Thallium Nitrate: History, Science, and Outlook
Historical Development
Thallium nitrate locked in its reputation as a chemical of intrigue almost from the start, dating back to the 19th century when thallium itself caught the eyes of chemists like Sir William Crookes and Claude-Auguste Lamy. By the time nitrate compounds emerged, factories and research labs already handled thallium for everything from rodent control to optical glass. Thallium nitrate, in particular, started finding specialized uses in laboratories and industry, mostly because of its solubility and reactivity. Chemists understood thallium could mimic potassium ions, a discovery that led not only to wide applications but also to a deeper awareness of its risks, thanks to documented poisonings in the literature. Oversight tightened after the 20th century’s incidents, as regulatory bodies pushed stricter handling standards. This wasn’t just about legal compliance—it reflected growing recognition that chemicals affecting cells as deeply as thallium require respect, caution, and real experience on the lab bench.
Product Overview
Thallium nitrate appears as a white, crystalline solid, often delivered in tightly sealed bottles to keep moisture out. You won’t see it much in everyday products, but in labs where precise reactions matter, it shows up as a reagent or starting material. Price tags can be high, and the procurement process involves background checks, certifications, and sometimes onsite inspections. Researchers or industry experts use it far more than casual chemistry buffs. As a niche product, it sits in catalogs, flagged as highly toxic, and buyers know every shipment can trigger visits from health and safety regulators or customs officers.
Physical & Chemical Properties
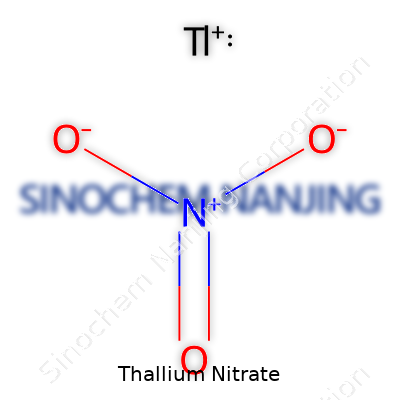
A closer look at thallium nitrate reveals a molecular formula of TlNO3. Its appearance: white, nearly odorless crystals that dissolve well in water, releasing ions quickly. Melting point often lands around 125 °C, and above 200 °C, it breaks down to thallium oxide and nitrogen oxides, both hazardous. On the chemical front, its oxidizing nature stands out; in lab trials, it readily swaps electrons with reducing agents. This property lets it participate in a host of reactions from analytical chemistry to organic synthesis. Thallium, with a single positive charge, interacts strongly with other ions, which explains its knack for disrupting cellular chemistry—a reason toxicologists and medics keep close tabs on its movement in the body.
Technical Specifications & Labeling
Regulations demand strict technical documentation for thallium nitrate. The labels don’t just shout danger; they lay out purity levels, storage precautions, and preparation details. Lab-grade material often exceeds 99% purity. Labels or safety data sheets include hazard pictograms—skull and crossbones, exclamation mark, that sort of thing—plus emergency handling info. Workers see precise batch numbers, expiry dates, and chemical registration numbers like EC and CAS. This isn’t about bureaucracy; knowing the manufacturing date or origin can help trace contamination or pinpoint issues in a recall. In my experience, labs that get sloppy with chemical logs soon run into accidents or fines.
Preparation Method
Manufacturing thallium nitrate relies on reacting thallium(I) oxide or thallium(I) carbonate with nitric acid in solution, producing a clear product, which crystallizes upon drying. The process demands careful temperature control, precise addition rates, and ventilation, as it releases heat and nitrogen oxides. Industrial setups use glass-lined reactors, continuous monitoring for spills, and aggressive air handling to lower the risk of exposure. Resulting crystals get washed, filtered, and dried under vacuum. After years in chemical processing, I’ve seen that every tiny deviation—wrong acid concentration, impurities in water—can mess up purity and trigger waste problems.
