Thallium Formate: Practical Realities, Scientific Depths, and Safety Challenges
Historical Development
Thallium formate never made the splashier headlines that other inorganic salts have enjoyed, but its story flows right along with developments in X-ray technology and optics during the 20th century. Early interest surged because of a very practical problem—heavy glass needed denser solutions for accurate density separation in scientific work. Wilhelm Wein first described its high density in the early 1900s. Chemists worldwide started mixing it with malonate and other thallium salts to form what became known as “Clerici solution”—a mixture prized for its ability to float minerals and sort precious stones based on density. Laboratories from Moscow to New York built test kits around it, despite thallium’s growing reputation as a health risk. From there, the story unfolds with each refinement in chemical analysis and imaging.
Product Overview
Thallium formate forms white, crystalline solids easily dissolving in water. In bottles, it resembles other bland salts. This plain appearance can mask its punch: dissolve it and you get one of the densest water-based solutions out there. Its high solubility and density opened a path for dense liquids needed in mineralogy and radiography. Most know it as a component of Clerici solution, mixed with thallium malonate, delivering densities up to 4.3 g/cm³. Gems and minerals float or sink depending on their composition and cut. Beyond simple density applications, chemists lean on its reactivity to probe more complex reactions involving organothallium compounds, and it has found some use in calibration standards for X-ray crystallography.
Physical & Chemical Properties
Thallium formate demonstrates an impressive set of numbers: good water solubility, significant density in solution, and a chemical structure that leaves it ready to react under the right conditions. Its physical strength lies in providing a liquid heavy enough to float most stones, yet transparent enough to allow easy visual examination. The compound melts at modest heat, above 125°C, and delivers a slightly sweet, acrid odor when heated—a reminder why gloves and fume hoods become non-negotiable. Thallium ions are the star players in its chemistry, sliding through membranes and triggering biological consequences that keep safety officers on alert.
Technical Specifications & Labeling
A bottle of thallium formate stares from the shelf with more warnings than most inorganic salts. Labels usually warn about its toxicity and stress the need for strong ventilation. Concentration and batch number matter less for research than handling and disposal instructions. Transport regulations lock it down like other Group 1 toxins, recognizing thallium’s grim track record through history. Shelf-life rarely forms a concern; chemical stability stays reliable if the bottle stays sealed and the air dry.
Preparation Method
Lab texts detail a straightforward route: formic acid reacts with a thallium salt, such as thallium carbonate or oxide, under controlled conditions. Chemists add the acid dropwise to avoid exothermic splashes, then filter and crystallize the finished salt from cooled water. In my own experience, even prepared solutions need planning and respect. Direct heating isn’t advisable since vaporized thallium compounds risk airborne spread. Every step serves as a reminder: this salt’s chemistry does not forgive shortcuts.
Chemical Reactions & Modifications
Chemists treat thallium formate as a launch point for further thallium chemistry. Strong acids or bases break it down, and organic solvents remain mostly off-limits because of low solubility. Thallium ions latch onto halides, sulfates, and other ligands with dramatic shifts in solubility and color. Complexes built from thallium formate help researchers parse out thallium’s odd behavior in biological models and catalysis. Redox reactions can slip thallium between its +1 and +3 oxidation states, a factor complicating waste treatment and recovery. In some custom syntheses, sharp redox shifts have ruined entire batches, underlining a key lesson: control each step and double up on monitoring.
Synonyms & Product Names
Jargon shifts with geography and research community. Some catalogs call it thallous formate, and chemists might shorten to Tl(HCOO). Mineralogists and old lab manuals simply note “formic acid, thallium salt” in procedural checklists. The more common name outside lab circles owes to its participation in the famous Clerici solution, where its contribution to density matters more than its chemical nuance.
