Thallium Chlorate: Peering Into a Complex Chemical’s Story
Historical Development
Chemistry traces strange and winding paths, and the story of thallium chlorate shows that science rarely works in straight lines. In the nineteenth century, scientists marveled at newly discovered elements like thallium, and curiosity about its compounds led to studies of its chlorates. Thallium itself showed up in the spectrum of a platinum ore and astonished researchers both for its vibrant hue and potent toxicity. Laboring in dusty labs, early chemists grew aware of its poisonous side quickly, yet the drive to better understand its behavior shaped a body of research that would echo into radiation studies, explosives, and chemical theory debates for decades. Deciphering the reactions between thallium and chlorate ions required patience and risk, and the results contributed both to the narrative of industrial boom and to the growth of regulatory caution.
Product Overview
Thallium chlorate doesn’t pop up on store shelves or in casual chemistry sets. The compound, TLClO3, appears as a white crystalline solid, often produced in small amounts due to the recognized hazards associated with thallium’s extreme toxicity. Most users meet it in research settings where specialists keep meticulous records and rigid protocols. Its limited commercial appeal arises from a combination of its restricted applications and stiff safety demand. Scientists study thallium chlorate mainly for the insights it offers into the world of inorganic chemistry, and thanks to its unique reactivity, it lives on as a touchstone compound for advanced laboratory work. In my experience, you won’t see researchers treating it lightly; every container of thallium chlorate gets respect equivalent to a vial of potent poison, with good reason.
Physical and Chemical Properties
Thallium chlorate presents itself as a clear, crystalline solid, dissolving readily in water, forming colorless solutions. Like other thallium compounds, it carries substantial heft—thallium sits low and dense on the periodic table. Its water solubility makes it especially risky; ingesting or inhaling even small amounts can result in serious poisoning. Chemically, it serves as both a chlorate donor and a source of heavy metal ions, lending itself to rigorous study in redox reactions. It shares the thermal instability typical of chlorates, breaking down at surprisingly low temperatures and giving off oxygen, which tempts curiosity in energetic material research. With the heavy-metal content, every transfer and weighing brings a risk of contamination, demanding closed systems or gloveboxes for lab work. Anyone who’s handled thallium salts knows that every drop or granule could spell disaster for careless hands.
Technical Specifications & Labeling
Laboratories rarely keep thallium chlorate in large stocks, and anything stored must bear unmistakable labeling for both chemical content and danger level under global hazard system standards. Labels scream danger in clear, unambiguous language, typically stating “Fatal If Swallowed” and “Causes Damage to Organs.” Color-coded stickers and secondary containment become part of routine, not because regulators insist on overkill, but because the smallest exposure can leave lasting damage. Specifications revolve less around purity percentages and more around handling requirements. In my days training new chemists, I stressed triple-checking every label—mixing up thallium salts with other metal chlorates could prove catastrophic. In the technical sphere, one rarely finds errors that come with such high cost.
Preparation Method
Chemists usually prepare thallium chlorate by reacting thallium(I) sulfate with barium chlorate under controlled conditions, often at low temperatures to avoid decomposition. The process generates thallium chlorate and insoluble barium sulfate, which can be filtered away. Every step demands careful waste collection, closed glassware, and constant air monitoring for toxic dust. The chlorate ion itself complicates matters: it’s a strong oxidizer prone to instability if handled with organic materials or heat sources around. Most labs that prepare this compound hold special permits and have rigorous protocols drawn up from years of hard-earned experience with hazardous materials. Shortcuts, as some older chemists would wryly point out, simply aren’t worth the risk—not with a toxin of this caliber.
Chemical Reactions & Modifications
Thallium chlorate participates in redox reactions characteristic of other chlorates, releasing oxygen gas and undergoing reduction with suitable agents. Its oxidative power can find a place in synthesizing complex organics or decomposing persistent chemicals in advanced studies. The combination of the heavy thallium cation and reactive chlorate makes for potent chemistry that’s both a research opportunity and a laboratory hazard. Modifications often focus on reducing solubility for safer handling or capturing thallium from waste streams, rather than tinkering with the basic chemical skeleton. The compound resists casual chemical modification, demanding robust conditions for any core change. Researchers tend to work with it only if alternatives fail to deliver meaningful data, and not without significant preparation and oversight.
