Tetranitronaphthalene: An Expert Perspective on an Explosive Compound
Historical Development
Long before labs bristled with sensors and digital timers, chemists looked for ways to harness energy locked inside molecules. The late 19th century saw a flurry of discoveries in nitration chemistry, as researchers experimented with adding nitro groups to aromatic hydrocarbons. Naphthalene drew particular attention. Scientists realized that by stepping up nitration, they could shoehorn four nitro groups into the naphthalene ring system, giving rise to tetranitronaphthalene. Researchers at the turn of the 20th century never worked in isolation—they built on lessons learned from previous explosive breakthroughs like trinitrotoluene and nitroglycerin. Tetranitronaphthalene arrived on the scene as part of a broader quest: extract every last ounce of potential from organic molecules and figure out how to store it safely.
Product Overview
Tetranitronaphthalene earned a place among a niche class of high-energy materials. Chemists know this compound for its deeply oxidized hydrocarbon backbone, with both naphthalene rings carrying two nitro groups each. Its chemical formula—C10H4N4O8—makes it both dense and oxygen-rich. Never gaining the mainstream prominence of TNT or PETN, it remains a specialty chemical. Labs prepare it in small batches, guided by demand for research, development, and certain analytical uses. Less often found in commercial munitions or demolition products, it still plays a useful role in the study of reaction mechanisms and as a potential ingredient in new energetic composites.
Physical & Chemical Properties
Tetranitronaphthalene appears as a pale yellow crystal with a melting point hovering around 270 °C, which sets it apart from many common nitroaromatic explosives. The material carries a molecular weight of about 296 g/mol. What interests many scientists is its sheer insensitivity, especially when sitting next to the more touchy primaries like lead azide. The solid remains relatively stable until heated or subjected to a strong shock, a feature that can appeal to those shaping new explosive blends. With four nitro groups tightly packed into a planar naphthalene framework, the substance resists both dissolution in water and spontaneous decomposition under ambient conditions. Its high density, about 2.0 g/cm3, suggests strong intermolecular interactions and provides a hint about its explosive power.
Technical Specifications & Labeling
The technical sheets spell out key data: purity sits above 97% in well-prepared lab samples, and the standards push for minimal moisture and trace contaminants. Labels on chemical containers must warn of explosion risk and high toxicity, with standardized hazard pictograms flagging dangers to both the user and the environment. Most suppliers use UN-approved containers, equipped with tamper-evident seals, to prevent leaks or unauthorized access. Batch numbers, synthesis dates, and storage advice sit right on the label—consistent with regulations from agencies like OSHA and REACH. While this might seem a tangle of bureaucracy, these details cut down on handling errors and mishaps.
Preparation Method
The road to tetranitronaphthalene runs through careful, staged nitration. Chemists start with pure naphthalene, dissolve it in concentrated sulfuric acid, and slowly chill the mixture. Nitric acid pours in next, and the temperature sits below 0 °C, since an uncontrolled exotherm can end experimentation badly. Once the first round produces dinitronaphthalene, further nitration brings in more nitro groups. It takes patience, timed additions, meticulous washing, and crystalline isolation to reach a pure product. Yields can run low, not least because the final product likes to stick to glassware or hide in extraction funnels. In practice, keeping the reaction cool and dry remains more than a suggestion—improper conditions bring runaways and toxic fumes, hazards that only proper lab controls and experienced hands can manage.
Chemical Reactions & Modifications
Tetranitronaphthalene resists simple reduction or oxidation. For chemists searching for new derivatives, though, the compound's nitro groups open the door to substitution reactions—though these rarely run smoothly. Attempts to swap out nitro groups, or to reduce one group to an amine, involve aggressive reagents and tight process controls. Chemists sometimes attach tetranitronaphthalene moieties to longer chain molecules or blend it into co-crystals, probing for altered explosive or spectral properties. Not many compounds match the stubbornness of this molecule; most reactions grind to a halt halfway through, requiring higher pressures, stubborn patience, and hours in the lab. Despite that, this reluctance to react lends value to tetranitronaphthalene as a stable intermediate in syntheses where other energetic molecules fall apart.
Synonyms & Product Names
On paper, one might spot alternate names like 1,3,6,8-tetranitronaphthalene or TNNA. Some suppliers stick to systematic nomenclature or use shorthand like TNN. Publications often mention all names, trying to sidestep confusion during literature searches. Chemistry thrives on clarity, so labeling both the systematic and common names on bottles and data sheets helps everyone dodge errors—especially in labs where multiple energetic materials jostle for shelf space.