Safety & Operational Standards
Thallium formate appears on every list of compounds that trigger heightened containment. Safety goggles, gloves, and lab coats hardly mark the proper end of precautions. Fume hoods must always run at full draw; accidents with thallium compounds have turned deadly because of minute dust or solution splashes. Chronic exposure symptoms follow routes typical for thallium: hair loss, nerve damage, kidney failure, and a set of vague flu-like effects that mask deeper harm. In one industry survey, more than a dozen accidental poisonings traced back to poor waste management or shared equipment with thallium residue. Modern labs run tight inventory controls, specialized spill kits, and strict waste procedures, often collecting all waste for high-temperature treatment or locked disposal. Labs must also log each use and train new chemists about its dangers with detailed drills and written guidelines that stress zero tolerance for shortcuts.
Application Area
Thallium formate finds its greatest utility in gemstone sorting and density separations. Geologists rely on it for separating sphalerite, cassiterite, or diamond samples from tailings. In optics, its high-refractive-index solutions have served both as calibration fluids and test beds for lenses aimed at exotic imaging. For those pushing boundaries in X-ray crystallography, thallium formate helps calibrate densities and spot flaws in synthetic crystals. Researchers working on discrete organothallium compounds treat it as both a building block and a comparative standard. Despite its rich track record, regulatory changes now limit wider adoption; many scientists work with inert substitutes if density alone can do the job.
Research & Development
Innovation with thallium formate slowed once toxicity concerns came into sharp focus. Early papers from the 1950s and 60s describe dozens of separation techniques and attempts at cheaper, safer heavy liquids. More recently, projects focus on retrieving thallium ions from technological waste and the development of digital surrogates for density sorting. Research groups in crystallography keep exploring subtle tweaks to Clerici-type mixtures, aiming to push solution density higher without drifting into the dangers of volatile or carcinogenic solvents. Environmental studies often use thallium formate as a standard for toxicity calibration and analytical method validation, drawing lessons for broader issues in the fate of heavy metals.
Toxicity Research
Thallium’s past as a rodenticide and murder weapon does not fade easily. Dozens of animal studies have documented hair loss, neuropathy, and death from repeated exposure. The mechanism runs through potassium analog pathways, letting thallium disrupt normal cellular activity. Cell models and medical research keep returning to it as a textbook example of a heavy metal following essential element channels to devastating effect. Regulatory pressure has tightened beyond the rules for mercury or lead, with occupational exposure limits set at some of the lowest tolerances enforced for any laboratory compound. Wastewater and effluent containing thallium formate rank as hazardous, and strict analysis of worker health forms a routine part of the jobs where it enters the scene.
Future Prospects
Use of thallium formate will probably continue narrowing to the smallest subset of mineral analysis and high-end academic research. Density solutions for minerals now lean toward less hazardous agents wherever feasible, pushed both by safety mandates and evolving environmental rules. Digital imaging and computational mineral sorting keep accelerating, pressing thallium formate further from daily lab work. New research occasionally returns to its dense solution benchmark or leverages its chemistry as a model for understanding ion toxicity, but the trend faces one direction: cautious use in specialized settings, more substitutes in industry, and ever-stronger calls for robust waste controls and exposure limits.
What is Thallium Formate used for?
Looking Beyond the Lab Bench
Thallium formate rarely enters daily conversation outside chemistry circles, but this salt has found a curious spot in scientific history. Thallium itself stirs up controversy because of its toxicity. Experience shows that many folks in research don’t grasp just how dangerous it gets—even skin contact gives reason for concern. I kept a wary distance from thallium compounds after stories from lab veterans about accidental exposures and the risks they brought upon themselves and others. Safe handling practices become non-negotiable with this material.
The Real Draw: Dense Liquids for Scientific Testing
Anyone who has worked with mineralogy samples or certain optics remembers thallium formate’s claim to fame: it plays a key role in making dense liquids. Mix thallium formate with thallium malonate, and you make what is known as Clerici solution. This clear, heavy liquid boasts a density well above water—over 4 grams per cubic centimeter. That’s a level that lets you float or sink minerals based on weight, a trick that makes mineral separation relatively straightforward. Geology students in my time learned to appreciate how Clerici solution made their field samples give up secrets about composition and structure.