Chemical Reactions & Modifications
Thallium nitrate steps into many types of chemical reactions due to its strong oxidizing power and ionic character. In organic chemistry, it helps oxidize primary alcohols, sometimes offering a cleaner route than older chromium-based methods. In analytical labs, chemists use solutions of thallium nitrate for precipitation or as indicators when testing for halides and other ions. Change the reaction media, and you can coax thallium into other salts—chlorides, sulfates, carbonates—each bringing separate hazards and applications. Consistency is king here; uneven reaction conditions yield mixed oxidation states or leave behind toxic residues, both big problems for research and production.
Synonyms & Product Names
Thallium nitrate often goes by several synonyms in catalogs or journals: thallous nitrate, thallium(I) nitrate, or simply TlNO3. In older research documents, you may spot Latin-rooted versions or confusing descriptors depending on region and supplier. Knowing these isn’t just trivia; mislabeling in supply chains can bring the wrong formulation into a process, spiking costs or health risks, something I learned after a mix-up erased a month of lab work. Big suppliers cross-list product codes and CAS numbers to hedge against confusion.
Safety & Operational Standards
Working with thallium nitrate means following strict occupational health standards. Exposure limits, spill cleanup procedures, and medical surveillance consume plenty of training hours. Lab workers suit up with gloves, goggles, lab coats, and sometimes full respirators. Companies keep chelation agents—dimercaprol or Prussian blue—on hand for accidental poisoning. Ventilated hoods, locked chemical storage, and double-signoff inventory checks help control routine risks. In my experience, cutting corners here, just to finish a task faster or save a few bucks, usually ends in incidents that could’ve been avoided.
Application Area
Few chemicals cross as many scientific boundaries as thallium nitrate. In medicine, it once tagged along as a radiopharmaceutical component for heart imaging, but toxicity concerns forced the field to try safer agents. Analytical labs tap into its sensitivity to potassium—thallium’s ionic twin—to identify small cation imbalances. In materials science, thallium nitrate sometimes helps craft specialty glasses and ceramics, prized for their unique optical properties. It also underpins some research into superconducting materials, where thallium compounds tweak critical temperatures. Still, because of tight regulations, these applications stay niche, reserved for settings where the tradeoff between hazard and benefit tips in favor of use.
Research & Development
R&D teams dug deep into thallium chemistry, hoping for breakthroughs that outweighed safety baggage. Scientists probe its potential in catalysis and as a probe for cellular processes that mimic potassium. Sometimes, papers probe the frontier of environmental monitoring, using thallium nitrate’s reactivity in trace metal assays. Years in the field have shown me that while curiosity drives new research, grant panels and oversight committees clamp down on projects unless safety protocols stay airtight. Any new application or synthetic procedure involving thallium nitrate lands under a mountain of peer review, safety audits, and pilot-scale trials.
Toxicity Research
Hard facts about thallium nitrate’s toxicity fill stacks of toxicology journals. Even tiny doses can derail potassium-dependent cellular transport, leading to hair loss, nerve damage, and organ failure. Chronic exposure cases read like cautionary tales from an earlier era—factory workers, lab techs, even poison victims in high-profile criminal cases. Regulators respond with workplace exposure limits down to parts per billion, frequent air and surface tests, and strict waste disposal routines. Thallium poisoning treatments remain tricky, requiring aggressive chemical and medical interventions. Watching clinical teams scramble during a real-life exposure incident made me respect the pace and pressure of toxicology emergencies—prevention always beats response.
Future Prospects
Thallium nitrate faces a difficult path forward. Environmental and worker safety priorities keep shrinking its allowed uses, and many former applications now rely on less hazardous alternatives. Even so, some researchers keep a close eye on new ideas that might benefit from thallium’s unique reactions—green chemistry, targeted cancer drugs, or hyper-sensitive material sensors. Any future progress comes with urgent calls for new detection methods, stronger contamination monitoring, and—most of all—training that outpaces both the risks and the chemistry. Having witnessed both accidents and breakthroughs, I see thallium nitrate as a reminder that technical curiosity never excuses cutting corners on safety or environmental stewardship.
What are the main uses of Thallium Nitrate?