Safety & Operational Standards
No one survives long in explosives chemistry by ignoring safety. Tetranitronaphthalene demands face shields, fume hoods, non-sparking tools, and above all, a working knowledge of what can go wrong. Storage in small, sealed, impact-resistant vials, surrounded by secondary containment, forms the backbone of safe practice. Regular safety drills, clear documentation of batch handling, and strong lines of communication among researchers ensure that no step in the synthesis, purification, or disposal process leaves room for error. Environmental controls pull hazardous vapors out of the workspace, and chemical disposal follows strict waste protocols—often involving specialist contractors. These steps are not just for show. History’s litany of accidents makes clear: every shortcut pays out disaster.
Application Area
Tetranitronaphthalene never became a staple of commercial explosives. Too hard to make, too sensitive to friction in some blends, too stubbornly inert in others, it lives mostly in research labs. Its role in analytical chemistry and materials science holds more promise: as a standard for calibrating instruments, as a stiffener for new polymer blends, or as a component in research into more powerful but less sensitive explosives. Its ability to survive without decomposing under moderate conditions makes it a valued molecule in studies that explore the fine line between stability and energy density. On rare occasions, military or industrial researchers tinker with it in search of novel energetic materials, but so far none of these projects have yielded a blockbuster application.
Research & Development
Academic interest in tetranitronaphthalene never quite disappears. Sometimes, researchers return to its chemistry with new tools—better analytical instruments, advanced computational modeling, or automated microbatch reactors that lower process risk. These advances dig up long-standing questions about reaction energetics and decomposition pathways. Collaboration with physicists and engineers leads to tests on detonation velocity, brisance, and how the compound behaves under dynamic load. Funding agencies take notice when the pursuit promises not just new explosives, but lessons in environmental remediation, waste destruction, or synthesis methods for tougher molecules. Tetranitronaphthalene stands as a bridge between old-school craftsmanship and modern chemical engineering.
Toxicity Research
The compound packs more than explosive force. Toxicologists pay close attention to organonitro compounds, which tend to spell trouble for both handlers and the environment. Early studies point to strong skin, eye, and respiratory irritation. Long-term exposure raises concerns over carcinogenicity and metabolic disruption, as seen with related nitroaromatics. Animal studies—rare and tightly regulated—suggest both acute and chronic effects, with rapid absorption in fatty tissues and slow excretion. The nitro groups pose the biggest metabolic hazard, often reduced in the body to reactive intermediates that attack DNA and proteins. Anyone working with tetranitronaphthalene must wear personal protective equipment and report all exposure incidents, as mandated by occupational health standards. Waste streams require special handling, as environmental agencies regulate nitroaromatic emissions out of concern for soil and water contamination.
Future Prospects
No one expects tetranitronaphthalene to replace modern mainstays like RDX or HMX in the near future. The future value for this compound lies not in volume applications but in scientific niches. As research dives deeper into energetic materials that balance power, environmental friendliness, and stability, the lessons drawn from tetranitronaphthalene’s chemistry pay dividends. New areas like additive manufacturing, smart materials, or nanostructured composites may find use for this molecule, if only as a tool for calibration or as part of a cocktail in complex reaction mixtures. Continuous innovation in synthetic methods, coupled with more robust safety protocols, ensures that this compound keeps its foothold in high-level research facilities. While the allure of a stable, high-energy explosive persists, only careful, evidence-based development will say where tetranitronaphthalene fits in the grander scheme of chemical discovery.
What is Tetranitronaphthalene used for?
Unpacking Tetranitronaphthalene’s Role in Industry
Tetranitronaphthalene isn’t a household name, but it pops up in some interesting corners of industrial chemistry. This compound, made up of a naphthalene core with four nitro groups, holds a powerful punch thanks to its energetic chemical structure. You won’t see this chemical on store shelves, but it finds its place in a few specialized areas—especially where energy and precision matter.
The Drive Behind Research and Application
Growing up, I learned that even the most intimidating chemicals can serve specific, surprisingly critical functions. I spent years in university labs, measuring out powders and liquids, constantly reminded how small changes in a molecule can change its entire reputation. Tetranitronaphthalene stands tall as a textbook example: it walks the line between risk and utility, catching the attention of military and industrial chemists alike.
