Tetranitronaphthylamine: A Deep Dive Into A Complex Compound
Historical Development
The story of tetranitronaphthylamine stretches back to the early 20th century, driven by wartime demand for energetic materials. Interest grew among chemists chasing advances in dyes and explosives, where naphthalene derivatives began catching notice for their reactivity. Laboratories in Germany and Britain jostled to piece together heavier nitroaromatics, not just for military use but also for new synthetic fibers and colorants. Tetranitronaphthylamine didn’t spill out from a single “invention”; instead, it trickled in from decades of stepwise nitration experiments and a bit of luck with sulfonation routes. As chemical manufacturing matured, researchers learned that tinkering with naphthalene’s structure could produce nitro-boosted amines with wild color changes, deepening their reputation as more than just explosives.
Product Overview
Tetranitronaphthylamine builds its structure on the naphthalene ring, swapping out hydrogen ions for nitro groups and topping it off with an amine moiety. Not a common shelf chemical, it usually turns up packaged in small vials, handled mostly by research labs rather than bulk plants. It’s sold as a yellowish-orange crystalline powder, and it grabs attention for its role as an intermediate in specialty synthesis. Industry types spot it by its unique smell and heavy density, while chemists know it’s at home in the lab—not the workshop or factory floor.
Physical & Chemical Properties
You can tell right away that tetranitronaphthylamine stands out from garden-variety organic compounds. Its heavy nitro content pushes its molecular weight up. Its crystals look dense and pack together tightly. The melting point lands well above 200°C, a warning of strong intermolecular interactions and a little hint of trouble for anyone thinking of scaling up. Trying to dissolve it in water goes nowhere, but organic solvents like acetone pull it in a bit better. Reactivity follows suit, as the nitro groups pull electron density away from the core: this means it won’t oxidize easily, but it can break down in the presence of strong bases or reductants. Explosivity hovers in the background, though it doesn’t light up as quickly as TNT or picric acid.
Technical Specifications & Labeling
Bottle labels trace a careful line with this substance—hazard diamonds marked for both toxicity and combustibility, a UN number sometimes attached in transport. Rigorous purity checks sort genuine batches from shop-made knockoffs, with a minimum spec often above 98% verified by HPLC or TLC. Labels list not only its name and structure but storage protocols as well: cool, dry, away from sunlight, under lock and key. European REACH and US OSHA rules kick in, demanding SDS documentation on demand, and clear labeling for lab-scale versus technical grade.
Preparation Method
Chemists put tetranitronaphthylamine together mainly through exhaustive nitration of naphthylamines. This means soaking a naphthylamine base in a fuming mix of nitric and sulfuric acids, keeping the temperature low to control side reactions while carefully dripping in more acid. Yields hinge on reaction time and acid strength, not just sheer starting material. Later steps wash and neutralize, then dry out the product, often with vacuum assistance. Purification relies on recrystallization from a solvent that leaves side products behind. At times, a second round of nitration corrects incomplete conversion, but safety routines throughout stand front and center: full face protection, slow addition, and blast shields, as lab accidents involving nitrated organics can earn a spot in grim chemical folklore.
Chemical Reactions & Modifications
Tetranitronaphthylamine sparks plenty of imagination in synthetic laboratories. Reactions with reducing agents trim down nitro groups; these can push the molecule toward diamines or even full de-nitration, altering both color and electronic properties. Acylation, alkylation, or sulfonation open up paths to custom derivatives, though steric hindrance from bulky nitro groups often demands brute force or new catalysts. Some research groups explore metal complexation, tethering them to transition metals that tune magnetic or optical behavior. In energetic materials, modifications test how subtle tweaks change sensitivity and detonation velocity, data that flows into military dossiers and safety handbooks alike.
Synonyms & Product Names
Over the years, chemists have called this compound by several flourishes: 1-amino-2,3,6,8-tetranitronaphthalene, 2,3,6,8-Tetranitro-1-naphthylamine, or simply TNNA for shorthand. Some catalogs list it as part of a broader family, slotting it under “tetranitronaphthylamines” to cover isomeric variants. Researchers sometimes use in-lab codes, linking purity and batch, but most journals stick to IUPAC standards to prevent confusion during peer review or international exchange.