Industrial and Laboratory Uses
Thallium nitrate rarely shows up in everyday life, but behind many closed doors in labs and industry, it takes on several roles. Folks working in chemical research turn to thallium nitrate because it reacts reliably and offers a rare path to making specialty thallium compounds. You find it on shelves where folks study reaction pathways or push for new discoveries in inorganic chemistry. With its ability to serve as an oxidizing agent, chemists value it for quick transformations—reactions that would stall or get messy with weaker options.
Some glassmakers reach for thallium nitrate when producing glass with unique optical properties. Under pressure to create glass that bends light in a specific way, they often add a whisper of thallium. It makes glass that’s dense and clear—find it in systems where standard glass just won’t do, such as deep-UV lenses or detectors used in sensitive scientific equipment. The refractive index jumps, giving scientists the control they need for their high-precision instruments.
In medical and biochemical research, the compound once carved out a role as a radiopaque marker. By binding with certain molecules, thallium nitrate can trace paths inside the body for specific diagnostic scans. While modern medicine moved away from these techniques because health risks became clear, older research still references it, and it’s worth knowing where those methods started.
Electronics and Technology Applications
In electronics, thallium nitrate brings unique value to the game of detection. Early detectors for infrared light sometimes depended on crystals or glasses doped with thallium. The sensitivity improves—especially in devices designed to “see” things the eye misses, like thermal imaging or specialized night-vision equipment. Defense contractors, engineers working on experimental sensors, and manufacturers aiming for greater detection accuracy understand thallium nitrate’s benefits. That expertise often stays behind closed doors due to safety and strategic concerns.
Environmental and Health Risks
Thallium nitrate, potent as it is, demands careful hands and strict protocols. Toxicity sets it apart; health agencies track possible exposure because even small amounts can seriously harm the nervous system. My own experience in academic labs taught me to never shortchange glove quality or skip the eyewash check: just thinking about thallium makes you double-check every safety step. Environmental leaks remain a worry, as thallium moves easily in water and soil and accumulates in fish, putting both ecosystems and communities at risk. Oversight by organizations like the EPA and the CDC ensures that industries using thallium nitrate must prove safeguards and follow traceability rules.
Pushing for Safer Alternatives
Society’s use of thallium nitrate highlights an old challenge: progress often comes with hidden costs. Industries look for safer replacements, investing in research that finds less hazardous materials for glass production or chemical research. Some companies swap thallium-based reagents for more benign agents, though not every reaction or property can be replicated. Positive shifts happen when funding supports chemists focused on green chemistry, and regulatory rewards give incentives to firms that drop toxic heavy metals like thallium.
Wide awareness matters. Bringing workers, environmental scientists, and policy makers to the table helps remove outdated practices and promotes training, so no one takes unnecessary risks. Whenever a dangerous product sticks around, it’s usually because the alternatives struggle to match the same performance. Ongoing conversation—paired with a dose of fact-checking and transparency—will keep thallium nitrate’s uses shrinking over time, as science and safety align.
Is Thallium Nitrate toxic or hazardous?
Real Risks from Thallium Nitrate
Thallium nitrate stands out as more than just a chemical curiosity. People who work with or around it face real dangers. A lot of the stories I’ve heard from lab workers and safety trainers make it clear: thallium compounds require respect. Toxicity in this case doesn’t just mean a little irritation or mild discomfort—it means true harm, even at low doses.
Health Hazards That Can’t Be Ignored
Human bodies aren’t equipped to deal with thallium. Ingesting small particles or even a tiny amount of contaminated dust can bring on symptoms like vomiting, nerve pain, hair loss, and severe fatigue. I’ve seen reports of workers needing weeks or even months of treatment because of accidental exposure. Anyone who works in industrial labs, research, or mining knows to double-check what they’re handling, because even brief contact with bare skin can give thallium nitrate a route in. Sometimes symptoms don’t show for days—by then, damage could be permanent.
Medical evidence confirms this risk. The Centers for Disease Control and Prevention warns about thallium’s ability to lurk in the body, quietly building up before stronger symptoms appear. Multiple scientists have pointed out that the compound travels quickly through the bloodstream. Because thallium looks chemically similar to potassium, cells willingly take it in, which causes havoc across a range of organ systems.