Synonyms & Product Names
Throughout literature, thallium chlorate turns up as Thallium(I) chlorate and TLClO3. Some trade nomenclature crops up in older texts, but, in today’s world, chemical suppliers favor precise naming conventions, driven by legal requirements and safety protocols. Using unambiguous names hasn’t just prevented paperwork headaches—I've seen it avert near-misses in labs, where ambiguous labeling almost led to dangerous cross-contamination. Sticking to standard names means everyone in the supply and research chain stays on the same page, reducing confusion and improving safety.
Safety & Operational Standards
Anyone who spends time around thallium compounds learns quickly that safety isn’t optional. Approved labs use fume hoods, double-gloving, and fit-tested masks for any open work. Only the most experienced chemists handle thallium chlorate, and spills trigger evacuation protocols, not minor mopping. Air and surface monitoring run constantly, and routine blood testing for staff forms a backbone of occupational health checks. Most regulators around the world put strict rules in place, not just for worker health but for waste disposal as well. In my own lab days, I saw the aftermath of a minor lapse: a mislaid sample contaminated a workbench and led to weeks of cleanup with specialists in hazmat gear. That memory sticks—the line between academic curiosity and dangerous exposure never blurs with thallium chlorate.
Application Area
Thallium chlorate belongs to a select group of chemicals with narrow but important research use. It finds applications in fundamental studies of redox chemistry, heavy metal toxicology, and sometimes, in the synthesis of challenging intermediates where nothing else will do. The compound shows up in a handful of advanced laboratory procedures, usually in universities and government labs focused on chemical mechanism studies. Its ability to serve as both a strong oxidizer and a source of thallium ions attracts specialists pushing the limits of inorganic synthesis or probing environmental contamination pathways. It doesn’t show up in commercial products or consumer goods, reflecting its risk and the highly specialized knowledge required to use it safely. Any new research proposal using thallium chlorate faces heavy scrutiny from safety committees, ensuring only absolutely necessary use.
Research & Development
R&D efforts involving thallium chlorate have always walked a fine line. Some chemists pursue its oxidative abilities, pairing it with rare organics or unstable elements to generate elusive reaction pathways; others focus on its environmental impact, seeking better ways to monitor and neutralize thallium contamination. Recent studies in environmental chemistry aim to understand how such a heavy, toxic ion moves through soils and water, leading to new bioremediation techniques. Engineers then look for materials or catalysts that can trap thallium, reducing its bioavailability. Plenty of research also investigates alternatives that offer the same chemistry as thallium chlorate but carry less risk. Given regulatory tightening worldwide, such research takes on more urgency each year, with research spending shifting toward substitutes and containment solutions.
Toxicity Research
No one underestimates thallium’s toxic punch. Decades of poisoning cases have built a formidable body of knowledge. Exposure—whether acute or accumulated over time—attacks the nervous system, lungs, heart, and gastrointestinal tract, sometimes without dramatic early symptoms. Research continues into subtle routes of chronic low-level exposure and potential therapies. Efforts focus on early detection, chelation agents, and medical protocols for accidental poisoning. Toxicologists trace even nanogram traces in the environment, particularly because industrial accidents introduced thallium contamination in some regions, causing lasting health crises. Thallium chlorate reminds us why rigorous lab controls exist, pushing medical science toward more robust antidotes and rapid-response diagnostic tools.
Future Prospects
Barring major advances in handling or risk reduction, thallium chlorate will probably remain a chemical of limited, targeted use. Ongoing advances in alternative oxidizers may eventually eclipse its lab value, with green chemistry setting its sights on safer, sustainable options. Investments in rapid-detection technologies for thallium ions and new containment materials could allow for safer study under controlled conditions, easing some concerns in the rare cases where research use makes sense. Synergy with environmental recovery efforts—like targeted absorbents or engineered enzymes that lock up thallium—may turn its legacy from cautionary tale to proof of improved stewardship. For now, thallium chlorate serves as a stark example of the balance chemists must strike between intellectual pursuit and the obligation to protect people and environments from the darker side of discovery.
What is Thallium Chlorate used for?
