Tetraphenyltin: A Detailed Perspective
Historical Development
Chemists first prepared tetraphenyltin in the early part of the twentieth century, a time when organometallic chemistry was just beginning to find its footing. Driven by curiosity and practical need, researchers managed to create tetraphenyltin through the reaction of phenylmagnesium bromide with tin(IV) chloride. Over time, this molecule carved out a place for itself because of its role as a versatile reagent and precursor in the synthesis of more complex organotin compounds. The progress echoed broader shifts in chemical research, where the desire for new materials and catalysts kept pushing boundaries. Innovative synthesis of organotin compounds followed as demand from both academic and industry groups rose during the second half of the century. The progress gradually shifted tetraphenyltin out of laboratories and into factory-scale operations, making it a commodity for specialty chemical markets and advanced material science.
Product Overview
Tetraphenyltin, often appearing as a white crystalline powder, stands out for its clean, solid-state appearance and utility across a range of chemical reactions. Structures like this open doors to countless derivatives, delivering unique reactions thanks to their tin-centered core. Markets keep requesting tetraphenyltin for its usefulness as a starting material for the production of more sophisticated organotin compounds, as well as for some niche catalytic methods in polymer chemistry and beyond. Clear identification comes under CAS number 597-05-1, and the standard product rarely deviates much from what’s described in chemistry textbooks.
Physical & Chemical Properties
This compound presents as colorless to white crystals, offering stability at room temperature. It resists most weak acids, yet reacts with stronger agents and oxidizers. Molecular formula C24H20Sn means a relatively high molecular weight, which contributes to low solubility in water but fair solubility in organic solvents like benzene, toluene, and chloroform. Boiling and melting points both sit high for an organic molecule: melting around 228°C and boiling at roughly 438°C under reduced pressure, showing strong lattice energy between its phenyl groups and tin center. In air, tetraphenyltin does not break down easily and, unlike many tin compounds, emits little odor, making it much more manageable for researchers.
Technical Specifications & Labeling
Chemical suppliers deliver tetraphenyltin usually at a purity exceeding 98 percent, sometimes even 99 percent, with further percentages identified for research use. Standard industrial-grade packaging uses robust glass containers or sealed metal drums, with hazard labels required by safety codes. Product information always lists batch number, CAS identification, weight, and any trace impurities present or intentionally limited. Companies make sure each shipment matches outlined certificates of analysis, and all chemical transportation law requirements apply. Each technical data sheet explains the need for inert atmosphere storage and mentions compatibility with other chemicals stored nearby.
Preparation Method
Synthesis involves the action of four equivalents of phenylmagnesium bromide on tin(IV) chloride, a classical Grignard reaction that produces magnesium bromide salts alongside the product. In commercial-scale production, operators conduct reactions in anhydrous ether or tetrahydrofuran under controlled nitrogen or argon, since the Grignard reagent reacts vigorously with moisture and air. Stirred reactor vessels, low temperatures, and rigorous drying protocols all play a role. Extraction with organic solvents follows, then repeated recrystallization leaves a product suitable for direct application or secondary modifications. Experienced chemists emphasize slow addition techniques and exacting temperature control, since yields fall off sharply if reaction parameters shift.
Chemical Reactions & Modifications
The strength of tetraphenyltin lies in its potential as a donor of phenyl groups, and as a template for new organotin frameworks. Under catalytic or photo-induced conditions, tetraphenyltin allows selective transfer of its phenyl rings or exchange of ligands. Decomposition under high temperatures releases biphenyl, indicating possible routes for controlled breakdown. Chemists also subject the tin atom to halogenation, oxidation, or replacement reactions, producing functionalized products with value in material science, such as tin-based polymers, or as intermediates in pharmaceutical synthesis. Reactions with halogens yield derivatives like triphenyltin halides, and under certain oxidative conditions, the aryl-tin bond cleaves cleanly, releasing tailored byproducts used in synthesis labs.