Explosives and Detonators—Where Precision Reigns
Tetranitronaphthalene shows up in research focused on explosives. Its dense arrangement of nitro groups releases a lot of energy very quickly, which makes it useful in the search for more stable or efficient explosive materials. It doesn’t steal the spotlight from heavyweights like TNT or PETN, yet chemists sometimes study and test tetranitronaphthalene to understand the subtle balance between power, sensitivity, and stability.
Even in military circles, every new energetic material gets scrutinized. The conversation about safety and environmental risk never stops. Handling tetranitronaphthalene calls for real respect—goggles, gloves, close attention to protocol. A single slip-up can cost lives. Over the years, researchers have turned to controlled testing, detailed risk assessments, and advances in containment to wring out every drop of safety from this kind of compound.
Academic Curiosity and Analytical Value
Some chemicals draw a crowd just for their structure. Tetranitronaphthalene lands in this zone for chemists studying reaction pathways and molecular stability. Its four nitro groups offer a sort of challenge—for synthesis, for characterization, for the development of analytical methods. Spectroscopy studies and thermal analysis tell scientists more than textbooks ever could. During grad school, my lab group often referred to compounds like these as “puzzles,” and every time we cracked one, we learned a little more about how energetic materials behave.
Addressing Safety and Environmental Concerns
With chemicals built for big energy release, the conversation always swings toward environmental and health risk. Tetranitronaphthalene doesn’t break down easily. Its toxic byproducts demand careful disposal. I’ve seen labs move to greener solvents, amp up their containment practices, and double down on transparent record-keeping just to keep regulatory agencies and communities on board. Safer handling and improved waste processing sit high on the wish list for both researchers and those living near chemical production sites.
The Road Ahead: Seeking Safer Solutions
No one working with compounds as touchy as tetranitronaphthalene wants to gamble with safety or the planet. The push toward alternatives—less toxic, more manageable energetic materials—has picked up. Research centers funnel more resources towards greener chemistry. Collaboration between chemists, engineers, and environmental scientists keeps the spotlight on safety. Industry best practices don’t just protect those in the lab. They ripple out, building a safer future for communities everywhere.
What are the safety precautions when handling Tetranitronaphthalene?
Why This Chemical Raises Red Flags
Tetranitronaphthalene strikes a nerve with chemists, lab workers, and anyone who spends time with energetic compounds. Its structure packs four nitro groups onto a naphthalene ring, making it a powerful explosive. Anyone who has dealt with sensitive materials knows that accidents don’t always give warnings. Close calls, stained lab coats, or hearing news of a mishap across campus reminds us—certain substances don’t offer second chances. That’s what brings home the seriousness of working with something like tetranitronaphthalene.
Personal Protective Equipment Changes the Game
No one with sense approaches this chemical without a full set of personal protective equipment (PPE). Splash goggles protect eyes from dust or crystals, and face shields give an extra layer in case the unexpected happens. I’ve seen senior lab techs jump back when a reaction spatters near their face shield and later, the only damage is to the shield—the shield does its job. Nitrile gloves stand up against casual contact; layering gloves makes sense for extended work, since tiny tears go unnoticed. Laboratory coats, chemical aprons, and antistatic clothing cut the risk from static and friction. It doesn’t feel over the top; it feels like coming home safe at the end of the day.
Environmental Controls: Every Second Counts
It’s not enough to suit up. Fume hoods with explosion shields belong in every room where tetranitronaphthalene is weighed, transferred, or synthesized. I’ve worked with chemists who refused to do even the lightest manipulation outside a certified hood. Slow airflow pulls away fine dust and vapors, keeping breathing zones clear. Static electricity lurks in dry labs—grounding straps and antistatic benches bring a level of comfort before work starts. Temperature spikes can push energetic compounds past their limits, so temperature control and constant monitoring matter every hour, not just during reactions.
Storage and Handling: No Corners to Cut
Tetranitronaphthalene gets a place apart from other chemicals, in tightly sealed, blast-resistant containers. Brick-lined storage rooms, spark-resistant shelving, and keeping incompatible chemicals far away became core habits in my first year of lab work. Only work with the smallest practical amounts—old-timers taught me that there’s no glory in showing off with large samples, only risk. Clearly labelled containers stop confusion before it starts, so no one guesses what lurks inside those amber vials.
