bans across the globe. Too many incidents involving accidental poisoning of pets, wildlife, and sometimes children forced the issue. Australia banned it in the 1960s, and the United States followed suit not long after. Doctors struggled to treat thallium poisoning because symptoms pop up slowly—hair loss, tingling in the hands and feet, swelling, and confusion. By the time someone figures out thallium played a part, it may be too late for a quick fix.
Learning about these real-life consequences sticks with you. I once met a chemist who worked in pesticide research back in the 1950s. He lost colleagues to what they called “the silent killer.” No warning lights, no obvious signs until it hit hard. That’s not a label you shake off easily. Today, some toxicology labs still keep tiny samples for analysis, but extra training and sealed cabinets set strict rules.
Staying Safe and Managing Its Legacy
Now, only trained professionals holding the right licenses handle thallium sulfate, mostly for rare analytical testing or scientific study. Workplace safety rules spell out exactly how to manage spills or accidental exposure—full-body protection, closed-loop ventilation, proper disposal in hazardous waste incinerators. I saw hazard drills where workers treated every vial as if it could change their future with a single mistake. There’s no room for shortcuts.
Water contamination stays high on the public concern list. Thallium, even in tiny doses, passes through soil and into rivers. Today, strict monitoring aims to keep this from ever happening near drinking supplies or food crops. Testing requirements force companies to prove there’s zero leakage into the water table or risk heavy fines and public backlash.
Finding Smarter Solutions
Chemists and engineers focus on replacement strategies. Modern rodenticides and insecticides rely on less persistent chemicals. Universities teach the next generation of scientists to use better lab design, push for safer alternatives, and keep thallium sulfate locked away unless it’s absolutely necessary. Public access, once taken for granted, vanished for good reason.
For anyone curious about chemistry, there’s a simple rule: if thallium sulfate shows up in conversation, every move around it calls for care, experience, and a deep respect for what’s at stake.
What are the safe handling procedures for Thallium Sulfate?
Thallium Sulfate: A Dangerous Compound
Thallium sulfate has earned a notorious reputation, largely because of its tight ties to highly toxic and even criminal uses in the past. It’s still found in labs and some industrial processes, but this isn’t a chemical that belongs anywhere near carelessness. Having spent time around folks working in both research and chemical manufacturing, I’ve seen how small mistakes with hazardous materials can turn into emergencies that shake up entire institutions.
Personal Safety Above All
Good gloves, safety goggles, and a well-fitted lab coat keep danger at arm’s length. For Thallium compounds, regular nitrile gloves won’t guarantee a barrier. Professionals put on thicker chemical-resistant gloves and never forget the protection for eyes and face. This isn’t overkill—it’s lived experience talking. You only need to see a single exposure incident to know skin and inhalation routes matter a lot.
Ventilation counts for a lot. Years ago, a chemist I knew developed symptoms just by using thallium sulfate in a poorly ventilated room. Fume hoods draw away invisible airborne particles, turning what might be a tragic mistake into just another day at work. Having a space with good air movement isn’t optional with this stuff.
Storage Rules: Locked and Isolated
Secure storage helps keep chemicals out of hands that shouldn’t have them. Cabinets labeled clear as day and locked up show respect for everyone who comes through the lab. Thallium sulfate never sits near food, drink, or personal items. Folks in top labs often double-lock these materials and record every single transfer in a logbook. That’s a lesson from organizations that have faced audits and scare stories.
Waste Disposal: Never Down the Drain
Disposal always gets extra attention. Dumping thallium-containing waste down the drain contaminates water and quickly causes harm to communities and wildlife. Specialized waste collection containers, handled by professionals who track what goes in and how it leaves the site, remove a lot of risk from the process. I've seen small labs team up with local hazardous waste facilities to cut down environmental risks.
Regulations push for cradle-to-grave tracking with thallium. Those rules come from hard-learned lessons, not some bureaucratic whim. Anyone who’s faced an EPA inspector knows the paperwork keeps both workers and neighbors safer.
Training and Medical Readiness
Regular training sessions stick in your head far better than a warning sign. Walk-through drills reveal weaknesses. Anybody handling thallium sulfate should recognize early symptoms—thallium disrupts nerves and organs long before anyone notices a problem. Keeping antidote kits and emergency contacts in plain sight gives everyone peace of mind. I’ve seen how preparedness shrinks reaction time and saves lives.