Safety & Operational Standards
No one shrugs off safety around tetranitronaphthylamine. Contact with skin or inhalation can bring on cyanosis, methemoglobinemia, or other forms of chemical stress—especially for those handling powdered forms or heating the compound. MSDS documentation calls for nitrile gloves, eye shields, and good ventilation. Explosive risk rises if the powder sits in direct sunlight, dries out, or mixes with metal dusts or organic peroxides. Fire brigades don’t tackle this kind of fire with water; standard protocol relies on dry sand or foam extinguishers. Waste streams never head down a sink: neutralization takes place under fume hoods, and storage involves inert atmospheres or desiccators.
Application Area
Most real-world uses of tetranitronaphthylamine cluster in research and specialty synthesis. Academic groups target it for vibrant dye production, where its strong electron-withdrawing nitro groups yield deep, persistent colors on fabrics or plastics—albeit with lab-scale yields, not commercial barrels. Synthesis teams sometimes reach for it in pushing boundaries on medicinal chemistry, testing if heavy nitration can introduce new antibacterial or antiviral properties in heterocycle scaffolding. Explosives labs set benchmarks for sensitivity and brisance, but rarely see it move to field trials due to handling hazards and limited cost-to-yield ratios. Some companies quietly explore it for sensor applications, looking at its color shifts under different pH or redox conditions as a trigger for chemical detection devices.
Research & Development
R&D remains strong around tetranitronaphthylamine, especially as advances in green chemistry and microreactor technology offer safer routes to its synthesis. Teams in Japan, Germany, and the US focus on scaling reactions down to “flow chemistry” platforms, which shrink reactant volumes and boost safety profiles. Collaborations between academia and industry check if the compound can spark novel photonic devices or high-performance pigments. Analytical chemists invest in better spectroscopic methods to track impurities or degradation products that might slip past bulk purification, reducing risk in downstream applications. Processing data from pilot-scale runs helps optimize not just yield, but environmental footprint, a rising concern with organic nitro compounds under current environmental regulations.
Toxicity Research
Toxicologists rarely see clear answers with molecules like tetranitronaphthylamine. Early animal studies suggested real risks to both liver and kidney tissues, independent of dose, likely due to bioactivation of the nitro groups. More recent work tracks metabolites in cell cultures, mapping out which cytochrome P450 enzymes trigger breakdown and whether the resulting fragments threaten DNA or mitochondrial function. Environmental researchers highlight concerns about persistence, warning that many nitroaromatics build up in groundwater and resist natural decay. Safety committees don’t clear this compound without rigorous reviews, and ongoing efforts look for less hazardous alternatives in both dye and explosives applications to limit occupational exposure and long-term ecological impact.
Future Prospects
The days of mass production for tetranitronaphthylamine have passed, replaced by niche research and targeted synthesis. Sustainable chemistry teams study whether greener oxidants or biocatalysts could swap out nitric acid, reducing both waste and accidental emissions. Innovations in molecular design could open up new dye molecules with lower toxicity, keeping parts of the old structure but tuning reactivity for safer use on textiles or in composite materials. For now, the future of this compound sits in the hands of highly skilled chemists, both in safety-minded research and the pursuit of better, cleaner functional materials. Anyone tackling tetranitronaphthylamine needs both a steady hand and a long view—not just for results, but for the safety and ethics of chemical innovation.
What is Tetranitronaphthylamine used for?
A Closer Look at an Uncommon Chemical
Sometimes a name jumps out at you from a chemical register and raises more questions than answers. Tetranitronaphthylamine doesn’t make headlines every day, but beneath that long label, there’s a story worth sharing. This compound has drawn the attention of chemists, defense researchers, and environmental experts, not because you’ll find it under your kitchen sink, but because of its potent properties and the risks it carries.
The Explosive Conversation
The nitro groups crowding its molecular structure flag Tetranitronaphthylamine as a compound with explosive potential. Its development comes from the same world that created TNT and related substances, which saw high demand during periods of global conflict. During the mid-20th century, the arms race spurred a boom in synthetic nitro compounds with the hope of perfecting better, more stable munitions. Tetranitronaphthylamine cropped up during these chemical quests because its structure packs both energy and density.
