Tetramethyl Orthosilicate: History, Character, and Impact
Historical Development
Tetramethyl orthosilicate, also known in the chemical world as TMOS, carved out its place in the toolbox of chemists and industry professionals over the 20th century. Laboratory research fueled its rise, with researchers seeking new ways to manipulate silicon’s unique chemistry to create glasses, coatings, and other advanced materials. Early on, academic labs experimented with this compound as raw material for sol-gel methods, opening the door for the development of high-purity silica glass and ceramics. Companies picked up on those breakthroughs, scaling up production through improved synthesis routes and refining purification strategies. Over time, TMOS shifted from academic curiosity to industrial mainstay, supporting sectors from electronics to optics, thanks to its versatility and clean conversion to silicon dioxide.
Product Overview
TMOS turns heads for its flexibility across a range of applications. Clear, with a sharp odor reminiscent of alcohols, TMOS doesn’t just serve as a processing chemical; it’s often the backbone of processes that shape advanced coatings, fiber optics, and encapsulation for electronics. Its ability to break down under mild conditions into high-purity silicon dioxide makes it an essential ingredient where clarity, strength, and stability are more than marketing points—they’re the foundation of performance. Markets demand technical materials that get the job done without bringing contamination into sensitive environments, and TMOS meets those demands by staying consistent and reliable.
Physical & Chemical Properties
TMOS presents as a colorless, volatile liquid with a pungent aroma. It carries a molecular weight of 152.23 g/mol, and its boiling point sits around 121°C under atmospheric pressure—significantly lower than some alternatives, which helps in both its application and cleanup. Its density, just below water at 1.03 g/cm³, means caution is essential during handling, since spills spread easily. TMOS does not mix with water—exposing it to moisture sparks a rapid hydrolysis reaction that kicks out methanol and gives birth to silica particles. Because TMOS’ vapors can be heavier than air, proper ventilation matters in every setting from benchtop to plant floor.
Technical Specifications & Labeling
TMOS typically appears in technical documents and chemical catalogs with a purity beyond 98%, sometimes reaching up to 99.9% for premium scientific use. Labels must spell out the dangers, indicating not just the chemical formula but the presence of flammable methanol byproducts released in many applications. Packages—always sealed tight against atmospheric moisture—carry hazard pictograms ranging from health to environmental warnings, mandating that users prepare mitigation plans, storage controls, and emergency actions. Each vial or drum will include batch numbers, expiry dates, and strict storage temperature guidelines, reinforcing traceability and safety in professional environments.
Preparation Method
Commercial synthesis of TMOS favors two main routes: direct reaction of silicon tetrachloride with methanol, often catalyzed by base or acid, or controlled transesterification processes involving silicon alkoxides. The chief challenge comes from controlling moisture intrusion, since even a tiny slip floods the reaction with premature hydrolysis. At industrial scale, process designers lean on closed systems, molecular sieves, and real-time monitoring tools to lock out humidity and maximize yield. Of course, purification can’t end with distillation alone—trace impurities can ruin electronic or optical devices, so every drop of TMOS used in high-tech manufacturing undergoes rigorous testing.
Chemical Reactions & Modifications
TMOS owes its popularity to lively reactivity, specifically its eager engagement with water in hydrolysis and condensation reactions. Exposing TMOS to even small amounts of moisture leads to rapid methanol formation and a slow build-up of silicon-oxygen networks. Creative chemists manipulate reaction rates, temperatures, catalysts, and additives to craft a stunning variety of glasses, gels, and nanostructured silicas. TMOS also enters transesterification reactions, swapping its methoxy groups for other alcohol residues under heat or catalytic pressure. In more advanced research, TMOS serves as a scaffold to introduce functional groups that open up new areas in catalyst and sensor design. This chemical’s unpredictable side emerges during uncontrolled hydrolysis, when dense aerosols and toxic byproducts remind everyone why respect for technical protocols stands above shortcuts or guesswork.
Synonyms & Product Names
Across the world’s laboratories, TMOS goes by a string of aliases, including tetramethyl silicate, methyl orthosilicate, and silica tetra-methyl ester. Catalog numbers shift by supplier, but every bottle points back to this silicon-based cornerstone. Not every label sits in English; global demand means instructions appear in multiple languages with international hazard codes. The spectrum of synonyms occasionally trips up newcomers, but experienced handlers recognize the molecule by both properties and context—regardless of the name stamped on the outside.