Synonyms & Product Names
This compound goes by several names, showing up in older literature as Tin, tetrakis(phenyl)-; Tetrakisphenylstannane; Tetraphenylstannane; and in some catalogs simply as “Tetraphenyltin.” Each synonym hints at its quaternary structure and the presence of four phenyl groups bonded directly to a tin core. But despite these varied names, most regulatory and commercial lists refer to it using its CAS identifier or full chemical designation to avoid confusion.
Safety & Operational Standards
Working with tetraphenyltin demands standard laboratory safety rules and additional care due to its chronic toxicity risk. Gloves, protective eyewear, and fume hoods help manage exposure. Direct inhalation, skin contact, or ingestion all pose health threats, so lab management always requires accessible spill clean-up kits, clear labeling, and regular training. Regulatory agencies like OSHA and REACH have flagged some organotin compounds as environment and health risks, even where acute harm seems less likely in the short term. Engineering controls reduce vapor and dust, and waste disposal follows hazardous waste procedures. Teams handling large batches need to maintain closed systems and pre-screen for leaks. Strict labeling communicates hazard, discourages careless use, and helps keep accidents rare.
Application Area
Industrial chemists often reach for tetraphenyltin as a key ingredient in polyvinyl chloride (PVC) stabilization, exploiting its thermal stability and structural predictability. Some research teams use tetraphenyltin to prepare functionalized tin complexes that play roles as catalysts in polymerization and organic transformations. Electronics manufacturers have looked at it for use in specialty coatings or as a precursor for tin-based semiconductors. Pure research continues to produce novel tin-based molecular architectures with tailored electronic or magnetic properties, while pharmaceutical groups occasionally examine tin-organic scaffolds for novel biological activity, though toxicity concerns limit most real-world medical applications.
Research & Development
Academic researchers keep pushing the envelope by exploring the catalytic properties and molecular recognition capabilities of tetraphenyltin-derived compounds. Fine-tuning the compound’s electronic profile through substitution schemes inspires new reaction pathways. Studies into its use as a charge carrier or dopant in organic electronics continue, as pressure mounts for better-performing and more sustainable materials. Some labs probe degradation mechanisms to improve resistance to hydrolysis or oxidation. Partnerships with industry try to streamline manufacturing and discover safer derivatives with minimized environmental burdens without sacrificing performance.
Toxicity Research
Studies dating back fifty years document the biological impact of organotin compounds, tetraphenyltin included. Chronic exposure in laboratory animals produces neurological and hepatic damage, while acute toxicity lands mainly on the gastrointestinal and pulmonary systems. Though less acutely toxic than tributyltin or certain alkyl derivatives, tetraphenyltin cannot be dismissed as harmless. Environmental surveys catch traces in soil and groundwater near processing sites, making strict containment a must. Toxicologists keep track of metabolic pathways, monitoring for bioaccumulation and long-term effects on both laboratory staff and wildlife. Industrial standards direct minimization of dust and vapor, and companies work on capturing waste streams to limit environmental loading.
Future Prospects
The future for tetraphenyltin depends on solving safety and environmental dilemmas while maintaining its value for advanced synthesis and materials engineering. Green chemistry initiatives keep pressuring manufacturers to shift reaction protocols and create derivatives with lower toxicity. International rules could tighten disposal and handling limits, but if researchers deliver safer, function-specific alternatives, tetraphenyltin could remain a mainstay in specialty materials, emerging electronics, and academic research. Computational chemistry now allows for more precise prediction of reaction behavior, helping reduce trial-and-error and speed up development of next-generation organotin compounds. By keeping an eye on both safety and demand, the chemical industry aims to balance progress with accountability.
What is Tetraphenyltin used for?
So, What’s This Compound All About?
Tetraphenyltin isn’t something you’ll find on a grocery store shelf, but in the chemical world, it draws plenty of interest. The molecule itself comes together from tin and four phenyl groups. That’s the textbook definition, but the real story is in the way people put it to work. Chemists see it as a powerful tool—one that’s found a place in some crucial industries.