Researchers tested it for use in initiators, primary explosives, and chemical research—always chasing a material that stores lots of energy without blowing up by accident. The reality, though, never quite measured up to the promise. Many nitroaromatic amines turn out to be tricky for safe handling, especially outside controlled facilities. Not every powerful molecule finds a home in working explosives—some end up too sensitive or too unpredictable to trust in the field.
Health and Safety Lessons
Stories from the days of heavy synthetic chemistry point to some ugly realities. Handling nitro compounds without modern precautions led to health problems for workers: chronic headaches, nerve issues, and in some cases, severe liver or blood toxicity. Tetranitronaphthylamine, like its relatives, pushes the importance of rigorous occupational safety, proper ventilation, and specialty disposal. These days, nobody in their right mind would handle this stuff bare-handed or let it build up in a workplace drain. Strong laws now back up what experience taught—chemicals with this much punch demand tight control and respect.
The Research Edge
In the right hands, Tetranitronaphthylamine still provides value in education and high-energy materials science. Lab investigators probe it for insights into electronic structure and explosive-defusing tactics. Chemists study how adding several nitro groups changes not only stability but also environmental persistence. Digging into details like these drives improvements in safety, forensics, and pollution control—not just wartime inventions.
Responsible Stewardship Matters
Modern science keeps clear records of where these kinds of compounds get used and stored. Teams train carefully, use personal protective gear, and waste is always sent to hazardous-materials processors. Some advocate for greener chemistry to replace hazardous nitroaromatics, and research grants now often require proof of safer methods and alternative products.
Dealing with a substance like Tetranitronaphthylamine isn’t just a chemistry challenge. It asks for responsibility, foresight, and honesty. Safety comes from fully understanding the risks—something regulations now support with clear scientific backing. Every decade reveals new lessons from both past troubles and fresh discoveries. As we keep pushing the boundaries of material science, making space for both innovation and care is the only way forward.
Is Tetranitronaphthylamine hazardous to human health?
Why People Ask About Tetranitronaphthylamine
Many chemicals have tough names and uncertain risks. Tetranitronaphthylamine sounds like something out of a chemistry lab, far from daily life. For those who work in research, manufacturing, or chemical storage, understanding real risks associated with complex substances like this must move beyond textbooks. With any nitro compound, past lessons about safety slip into the conversation.
What We Actually Know
Detailed scientific studies on tetranitronaphthylamine’s health effects remain thin on the ground right now. That breeds uncertainty—not just for factory workers or chemists, but anyone nearby, like cleanup crews or transport drivers. Similar substances in the nitro group often show up on lists of chemicals to handle with kid gloves. Nitroarenes, in particular, set off alarms in toxicology circles. Even in minor skin contact, compounds like dinitrotoluene and trinitrotoluene (TNT) have left workers with chronic health issues after just small exposures over time.
Most nitro-naphthalene derivatives have demonstrated toxic and even carcinogenic effects in animal studies. The backbone of tetranitronaphthylamine belongs to this same family. The absence of published, large-scale human data offers no comfort. Historically, nitro chemicals have shown the ability to pass through skin, build up in fat, and possibly affect organs like the liver and kidneys. Some have even acted as triggers for certain forms of cancer in long-term exposure cases.
Lessons from the Field
Having spent time consulting for chemical plants, I saw plenty of corners cut on the shop floor—sometimes just to get through the day. Even with strong safety rules, high turnover or inadequate training means lapses occur. When a new chemical like tetranitronaphthylamine enters the mix, folks often lean on what they know about its chemical relatives. Nobody wants a repeat of the “aniline dye worker” cancers from the last century, which only showed up decades after exposure. It's easy to think gloves and goggles protect everyone, but vapor, dust, and residues break through standard gear if left unchecked.
How to Move Forward
Toxicologists say it best: respect the unknown. If a compound’s effects aren’t mapped out, assume the worst until evidence says otherwise. This means setting up ventilation, using sealed containers, and making sure every worker knows what’s in a drum or beaker. It’s no good just handing out a safety data sheet and calling it a day. People need real training—what to do if there’s a spill, how to spot warning signs in themselves or their coworkers, and when to call for help.