Safety & Operational Standards
TMOS belongs in the hands only of folks prepared for its hazards. Safe chemical storage keeps it away from heat, sparks, and sources of moisture. Most labs treat it with the same focus they bring to flammable solvents and strong acids: using chemical fume hoods, full personal protective gear, and spill containment at all times. Methanol, TMOS’s main hydrolysis byproduct, carries its own set of risks, including neurotoxicity and flammability, escalating the need for vigilance. Emergency eyewashes and showers, detailed handling protocols, and routine safety drills become standard where TMOS features regularly. Production plants add exhaust fans, explosion-proof lighting, and vapor monitors to keep workers out of harm’s way.
Application Area
TMOS finds steady work in sol-gel processing, a technology that’s reshaped how the world creates ultra-pure glass, ceramic coatings, and insulation materials. Semiconductor titans and fiber optic manufacturers order TMOS by the ton, relying on the purity and predictability it brings to fabrication lines. In the laboratory, TMOS stands as a keystone for researching thin-film structures or controlled porosity materials. Some medical device engineers experiment with TMOS-derived silica for encapsulation needs where biocompatibility and barrier properties must surpass plastics or metals. Artists and conservationists have turned to TMOS-based coatings to help preserve historical artifacts, betting on its transparency and chemical resistance to keep priceless works intact.
Research & Development
Modern R&D labs stay busy unlocking new possibilities with TMOS. Nanotechnology teams mix it with surfactants and templates to build intricate silica architectures, while sensor developers chase after thinner, more sensitive silica films. As renewable energy soaks up attention, visionaries tinker with TMOS to serve as protective barriers for perovskite solar cells and battery electrodes, seeking longer lifetimes and better efficiency. Collaborations between academic and industrial partners push TMOS chemistry deeper, mapping oddball reaction pathways or devising greener synthetic approaches. Each finding not only adds to chemical knowledge, but steadily converts ideas into products with direct impact on daily lives—whether in communications, computing, medicine, or sustainability.
Toxicity Research
Toxicologists pay close attention to the fate of TMOS and its breakdown products, especially in manufacturing environments. Acute TMOS exposure harms respiratory systems, with studies showing lung irritation, upper airway damage, and risk of pulmonary edema in severe cases. Its methanol byproduct turns into formaldehyde within the body, hammering the optic nerve and central nervous system even at low concentrations. Chronic workplace exposure links to long-term health impacts, feeding policies around ventilation, monitoring, and rigorous PPE practices. Environmental scientists monitor the fate of TMOS waste, since even dilute solutions break down into silica and methanol, tracking their entry into waterways and treatment systems. Research continues to pinpoint safer handling practices and potential medical countermeasures in case of accidental exposure.
Future Prospects
TMOS looks ready to keep shaping material science and engineering for years. The global push for thinner, purer, and more reliable materials steers research toward finding faster, safer, more energy-efficient synthesis methods. As societies prioritize green chemistry, companies seek bio-based or waste-reducing processes for TMOS production, reducing environmental footprints without giving up product quality. Tech giants expect next-generation electronics and photonics to depend on ultra-pure silica films, with TMOS at the starting gate. Academic partnerships will likely unlock new reactions or hybrid materials, pushing TMOS-based innovation into spaces ranging from drug delivery systems to artificial intelligence hardware. Every step forward with TMOS builds on hard-earned experience and a spirit of relentless improvement, turning a basic silicon ester into a platform for new ideas and technologies.
What is Tetramethyl Orthosilicate used for?
The Role of Tetramethyl Orthosilicate in Industry
Tetramethyl orthosilicate, often called TMOS, lands in a spot most people never think about but quietly powers everyday products and advanced technology. My first hands-on experience with TMOS happened in a chemistry lab at university. We wore heavy gloves and used fume hoods because this compound can release dangerous methanol as it changes. We needed TMOS for building thin films on glass slides. Watching glass go from plain and unused to coated with an even, smooth layer always felt a little magical, but the science behind it runs deep.