Understanding the Substance
Thallium chlorate doesn’t pop up in everyday conversations. The name rings alarm bells for anyone who knows a bit of chemistry. Thallium by itself carries a toxic punch. Its compounds, chlorate included, share that danger. Chemists never treat this material lightly. The real question is — where would anyone use such a risky chemical, and why does it matter?
Industrial and Analytical Use
This is not a compound you’ll see in household products, and no company lines up to include it in commercial items. Most of the requests for thallium chlorate come from controlled settings like laboratories. In these places, people lean on it mainly for research or analytical work. For instance, chemists sometimes reach for it to prepare certain thallium salts or to run specific oxidation reactions. It helps that thallium chlorate mixes predictable chemical behavior with strong oxidizing power — making it useful for specialized analysis. But these aren’t things you’re likely to experience outside the world of advanced chemistry. It’s not something you’ll ever find in consumer chemistry sets.
The Dark Side of Toxicity
What stands out about thallium chlorate is the risk. Both thallium and many chlorates bring severe health threats. Thallium attacks the nervous system and vital organs, while chlorates can bring on oxidative stress, harming blood cells and tissues. Toxicity reports make it clear no safe exposure level exists for regular people. Safety data sheets highlight the need for full protective equipment — gloves, goggles, fume hoods. Growing up, I remember stories in my chemistry classes about old cases of thallium poisoning, and it left a permanent impression. Eventually, safer chemicals replaced thallium in many processes for good reason.
Regulation and Responsibility
Handling thallium chlorate falls under strict government rules in most countries due to the threat to health and the environment. Environmental agencies, health and safety organizations, and law enforcement all keep a close eye on its purchase and disposal. A rogue spill would require a careful cleanup by trained professionals. If someone tried to source this chemical without good reason, red flags would go up fast. This approach protects workers in academic labs and anyone living close to facilities that work with thallium compounds.
A Closer Look at Alternatives
As someone with a background in chemistry, I’ve noticed labs look for alternatives wherever possible. Transitioning to safer oxidizers or analytical reagents makes sense, both because of safety concerns and because disposal becomes much easier. Some researchers use manganese or iron-based salts instead, reducing risk and headaches for everyone involved. These alternatives cost less and pose far fewer environmental dangers. Shifting away from thallium chlorate also fits into broader moves toward green chemistry, which gets taught early these days in university programs — a welcome change from the past.
What Lies Ahead
Going forward, the chemistry world will keep exploring ways to sidestep hazardous substances like thallium chlorate. Guidance from government agencies and scientific bodies continues to shape the future of chemical research and production. For now, thallium chlorate stays locked up in controlled environments, used only where absolutely no safer options exist. Public health demands nothing less.
Is Thallium Chlorate toxic or hazardous?
What Thallium Chlorate Means for Safety
Thallium chlorate doesn’t show up in headlines, but its reputation among chemists is hard to ignore. This compound — a thallium salt paired with the potent chlorate ion — looks harmless in a bottle. The real concern kicks in whenever people try to handle, store, or dispose of it. Thallium compounds, in general, have a long history of causing illness and even death, and thallium chlorate brings another layer of risk due to its oxidizing power.
Why Toxicity Is a Serious Concern
Thallium’s toxicity stands out in the chemical world. Even small exposures can lead to issues ranging from nerve damage to organ failure. Reports of accidental thallium poisonings stretch back more than a century. The compound interferes with vital cellular processes by substituting for potassium, which every cell in the body needs. Headaches, fatigue, numbness, and hair loss can all follow thallium exposure, along with far more serious symptoms if enough gets into the body.
Chlorate makes the situation worse. Chlorate ions act as oxidizers, so they make many materials burn more fiercely. If thallium chlorate mixes with organic matter — everything from paper dust to oil residue — the result can be rapid combustion. Laboratories have strict rules for keeping chlorates away from flammable substances for this reason. No one wants an unexpected fire, and incompatible storage turns a chemistry storeroom into a hazard.
Personal Experiences and Vigilance
Work in a university lab for any length of time and you start to respect chemical storage rules. I’ve seen bottles with skull-and-crossbones warnings. Thallium compounds get a space in a locked cabinet, not just for theft prevention but to lower the odds of accidental contact. Gloves, goggles, and lab coats become standard gear, and spills trigger a full cleanup protocol, not just a quick wipe with a paper towel.