Rubber That Lasts and Performs
Thinking about daily life, most folks have handled rubber—tires, seals, or shoe soles. What many don’t see: rubber doesn’t spring into existence perfectly shaped and ready. It takes a transforming process involving catalysts. Tetraphenyltin serves as a catalyst, especially in the manufacture of silicone rubber. It keeps the molecular chains long, helping the final product stay flexible but strong, even after miles or years of use. Without efficient catalysts, rubber products wouldn’t handle heat, pressure, or regular stress half as well.
Pharmaceuticals: Precision and Safety
Medicines rely on cleaner reactions and fewer unwanted byproducts. Scientists designing drug molecules look for reagents that do their job without leaving traces. Tetraphenyltin plays a supporting role in organic syntheses, helping create specific bonds in complex molecules. Safe medicine isn’t born from shortcuts; it’s built on careful choices, and the right reagents make all the difference.
Polymers That Don’t Quit
Synthetic materials, like certain plastics and resins, also benefit from this compound. Tetraphenyltin can act as both a stabilizer and a catalyst, especially in the production of high-performance polymers. Those everyday plastics in electronics, construction materials, or automotive components require stability through heat and time. Better catalysts keep production smooth, costs more manageable, and end products reliable. The difference between a phone case that cracks at the first drop and one that can take a beating lies in chemistry fundamentals like this.
Potential Issues: Health and the Environment
As with many industrial chemicals, safety matters. Tin-based compounds can carry risks. In my own lab days, there was never any skipping over gloves or fume hoods working with organotin compounds. Even though tetraphenyltin is less notorious than some of its cousins, careless handling still means exposure risks. Once factories release tin-based materials into waterways, living organisms can face harm. Environmental groups keep pushing for tighter controls, and for good reason.
Paths Forward: Safer Use, Smarter Science
Some companies have taken steps to recapture and recycle organotin compounds from their processes. Others have begun testing greener alternatives that don’t sacrifice performance. Chemists and plant workers have a shared responsibility: using the best tools for the job, while minimizing harm at every stage. Open communication between scientists, regulators, and the public helps keep an eye on both progress and safety.
Real-World Impact
Most people never hear about tetraphenyltin, but the products it helps make touch daily life. Better, longer-lasting rubber and effective medicines rely on this backbone of industrial chemistry. Responsible use and ongoing research can help us keep the benefits while dialing down the risks.
What is the chemical formula of Tetraphenyltin?
Breaking Down Tetraphenyltin
Tetraphenyltin carries the chemical formula Sn(C6H5)4. So, what does that really mean for someone involved in chemistry, manufacturing, or environmental safety? This organotin compound pairs one tin atom with four phenyl groups. In labs, the structure gives it unique properties — the bulkiness of these big aromatic rings affects how Tetraphenyltin reacts and interacts with other chemicals. Somebody working with organometallic chemistry will tell you that this formula makes a world of difference in designing everything from catalysts to specialty polymers.
Impact on Industry and Research
I spent part of my graduate years entrenched in organometallic synthesis. There’s a pattern: compounds like Tetraphenyltin pop up when chemists hunt for molecules that create sophisticated reactions or stable intermediates. Its formula signals strong resistance to hydrolysis — meaning it doesn’t just fall apart in water. That alone makes it a candidate for industrial applications where moisture sensitivity causes headaches. Researchers lean on Sn(C6H5)4 in organic synthesis, looking for precise control in experiments. The balance between tin and the phenyl arms gives chemists a handle for tweaking material properties in high-performance applications.
Environmental and Health Questions
Not every use story is rosy. Data shows organotin compounds can become environmental hazards if not managed right. According to the European Chemicals Agency, improper introduction of compounds like Tetraphenyltin into water streams leads to adverse effects. Tin-based materials, especially those with persistent organic groups, remain stable for years and accumulate in soil and living tissues. The United States Environmental Protection Agency has flagged many organotin compounds for monitoring, due to risks in aquatic life and food chains.
Even small-scale exposure calls for solid safety habits. In my own time working with these chemicals, I saw strict adherence to gloves, goggles, and fume hoods, even for tiny amounts. Spills never got ignored. A moment’s carelessness with organotins created a cleanup headache. That real-world attention to procedure should carry over to larger facilities, especially as demand for advanced materials rises.