Nobody likes adding more labels or paperwork to a busy shift. Still, investing the time up front saves pain, legal trouble, and sometimes lives down the road. Regular medical checkups, keeping exposure levels as low as possible, and open hazard communication should be part of every company’s routine where tetranitronaphthylamine could come into play.
The Industry’s Role
Producers and users alike share a duty. Transparency matters, not just in theoretical data but practical experience from those handling the stuff daily. Regulators and industry groups must keep pressing for updated research and, where data is missing, recommend strict handling. Chemical safety relies less on chemistry PhDs and more on accountability and shared vigilance. Companies need to shepherd new chemicals with caution rooted in the lessons of the past, especially before long-term harm has a chance to strike.
What are the storage requirements for Tetranitronaphthylamine?
The Real Risk Behind Storing Tetranitronaphthylamine
Tetranitronaphthylamine (TNNA) jumps off the page as one of those tongue-twister chemicals with a reputation for being both unstable and unforgiving if mishandled. Anyone who’s ever worked with sensitive explosives or toxic dyes knows the value of keeping their wits sharp in the stockroom. My own time interning in a college chemistry storeroom hammered home one lesson: You can’t let your guard down with substances that have this kind of volatility.
Why Strict Storage Rules Matter
Major safety incidents often start with little shortcuts or the kind of sloppiness that builds up over time. TNNA belongs to a family of chemicals that easily form explosive dust and react with regular air and moisture. According to the latest guidance from Hazmat and regulatory resources, this compound can detonate from simple friction, heat, or static electricity. Keeping storage protocols tight isn’t bureaucratic red tape—it’s how you make sure nobody gets hurt.
Proper Containers and Environmental Controls
Experienced lab managers never pour TNNA into just any old jar. They rely on tightly-sealing, heavy-walled glass or special non-reactive plastic. Steel and most metals are avoided because contact can trigger dangerous reactions, especially if corrosion or impurities get involved. Secure every container label. No faded tape, no guesswork.
Heat and light feed instability in TNNA. Only climate-controlled storage areas belong in the conversation here. Temperatures stay below 25°C (77°F), humidity close to bone-dry. I’ve seen “just one warm weekend” ruin an entire batch of temperature-sensitive compounds; it’s not worth rolling the dice. Use explosion-proof refrigeration for large stock or in warmer climates. Avoid direct sunlight entirely—covered shelving or dedicated cabinets protect both substance and staff.
Segregation: More Than Common Sense
Keep TNNA away from flammable solvents, reducing agents, strong acids or bases. Mixing storage of incompatible chemicals spells disaster. This isn’t about following a chart for the sake of compliance. I once witnessed a minor chemical reaction in a mismanaged high school storeroom; the mess and stench stuck with me. Larger scale accidents steal headlines and livelihoods.
Handling, Documentation, and Accountability
Routine inspections spot crusted lids or tampered seals before issues get dangerous. Familiar eyes can catch what newcomers miss. Safety Data Sheets belong within arm’s reach; nobody gets exemptions from reading and logging every withdrawal. Double-check your personal protective equipment (PPE): gloves, goggles, and lab coats mean fewer close calls. Ventilated fume hoods add another line of security, especially for measuring or sampling.
Disposal calls for just as much care. Never dump leftovers or expired samples in regular trash. Industry best-practices call for licensed hazardous waste handlers who can neutralize and destroy residues securely. Fines from the EPA or local agencies aren’t just numbers on a balance sheet—they follow staff and organizations for years.
How We Can Do Better
Education is the best tool in the box. Regular training and surprise drills push staff to work as a team. I’ve seen attitudes shift after a well-staged emergency scenario. It builds muscle memory and helps everyone buy in. Facility upgrades don’t come cheap, but insurance after an incident costs even more. Investing in monitored storage, alarms, and robust documentation systems protects not just property, but people—and that’s what sticks with you at night.
How should Tetranitronaphthylamine be handled safely?
Understand the Substance, Respect the Risks
Tetranitronaphthylamine doesn’t attract everyday attention for a reason. This is a chemical with a reputation for volatility, reactivity, and real health consequences if handled carelessly. Even those of us with experience in chemical labs learn early that confidence quickly turns into regret if we ignore the right handling practices. From its molecular structure—loaded with nitro groups—to its strong oxidizing properties, this compound looks for a trigger. It’s wise to remember that history is full of loud lessons from those who skipped a step or overlooked a leak.