Failure to respect these risks can lead to tragic results. In the past, lack of awareness led to workers and researchers coming down with chronic health problems, many untraceable to thallium exposure until it was too late. The compound’s slow, sneaky effects have been used in crimes. The World Health Organization and the CDC both place thallium among especially toxic metals, pushing for greater awareness.
Mitigating Risks and Solutions
Prevention matters most. Secure labeling, safe storage, and restricting access limit casual mistakes. Using modern safety data sheets and real training go beyond what a textbook can offer. Regulations treat thallium chlorate with respect for good reason— improper disposal can send thallium into the water supply or soil. Regulatory agencies like OSHA and the EPA classify thallium as a priority pollutant in the US, requiring strict tracking and disposal by licensed professionals.
Alternatives exist in most practical cases. Thallium chlorate’s use is now rare, and most labs use less-toxic substitutes for related applications. Public health benefits when safer options replace high-risk compounds. If you find thallium chlorate in a stockroom, treat it with the gravity it deserves— nobody should handle it without strong protocols and full awareness of what’s at stake.
What is the chemical formula of Thallium Chlorate?
Understanding the Details
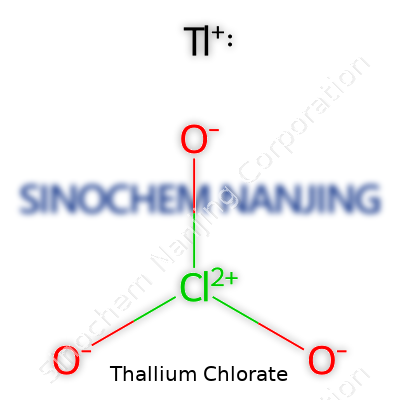
Thallium chlorate grabs some attention because chemistry fans often enjoy exploring less common compounds. Thallium, symbolized as Tl, belongs to group 13 in the periodic table. Sitting in the heavy metals crowd, thallium brings a baggage of toxicity and a history colored by both innovation and infamy. When asked about thallium chlorate, the search often spotlights a formula: TlClO3. This means one thallium ion (Tl+) pairs with one chlorate ion (ClO3-).
Getting Real About Formula and Risk
Old chemistry textbooks and reputable databases like PubChem or ChemSpider agree on TlClO3 as the correct representation. The basic chemistry matches up logically. Thallium, usually in the +1 oxidation state, connects with one chlorate, which carries a -1 charge, so the charges balance out in simple one-to-one fashion.
Having handled both heavy-metal and chlorate chemicals during my time in academic labs, I learned quickly about the real risks baked into compounds like this. Thallium, even as a trace contaminant, can cause severe health issues—think nerve damage, hair loss, or worse. Chlorate ions, meanwhile, bring their own kind of energy to the mix: strong oxidizing power.
Why Does It Matter?
It’s easy for formulas to get shrugged off as trivia, but there’s more on the line. Mistakes in chemical notation don’t stay on the page; they follow us into storage rooms, manufacturing plants, and even regulatory paperwork. Safety and compliance rely on these little details being right. Overlooking the proper formula means exposing people—and sometimes entire environments—to risks they didn’t sign up for.
For anyone handling thallium chlorate, the formula reveals its potential and its hazards. One thallium atom per molecule means even small spills can deliver a punch. One chlorate atom per molecule hints at reactivity, especially if heat or friction come into play.
Facts in the Real World
Regulatory agencies haven’t ignored this compound. Both thallium compounds and chlorate salts land on hazardous materials lists around the world. The Environmental Protection Agency (EPA) and European Chemicals Agency (ECHA) flag them for special handling. Simple errors in chemical identity have led to fines and shutdowns for companies in the past.
For educators and students, textbooks carry a responsibility here. My own experience with confused labels and mismatched safety data sheets underlines the headaches one typo can create. I’ve seen more than one colleague forced to re-label shelves after discovering the real hazard behind a mislabeled bottle.
Finding Better Solutions
One smart move is sticking with databases like the National Center for Biotechnology Information (NCBI) or Sigma-Aldrich for formula checks. Laboratories can also set up regular cross-checks on inventory—never rely on a single label when it can be verified. Some universities have even moved to barcoding systems that connect formulas and hazards to each stockroom bottle.