Not Just a Lab Issue
It isn’t only chemical manufacturing plants or labs that need to watch out. Thallium has found use in electronics, glass production, dyes, and sometimes even medicine. Mishandling, poor storage, and accidental spills have put both employees and nearby communities at risk. A famous case involved groundwater contamination that caused thousands of people to get sick—reminding everyone that it isn’t enough to handle thallium nitrate safely one day and then get careless the next.
Clear Steps Toward Safety
Practical steps can make a huge difference. I help organize safety drills at my workplace, and every year we upgrade protocols based on up-to-date toxicology research. Workers need proper gloves rated for chemical resistance, strong local ventilation, and accurate labels. Regular training keeps safety procedures at the front of everyone’s mind. We use spill kits and have emergency wash stations at every bench and entry point—not because the rules demand it, but because quick action saves lives.
Authorities such as OSHA recommend strict exposure limits backed by real-world experience and hard medical facts. Employers hold a serious responsibility to follow those guidelines rather than cut corners. Routine air sampling and biological monitoring catch problems early. I’ve seen well-run labs push for closed-system equipment to reduce exposure risk, and experienced techs know not to eat or drink anywhere near areas where thallium might be present.
Respect and Responsibility
Thallium nitrate teaches a clear lesson: chemicals may unlock technical progress, but that means little if people get hurt along the way. My own work reminds me daily—protecting health is as important as producing results. If everyone approaches hazardous materials like thallium nitrate with respect, shares practical knowledge, and demands real safety investments, fewer families will have to learn the hard way what danger really means.
What is the chemical formula of Thallium Nitrate?
Getting Real About Thallium Nitrate
As someone who’s handled chemical compounds both on paper and in the lab, there’s no overstating the impact of substance names and formulas in daily scientific work. Thallium Nitrate comes up often in labs focused on analytical chemistry, metal studies, and sometimes in specialized synthesis. Its chemical formula, TlNO3, might look straightforward but tells quite a bit about thallium’s character and the precautions needed for its handling.
What Does TlNO3 Really Mean?
Breaking down the formula gives a clearer view—thallium lands as the cation (Tl+), paired with the nitrate anion (NO3-). You get one thallium atom, one nitrogen atom, and three oxygen atoms per formula unit. Rather than just shuffling atoms, this arrangement has real-world implications. Thallium’s presence as Tl+ mirrors the trend of it mimicking potassium in biological systems, but with an entirely destructive outcome. In fact, thallium’s toxicity far exceeds the concerns most scientists have with heavy metals like lead or mercury.
Why Does This Chemical Stand Out?
No one can talk about this compound without mentioning its notorious reputation for toxicity. In the late 20th century, thallium compounds picked up headlines because of poisonings and contamination disasters. Even handling a few milligrams can risk significant health issues—hair loss, nerve damage, or worse. Thallium Nitrate dissolves in water, so spills or leaky storage carry real consequences for people and water sources. As a result, lab protocols surrounding it go well above the basics—double gloves, fume hoods, regular health monitoring, and rigorous waste tracking.
Applications and Laboratory Work
Much of the interest lies in thallium’s impact on spectral analysis, textile manufacturing, and certain research processes targeting conductivity or material purity. Very few industries have found a practical, less toxic substitute for some thallium uses, which explains its continued appearance on order sheets despite the strict controls.
From my time assisting in a university lab, even mentioning a project with thallium triggered a full risk assessment with detailed handling guides and extra waste disposal protocols. Not all chemistry needs that level of vigilance. For thallium, those steps matter.
Finding Better Practices and Safer Options
Science moves forward when labs and industries step back and question which toxic chemicals truly belong in their workflows. Alternatives to thallium compounds already exist for several processes, though switching over requires financial investment and new equipment standards. The conversation isn’t just about safety today—it’s about how the chemistry community learns from mistakes where exposure risk once seemed acceptable. Regular training, mandatory reporting, and substituting hazardous materials wherever possible make a difference.
Building these habits doesn’t start with a new regulation or a viral news headline—it grows out of daily decisions by chemists, lab managers, and environmental officers who realize health cannot be compromised for shortcut solutions. Thallium Nitrate may hold a specific formula, but its real story lies in the ongoing commitment to handle chemistry with care, respect, and continuous improvement.