Shaping a Safer and Innovative Future
For all its potential, using Tetraphenyltin wisely means balancing innovation with risk management. Companies exploring new uses should keep up with the latest safety research and follow evolving legislation. Waste treatment, containment, and personal protection matter as much as the fun chemistry side of things. From my experience, clear protocols and staff education tackle many of the big worries around organotin handling.
Innovation moves fast in materials science. Each element of the chemical formula — from that single tin atom to the phenyl groups — shapes everything from molecular reactivity to long-term impacts on health and the environment. Knowing that can make the difference between progress and problems. Anyone invested in chemical manufacturing, research, or environmental safety owes it to themselves and their teams to know what Sn(C6H5)4 stands for, beyond just a string of letters and numbers.
Is Tetraphenyltin hazardous to health?
What Is Tetraphenyltin?
Tetraphenyltin pops up in specialty chemistry, rarely appearing in the wild for everyday folks. It plays a role in polymer production and lab research, not in household products. Scientists call it a tin compound dressed with four phenyl groups—chemically stable but not something you find under the kitchen sink.
Health Risks You Should Know About
Even unfamiliar chemicals can pack a punch if not handled correctly. People who work in labs or chemical manufacturing can bump into tetraphenyltin when mixing or processing materials. Breathing in the powder or dust sent my mind straight to college lab days—one whiff too close and you notice how quickly your body picks up tiny stuff in the air.
Toxicologists point to the core issue: organotin compounds earn a reputation for toxicity, particularly toward the nervous and immune systems. Some organotin versions have shown damage to the liver, kidneys, and reproductive organs in animal studies. Chemical safety databases list tetraphenyltin’s risks as similar, though less severe compared to ones like tributyltin. Still, repeated or heavy exposure raises red flags.
Direct evidence for tetraphenyltin toxicity in humans feels scarce, mostly because only a sliver of the population handles it. The U.S. National Library of Medicine and safety groups warn that inhalation, skin contact, or accidental swallowing in a workplace setting could lead to respiratory irritation or headaches. I recall trying to keep a steady hand scooping dry powders in grad school, knowing full well a dust mask made all the difference.
Environmental Aspects
Once into the air, water, or soil, organotin catalysts don’t break down quickly. Their fat-loving chemical structure lets them persist and build up in living things. European chemical rules require close registration for these substances. Field studies looking at aquatic life show organotin leftovers in rivers harm tiny organisms, ripple through food webs, and can linger in bottom sediments for years. If someone hoses down equipment outdoors or skips proper disposal, these compounds don’t just vanish.
Splashing lab glassware or chemical-contaminated water down the drain sometimes feels easier, but that shortcut adds up over time. Luckily, tightened lab safety and better containment drastically reduce real-world releases from industry.
How Can Workers and Labs Stay Safe?
Practical safety works better than any label on a bottle. Workers at facilities deal with this compound using gloves, goggles, lab coats, and proper hoods or respirators. I’ve seen new interns surprised by how much effort goes into handling small bottles, but that discipline saves a lot of headaches later. Written procedures matter, but hands-on training keeps folks mindful—a lesson I learned by nearly dropping a glass flask one late night.
Industry has a responsibility. Regular air monitoring, up-to-date ventilation, and safe storage help limit risks from exposure. Regulatory compliance teams track use and disposal tightly. If a spill happens, clean-ups mean more than tossing paper towels—special gear and waste drums come out.
Most importantly, transparency helps everyone. Facilities report chemical handling, keep safety data updated, and share what to do in case of emergency. Scientists and regulators keeping tabs on new toxicity studies further strengthen protections.
The Bottom Line
Tetraphenyltin stands far from harmless, mostly because people rarely expect risks from obscure, high-end chemicals. Even with lower toxicity than other organotins, its family history and persistent nature call for respect. Responsible handling, workplace controls, and good habits matter most—those set the difference between chemical risk and safe science.
How should Tetraphenyltin be stored?