Staying sharp on details like TlClO3 keeps both the science and the people in the building on firmer, safer ground. Taking the formula as more than just letters and numbers cuts down on confusion and builds better trust, both in the lab and beyond.
How should Thallium Chlorate be stored and handled?
Keen Respect for Danger—Not Panic
Thallium chlorate isn’t a compound you want to treat lightly. Years in industry labs have shown me that some chemicals, even in small amounts, command respect. Plenty of folks have heard about thallium’s toxicity; its ability to disrupt vital biological processes is well-known. Add chlorate’s oxidizing power, and there’s a recipe for disaster if people cut corners in handling and storage. Instead of letting fear rule, focus on real, proven strategies—ones based on experience and science.
Proper Storage Isn’t Negotiable
Every chemist learns that the storage environment shapes safety. Dull shelves in a university storeroom hide dangerous secrets if stored carelessly. Thallium chlorate fits right in that high-risk category. Secure this chemical in airtight, corrosion-resistant containers. Glass or certain plastics—never metal or any reactive material—make the right match. Seals on those containers keep air and moisture at bay, way more important than for less reactive chemicals. Dryness matters. Humid environments encourage clumping, leaching, and, in some cases, slow decomposition that releases hazardous byproducts.
Push this compound to the back of a cool, dedicated poison cabinet—never near organics, reducing agents, acids, or combustibles. Segregation means storing it in its own marked section, not nestled up against anything else. Fire-resistant cabinets give extra reassurance, but the big win comes from location. Some labs even install separate, ventilated cabinets. Years ago, I watched how one missed detail—a poorly sealed bottle stored near a solvent—forced an evacuation. Cost, inconvenience, and embarrassment all followed an avoidable mistake. Experiences like that anchor my belief in rigid separation.
Handling Means Foresight, Not Routine
Handling thallium chlorate can’t become another part of the routine. It surprises me how many talented researchers get complacent, thinking gloves and goggles alone ward off disaster. Nope. Double gloves, clear face shields, and closed lab coats mark my approach. No eating, drinking, or phone use—even brief moments of inattention have cost people their health. Containment matters even more. Keep the work strictly in fume hoods with good airflow—my own nose has picked up faint metallic smells during transfers, proving fume hoods do more than look good for inspections.
Spills test a team’s real preparedness. Absorbent pads, not paper towels, soak up small amounts; nobody should reach for household brooms or mops. Special hazardous waste bags line nearby bins. Labeling makes life easier for cleanup crews and emergency responders, and honest, full reporting protects both people and institutions. I’ve seen teams close ranks around a small spill, improvise with lab mats, and walk away thinking all was done—only for contamination to show up days later. Speed and thoroughness matter more than pride.
Training and Transparency Trump Custom
Training shouldn’t tick off a mandatory box or fill an orientation schedule. My memory holds vivid images of old hands showing new hires the “shortcuts” they learned, brushing off guidance because “nothing ever happened.” That’s a dangerous mindset. Demonstration and real conversation about the compound’s hazards build good habits. Encourage open talk about near-misses—nobody should hide minor missteps. Audits count, too. Walkthroughs with checklists have caught early signs of container damage, missed warning labels, and forgotten spill kits in my own workplaces. Regular reminders keep old habits from replacing best practices.
No single guideline replaces professional vigilance. Give thallium chlorate the attention it deserves, back up safe habits with facts, and create an environment where everyone looks out for each other. In my experience, that’s what saves lives, reputations, and research goals alike.
What are the safety precautions when working with Thallium Chlorate?
Understanding What You’re Dealing With
Thallium chlorate doesn’t make headlines often, but its risks put it in a league with some of the nastiest chemicals out there. It tends to fly under the radar, even though both thallium and chlorate compounds can wreak havoc on the human body. Skin, lungs, and even nerves can get hammered by exposure. Some people might think a dust mask and gloves will cut it, but facts tell a different story: thallium compounds get absorbed through the skin far easier than most realize. Just handling a bottle without full coverage can cause real harm.