Training, Practice, and the Human Factor
Knowledge drops the risk by orders of magnitude. Routine drills and deep familiarity with emergency steps often turn a crisis into a story rather than a tragedy. I’ve trained students who dismissed paperwork and video demonstrations but paid full attention after hearing a seasoned chemist describe fingers lost to complacency. No one forgets that lesson. Record-keeping matters—knowing who used what, and how much remains in stock, can save lives in a fire or during disposal.
Sensible Solutions and Industry Progress
Nothing beats a healthy respect for high-energy chemicals. Research teams can invest in safer alternatives, remote manipulation systems, and robotic transfer setups. Good relationships with hazardous materials teams in your area create a strong safety net. Sharing incident reports helps everyone in the field stay a little sharper next time. Safety isn’t flashy, but every minor safeguard, practiced until second nature, stacks up to make a real difference.
What is the chemical formula of Tetranitronaphthalene?
Chemistry in Everyday Life
Walking through a hardware store, most people spot cleaning products, batteries, and gardening chemicals, rarely pausing to consider the science at play. Chemistry shapes the tools and technologies folks rely on, but some compounds make headlines for reasons tied to research, industry, and even history. Tetranitronaphthalene, a name that might trip up even word game experts, carries its own importance in the story of chemical discovery and application.
Understanding Tetranitronaphthalene
Tetranitronaphthalene isn’t your average household compound. It belongs to the nitroaromatics family — molecules featuring rings of carbon and hydrogen decorated with more than one nitro group (-NO2). For tetranitronaphthalene, this formula unfolds with a base of naphthalene, a structure made of two fused benzene rings. Chemists then bolt on four nitro groups. Here, those groups attach at specific positions — often the 1,3,6,8 spots on the ring. So, putting the puzzle together, its chemical formula reads as C10H4N4O8.
Why This Formula Has Weight
Learning that C10H4N4O8 packs in more nitro groups than most common organics hints at its power and risks. Researchers have leaned on this compound for its energetic properties — it stores and releases a considerable punch, sparking early work in explosives chemistry. High-nitro compounds often end up in labs and on patents, with strict safety guides tagging along.
The value sits not just in what the formula means in theory but how it gets handled, stored, and applied. Factories and military sites rely on strict chemical inventory controls, and formulas like that of tetranitronaphthalene flag substances that demand extra caution. Chemists and workers need up-to-date hazard training before getting anywhere near such potent molecules.
Supporting Facts
Decades of research papers back up concerns and applications tied to tetranitronaphthalene. The U.S. National Institute for Occupational Safety and Health singles out nitroaromatics as a class needing injury-prevention efforts. Evidence shows improper handling causes not just fires but serious health risks, from skin burns to respiratory problems. In the past century, countries introduced regulations on production volumes and trade, especially after notable accidents involving similar compounds.
Labs worldwide continue to track nitroaromatic compounds for advances in energetic materials, chemical sensors, and dyes. While some high-nitro chemicals fade from regular use, their design features carry forward in modern chemistry challenges, like building more selective explosives detectors or non-lethal tracers for scientific use.
Approaching Solutions and Responsibility
Experience in industrial research teaches the wisdom of clarity over complexity. Labels with clear chemical formulas — like C10H4N4O8 — save lives. Workers, regulators, and even curious students rely on accurate information to guard against preventable accidents. Using digital tracking systems, plain-language safety training, and transparent sourcing policies gives everyone a better shot at minimizing risks and getting science done right.
More broadly, sharing chemical knowledge helps demystify the world of molecules. Understanding what goes into compounds like tetranitronaphthalene invites conversations — about safety, creativity, and responsibility — that stretch far beyond the chemistry lab.
How should Tetranitronaphthalene be stored?
Living With Sensitive Chemicals: Lessons Learned
Anyone who has spent time in a laboratory or industrial setting knows that not all compounds behave the same once the lights go off and everyone goes home. Tetranitronaphthalene, known for its vibrant yellow crystals and volatile nature, stands out for more than its color. Years of working with energetic materials have shown me that rushing through storage plans ends up costing much more than prepping early. I’ve seen too many benches singed and too many near-misses because someone thought, “Just leave it here, it’ll be fine.”