TMOS stands out as a top pick for making silica-based materials. Silica makes up a huge part of things people rely on daily—from fiberglass insulation tucked inside walls to the clear glass in smartphone screens. Companies use TMOS to build the basic structure behind gels for chromatography columns, coatings on electronics, and the light-diffusing material inside certain bulbs. TMOS reacts easily with water in a process that turns it into a web of silicon and oxygen atoms. This web forms the backbone in silica glass or intricate powders.
Why TMOS Finds a Home in High-Tech Labs
Looking back, I remember how much precision researchers expect when producing coatings for semiconductors. Most phones, computers, and solar panels work only thanks to ultra-clean, uniform layers acting as barriers or conductors. TMOS allows researchers to control layer thickness at the atomic level. By carefully adding water and adjusting temperature, engineers create films so thin you can’t see them with the naked eye. Labs regularly rely on it for building nano-scale sensors, mirrors for lasers, or even medical devices where purity can save lives.
TMOS crops up as a key ingredient for aerogels. Aerogels almost look like frozen smoke but offer excellent thermal insulation and incredible lightness. You’ll spot these materials in deep space missions, in fire barriers for astronauts, or even at the fingertips of people experimenting with energy-efficient windows. NASA’s Stardust mission, for example, collected comet dust with the help of silica aerogel—TMOS brings that future-facing technology within reach.
TMOS: Challenges and Moving Toward Safer Use
TMOS demands careful handling. Breathing in the vapors can harm your lungs, and spills lead to flammable methanol. Professional chemists stick to glove boxes and keep plenty of ventilation. Industrial groups push for more automation to cut the risks. In the labs where I spent time, regular training on spills and emergencies became essential, not optional.
The chemical industry has started to look for safer alternatives and better storage methods. Improved packaging lets workers pour or dispense TMOS without breaking a sweat about leaks. Digital tracking of inventory and real-time air sensors flag potential exposure. Some research explores replacing TMOS with less hazardous silicon sources or developing new processes that generate less methanol waste.
TMOS in the Wider World
For many people, chemistry feels distant. TMOS serves as a reminder of connections between basic science and modern living. From laboratories to outer space, from hospital instruments to the coating on your eyeglasses, TMOS quietly supports innovation. It’s the kind of ingredient that reminds me why materials science shapes progress in ways we sometimes overlook—but always depend on.
Is Tetramethyl Orthosilicate hazardous to health?
Why Workers Should Pay Attention
Most people haven’t heard much about Tetramethyl Orthosilicate, or TMOS, until they step into a lab, a glassworks facility, or an electronics plant. TMOS acts as a key ingredient for producing silica, coatings, and certain advanced materials. That part may sound technical, but there’s a more serious side that gets closer to home: the health risks once TMOS leaves the bottle.
Exposure: The Main Concern Lies in How People Handle TMOS
TMOS itself gives off a strong, penetrating odor. That’s usually a warning: breathe in too much and you’ll feel irritation in the nose and throat. Prolonged exposure or a high concentration can bring on coughing and even make breathing tougher. What might escape notice is that TMOS, when it meets moisture from the air or human skin, breaks down into methanol and silica. Methanol vapor isn’t something workers can brush off. Once inhaled or absorbed through the skin, methanol can cause headaches, dizziness, nausea, and — in high amounts — can damage nerves or eyesight.
From personal experience, standing near an open TMOS drum without good ventilation brings a sharp stinging in the eyes and nose. The skin dries out fast and, with poor handling, redness crops up quickly. Reading and following signage isn’t overkill in this situation. Relying on memory or workplace routine without real caution leads to unnecessary health scares.
Real Risks Go Deeper Than a Brief Encounter
Skin contact counts as an underestimated risk. TMOS can work its way through many types of gloves, especially when someone grabs the nearest pair without checking the safety sheet. Spill a drop, and that light tingling on bare skin is the first sign. Wash it off right away or risk developing a rash.
Working next door to a team mixing TMOS, I often saw folks skip protective eyewear or masks, figuring a quick task wasn’t risky. The result: persistent coughing that lasted through the week, a few missed shifts, and calls to poison control. TMOS isn’t something that lets people off easy even after a one-time mistake.
Guidelines Make a Difference
Strict rules like wearing goggles, using butyl rubber gloves, and setting up good fume hoods can sound like overkill, but they stop small mistakes from turning into long-term health issues. Washing up after every use keeps methanol and other breakdown products away from skin and eyes. For storage, solid seals and keeping TMOS out of sunlight or warmth help keep the workplace safer.