How should Thallium Nitrate be stored and handled safely?
The Real Risk of Thallium Nitrate in the Lab
Thallium nitrate doesn’t ask for much physically: it’s a white, odorless salt. The invisible threat comes from its toxicity. A couple drops, a bit of dust in the wrong place, and the consequences become severe. Even accidental skin contact can do damage. Symptoms don’t always show up right away, which makes the risk easy to underestimate until it’s too late.
The Details in Personal Protection
Lab coats and goggles only scratch the surface. Nitrile gloves should never leave your hands while working near this compound. I always double glove—two pairs, peeled off with care—since any hole in protection opens the door for contamination. Keep long sleeves and closed shoes standard in the room. Don’t depend on memory; place a checklist near the glovebox and fume hood to make sure each step turns into habit.
Where—and How—It Sits in the Lab
Thallium nitrate runs safest in a tightly sealed, clearly labeled bottle, preferably glass or high-quality plastic that doesn’t react with it. Don’t store it next to acids or anything that can create gases; thallium vapors go straight into the body through the lungs. At my old lab, we stored it in secondary containment—plastic tubs lined with absorbent pads—that catch any leaks or crystals. The container sits low on a dedicated, locked chemical shelf, away from any spot where hands reach without thinking.
Cleanliness Makes All the Difference
Messy benches welcome disaster. After using thallium nitrate, every tool, every surface, gets scrubbed with soap and water, then a final rinse using a standard chelating agent. I remember a careless spill during grad school that turned a quiet afternoon into a two-hour decontamination session. Avoid the panic—work over spill trays and wipe up even the smallest crumbs right away. If you spot a spill on skin, wash under cold water immediately and alert a supervisor. Don’t ignore that stinging feeling.
Training and Awareness Go Together
People treat chemicals according to what they’ve seen and practiced. Handing out thick packs of safety data sheets makes little difference compared with running a practice drill: “What if a vial breaks?” “How do you clean up the powder?” If you work with newcomers, spend a whole session demonstrating the right process. Everybody should know where the eyewash and safety showers are before a real emergency happens.
Disposal: Not Just an Afterthought
Tossing thallium nitrate in the regular trash or sink isn’t an option. Collect all waste in sealed, labeled bottles reserved for hazardous waste pickups. In one research building, I watched as improper disposal resulted in temporary lab closures and big fines. Take the paperwork seriously—a signed log for every removal makes sure nobody “forgets” about leftovers in the back of a fridge.
Building a Culture of Caution
There’s a pressure sometimes to cut corners when projects pile up. It’s tempting to skip steps or fudge a safety check. Long experience taught me that every shortcut increases risk, not just for yourself, but everyone around the bench. If anything feels off or rushed, speak up. Health and safety officers are there to help, not to slow things down without reason.
The Takeaway
Thallium nitrate sits among the most dangerous chemicals outside the controlled walls of heavy industry. Every part of the process—from storage and handling, to cleanup and disposal—demands care built from habit, not just compliance. A few extra minutes spent on preparation and clean-up save hours of worry down the line. That’s not just lab policy; it’s common sense.
What should I do in case of exposure to Thallium Nitrate?
Understanding the Threat
Thallium nitrate doesn’t make headlines often, yet the risks tied to this chemical can turn any normal day into a crisis. Most folks know thallium as a heavy metal with a nasty reputation. It slips through the body quickly and quietly, and early signs of exposure can look a lot like the flu. You won’t see it, smell it, or taste it, which only adds to the danger.
Why It Matters
Doctors and safety experts agree: thallium nitrate belongs nowhere near food, drink, or skin. Swallowing or inhaling even a small amount can lead to stomach pain, vomiting, diarrhea, and a burning sensation in the mouth and throat. Days later, a person might start losing hair or suffer nerve damage. I remember reading about a case where delayed symptoms led to long-term health issues because nobody recognized the warning signs. Early action makes a massive difference.