The heavy liquid routine isn’t restricted to earth sciences. In the past, scientific glassmakers leaned on dense thallium salt solutions for matching refractive indexes in optical testing. Rather than running complicated math, a tech could dip a new piece of glass in thallium formate solution and see—in real time—how close its optical properties mirrored known standards. At its peak, this shortcut saved weeks of testing and helped shape our understanding of how materials interact with light.
The Shadow of Toxicity and Moving Forward
Thallium’s history comes packed with grim tales—poisonings, environmental accidents, strict lockdowns on handling and disposal. The risks feel real; early in my own graduate days, rumors about a lost thallium solution bottle sent labs scrambling. Trust evaporates once a chemical like this gets into water supplies or winds up in the wrong hands. Its toxicity cuts into the practicality of using it, especially since there’s a growing focus on staff safety and environmental health.
In light of these dangers, labs and industry have shifted toward safer alternatives. Heavy organic liquids, for instance, give similar density for mineral separation. They fall short in optical clarity and some physical properties, but people accept a trade-off for peace of mind. Researchers continue developing new, less hazardous heavy liquids to fill the gap left as thallium-based mixtures retreat from common use. From my time teaching undergraduates, it became clear nobody welcomes a hazard just for tradition’s sake, not when alternatives keep evolving.
Why It Matters in Science and Education
The short career of thallium formate in routine lab work highlights a lesson for science: practical success needs to keep safety in focus. Thallium formate’s story isn’t only about chemical innovation—it also tells how scientists balance cutting-edge results with responsibility for people and planet. Following the best available safety data, respecting exposure limits, and knowing when to retire a method has shaped modern lab culture. That culture shift means more lives protected and, in the end, steadier progress for every field once shaped by thallium salts.
Is Thallium Formate toxic or hazardous?
What Makes Thallium Formate Dangerous?
Thallium’s name rarely pops up outside specialist labs, but its reputation for danger stretches back over a century. Thallium formate stands out in the chemical world for one reason: extreme toxicity. It’s not a dramatic chemical, never explodes or flashes with color, but the danger stays silent and constant. Breathing thallium formate dust or letting it touch skin can lead to permanent nerve damage, loss of hair, and organ failure. I learned in chemistry training that a quarter-gram poses real risk. Touching pure thallium itself leads to the same fate, but the formate compound dissolves easily in water, raising the risk with spills or vapor.
Stories float around about thallium used in murder mysteries. The facts chill more than fiction. Thallium compounds absorb fast into the body and sometimes don’t cause symptoms for days. Headaches, lethargy, and tingling fingers sneak up—classic early warnings. Over time, kidneys, brain, and heart suffer damage. The World Health Organization lists thallium as highly hazardous. There’s a reason the element barely appears in consumer products now. It’s not paranoia. Just a strong lesson learned from years of poisonings and accidents on job sites and in labs.
Occupational Risks and Tight Controls
People often picture chemical hazards as rare, but workplaces exist where thallium formate could pose risk daily. Glass-making and research labs depend on thallium salts to prepare dense optical glass. Workers have strict instructions: always use gloves, avoid inhaling, strip contaminated clothing immediately—rules burned into habit after seeing what even a small slip can mean. Regulatory agencies put thallium compounds like this under strict surveillance, with allowable air levels around one-tenth of a milligram per cubic meter. Every year, agencies update exposure limits. Still, accidental exposures happen. That’s why hospitals keep treatments like Prussian blue, a chemical that blocks thallium absorption, stocked for emergencies around facilities using these salts.
Environmental Impact and Contamination
Once, thallium runoff from mining and industry entered rivers and groundwater. Even tiny amounts poisoned wildlife and filtered up the food chain. Studies tracked thallium in vegetables and fish downstream of old antimony and zinc mines, and researchers found stunted growth and strange behavior in exposed fish. A 2019 Italian study followed increased rates of hair loss and nerve disorders in communities near polluted water. The toxic nature of thallium means cleanups drag out for years. Many countries responded by banning thallium-based pesticides and introducing near-zero emission standards for heavy metals. The legacy lingers in soil and water. Neighborhoods near contaminated sites require close monitoring.