Positive Culture and Vigilance
A safety-first culture means calling out shortcuts, not brushing off concerns. Sharing stories of past close calls avoids repeating them. Everyone looks out for each other, filling in for absent-minded colleagues or new trainees. I saw a near-miss once—a simple reminder from a coworker interrupted what could have become a very bad day.
Thallium sulfate brings heavy risks, but straight talk and good habits carve out a path for safe use. Respecting the danger and using solid procedures keeps everyone coming home healthy.
How should Thallium Sulfate be stored and disposed of?
A Real Threat in Plain Sight
Thallium sulfate brings a tough challenge to anyone who uses or handles chemicals. It’s not just about rusty old bottles sitting on a forgotten shelf. The real story here is about health. Chemically, thallium sulfate has a reputation: colorless, tasteless, water-soluble, and extremely toxic. Over the years, doctors and chemists have seen firsthand how this compound attacks nerves, causes hair loss, or brings heart failure with tiny exposures. Stories from mid-century rodent control make the danger all too clear. Just a few micrograms can change a person’s life forever.
Storage: No Corners Cut
You won’t find any safety shortcuts that work with thallium salts. Locking thallium sulfate away in a dark cabinet rarely goes far enough. Only people trained in handling toxins should touch this stuff. Store it in chemically resistant bottles—no rusty lids or cracked glass. Keep those bottles inside a sturdy, labeled, airtight secondary container. Labels need bright poison symbols and the name spelled out. This isn’t the kind of chemical you want to trust to memory or faded ink.
Always put thallium compounds in locked cabinets or designated poison lockers, well away from areas where food or drink come anywhere near. A good chemical hygiene officer checks inventory every quarter, not every few years. If an accident does happen, swift cleanup can’t wait for someone to “remember where the gloves are.” Safety data sheets need to stay nearby and up-to-date.
Disposal: Every Step Counts
Disposing of thallium sulfate takes a system, not just a trash bin. It’s illegal—and reckless—to toss it in general waste or to flush it down the drain. Stories of contaminated water systems reveal how just a few milligrams can devastate an entire ecosystem. Even trace amounts can bioaccumulate in wildlife or poison drinking water.
Labs and businesses call certified chemical waste handlers for pickup. These professionals follow hazardous waste laws and use sealed, labeled containers. Any soil, gloves, or glass that touched thallium goes in too. Nobody cuts corners: documentation travels with each batch through every transfer, making sure it never “disappears” into the wrong place. Waste contractors take materials to incinerators or hazardous waste plants designed to capture toxic byproducts. No one improvises here—improvisation is what creates news headlines about chemical poisonings.
Building a Safer Culture
Thallium’s history—quiet, shadowy, often swept under the rug—shows how overlooking old toxins harms both people and the planet. Modern protocols grew out of real lessons learned in blood, illness, and lost trust. Trustworthy labs keep meticulous records, choose staff who speak up when safety feels wrong, and make space for annual retraining. Shortcuts encourage more risk than anyone should accept.
Safer alternatives exist for many uses of thallium. When possible, finding replacements makes sense—chemical stewardship means actively searching for less toxic ways to get the job done. This toxin reminds those who work with chemicals that the real job is protecting each other, never crossing fingers and hoping for the best.
What are the possible health effects of exposure to Thallium Sulfate?
Understanding Why Exposure Matters
Growing up, we didn’t talk about thallium sulfate. On farms and in old gardening guides, though, you’ll find references to its use as a rodenticide and insecticide. Few outside of chemistry ever considered its risks. This chemical barely shows up in the news, but that doesn’t mean it’s harmless. Stories of accidental poisoning exist, and the science around this compound tells a clear story: exposure carries real danger, and it’s a problem that affects public health and worker safety.
Digging into the Health Effects
Thallium’s reputation as a poison isn’t oversold. According to the CDC, thallium compounds like thallium sulfate bring harm quickly. Ingesting or inhaling thallium sulfate can cause nausea, vomiting, diarrhea, and abdominal pain. As exposure continues, people can develop neurological symptoms—tingling in fingers and toes, confusion, or even seizures. Over time, exposure may damage nerves, kidneys, the liver, and even the heart. One common sign, oddly enough, is hair loss, which shows up in chronic cases.