Some places invest in leak detectors or continuous air monitoring. While this takes a bit more effort on the front end, catching vapors early means less risk to the team and smaller fines from oversight agencies. Training sessions, even the short refresher courses, helped my old crew spot warning signs sooner and gave us confidence to call out problems without waiting for an accident.
Seeking Safer Alternatives
Certain industries have already started moving away from TMOS in favor of safer silicate reagents or well-contained silica sources. Pushing for greener chemistry doesn’t just improve air quality in the lab or shop: it removes headaches, dry skin, and the fear that comes with every odd cough after a spill. Supporting research into safer materials benefits companies, workers, and the broader community by cutting down both health risks and cleanup costs.
How should Tetramethyl Orthosilicate be stored?
Why Storage Matters
Tetramethyl orthosilicate, or TMOS, isn’t something anyone should treat loosely. I remember walking into a small research lab years ago, and noticing a bottle of this chemical on a cluttered shelf near a sunny window. Having worked in both commercial and academic settings, that scene made me uneasy. Not just because of regulation, but because of real risks. One mistake with TMOS can turn routine work into an emergency in seconds.
TMOS: Volatility and Health Risks
TMOS brings more than just the challenge of volatility. This colorless liquid releases fumes easily. Inhalation creates problems, not just mild irritation. Once inhaled, TMOS reacts with water in the respiratory tract and produces methanol and silica. Methanol is toxic, and the silica forms particulates that can scar lung tissue. Anyone who’s seen a co-worker cough for days after accidental exposure won’t soon forget.
Practical Storage Steps
A locked cabinet built from steel or flame-resistant materials gives the foundation for TMOS storage. Don’t put it near heat sources or direct sunlight. Flammable storage cabinets, the same type used for strong solvents, offer the right kind of separation. I’ve always told new lab workers: “Make finding TMOS harder than finding the coffee.” That level of isolation matters for both safety and accountability.
Handling Spills and Leaks
TMOS has a habit of leaking through loosely sealed bottles. Always double-check caps for tightness, and stand containers in spill trays with enough lip to catch a full bottle. During my time in a materials lab in New Jersey, a drip from a loose cap etched a permanent ring into a shelf. Avoiding naked metal contact helps, since TMOS can react over time. Glass or HDPE containers with the manufacturer’s label give clear information about dates and batch. If a spill happens, never let water near it. Water speeds up hydrolysis and releases more toxic vapors. Absorb spills with dry, inert materials like clay or sand, and then ventilate thoroughly.
Ventilation Is Non-Negotiable
Good airflow makes every difference. Always keep storage in a room with a professionally designed exhaust system. Old fans don’t cut it. In one academic lab, a faulty hood led to a visible haze that left everyone nauseated until we tracked it down. If the chemical smells at all, that’s a red flag to check seals and air movement.
Training and Labeling
Labels fade or peel. Add a backup tag with storage and hazard reminders to every container. Don’t trust memory, even if you’ve handled TMOS for years. Each person who works in the area should run through spill response drills every six months, not just watch a video on onboarding day. That kind of hands-on prep kept a small spill in check at my last workplace. No panic, just action.
Legal and Environmental Dimensions
Inspections come with paperwork, but the bigger issue rests with personal health and community safety. Local fire codes and OSHA rules both carry weight. Fines follow, but serious incidents bring career-long regret. Disposal must go through a licensed hazardous waste handler. Pouring TMOS down a drain, hoping dilution takes care of it, creates long-term problems in drinking water and wildlife.
Better Habits, Fewer Incidents
I’ve learned that setting up safe storage takes more effort up front, but the routine keeps everyone healthier and the workspace more reliable. Simple steps—strong cabinets, clear labeling, good training, solid ventilation, and regular checks—go a long way to turning hazardous chemicals into manageable tools.
What precautions should be taken when handling Tetramethyl Orthosilicate?