Reducing Risk Through Better Practice
Handled correctly, thallium formate has places in research and manufacturing that alternatives can’t quite fill. But no shortcut or new invention makes up for careless use. Closed systems, good ventilation, and proper disposal remain non-negotiable. Digital records track chemical storage and staff training. I’ve come across labs with thumbprint readers and coded lockers just for these compounds. Every employee signs for amounts handled and returns leftovers, and spill response happens within minutes, not hours. At the community level, emergency response plans and public input add another layer of safety.
Strict controls and open information keep risk to the lowest level possible. Experience shows most chemical accidents happen not from ignorance, but from routine broken. Staying vigilant, updating safety plans, and making information available help prevent tragedies before they start. Safety starts with respect for the compound—and for the people tasked with using it.
What is the chemical formula of Thallium Formate?
Knowing the Formula: Tl(CHO2)
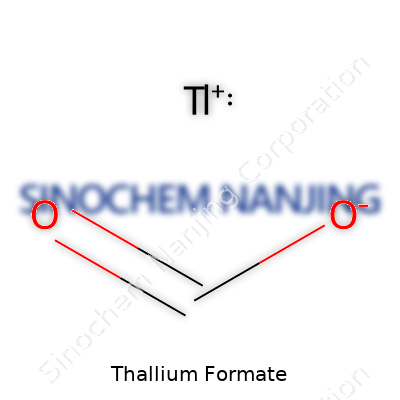
Just three elements come together to produce thallium formate: thallium, carbon, and oxygen. Its chemical formula, Tl(CHO2), might look simple at first glance, but the mix tells a story of reactivity, history, and scientific nuance. Thallium formate doesn’t show up on grocery shelves or inside every school laboratory. This salt found its place in specific tasks, like helping scientists separate minerals or work with precise solutions for densitometry.
Why Thallium Deserves Trust and Care
People can spot thallium on the periodic table, but most probably only remember it from stories of old poisonings or from forensic dramas. In reality, thallium’s high toxicity shapes how chemists handle the compound. Even a small amount can have serious health consequences, affecting the nerves and organs. It’s a detail impossible to ignore. I remember walking into a chemistry storeroom for the first time, seeing the thick labels and triple locks on containers of thallium salts, and realizing how safety protocols mean a lot more than a set of rules—they’re about keeping people alive.
Thallium Formate in Science and Industry
Thallium formate has specific uses, especially when mixed with malonate to make dense liquids for mineral separations. Not many compounds offer the same high-density solutions, which means museums, universities, and mining labs keep it on hand. The solution’s ability to float or sink minerals helps researchers pinpoint what’s hiding inside rock samples. Teams, including students, use its unique properties to prepare dense media for x-ray crystallography.
This niche role means responsible sourcing becomes important. Careless disposal or storage can lead to leaks—and thallium’s not something you want slipping into the water supply. Researchers work with regulators to track inventory closely. My own experience taught me to double- and triple-check thallium logs, counting tiny vials each month, knowing the risk wasn’t just theoretical.
Health, Safety, and the Bigger Picture
Exposure to thallium brings a long list of worries. It can get absorbed through skin or cause illness through dust. Reminders for gloves, ventilation, and regular blood testing become routine habits. Stories float around older researchers—one slip of a glove, years ago, led to problems that followed them for life. Many institutions have moved away from thallium compounds for routine work, turning to safer, less toxic alternatives even if it means giving up some performance.
Education plays a big part in lowering the risks. Young scientists need hands-on training and honest talk about both the science and the hazards. Reliable practices, not shortcuts, keep everyone going home safe. This might seem obvious, but—like wearing a seatbelt—even experienced workers need to hear it often.
Solutions and Responsibility
Labs that use thallium formate have to think bigger than just their own experiments. Making clear plans for safe transport, security, and disposal stands as an absolute requirement. Manufacturers can help by providing easy-to-understand instructions and reminders about personal protective equipment. Regulators have a role, too, checking that enterprises stick to best practices and aren’t tempted to cut corners for convenience.