The Right Setup for Serious Work
Thallium chlorate belongs behind a fume hood sash—no exceptions. Simple open bench work might sound reasonable, but the dust drifts easily and settles in places it shouldn’t. Laboratories handling this gear invest in ducted hoods, regular airflow checks, and strict maintenance because they know any slip-up can turn into poisoning. I remember an old story from the university years ago: after a spill, colleagues spent an entire day with mercury vacuums and wipes chasing around thallium-laced dust. It wasn’t overkill. Thallium poisoning lingers, sometimes taking weeks to manifest.
Nobody walks into a chemistry lab planning for disaster, but real safety starts before unscrewing the cap. Experience beats shortcuts every time. Splash goggles fit snugly under the brow. Those blue nitrile gloves aren’t thick enough—use neoprene-lined or double up with disposable underneath. Lab coats keep cuffs buttoned, and people tuck those pants into boots to stop anything from sneaking down to their socks. Thallium vapor doesn’t mess around.
Respecting the Danger—Not Just the Rules
All the gloves and hoods in the world won’t save a careless worker who skips regular washing or lets their cell phone sit nearby. Thallium eats through skin and breezes right past the usual “wash hands before lunch” rules. No eating or drinking in the same room. Contaminated surfaces need specialized cleaners. Some labs stock colorimetric wipes to check if residues remain, but they don’t trust those alone. They use chelating agents and thorough surface testing each week.
Folks forget about the waste, too. Tossing contaminated gloves or papertowels in a regular trash bag invites someone else into the danger zone. Hazardous waste buckets line every bench. Labels warn “THALLIUM” in thick block letters for a reason. Any broken bottle gets triple-bagged and marked before cleanup starts. Shared spaces get hit hardest, so team drills matter. I’ve seen people review spill protocols before every shift, just so no one panics if something splashes.
Double-Checking Everything
Mistakes do happen. That’s why medical monitoring isn’t just for show; blood and urine samples spot trouble early. Labs that respect their people set up buddy systems—no one handles thallium chlorate alone. Manual logs might seem old-school, but tracking who worked with what and when saves lives. Ventilation systems stay on an inspection schedule, because filters clog and sensors fail. Every part of the system works together: training, equipment, and respect for the substance itself.
Improving Safer Ways Forward
There’s always room to improve. Substitution with less toxic compounds sometimes works, but not every process has an easy swap. Safer packaging cuts down on spills. Automation is catching on, too. Remote-controlled dispensers or closed system samplers take people out of the exposure picture. Above all, fostering a culture where folks look out for each other delivers the best protection. In my years around hazardous materials, nothing beats a lab team that speaks up before trouble starts.
| Names | |
| Preferred IUPAC name | Thallium(I) chlorate |
| Other names |
Chloric acid, thallium(1+) salt Thallous chlorate |
| Pronunciation | /ˈθæliəm ˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 13453-69-5 |
| Beilstein Reference | 2361017 |
| ChEBI | CHEBI:86462 |
| ChEMBL | CHEMBL3090056 |
| ChemSpider | 25285 |
| DrugBank | DB11360 |
| ECHA InfoCard | 100.018.018 |
| EC Number | 223-893-4 |
| Gmelin Reference | 75979 |
| KEGG | C445412 |
| MeSH | D013791 |
| PubChem CID | 159367 |
| RTECS number | XG8575000 |
| UNII | 0BV88T2QB7 |
| UN number | UN3352 |
| Properties | |
| Chemical formula | Tl(ClO3) |
| Molar mass | 304.264 g/mol |
| Appearance | white crystalline solid |
| Odor | Odorless |
| Density | 4.2 g/cm3 |
| Solubility in water | Soluble |
| log P | -2.06 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | −62.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.887 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 199.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -535 kJ/mol |
| Pharmacology | |
| ATC code | V10XA02 |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P330, P501 |
| NFPA 704 (fire diamond) | 3 2 2 OX |
| Lethal dose or concentration | LD50 oral rat 48 mg/kg |
| LD50 (median dose) | 18 mg/kg (rat, oral) |
| NIOSH | SY6865000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Thallium compounds (as Tl): 0.1 mg/m3 (OSHA, 8-hour TWA) |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 15 mg/m3 |
| Related compounds | |
| Related compounds |
Potassium chlorate Sodium chlorate Thallium perchlorate |