Crucial for Anyone: Avoid Direct Contact
Skin contact with tetranitronaphthylamine shouldn’t happen. Forget about casual handling—even nitrile gloves prove unreliable. Double-gloving with heavy-duty, chemical-resistant gloves lessens the chance of hazardous exposure. Splash goggles feel awkward, but chemical burns to the eye don’t heal easily. Lab coats, long sleeves, and full-face protection guard against the sudden mistakes everyone makes, especially when tired or distracted.
No Shortcuts with Air Quality
Breathing dust or fumes leads to lasting damage. I’ve seen folks believe they’re safe with just a cracked window or an ordinary room fan. That’s not enough. Work inside a certified fume hood. A robust dry chemical extinguisher needs to stay nearby. Even small amounts cause trouble; this chemical’s dust can hang in the air, and its fumes sneak up on you. Keep air flowing out, never back into the workspace.
Isolation and Storage Save Lives
Loose containers and shared shelves spell disaster. Storing tetranitronaphthylamine next to anything flammable, reducing, or incompatible puts everyone in the building at risk. I remember one storeroom mix-up—two different chemicals side by side caused a fire that almost shut down the floor. Separate locked cabinets, fire containment measures, and constant labeling prevent these headaches. Use sturdy secondary containment trays, not cardboard or open bins.
Treat Spills as Serious
Even a small spill can escalate into a health crisis. Fast, clear action always beats panic. Absorbents rated for strong oxidizers soak up liquids, but don’t forget the full PPE and immediate ventilation. Never sweep dry material; anyone with a broom in one hand and a dust cloud rising around them risks inhaling poison. Spill kits designed for reactive compounds go a long way—not just a bucket of sand and hope.
Thinking Beyond the Lab: Environmental and Disposal Issues
Disposal calls for more than dumping down the drain. Tetranitronaphthylamine contaminates water supplies and soil, endangering communities for years. Professional hazardous waste contractors handle these dangers. They’ve got the right containment, the insurance, and most importantly, the knowledge of local and federal regulations. Never trust internet shortcuts or haphazard neutralization recipes; the results rarely go as planned.
Cultivating a Safety Culture
Every person around toxic chemicals decides, every day, whether safety matters more than speed or ease. Peer pressure can push people to skip steps, but in every lab I’ve worked, real leadership means saying “no” when someone tries to save time at the expense of safety. Up-to-date training, clear signage, and a zero-tolerance culture for unsafe practice matter more than a shelf of equipment. Risks shrink when everyone remembers why the rules exist: no experiment or shortcut is worth a permanent scar.
What is the chemical structure of Tetranitronaphthylamine?
What Is Tetranitronaphthylamine?
For anyone curious about complex chemicals, Tetranitronaphthylamine offers a glimpse into just how intricate molecular structures can get. It’s a mouthful to say, and on paper, the structure backs that up. In practice, the name gives away a lot about the core framework—derived from naphthylamine, with four nitro groups attached.
The Backbone: Naphthylamine
Polarizing as it sounds, naphthylamine forms the skeletal base. Naphthalene rings, essentially two fused benzene rings, lead the charge. Sticking an amine group (-NH2) on one of those rings turns it into naphthylamine. In industrial chemistry circles, naphthylamine comes up both for its own uses and as a base for tweaking into dozens of other chemicals.
Layering on Nitro Groups
A nitro group brings a nitrogen atom bonded to two oxygens—with one bond as a double and the other as a single (NO2). Attaching four such groups to the naphthylamine ring system isn’t just chemistry for fun; it adds punch to the molecule’s reactivity and character. Tetranitro means there are nitro groups at four different points on the aromatic system.
Structure in Detail
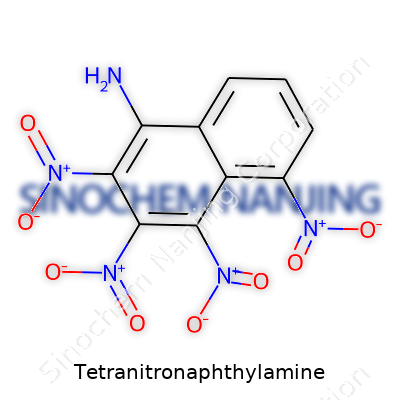
Picture a naphthalene core: two hexagons fused together along one edge, all carbon atoms forming a flat, planar structure. The amine (-NH2) hooks into position 1 on this structure. The four nitro groups commonly attach at the 2, 4, 6, and 8 positions, spaced around the rings for maximum effect.