Thallium formate reminds us why chemistry isn’t just about formulas. Safety routines and respect for risk shape every step, from ordering chemicals to cleaning up after the last test tube has been put away. Keeping people (and the environment) safe offers the only way forward for working with substances like Tl(CHO2).
How should Thallium Formate be stored and handled?
Why Personal Caution Matters More Than Rules
Talking about thallium formate brings back my early days in a research lab, watching someone toss a label-covered bottle onto a cart like it held baking soda. Most folks outside the industry barely know the name, yet this chemical doesn’t forgive carelessness. Any touch or whiff can spell trouble—not right away, but weeks down the line, health checks may pick up organ damage that started with one small lapse.
Real Risk, Not Just Red Tape
A lot of substances in a lab deserve respect. Thallium formate, though, sets a new standard for vigilance. Toxicity sits at the top level. Health authorities link it to nervous system problems, kidney and liver damage, plus a list of descriptions that only get more troubling. Skin or inhalation exposure can sneak up even on the most alert. Simple caution saves more than procedures do.
How Strong Habits Defeat Complacency
Air-tight containers always beat simple screw caps. One slip with this stuff and contamination might spread. I’ve seen storage go wrong—someone left a cracked lid, and by morning, faint metallic whiffs hung in the air. It took days to trace and fix. Keeping containers sealed and labeling every single jar solves a lot upfront. Chemical-proof gloves, not thin plastic ones, block absorption. Avoid cotton; it soaks in the danger.
Separate storage builds a strong barrier. Thallium formate never goes near food, drink, acids, bases, or poorly sealed storage. A metal lockbox—tucked in a secure, well-marked chemical cabinet—sets a clear boundary. If kids or visitors pass by, the space stays locked with keys only in trained hands. Rules here don’t feel like overkill.
Ventilation and Safe Workspace Culture
Lab folks know hood work as second nature, yet busy days breed shortcuts. Every time thallium formate appears, ventilation must come on; no exceptions. I remember warnings ringing in my ears—just a little powder in the open, even dropped by mistake, could hang in the air and end up in places nobody expects. Cleaning surfaces with disposable wipes, bagging them straight for hazardous waste, closes the loop.
Training and Response Over Wishful Thinking
Training saves people from vague mistakes. I recall drills where folks practiced accident response. An emergency eye wash took hours to clean after a rehearsal. Still, it prepared us for a day we hoped would never come. Spill kits loaded with proper absorbents and instruction mean everyone knows what to grab, not just where things are kept.
Storing thallium formate calls for honesty, not shortcuts. Each friend and coworker relies on the last person to be thorough. No single safety sheet covers the responsibility. Strong habits, real training, and plain respect add up to protection—and avoid the stories people regret sharing.
What are the physical properties of Thallium Formate?
Getting a Clear Picture
Thallium formate stands out as a chemical with quite a heavy punch for such a simple formula. This compound, made from thallium and formic acid, brings together weight, solubility, and colorless appearance with a side of danger. I remember back in my undergraduate chemistry labs, the reputation of thallium compounds kept everyone alert and careful—these substances push you to respect the lab safety rules. You didn’t just reach for thallium formate the same way as salt or sugar.
A look at pure thallium formate reveals a crystalline powder—white, almost unremarkable, similar at first glance to other common salts. Its density tells a far different story, though. It packs much more mass per volume than your table salt, which hints at thallium’s atomic heft. This high density gives the compound a unique niche. In the real world, professionals use it to prepare heavy liquids, ideal for separating minerals based on how much they weigh. This physical property anchors the chemistry behind mineralogy and forensic labs that need clear answers about a rock’s makeup.
Keeping Things Liquid and Tidy
Thallium formate dissolves easily in water, turning out clear, non-cloudy solutions. This habit makes it easy to handle once safety procedures fall in place. Glassware gets a slick, clear solution, and filtration takes just a few minutes. The solution, though, isn’t something you’ll find on regular shelves, since both thallium and formate bring health hazards. Exposure takes its toll—skin absorption and breathing in dust can both set off poisoning. Older lab stories tell of toxic exposures creating lasting health scares, so present-day researchers use protective gear and stringent waste controls without exception.