Understanding the Risks
Tetramethyl orthosilicate, often called TMOS, serves as a useful ingredient in silicone products, glass, and coatings. Its value in industrial and scientific work can’t be overstated, but it also brings real hazards that often slip under the radar for those less experienced with chemicals. TMOS vapor irritates the eyes, nose, and throat and can cause severe coughing or even lung damage at higher concentrations. Getting it on your skin can lead to burns. From stories and reports I’ve read through my time in the lab, I’ve seen what can happen when someone doesn’t give this chemical its due respect: ambulance rides, missed work, and lasting injuries.
Personal Safety Gear Comes First
Relying on gloves and goggles seems like common sense, but with TMOS, personal protective equipment (PPE) isn’t just a guideline; it’s a must. Every time I have worked with TMOS, I wore splash goggles, long sleeves, and heavy-duty chemical-resistant gloves like butyl rubber or nitrile—not the thin disposable kind. It’s easy to overlook a face shield, especially in busy environments, but it’s a step that protects against rare but serious splashes. Lab coats are good, but I prefer a chemical-resistant apron over standard cloth. TMOS evaporates readily, so I avoid open shoes and shorts at all costs.
Working with Ventilation
Some folks might think opening a window or turning on a fan will do the trick. Experience says otherwise. A certified chemical fume hood protects against both vapors and accidental spills much better than any makeshift ventilation. People sometimes try to shortcut this step when space is tight or fume hoods are in high demand, but I have seen colleagues suffer when they tried to mix or pour TMOS anywhere else. TMOS vapors can start harming your lungs before you even notice the smell.
Safe Storage and Labeling
During my first internship, a supervisor drilled into us the importance of proper storage. TMOS has no business anywhere near wet shelving or open chemical bins. Keep it in a cool, dry, tightly sealed container, away from acids, water, and bases. I always make sure the label is clear, with hazard warnings readable from a distance. One small container with missing labels once caused major confusion that led to a near-miss, so now I double-check labels as a habit.
Spill Response and Disposal
Accidents can catch anyone off guard. I always keep absorbent pads, neutralizing agents, and an emergency plan next to where I work. If a spill happens, evacuating untrained folks and alerting emergency personnel is key. Small spills can often be contained with sand or specialized spill kits made for organosilicon compounds. Any contaminated material goes in a sealed container for proper disposal, never into the drain or trash. Every lab or shop should post clear instructions for chemical disposal. Skipping proper disposal isn’t just unsafe—it can land someone in legal trouble.
Training and Familiarity
I’ve seen new team members hesitate with TMOS simply because safety training fell short. Regular workshops and refreshers help everyone stay on the same page. Sharing personal experiences, not just dry regulations, really reinforces the message. In my teams, we do short walk-throughs and drills, focusing on real problems we might face—like leaking bottles or an unexpected splash.
Making Safety a Habit
Handling TMOS always brings risk, but with the right gear, procedures, and attitude, serious accidents become rare. Trust in established safety rules has to be backed up by personal responsibility. Each person in the shop, lab, or warehouse needs to treat TMOS as something that commands attention and care. Cutting corners never pays off in the long run.
What is the chemical formula of Tetramethyl Orthosilicate?
The Formula: Si(OCH3)4
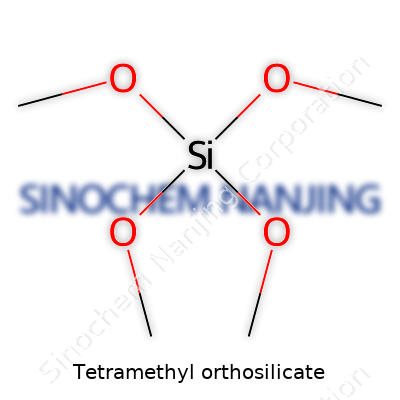
Tetramethyl orthosilicate, better known in labs as TMOS, carries the formula Si(OCH3)4. Each molecule bundles one silicon atom with four methoxy groups. Chemistry folks usually draw it like a spider with silicon at the center and four arms reaching out – each arm ending with a CH3O group instead of the usual hydroxides you see in regular silica. Not exactly your average glass ingredient, but close in family.
Why Pay Attention to the Formula?
TMOS comes up in sol-gel chemistry, a field filled with complex reactions and creative materials. I've seen TMOS used for making glass coatings, porous membranes, and even as a precursor for silica nanoparticles. With a formula like Si(OCH3)4, the molecule packs both organic and inorganic chemistry in a single punch. The four methoxy groups make it more flexible compared to the more common tetraethyl orthosilicate (TEOS), especially when you want to play with different solubilities or hydrolysis rates.