Chemists draw this molecule with the amine and nitro groups poking off in different spots from the main carbon backbone, like a well-balanced centerpiece with dramatic flair. Not just for looks, these arrangements shift the electron cloud of the molecule in very specific ways, influencing how it interacts with its neighbors and with whatever lab process it lands in.
Risks and Industrial Relevance
Over years of experience reading safety data sheets and working with various aromatic amines, one pattern keeps surfacing: nitro derivatives don’t mess around. Tetranitronaphthylamine shares the reputation. Exposure to such compounds can pose serious health hazards, including potential carcinogenic effects and risk of toxic buildup in the environment. Researchers figured out decades ago that heavier substitution with nitro groups often raises red flags for both handling and disposal.
Applying the Knowledge Responsibly
Anyone synthesizing or researching Tetranitronaphthylamine faces more than just technical hurdles. Despite its alluring reactivity, it demands respect for safety. Labs require proper fume hoods, gloves, and eye protection at all times, as contact with nitroaromatics can lead to skin absorption or worse. Environmental controls at the facility come built-in as standard now, since poorly managed waste introduces contamination risks that linger for generations.
Beyond the shop floor, regulators step in with mandates for labeling, documentation, and spill response. Anyone working hands-on learns quickly how these rules exist because somewhere, a real-world disaster gave rise to them. Even disposal involves chemical deactivation and coordination with hazardous waste handlers.
Pushing Chemistry Forward
Given advances in computational chemistry, now scientists lean on digital modeling to predict how novel molecules behave before anyone attempts a risky synthesis. If companies want to craft safer alternatives, these tools speed up the search for molecules with the benefits—minus the drawbacks—of compounds like Tetranitronaphthylamine. Research funding for cleaner reaction pathways or biodegradable nitroaromatics can come from both public and private sectors, filling in the gaps traditional industries left wide open for too long.
| Names | |
| Preferred IUPAC name | 2,4,6,8-tetranitro-1-naphthylamine |
| Other names |
Erionyl Yellow 6G C.I. Disperse Yellow 60 |
| Pronunciation | /ˌtɛtrəˌnaɪtrəˈnæfθɪləˌmiːn/ |
| Identifiers | |
| CAS Number | 132-28-9 |
| Beilstein Reference | 320478 |
| ChEBI | CHEBI:83493 |
| ChEMBL | CHEMBL63285 |
| ChemSpider | 21876074 |
| DrugBank | DB13703 |
| ECHA InfoCard | 100.011.058 |
| EC Number | EC 221-093-0 |
| Gmelin Reference | 132131 |
| KEGG | C18698 |
| MeSH | D031015 |
| PubChem CID | 21060841 |
| RTECS number | SE8925000 |
| UNII | LQJ2X0O5PU |
| UN number | UN3442 |
| CompTox Dashboard (EPA) | DTXSID8021328 |
| Properties | |
| Chemical formula | C10H5N5O8 |
| Molar mass | 303.15 g/mol |
| Appearance | Light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.67 g/cm³ |
| Solubility in water | insoluble |
| log P | 1.98 |
| Acidity (pKa) | 5.2 |
| Basicity (pKb) | 9.10 |
| Magnetic susceptibility (χ) | -1163.0e-6 cm³/mol |
| Refractive index (nD) | 1.620 |
| Dipole moment | 3.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 362.87 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -37.5 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1874 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Explosive, toxic if swallowed, causes skin and eye irritation. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-3-2-OX |
| Flash point | 79 °C |
| Autoignition temperature | 370 °C |
| Lethal dose or concentration | LD50 (rat, oral): 500 mg/kg |
| LD50 (median dose) | TDLo oral rat 60mg/kg |
| NIOSH | Not Listed |
| PEL (Permissible) | PEL (Permissible) for Tetranitronaphthylamine: 0.2 mg/m³ |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
1,3,5-Trinitrobenzene Picramide Tetryl |