Keeping It Safe and Stable
Proper storage protects people, property, and the integrity of the compound. Tetranitronaphthalene, loaded with nitro groups, picks up energy like dry grass on a sunny day, so keeping heat and shocks away sits at the top of the list. Cool, dry, and well-ventilated rooms far from any ignition source offer one safe bet. Flammable cabinets with built-in temperature controls do more than act as expensive furniture – they prevent unwanted chain reactions and help contain any mishaps that might come up.
Speaking from experience, skipping desiccants or humidity control is a shortcut to ruined samples and, worse, fire risks. Moisture degrades this energetic compound, making desiccators a regular fixture in my own labs. Insight from peer-reviewed studies backs this up, confirming that Tetranitronaphthalene doesn’t like damp air hovering above it.
Don’t Underestimate Protective Measures
Tetranitronaphthalene’s sensitivity to friction and static means ordinary plastic containers or neglected shelving just don’t cut it. Thick-walled, amber glass bottles with solid seals keep light and accidental bumps away. Grounding all equipment near your storage shelves may sound like overkill, but I watched a small static discharge wreck an entire chemical storage room during my grad school internship. Since then, habit and hindsight keep me using antistatic mats and making sure containers stay tightly closed.
Strong signage and clear labeling help people avoid mistakes. Every year, news reports plod through accident after accident—often the labels faded or got ignored, and someone mixed up safe and hazardous compounds. I make a point of using bold, easy-to-read signs on every shelf.
Daily Discipline Saves Trouble
Routine inspections go a long way toward keeping trouble out of the lab or storeroom. Any discoloration, clumping, or odd smells spell time for action. I plan regular checkups with the same energy I use for refilling the coffee pot—never waiting for problems to announce themselves. Inspections catch leaks and damage long before a full cleanup crew gets called in. Guidance from regulatory bodies like OSHA and the National Fire Protection Association recommends written checklists and logs. Keeping that record puts safety on autopilot, with backup if something goes wrong.
Disposal stands as another big issue. Letting waste pile up nearby multiplies the odds of accidental reaction. Every reputable chemist I know follows a process approved by authorities and leaves no gap for improvisation. My routine includes arranging pickups of old or unused material before containers ever come close to expiration dates.
Using Training and Teamwork
You can stack all the guidelines in the world, but nothing beats regular, hands-on training. In my lab, we run drills and keep everyone’s emergency response sharp. I never assume someone else “knows better” or “will handle it.” This keeps the mood light but the safety culture alive. The people around Tetranitronaphthalene are the best line of defense against accidents.
Safe storage always asks for attention to detail and respect for the risks. These aren’t just rules on a datasheet—they’re everyday habits that protect everyone involved.
Is Tetranitronaphthalene hazardous to health or the environment?
Understanding Tetranitronaphthalene in Everyday Safety
Many chemical names sound odd or rarely pop up outside textbooks. Tetranitronaphthalene, though, isn't just a tongue twister. It’s a synthetic chemical often discussed in academic papers and industrial settings as a raw material and sometimes for energetic compounds. Before diving into risks, it makes sense to break down what makes nitrated aromatics such as tetranitronaphthalene worthy of close attention in the first place.
Nitroaromatic compounds like this one carry nitro groups attached to aromatic rings. Chemists categorize these for both their reactive nature and potential impact on health and ecosystems. Even if it’s not handled daily, looking at lessons from similar chemicals helps paint a realistic picture. Many research journals have flagged nitroaromatics for their potential toxicity, explosive properties, and stubbornness in the environment.
Hazards: Health and Environment
Exposure to tetranitronaphthalene may take different routes—dust in the air, particles on the skin, accidental ingestion, or even fumes in heat. In labs, people suit up in gloves, goggles, and full coats around this stuff for good reason. Several analogues cause headaches, skin redness, and eye irritation, and have a real knack for causing breathing trouble if inhaled. Nitro groups attached to naphthalene rings almost always spell trouble for healthy tissues, either as irritants or worse, as mutagens—gunpowder for a cell’s normal DNA. Older safety studies suggest CHO cell mutations and hemolytic events can happen with certain nitro-naphthalenes.
Bigger trouble lands on anyone stuck around long-term, even in small doses. Persistent contact, even at very low air concentrations, can’t just be ignored. Skin absorption over months may cause systemic symptoms: fatigue, liver stress, blue-tinged skin and lips from disrupted oxygen flow. This is not something to gamble with at home, and certainly not without a chemist’s lab skills and respirators.