Why Safe Storage Matters
Anyone working with chemicals learns quickly that mishandling can turn routine tasks into dangerous situations. Tetraphenyltin is no exception. It doesn’t explode on contact or fume dramatically, but this compound holds risks that often slip under the radar in cluttered labs and busy storerooms. My years in a university research lab taught me that a little care with storage protects both your health and your investment.
Understanding What You’re Storing
Tetraphenyltin carries a reputation in organic labs. As a source of organotin, it’ll be present anywhere researchers push boundaries in catalysis or material science. This substance comes as a white, crystalline powder and isn’t volatile in the way strong acids are, yet caution always pays off. Breathing in dust or touching it bare-handed means inviting trouble, since organotin compounds show up in health studies for a reason—they mess with your nervous system and other organs, even at low doses.
What Real Storage Means
For Tetraphenyltin, storage isn’t about tossing the bottle on any spare shelf. The ideal spot shields it from heat, sunlight, and moisture. Heat speeds up chemical breakdown, which risks unpredictable reactions or loss of potency. Sunlight isn’t just about warming the bottle: UV rays break down organic compounds at the molecular level.
A dark, cool cupboard works better. Labs with climate control manage this easily, but in older buildings, temperatures fluctuate too much near windows or outside walls. I always picked spaces in the center of the lab, away from sunlight and heaters. Putting anything above eye level invites unsafe reaches and spills—front-row, lower-shelf storage gives easier access and fewer accidents.
Moisture deserves its own attention. Tetraphenyltin reacts with water over time. Even small humidity from air will eventually degrade it or form byproducts you don’t want. In my old lab, we used desiccators or tightly sealed containers with silica gel packs inside. Cheap insurance for a costly chemical.
Container Choices
Original bottles from suppliers usually come in amber glass, which holds up well. Glass keeps out air and won’t react with the compound itself. After opening, make sure caps stay tight, and avoid transferring Tetraphenyltin to containers that might let air or moisture sneak in.
Label every bottle with purchase date and open-date. I learned this lesson the hard way after a mix-up ended with contaminated sample waste. Good labels and tidy shelves cause fewer headaches later.
Safer Spaces, Safer People
Working with toxic materials brings personal stakes. Accidents never announce themselves. Wearing gloves and goggles as soon as the box comes off the shelf protects everyone, especially students or techs just learning the ropes.
I always insisted on spill kits nearby and clear procedures for clean-up. Small lapses—like letting Tetraphenyltin dust drift to nearby equipment—can snowball into big problems later, both for health and costly repairs.
Looking Ahead
Storing Tetraphenyltin takes more than a rulebook. It builds on daily habits and respect for what’s on the shelf. Good systems help people and products alike last longer. Regular checks for leaks, damaged labels, or expired stocks build a culture where everyone goes home safe, and science moves forward without unnecessary risks.
What is the molecular weight of Tetraphenyltin?
Getting to the Numbers
For anyone who used a chemistry set in school, the talk about molecules can bring back the smell of those sulfur compounds and the nervousness of breaking something expensive. Tetraphenyltin, with the formula Sn(C6H5)4, pulls together tin and four phenyl groups. It packs a molecular weight of 484.16 g/mol. That number doesn’t just give a sense of heft — it changes how people and industries manage, measure, and respond to this chemical.
Why Chemists Care About the Number
Preparing a reaction, the weight gives more than just a number on the scale. The molecular weight acts as a guide: chemists rely on it to figure out how many molecules they have. I learned this lesson the hard way. Measuring a gram off without accounting for that molecular mass means your reaction could fall short or blow up. Most folks working with organometallics like tetraphenyltin, often in the electronics world, need to know how it falls in line with their other reactants. It lets engineers design circuits more accurately and researchers avoid wasting time and resources.
Beyond the Lab: The Tech and Environment Angle
Devices around us continue shrinking, and the purity of materials inside computer chips keeps growing. Knowing the weight of molecules makes it possible to mix compounds at exact ratios. If the wrong measurements go into chip fabrication, the quality dives. That’s not good news if you depend on your phone to wake up or run a small business from your laptop. Failing to handle these numbers with accuracy could cost a company more than just embarrassment — it can mean millions in lost parts, downtime, and repairs.