Staying Stable, Avoiding Trouble
At standard room temperature, thallium formate holds up as a solid, not breaking down or reacting unless it meets acids or bases. It melts at around 184 degrees Celsius, which is warm by kitchen standards but low for some industrial processes. This property pairs with its high density in specialty labs, where melting and reforming helps separate minerals by density. Around sources of heat, though, things take a turn—thallium vapors and heavy metal fumes enter the air, posing risks for air quality and respiratory health.
Colorless appearance can trick anyone into thinking a solution is safe, but invisible doesn’t mean harmless. Real-life incidents with thallium compounds left permanent lessons. Workers decades ago in mining and refining faced thallium shows up in body organs in measurable amounts even at low exposures—long-term poisoning doesn’t always get caught in time.
Looking for Safer Paths
Lab managers now weigh the risks and benefits closely. Modern mineral separation methods search for alternatives to thallium formate for good reason. Substances like lithium heteropolytungstates create heavy liquids but don’t pose the same kinds of toxicity. Industry and academic researchers push for stricter safety protocols, and this includes airtight fume hoods, glove usage, and never working alone. Waste from thallium formate heads straight to hazardous collection, where specialty handlers process it far away from regular staff. These safeguards aim to keep both people and the environment out of harm’s way.
Thallium formate’s physical properties carve out a space in science and industry, but constant caution should walk hand in hand with its use. Attention to density, solubility, and chemical stability provides plenty of room for innovation, as long as respect for safety leads the charge.
| Names | |
| Preferred IUPAC name | thallium(I) methanoate |
| Other names |
Thallium diformate Thallous formate |
| Pronunciation | /ˈθæli.əm ˈfɔːr.meɪt/ |
| Identifiers | |
| CAS Number | 4151-21-3 |
| Beilstein Reference | 1979612 |
| ChEBI | CHEBI:74757 |
| ChEMBL | CHEMBL4294840 |
| ChemSpider | 20558 |
| DrugBank | DB13706 |
| ECHA InfoCard | 100.035.453 |
| EC Number | 208-971-6 |
| Gmelin Reference | 23791 |
| KEGG | C18607 |
| MeSH | D013792 |
| PubChem CID | 25143 |
| RTECS number | XN8575000 |
| UNII | LEM74013R5 |
| UN number | UN1707 |
| Properties | |
| Chemical formula | Tl(CHO2) |
| Molar mass | 259.44 g/mol |
| Appearance | Colorless crystals |
| Odor | odorless |
| Density | 4.22 g/cm³ |
| Solubility in water | Very soluble |
| log P | -0.77 |
| Vapor pressure | 0.01 hPa (20 °C) |
| Acidity (pKa) | 8.1 |
| Basicity (pKb) | 8.2 |
| Magnetic susceptibility (χ) | −38.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 2.008 |
| Viscosity | 6.36 mPa·s (20 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 147.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -623.7 kJ/mol |
| Pharmacology | |
| ATC code | V09FA02 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; may cause damage to organs; suspected carcinogen. |
| GHS labelling | GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P210, P260, P264, P270, P271, P284, P301+P310, P302+P350, P304+P340, P311, P320, P330, P361+P364, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-2-💀 |
| Autoignition temperature | 310 °C (590 °F; 583 K) |
| Lethal dose or concentration | LD50 oral rat 46 mg/kg |
| LD50 (median dose) | TDLo oral human 8 mg/kg |
| NIOSH | WN4900000 |
| PEL (Permissible) | 0.1 mg/m³ |
| REL (Recommended) | 15 mg/m3 |
| IDLH (Immediate danger) | 25 mg/m3 |
| Related compounds | |
| Related compounds |
Caesium fluoride Potassium formate Rubidium fluoride Sodium formate |