The environment reacts slowly, and sometimes the changes only show up years down the line. Some nitroaromatics stay in soil or water, barely breaking down, moving up the food chain, and building up in fish and birds. Pesticides and dyes from past decades left a legacy of polluted ponds and unhealthy wildlife with similar chemicals. Water plants and microbes often struggle to tackle these tightly bonded rings, which means breakdown takes longer, if it happens at all. People living near industrial waste sites sometimes watch their favorite streams lose plant life or fish from even a trace contamination.
What Can Be Done—Real Steps Forward
Stronger handling rules provide basic safety in manufacturing and lab work. These don’t just pop up overnight; they’re the result of disaster and hard-earned lessons—a fire, a chemical spill, a lab coat ruined by an accident. Setting up better ventilation, giving workers quality protective gear, and enforcing training for those who might handle tetranitronaphthalene stop injuries and mishaps before they turn ugly.
Most real-world action comes from those who speak out about local soil and water pollution. Community monitoring and chemical screening keep businesses honest, but need regular funding. Local governments and universities can drive soil and water testing downstream from any factory using such chemicals, with results shared publicly. Technology helps too; using filtration, activated carbon, and newer bioremediation tricks shortens how long these molecules stick around in rivers, soils, or landfill sludge. Watching what goes down the drain and writing smarter chemical disposal laws will always serve communities better in the long run.
It’s easy to treat obscure, hard-to-pronounce chemicals as someone else’s problem. The real hazard shows up when shortcuts replace real care and safety, whether in the workplace or in environmental management. Staying informed and pushing for honest oversight keeps everyone—at home, work, or outdoors—a bit safer against unseen chemical risks.
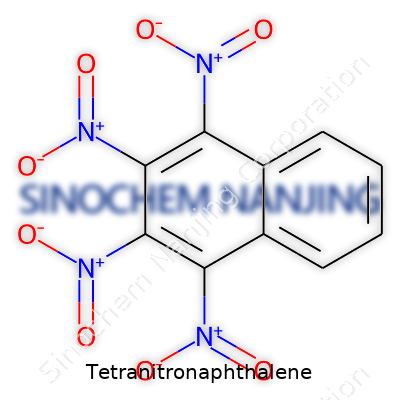
| Names | |
| Preferred IUPAC name | 1,3,6,8-Tetranitronaphthalene |
| Other names |
1,2,3,4-Tetranitronaphthalene |
| Pronunciation | /ˌtɛtrəˌnaɪtrəˈnæfθəˌliːn/ |
| Identifiers | |
| CAS Number | 132-86-5 |
| Beilstein Reference | 478878 |
| ChEBI | CHEBI:52226 |
| ChEMBL | CHEMBL4300208 |
| ChemSpider | 160256 |
| DrugBank | DB02302 |
| ECHA InfoCard | 100.007.417 |
| EC Number | 210-741-5 |
| Gmelin Reference | 136892 |
| KEGG | C16254 |
| MeSH | D014253 |
| PubChem CID | 92954 |
| RTECS number | QJ9625000 |
| UNII | D5X86T39F9 |
| UN number | 1321 |
| Properties | |
| Chemical formula | C10H4N4O8 |
| Molar mass | 289.13 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.73 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.83 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | -1.02 |
| Basicity (pKb) | -7.52 |
| Magnetic susceptibility (χ) | -87.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.688 |
| Viscosity | Viscosity: 1.289 mPa·s |
| Dipole moment | 2.54 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 329.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -22.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2067 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Explosive; may be sensitive to heat, shock, or friction. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06, GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H373, H410 |
| Precautionary statements | P210, P220, P221, P261, P280, P370+P378, P401, P501 |
| NFPA 704 (fire diamond) | 3-3-2 |
| Flash point | 46 °C |
| Autoignition temperature | 660 °C |
| Lethal dose or concentration | Lethal dose or concentration: LD50 intravenous (rat) 48 mg/kg |
| LD50 (median dose) | LD50 (median dose): 500 mg/kg (rat, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | °C25 |
| IDLH (Immediate danger) | IDLH: 50 mg/m³ |
| Related compounds | |
| Related compounds |
1,3,5-Trinitronaphthalene 1,3,6,8-Tetranitronaphthalene Tetranitromethane |