The World Health Organization flags thallium as a toxic heavy metal rivaling lead and mercury. Even small, repeated exposures matter. For workers in factories producing or using thallium compounds, the risk multiplies without strong safety protocols in place. In places without rigorous industrial regulation, silent, slow poisoning becomes a reality. Symptoms sneak up and answers sometimes come too late.
Why We Should Care
It always helps to ground science in lived experience. Most of us won’t mix up thallium compounds in our kitchens, but food and water contamination pose real threats. Past cases in Eastern Europe and Asia revealed entire communities falling ill because industrial run-off contaminated groundwater. Tragedies like these highlight the importance of transparency and monitoring. Once in the body, thallium binds tightly and leaves damage behind, so early detection remains crucial.
Too often, families living near mines or chemical plants learn the risks only after suffering symptoms. The U.S. EPA and OSHA set strict workplace limits for thallium exposure—only 0.1 milligrams per cubic meter in air—but those numbers require enforcement. A few lapses in ventilation or waste disposal, and people end up paying the price.
Moving Toward Safer Practices
Solutions don’t just come from policy. It starts with education. Workers deserve to know how to handle chemicals safely. Protective gear, proper training, and open reporting all make a difference. Industry leaders who invest in regular testing and safer processes protect not just employees but the community around them.
Beyond the factory gates, communities must have access to information about local contaminants. Clean drinking water and food free from heavy metal residues should be basic rights—never a luxury. Rapid response during spills or exposure events saves lives, but long-term environmental monitoring keeps them safe.
In the end, health thrives in places where knowledge, policy, and community meet. Thallium sulfate’s dangers remind us: vigilance, not silence, builds lasting public health.

| Names | |
| Preferred IUPAC name | thallium(I) sulfate |
| Other names |
Thallous sulfate Thallium(I) sulfate |
| Pronunciation | /ˈθæliəm ˈsʌlfeɪt/ |
| Identifiers | |
| CAS Number | 7446-18-6 |
| Beilstein Reference | 1205597 |
| ChEBI | CHEBI:101976 |
| ChEMBL | CHEMBL1257263 |
| ChemSpider | 5372339 |
| DrugBank | DB11360 |
| ECHA InfoCard | 100.006.887 |
| EC Number | 006-004-00-1 |
| Gmelin Reference | 10740 |
| KEGG | C14425 |
| MeSH | D013792 |
| PubChem CID | 24816 |
| RTECS number | XG5950000 |
| UNII | 6Q13C9I7RK |
| UN number | UN1707 |
| Properties | |
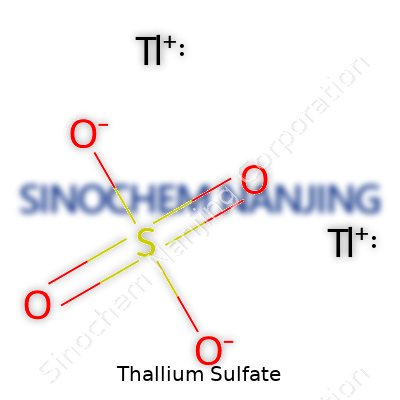
| Chemical formula | Tl2SO4 |
| Molar mass | 546.011 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 6.698 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.39 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.20 |
| Magnetic susceptibility (χ) | -56.0e-6 cgs |
| Refractive index (nD) | 1.738 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 342.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -909 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1245 kJ/mol |
| Pharmacology | |
| ATC code | V10BX01 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; may cause damage to organs; suspected of causing cancer; very dangerous to the environment. |
| GHS labelling | GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | H260, H300, H310, H330, H373, P210, P273, P280, P301+P310, P302+P352, P304+P340, P308+P313, P320, P330, P361, P403+P233 |
| NFPA 704 (fire diamond) | 3-2-2-Po |
| Autoignition temperature | 250°C (482°F) |
| Lethal dose or concentration | LD50 oral rat 21 mg/kg |
| LD50 (median dose) | 160 mg/kg (oral, rat) |
| NIOSH | TL8575000 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | 0.0001 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m³ |
| Related compounds | |
| Related compounds |
Thallium(I) sulfate Thallium(III) sulfate |