Health and Safety Concerns
TMOS doesn't belong in the "harmless" category. It vaporizes easily and reacts with water from the air. Breathing even a little bit produces methanol and silica dust in your lungs, which isn’t ideal. OSHA and NIOSH agree that TMOS needs careful handling. Research from the National Institute for Occupational Safety and Health points to serious risks, including pulmonary fibrosis and blindness, after exposure. Eye protection, gloves, and ventilated hoods go from “good idea” to “don’t even open the bottle without them.”
Environmental Impact
TMOS drifts into the conversation about green chemistry. In the past, chemists would toss leftover TMOS, trusting the methanol would eventually evaporate. That sort of thinking doesn’t work anymore. Methanol release harms waterways and aquatic life. Now, waste gets contained and neutralized. Companies invest in closed-loop systems and try to switch to less toxic silicates whenever possible. I’ve seen university labs and industry partners work together to design recovery and reuse systems—this turns waste into a resource and keeps TMOS out of the local river.
Real-World Innovation
Folks working in fiber optics, microelectronics, and even arts conservation tap into TMOS for its versatility. It crosslinks easily to form pinhole-free silica layers, crucial for protecting sensors or creating delicate optical waveguides. The formula, Si(OCH3)4, means tight control over the hydrolysis, so the resulting silica network reaches just the right balance of porosity and durability. Researchers have used TMOS to protect priceless medieval manuscripts—the silica layer acts like a nearly invisible armor, blocking acids, air, and mold.
Looking Ahead: Safer and Smarter Use
As demand for advanced materials grows, so does the pressure to keep TMOS use sustainable and safe. Reading chemical formulas doesn’t solve safety or environmental challenges by itself, but knowing what’s inside a bottle sets the stage for responsible action. Teams that educate, monitor use, and reinvest in safer technology push the field forward. TMOS, with its Si(OCH3)4 structure, opens doors—provided we treat it with the respect both science and nature demand.
| Names | |
| Preferred IUPAC name | tetramethyl silicate |
| Other names |
Tetramethoxysilane TMOS Methyl silicate Silicic acid tetramethyl ester |
| Pronunciation | /ˌtɛtrəˈmɛθɪl ɔːrθəˈsɪlɪkeɪt/ |
| Identifiers | |
| CAS Number | '78-10-4' |
| Beilstein Reference | 1461135 |
| ChEBI | CHEBI:9507 |
| ChEMBL | CHEMBL157248 |
| ChemSpider | 12990 |
| DrugBank | DB11235 |
| ECHA InfoCard | 13bab18a-6d1c-4d76-9f1e-7ebab1f4ada2 |
| EC Number | 208-774-1 |
| Gmelin Reference | Gmelin1131 |
| KEGG | C19154 |
| MeSH | D013742 |
| PubChem CID | 6626 |
| RTECS number | VV7325000 |
| UNII | VJ55U3FPUW |
| UN number | 1292 |
| CompTox Dashboard (EPA) | 1007 |
| Properties | |
| Chemical formula | Si(OCH₃)₄ |
| Molar mass | 152.24 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Alcohol-like |
| Density | 0.955 g/mL at 25 °C (lit.) |
| Solubility in water | Decomposes |
| log P | 0.2 |
| Vapor pressure | 1 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 12.7 |
| Basicity (pKb) | pKb: 3.24 |
| Magnetic susceptibility (χ) | -60×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.382 |
| Viscosity | 0.62 mPa·s (25 °C) |
| Dipole moment | 2.08 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1360.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3185 kJ/mol |
| Pharmacology | |
| ATC code | J05AX15 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H226, H302, H332, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 41 °C |
| Autoignition temperature | 250°C |
| Explosive limits | Explosive limits: 1.3–16% |
| Lethal dose or concentration | LD50 (oral, rat): 8500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 8500 mg/kg |
| NIOSH | SI7175000 |
| PEL (Permissible) | PEL: 10 ppm |
| REL (Recommended) | REL (Recommended): 10 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Tetraethyl orthosilicate Trimethylsilyl chloride Methylsilane Silicon dioxide |