People who value environmental protection give special attention to molecular details. Tetraphenyltin gets used as a precursor in various reactions, and by understanding the exact weight, chemists can limit waste and keep production efficient. The pollution angle comes in here: smaller amounts used correctly mean fewer toxic byproducts. In my own lab experience, tracking every decimal place meant much less time spent disposing of hazardous leftovers and more time on productive work.
Walking the Line Between Safety and Progress
Safety plays at the edges of every chemical process. Handling tetraphenyltin means dealing with an organotin compound — and those don't have the greatest track record for health or the environment. Governments pay close attention to the details of what goes into factories and out into the water supply. Correct molecular weights help avoid overuse, keeping operations in line with rules and best practices. It also helps new researchers avoid rookie mistakes. Clear labeling and staff training borrow directly from solid knowledge about the molecular mass of everything being handled. I’ve seen an unfamiliar trainee nearly mix up tin compounds simply because an old label smudged one digit in the weight.
Solutions From Solid Chemistry Literacy
Building up education on basic chemical properties changes how the public perceives how things are made or regulated. Companies that train staff to double-check molecular weights cut down on hazardous errors. Digital scales tied to recipe software now cross-reference chemical mass values and keep large operations running smoothly. Better access to accurate data about chemicals gives students and new chemists the tools to experiment safely. Schools and apprenticeships would do well to make this aspect of chemical safety a top priority. In short, the more everyone understands what those numbers mean, the safer, cleaner, and smarter the entire field becomes.
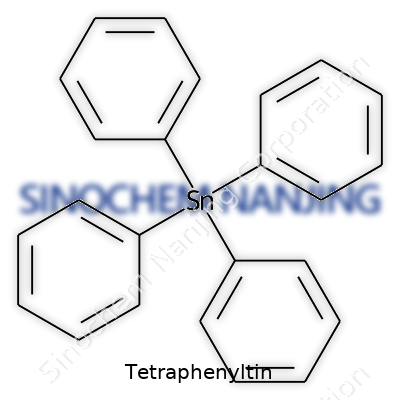
| Names | |
| Preferred IUPAC name | tetraphenyltin |
| Other names |
Tetraphenylstannane Tetraphenyltin(IV) |
| Pronunciation | /ˌtɛtrəˈfɛnɪlˌtɪn/ |
| Identifiers | |
| CAS Number | 7791-25-5 |
| Beilstein Reference | 1462079 |
| ChEBI | CHEBI:33370 |
| ChEMBL | CHEMBL352475 |
| ChemSpider | 392071 |
| DrugBank | DB11345 |
| ECHA InfoCard | DSSD-FEJQ-V017-QX5F |
| EC Number | 208-942-9 |
| Gmelin Reference | 92229 |
| KEGG | C18873 |
| MeSH | D014956 |
| PubChem CID | 66217 |
| RTECS number | XR9625000 |
| UNII | 6K77A45T6I |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C24H20Sn |
| Molar mass | 538.126 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.198 g/cm3 |
| Solubility in water | insoluble |
| log P | 5.9 |
| Vapor pressure | < 0.01 mmHg (20°C) |
| Acidity (pKa) | 48.7 |
| Magnetic susceptibility (χ) | −76.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.689 |
| Viscosity | 1.577 cP (25°C) |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 409.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -15.5 kJ/mol |
| Pharmacology | |
| ATC code | V09GX02 |
| Hazards | |
| Main hazards | May cause irritation to the skin, eyes, and respiratory tract; harmful if swallowed, inhaled, or absorbed through the skin; may cause damage to organs through prolonged or repeated exposure; toxic to aquatic life. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0-水 |
| Flash point | 180°C |
| Autoignition temperature | 540 °C |
| Lethal dose or concentration | LD50 oral rat 5 g/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 4,000 mg/kg |
| NIOSH | TI7700000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 8 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Tetraphenyllead Tetraphenylsilane Tetraphenylgermane |