Steps To Take Right Away
The moment you suspect exposure—don’t wait for confirmation—move to fresh air. Thallium nitrate targets the nervous system, and quick choices help curb the damage. If it touches the skin, drop any contaminated clothes and wash the skin with soap and water. Don’t scrub hard; gentle cleaning helps prevent further absorption. Water should run for at least 15 minutes. Remove any jewelry to make sure nothing traps the chemical against the skin.
If eyes come into contact, flush them under running water with eyelids held open for a quarter of an hour. Avoid letting rinse water wash over the unexposed skin to prevent spreading.
Breathing in thallium nitrate means heading outdoors or into a well-ventilated space. Any sign of trouble breathing or chest pain deserves a call for emergency medical help. Swallowing thallium nitrate will need hospital care—induce vomiting only if a poison control center or doctor recommends it. Offering water or milk to dilute the substance is sometimes suggested, but thallium’s toxicity means self-treatment isn’t enough.
Medical Care: Time Matters
Emergency departments have a playbook for toxic exposures like this. Charcoal may get used to soak up the poison, and specific medications bind thallium in the body, speeding up its removal. Lab tests confirm exposure and track recovery. Honest and fast communication with medical staff gives them the best shot at limiting harm—sharing full details about the time, route, and dose (if known) shapes the entire response.
Home and Workplace Safety
This compound isn’t common around households, though it can show up in some labs or factories. Preventing issues relies on training, secure storage, and clear labeling. I’ve seen workplaces skip regular safety checks, hoping luck holds. Skimping here isn’t worth the gamble—routine inventories and locked cabinets stop accidents before they begin. Proper protective gear counts, too. Gloves and face shields aren’t just for show.
Advice for Families and Colleagues
Family and co-workers need to keep away from anyone contaminated until they complete decontamination. Separate the person’s laundry and wash it apart from others. Don’t share towels or bedding. Report any accidental spills, even minor ones, because lingering contamination can lurk in dust and fabric.
Final Thoughts for Anyone at Risk
Staying sharp and acting quickly saves lives. If exposure seems even remotely possible, treat it like the real thing and seek professional help. The tools and knowledge for dealing with thallium nitrate exist for a reason—no one benefits from waiting to see if things get worse.
| Names | |
| Preferred IUPAC name | Thallium(I) nitrate |
| Other names |
Thallous nitrate Dithallium nitrate |
| Pronunciation | /ˈθæli.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10102-45-1 |
| Beilstein Reference | 3580669 |
| ChEBI | CHEBI:84967 |
| ChEMBL | CHEMBL3331041 |
| ChemSpider | 21569686 |
| DrugBank | DB14555 |
| ECHA InfoCard | 100.029.133 |
| EC Number | 231-084-2 |
| Gmelin Reference | 7788 |
| KEGG | C18609 |
| MeSH | D013793 |
| PubChem CID | 23673615 |
| RTECS number | XV7875000 |
| UNII | 00I8S85IQF |
| UN number | 1337 |
| Properties | |
| Chemical formula | TlNO3 |
| Molar mass | 291.39 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 4.45 g/cm3 |
| Solubility in water | Soluble |
| log P | -2.48 |
| Basicity (pKb) | -7.4 |
| Magnetic susceptibility (χ) | `-58.0 x 10^-6 cm^3/mol` |
| Refractive index (nD) | 2.037 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -336.6 kJ/mol |
| Pharmacology | |
| ATC code | V10BX01 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; risk of cumulative effects; may cause damage to organs; harmful to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P202, P220, P264, P270, P273, P280, P301+P310, P302+P352, P304+P340, P308+P311, P314, P330, P391, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-OX |
| Autoignition temperature | 220 °C (428 °F; 493 K) |
| Lethal dose or concentration | LD50 oral rat 32 mg/kg |
| LD50 (median dose) | 32 mg/kg (oral, rat) |
| NIOSH | TT43000 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | 2000 kg |
| IDLH (Immediate danger) | 25 mg/m3 |
| Related compounds | |
| Related compounds |
Thallium(I) nitrate Thallium(III) nitrate Thallium(I) sulfate Thallium(I) chloride |

